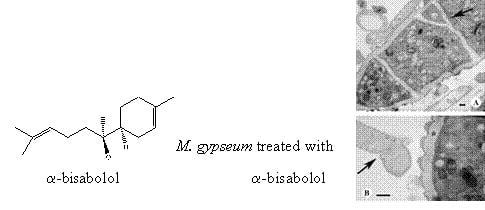A Multi-Target Approach toward the Development of Novel Candidates for Antidermatophytic Activity: Ultrastructural Evidence on α-Bisabolol-Treated Microsporum gypseum
Abstract
:1. Introduction

2. Results and Discussion
2.1. Antifungal Activity
| Kojic Acid | β-Arbutin | Azelaic Acid | Hydroquinone | Nicotinamide | α-Bisabolol | Glycine | Glutathione | Ascorbyl TIP | ||
|---|---|---|---|---|---|---|---|---|---|---|
| M. gypseum | 100 | 3.5 ± 0.1 | 7.9 ± 0.2 | 6.9 ± 0.2 | 17.1 ± 0.1 | 9.8 ± 0.1 | 79.8 ± 0.4 | + | + | 16.6 ± 0.3 |
| 200 | 7.5 ± 0.2 | 27.9 ± 0.1 | 19.8 ± 0.1 | 51.2 ± 0.1 | 18.3 ± 0.4 | 83.7 ± 0.2 | 0.8 ± 0.1 | + | 21.3 ± 0.2 | |
| M. canis | 100 | 22.6 ± 0.3 | −11.8 ± 0.2 | −5.9 ± 0.1 | 20.0 ± 0.1 | 2.4 ± 0.1 | 34.7 ± 0.2 | 0.0 | 0.0 | + |
| 200 | 29.0 ± 0.1 | −2.9 ± 0.2 | 5.9 ± 0.3 | 46.7 ± 0.2 | 12.0 ± 0.1 | 43.1 ± 0.2 | + | 3.8 ± 0.3 | + | |
| T. violaceum | 100 | 18.2 ± 0.2 | −2.8 ± 1.4 | −8.2 ± 0.9 | 20.0 ± 1.2 | 3.4 ± 1.8 | 31.0 ± 0.8 | + | 9.8 ± 2.3 | + |
| 200 | 19. 9 ± 1.1 | −5.0 ± 0.7 | 2.5 ± 1.6 | 30.6 ± 1.3 | + | 38.0 ± 1.8 | 0.0 | 12.0 ± 1.4 | 15.6 ± 2.0 | |
| N. cajetani | 100 | 2.7 ± 1.1 | 6.9 ± 1.6 | 0.9 ± 1.9 | 0.0 | 14.4 ± 0.7 | 60.1 ± 1.3 | 0.0 | + | + |
| 200 | 8.0 ± 1.0 | 7.6 ± 1.4 | 0.0 | 4.2 ± 0.3 | 25.1 ± 0.6 | 62.7 ± 1.5 | + | 5.2 ± 1.3 | 0.0 | |
| T. mentagroph | 100 | 0.0 | −1.5 ± 0.5 | 0.0 | 29.7 ± 1.8 | 50.8 ± 1.8 | 67.0 ± 1.3 | 6.1 ± 0.6 | + | 9.8 ± 1.1 |
| 200 | 6.3 ± 1.2 | 0.0 | 9.3 ± 0.8 | 59.4 ± 1.4 | 78.0 ± 3.1 | 70.9 ± 1.7 | + | + | 19.6 ± 0.8 | |
| E. floccosum | 100 | 1.9 ± 1.7 | −1.6 ± 0.5 | 0.3 ± 0.1 | 2.5 ± 0.2 | 7.8 ± 0.7 | 31.8 ± 1.1 | + | 0.6 ± 0.2 | + |
| 200 | 2.5 ± 0.9 | −1.3 ± 0.5 | 3.9 ± 1.0 | 5.4 ± 0.8 | + | 33.1 ± 1.9 | + | 0.0 | 6.4 ± 0.9 | |
| A. gypseum | 100 | 10.0 ± 1.2 | 0.0 | 7.4 ± 1.4 | −11.0 ± 0.9 | 1.9 ± 1.2 | 62.0 ± 1.7 | + | 0.6 ± 2.1 | 0.7 ± 0.5 |
| 200 | 11.3 ± 1.2 | 2.3 ± 1.8 | 3.7 ± 1.4 | 2.4 ± 1.1 | 7.1 ± 0.6 | 65.8 ± 1.3 | + | 1.3 ± 1.0 | + | |
| T. rubrum | 100 | −0.4 ± 0.8 | 6.1 ± 1.4 | 0.8 ± 0.5 | −2.3 ± 1.9 | 4.5 ± 0.3 | 14.6 ± 1.9 | + | 2.6 ± 0.2 | 0.0 |
| 200 | 2.9 ± 0.7 | 6.8 ± 1.3 | 1.6 ± 0.7 | 2.9 ± 0.6 | 10.8 ± 1.1 | 37.4 ± 1.4 | + | + | + | |
| T. tonsurans | 100 | 0.0 | 4.4 ± 0.5 | −2.6 ± 1.2 | 40.0 ± 1.1 | 9.3 ± 1.3 | 69.6 ± 1.9 | + | + | + |
| 200 | 5.0 ± 0.5 | 11.1 ± 0.7 | 0.0 | 55.00 ± 1.5 | + | 71.7 ± 1.2 | 0.0 ± 0.8 | + | + |
| α-Bisabolol | Fluconazole | |
|---|---|---|
| IC50 μg/mL | ||
| M. gypseum | 35.24 ± 0.9 | 18.5 ± 0.2 |
| M. canis | >200 | 29.6 ± 0.3 |
| T. violaceum | >200 | 31.03 ± 0.9 |
| N. cajetani | 85 ± 0.6 | >200 |
| T. mentagrophytes | 60.51 ± 0.3 | 3.53 ± 0.8 |
| E. floccosum | >200 | 0.08 ± 0.6 |
| A. gypseum | 81.69 ± 0.4 | >200 |
| T. rubrum | >200 | 37.16 ± 0.6 |
| T. tonsurans | 49.38 ± 0.7 | 19.41 ± 0.5 |
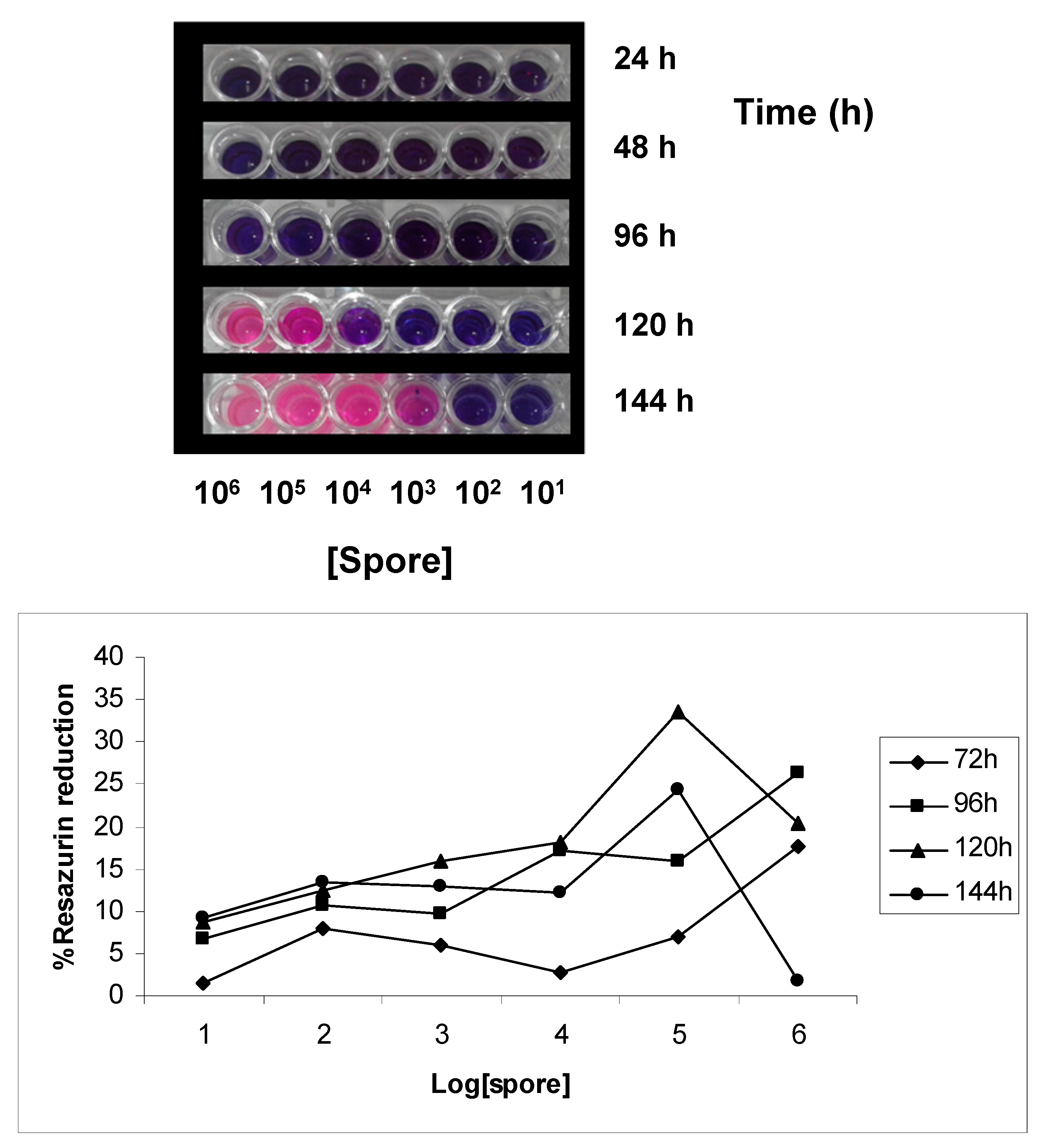
2.2. Effect of Time and Spore Density on Percent Reduction of Resazurin
2.3. Inhibition of Spore Germination by Resazurin Assay
| α-Bisabolol Concentration | %Inhibition of Spore Germination |
|---|---|
| 100 μg/mL | 52.08 ± 0.9 |
| 200 μg/mL | 59.32 ± 1.3 |
2.4. TEM


3. Experimental Section
3.1. Chemicals
3.2. Microorganisms
3.3. Antifungal Activity
3.4. Determination of Optimal Spore Concentration-Growth in Sabouraud Dextrose Broth in Resazurin Assay
3.5. Evaluation of Inhibition of Spore Germination of α-Bisabolol
3.6. Spectrophotometric Measurement and Visual Inspection
3.7. Transmission Electron Microscopy (TEM)
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Havlickova, B.; Czaika, V.A.; Friedrich, M. Epidemiological trends in skin mycoses worldwide. Mycoses 2008, 51, 2–15. [Google Scholar] [CrossRef] [PubMed]
- Jamalian, A.; Shams-Ghahfarokhi, M.; Jaimand, K.; Pashootan, N.; Amani, A.; Razzaghi-Abyaneh, M. Chemical composition and antifungal activity of Matricaria recutita flower essential oil against medically important dermatophytes and soil-borne pathogens. J. Mycol. Med. 2012, 22, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Mares, D. Electron microscopy of Microsporum cookie after “in vitro” treatment with protoanemonin: A combined SEM and TEM study. Mycopathologia 1989, 108, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Romagnoli, C.; Mares, D.; Fasulo, M.P.; Bruni, A. Antifungal effects of α-terthienyl from Tagetes patula on five dermatophytes. Phytother. Res. 1994, 8, 332–336. [Google Scholar] [CrossRef]
- Romagnoli, C.; Bruni, R.; Andreotti, E.; Rai, M.K.; Vicentini, C.B.; Mares, D. Chemical characterization and antifungal activity of essential oil of capitula from wild Indian Tagetes patula L. Protoplasma 2005, 225, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Mares, D.; Romagnoli, C.; Tosi, B.; Andreotti, E.; Chillemi, G.; Poli, F. Chicory extracts from Cichorium intybus L. as potential antifungals. Mycopathologia 2005, 160, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Mares, D.; Romagnoli, C.; Sacchetti, G.; Vicentini, C.B.; Bruni, A. Morphological study of Trichophyton rubrum; ultrastructural findings after treatment with 4-amino-3-methyl-1-phenylpyrazolo-(3,4-c)isothiazole. J. Mycol. Med. 1998, 36, 379–385. [Google Scholar] [CrossRef]
- Mares, D.; Romagnoli, C.; Tosi, B.; Benvegnù, R.; Bruni, A.; Vicentini, C.B. Mannan changes induced by 3-methyl-5-aminoisoxazole-4-thiocyanate, a new azole derivative, on Epidermophyton floccosum. Fungal Genet. Biol. 2002, 36, 47–57. [Google Scholar]
- Vicentini, C.B.; Forlani, G.; Manfrini, M.; Romagnoli, C.; Mares, D. Development of New Fungicides against Magnaporthe grisea: Synthesis and Biological Activity of Pyrazolo[3,4-d][1,3]thiazine, Pyrazolo[1,5-c][1,3,5]thiadiazine, and Pyrazolo[3,4-d]pyrimidine Derivatives. J. Agric. Food Chem. 2002, 50, 4839–4845. [Google Scholar]
- Vicentini, C.B.; Romagnoli, C.; Andreotti, E.; Mares, D. Synthetic Pyrazole Derivatives as Growth Inhibitors of Some Phytopathogenic Fungi. J. Agric. Food Chem. 2007, 55, 10331–10338. [Google Scholar] [CrossRef] [PubMed]
- Kurahashi, Y. Melanin biosynthesis inhibitors (MBIs) for control of rice blast. Pestic. Outlook 2001, 12, 32–35. [Google Scholar] [CrossRef]
- Mares, D.; Romagnoli, C.; Andreotti, E.; Forlani, G.; Guccione, S.; Vicentini, C.B. Emerging antifungal azoles and effects on Magnaporthe grisea. Mycol. Res. 2006, 110, 686–696. [Google Scholar] [CrossRef] [PubMed]
- Parvez, S.; Kang, M.; Chung, H.S.; Cho, C.; Hong, M.C.; Shin, M.K.; Bae, H. Survey and mechanism of skin depigmenting and lightening agents. Phytother. Res. 2006, 20, 921–934. [Google Scholar] [CrossRef] [PubMed]
- Alexis, A.F.; Blackcloud, P. Natural ingredients for darker skin types: Growing options for hyperpigmentation. J. Drugs Dermatol. 2013, 12, 123–127. [Google Scholar]
- Ishikawa, M.; Kawas, I.; Ishii, F. Glycine inhibits melanogenesis in vitro and causes hypopigmentation in vivo. Biol. Pharm. Bull. 2007, 30, 2031–2036. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Jun, H.; Jung, E.; Ha, J.; Park, D. Whitening effect of alpha-bisabolol in Asian women subjects. Int. J. Cosmet. Sci. 2010, 32, 299–303. [Google Scholar] [CrossRef] [PubMed]
- Solano, F.; Briganti, S.; Picardo, M.; Ghanem, G. Hypopigmenting agents: An updated review on biological, chemical and clinical aspects. Pigment Cell Res. 2006, 19, 550–571. [Google Scholar] [CrossRef] [PubMed]
- Tabanca, N.; Demirci, B.; Crockett, S.A.L.; Baser, K.H.C.; Wedge, D.E. Chemical Composition and Antifungal Activity of Arnica longifolia, Aster hesperius, and Chrysothamnus nauseosus Essential Oils. J. Agric. Food Chem. 2007, 55, 8430–8435. [Google Scholar] [CrossRef] [PubMed]
- Pauli, A. α-Bisabolol from Chamomile—A specific ergosterol biosynthesis inhibitor? Int. J. Aromather. 2006, 16, 21–25. [Google Scholar] [CrossRef]
- De Lucca, A.; Pauli, A.; Schilcher, H.; Sien, T.; Bhatnagar, D.; Walsh, T.J. Fungicidal and bactericidal properties of bisabolol and dragosantol. J. Essent. Oil Res. 2011, 23, 47–54. [Google Scholar] [CrossRef]
- Tabassum, N.; Vidyasagar, G.M. Antifungal investigations on plant essential oils. Int. J. Pharm. Pharm. Sci. 2013, 5, 19–28. [Google Scholar]
- Inoue, S.; Turgeon, B.G.; Yoder, O.C.; Aist, J.R. Role of fungal dynein in hyphal growth, microtubule organization, spindle pole body motility and nuclear migration. J. Cell Sci. 1998, 111, 1555–1566. [Google Scholar] [PubMed]
- Beaudouin, J.; Gerlich, D.; Eils, N.D.R.; Ellenberg, J. Nuclear Envelope Breakdown Proceeds by Microtubule-Induced Tearing of the Lamina. Cell 2002, 108, 83–96. [Google Scholar] [CrossRef]
- Mares, D. Fungal morphogenesis induced by natural and synthetic substances: Herniarin-induced alterations in the dermatophyte Microsporum cookie Ajello. Plant Biosyst. 2005, 139, 323–334. [Google Scholar] [CrossRef]
- Howard, R.J.; Aist, J.R. Cytoplasmic microtubules and fungal morphogenesis: Ultrastructural effects of methyl benzimidazole-2-yl carbamate determined by freeze substitution of hyphal cells. J. Cell Biol. 1980, 118, 109–120. [Google Scholar] [CrossRef]
- Georgopapadakou, N.H. Antifungals: Mechanism of action and resistance, established and novel drugs. Curr. Opin. Microbiol. 1998, 1, 547–557. [Google Scholar] [CrossRef]
- Odds, F.C.; Brown, A.J.P.; Gow, N.A.R. Antifungal agents: Mechanisms of action. Trends Microbiol. 2003, 11, 272–279. [Google Scholar] [CrossRef]
- Rampersad, S.N. A rapid colorimetric microtiter bioassay to evaluate fungicide sensitivity among Verticillum dahliae Isolates. Plant Dis. 2011, 95, 248–255. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romagnoli, C.; Baldisserotto, A.; Malisardi, G.; Vicentini, C.B.; Mares, D.; Andreotti, E.; Vertuani, S.; Manfredini, S. A Multi-Target Approach toward the Development of Novel Candidates for Antidermatophytic Activity: Ultrastructural Evidence on α-Bisabolol-Treated Microsporum gypseum. Molecules 2015, 20, 11765-11776. https://0-doi-org.brum.beds.ac.uk/10.3390/molecules200711765
Romagnoli C, Baldisserotto A, Malisardi G, Vicentini CB, Mares D, Andreotti E, Vertuani S, Manfredini S. A Multi-Target Approach toward the Development of Novel Candidates for Antidermatophytic Activity: Ultrastructural Evidence on α-Bisabolol-Treated Microsporum gypseum. Molecules. 2015; 20(7):11765-11776. https://0-doi-org.brum.beds.ac.uk/10.3390/molecules200711765
Chicago/Turabian StyleRomagnoli, Carlo, Anna Baldisserotto, Gemma Malisardi, Chiara B. Vicentini, Donatella Mares, Elisa Andreotti, Silvia Vertuani, and Stefano Manfredini. 2015. "A Multi-Target Approach toward the Development of Novel Candidates for Antidermatophytic Activity: Ultrastructural Evidence on α-Bisabolol-Treated Microsporum gypseum" Molecules 20, no. 7: 11765-11776. https://0-doi-org.brum.beds.ac.uk/10.3390/molecules200711765