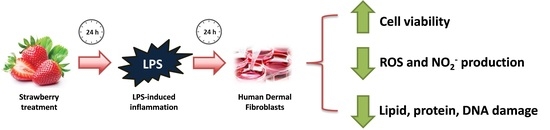
Protective Effect of Strawberry Extract against Inflammatory Stress Induced in Human Dermal Fibroblasts
Abstract
:1. Introduction
2. Results and Discussion
2.1. Strawberry Fruit Analysis
2.2. Strawberry and LPS Effects on Cell Viability
2.3. Strawberry Treatment Reduced ROS Intracellular Production
2.4. Strawberry Treatment Decreased NO2− Accumulation
2.5. Strawberry Treatment Reduced Protein, Lipid and DNA Damage
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Strawberry Fruit Preparation and Analysis
3.3. Cell Culture and Strawberry/LPS Treatments
3.4. Cell Viability Assay
3.5. TALI® ROS Concentration Assay
3.6. Determination of Nitrite Production
3.7. Measurements of the Protein and Lipid Oxidative Damage
3.8. Immunoblotting Assay for DNA Damage Evaluation
3.9. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Joseph, S.V.; Edirisinghe, I.; Burton-Freeman, B.M. Berries: Anti-inflammatory Effects in Humans. J. Agric. Food Chem. 2014, 62, 3886–3903. [Google Scholar] [CrossRef] [PubMed]
- Trachootham, D.; Lu, W.; Ogasawara, M.A.; Nilsa, R.D.; Huang, P. Redox regulation of cell survival. Antioxid. Redox Signal. 2008, 10, 1343–1374. [Google Scholar] [CrossRef] [PubMed]
- Ray, P.D.; Huang, B.W.; Tsuji, Y. Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling. Cell Signal. 2012, 24, 981–990. [Google Scholar] [CrossRef] [PubMed]
- Elliott, C.G.; Forbes, T.L.; Leask, A.; Hamilton, D.W. Inflammatory microenvironment and tumor necrosis factor alpha as modulators of periostin and CCN2 expression in human non-healing skin wounds and dermal fibroblasts. Matrix Biol. 2015, 43, 71–84. [Google Scholar] [CrossRef] [PubMed]
- Landén, N.X.; Li, D.; Ståhle, M. Transition from inflammation to proliferation: A critical step during wound healing. Cell. Mol. Life Sci. 2016, 73, 3861–3885. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Suarez, J.M.; Giampieri, F.; Cordero, M.; Gasparrini, M.; Forbes-Hernández, T.Y.; Mazzoni, L.; Afrin, S.; Beltrán-Ayala, P.; González-Paramás, A.M.; Santos-Buelga, C.; et al. Activation of AMPK/Nrf2 signalling by Manuka honey protects human dermal fibroblasts against oxidative damage by improving antioxidant response and mitochondrial function promoting wound healing. J. Funct. Foods 2016, 25, 38–49. [Google Scholar] [CrossRef]
- Afrin, S.; Giampieri, F.; Gasparrini, M.; Forbes-Hernandez, T.Y.; Varela-López, A.; Quiles, J.L.; Mezzetti, B.; Battino, M. Chemopreventive and Therapeutic Effects of Edible Berries: A Focus on Colon Cancer Prevention and Treatment. Molecules 2016, 21, 169. [Google Scholar] [CrossRef] [PubMed]
- Davies, K.J. Oxidative stress, antioxidant defenses, and damage removal, repair, and replacement systems. IUBMB Life 2000, 50, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Kohen, R.; Nyska, A. Oxidation of biological systems: Oxidative stress phenomena, antioxidants, redox reactions, and methods for their quantification. Toxicol. Pathol. 2002, 30, 620–650. [Google Scholar] [CrossRef] [PubMed]
- Forbes-Hernández, T.Y.; Giampieri, F.; Gasparrini, M.; Mazzoni, L.; Quiles, J.L.; Alvarez-Suarez, J.M.; Battino, M. The effects of bioactive compounds from plant foods on mitochondrial function: A focus on apoptotic mechanisms. Food Chem. Toxicol. 2014, 68, 154–182. [Google Scholar] [CrossRef] [PubMed]
- Giampieri, F.; Alvarez-Suarez, J.M.; Battino, M. Strawberry and human health: Effects beyond antioxidant activity. J. Agric. Food Chem. 2014, 62, 3867–3876. [Google Scholar] [CrossRef] [PubMed]
- McCarty, M.F.; DiNicolantonio, J.J.; O’Keefe, J.H. Capsaicin may have important potential for promoting vascular and metabolic health. Open Heart 2015, 2, e000262. [Google Scholar] [CrossRef] [PubMed]
- Giampieri, F.; Tulipani, S.; Alvarez-Suarez, J.M.; Quiles, J.L.; Mezzetti, B.; Battino, M. The strawberry: Composition, nutritional quality, and impact on human health. Nutrition 2012, 28, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Giampieri, F.; Alvarez-Suarez, J.M.; Mazzoni, L.; Romandini, S.; Bompadre, S.; Diamanti, J.; Capocasa, F.; Mezzetti, B.; Quiles, J.L.; Ferreiro, M.S.; et al. The potential impact of strawberry on human health. Nat. Prod. Res. 2013, 27, 448–455. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Kim, S.; Namgung, H.; Jo, Y.H.; Bao, C.; Choi, H.K.; Auh, J.H.; Lee, H.J. Ellagic acid identified through metabolomic analysis is an active metabolite in strawberry (‘Seolhyang’) regulating lipopolysaccharide-induced inflammation. J. Agric. Food Chem. 2014, 62, 3954–3962. [Google Scholar] [CrossRef] [PubMed]
- Alarcón, M.; Fuentes, E.; Olate, N.; Navarrete, S.; Carrasco, G.; Palomo, I. Strawberry extract presents antiplatelet activity by inhibition of inflammatory mediator of atherosclerosis (sP-selectin, sCD40L, RANTES, and IL-1β) and thrombus formation. Platelets 2015, 26, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Amatori, S.; Mazzoni, L.; Alvarez-Suarez, J.M.; Giampieri, F.; Gasparrini, M.; Forbes-Hernandez, T.Y.; Afrin, S.; Errico Provenzano, A.; Persico, G.; Mezzetti, B.; et al. Polyphenol-rich strawberry extract (PRSE) shows in vitro and in vivo biological activity against invasive breast cancer cells. Sci. Rep. 2016, 6, 30917. [Google Scholar] [CrossRef] [PubMed]
- Mayer, H.; Tharanathan, R.N.; Weckesser, J. Analysis of Lipopolysaccharides of Gram-Negative Bacteria. Methods Microbiol. 1985, 118, 157–207. [Google Scholar]
- Giampieri, F.; Forbes-Hernandez, T.Y.; Gasparrini, M.; Alvarez-Suarez, J.M.; Afrin, S.; Bompadre, S.; Quiles, J.L.; Mezzetti, B.; Battino, M. Strawberry as a health promoter: An evidence based review. Food Funct. 2015, 6, 1386–1398. [Google Scholar] [CrossRef] [PubMed]
- Malpass, G.E.; Arimilli, S.; Prasad, G.L.; Howlett, A.C. Complete artificial saliva alters expression of proinflammatory cytokines in human dermal fibroblasts. Toxicol. Sci. 2013, 134, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Awang, M.A.; Firdaus, M.A.; Busra, M.B.; Chowdhury, S.R.; Fadilah, N.R.; Wan Hamirul, W.K.; Reusmaazran, M.Y.; Aminuddin, M.Y.; Ruszymah, B.H. Cytotoxic evaluation of biomechanically improved crosslinked ovine collagen on human dermal fibroblasts. Biomed. Mater. Eng. 2014, 24, 1715–1724. [Google Scholar] [PubMed]
- Tardif, F.; Ross, G.; Rouabhia, M. Gingival and dermal fibroblasts produce interleukin-1 beta converting enzyme and interleukin-1 beta but not interleukin-18 even after stimulation with lipopolysaccharide. J. Cell. Physiol. 2004, 198, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Wheater, M.A.; Falvo, J.; Ruiz, F.; Byars, M. Chlorhexidine, ethanol, lipopolysaccharide and nicotine do not enhance the cytotoxicity of a calcium hydroxide pulp capping material. Int. Endod. J. 2012, 45, 989–995. [Google Scholar] [CrossRef] [PubMed]
- Tulipani, S.; Mezzetti, B.; Battino, M. Impact of strawberries on human health: Insight into marginally discussed bioactive compounds for the Mediterranean diet. Public Health Nutr. 2009, 12, 1656–1662. [Google Scholar] [CrossRef] [PubMed]
- Buendía, B.; Gil, M.I.; Tudela, J.A.; Gady, A.L.; Medina, J.J.; Soria, C.; López, J.M.; Tomás-Barberán, F.A. HPLC-MS analysis of proanthocyanidin oligomers and other phenolics in 15 strawberry cultivars. J. Agric. Food Chem. 2010, 58, 3916–3926. [Google Scholar] [CrossRef] [PubMed]
- Giampieri, F.; Alvarez-Suarez, J.M.; Tulipani, S.; Gonzàles-Paramàs, A.M.; Santos-Buelga, C.; Bompadre, S.; Quiles, J.L.; Mezzetti, B.; Battino, M. Photoprotective potential of strawberry (Fragaria × ananassa) extract against UV-A irradiation damage on human fibroblasts. J. Agric. Food Chem. 2012, 60, 2322–2327. [Google Scholar] [CrossRef] [PubMed]
- Giampieri, F.; Alvarez-Suarez, J.M.; Mazzoni, L.; Forbes-Hernandez, T.Y.; Gasparrini, M.; Gonzàlez-Paramàs, A.M.; Santos-Buelga, C.; Quiles, J.L.; Bompadre, S.; Mezzetti, B.; et al. An anthocyanin-rich strawberry extract protects against oxidative stress damage and improves mitochondrial functionality in human dermal fibroblasts exposed to an oxidizing agent. Food Funct. 2014, 5, 1939–1948. [Google Scholar] [CrossRef] [PubMed]
- Ariza, M.T.; Martínez-Ferri, E.; Domínguez, P.; Medina, J.J.; Miranda, L.; Soria, C. Effects of harvest time on functional compounds and fruit antioxidant capacity in ten strawberry cultivars. J. Berry Res. 2015, 5, 71–80. [Google Scholar] [CrossRef]
- Giampieri, F.; Alvarez-Suarez, J.M.; Mazzoni, L.; Forbes-Hernandez, T.Y.; Gasparrini, M.; Gonzàlez-Paramàs, A.M.; Santos-Buelga, C.; Quiles, J.L.; Bompadre, S.; Mezzetti, B.; et al. Polyphenol-rich strawberry extract protects human dermal fibroblasts against hydrogen peroxide oxidative damage and improves mitochondrial functionality. Molecules 2014, 19, 7798–7816. [Google Scholar] [CrossRef] [PubMed]
- Schieber, M.; Chandel, N.S. ROS function in redox signaling and oxidative stress. Curr. Biol. 2014, 24, R453–R462. [Google Scholar] [CrossRef] [PubMed]
- Park, G.J.; Kim, Y.S.; Kang, K.L.; Bae, S.J.; Baek, H.S.; Auh, Q.S.; Chun, Y.H.; Park, B.H.; Kim, E.C. Effects of sirtuin 1 activation on nicotine and lipopolysaccharide-induced cytotoxicity and inflammatory cytokine production in human gingival fibroblasts. J. Periodontal Res. 2013, 48, 483–492. [Google Scholar] [CrossRef] [PubMed]
- Mo, C.; Wang, L.; Zhang, J.; Numazawa, S.; Tang, H.; Tang, X.; Han, X.; Li, J.; Yang, M.; Wang, Z.; et al. The crosstalk between Nrf2 and AMPK signal pathways is important for the anti-inflammatory effect of berberine in LPS-stimulated macrophages and endotoxin-shocked mice. Antioxid. Redox Signal. 2014, 20, 574–588. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.Y.; Kim, H.J.; Han, J.S. Anti-inflammatory effects of calcium citrate in RAW 264.7 cells via suppression of NF-κB activation. Environ. Toxicol. Pharmacol. 2015, 39, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Zong, Y.; Sun, L.; Liu, B.; Deng, Y.S.; Zhan, D.; Chen, Y.L.; He, Y.; Liu, J.; Zhang, Z.J.; Sun, J.; et al. Resveratrol inhibits LPS-induced MAPKs activation via activation of the phosphatidylinositol 3-kinase pathway in murine RAW 264.7 macrophage cells. PLoS ONE 2012, 7, e44107. [Google Scholar] [CrossRef] [PubMed]
- Español, A.J.; Goren, N.; Ribeiro, M.L.; Sales, M.E. Nitric oxide synthase 1 and cyclooxygenase-2 enzymes are targets of muscarinic activation in normal and inflamed NIH3T3 cells. Inflamm. Res. 2010, 59, 227–238. [Google Scholar] [CrossRef] [PubMed]
- DeLeve, L.D.; Kaplowitz, N. Importance and regulation of hepatic glutathione. Semin. Liver Dis. 1990, 10, 251–266. [Google Scholar] [CrossRef] [PubMed]
- Ciolino, H.P.; Levine, R.L. Modification of proteins in endothelial cell death during oxidative stress. Free Radic. Biol. Med. 1997, 22, 1277–1282. [Google Scholar] [CrossRef]
- Babujanarthanam, R.; Kavitha, P.; Mahadeva Rao, U.S.; Pandian, M.R. Quercitrin a bioflavonoid improves the antioxidant status in streptozotocin: Induced diabetic rat tissues. Mol. Cell. Biochem. 2011, 358, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Häcker, H.; Redecke, V.; Blagoev, B.; Kratchmarova, I.; Hsu, L.C.; Wang, G.G.; Kamps, M.P.; Raz, E.; Wagner, H.; Häcker, G.; et al. Specificity in Toll-like receptor signalling through distinct effector functions of TRAF3 and TRAF6. Nature 2006, 439, 204–207. [Google Scholar] [CrossRef] [PubMed]
- Slinkard, K.; Singleton, V.L. Total Phenol analysis: Automation and comparison with manual methods. Am. J. Enol. Viticult. 1977, 28, 49–55. [Google Scholar]
- Dewanto, V.; Wu, X.; Adom, K.K.; Liu, R.H. Thermal processing enhances the nutritional values of tomatoes by increasing the total antioxidant activity. J. Agric. Food Chem. 2002, 50, 3010–3014. [Google Scholar] [CrossRef] [PubMed]
- Jastrebova, J.; Witthoft, C.; Grahn, A.; Svensson, U.; Jagerstad, M. HPLC determination of folates in raw and processed beetroots. Food Chem. 2003, 80, 579–588. [Google Scholar] [CrossRef]
- Patring, J.D.; Jastrebova, J.A.; Hjortmo, S.B.; Andlid, T.A.; Jägerstad, I.M. Development of a simplified method for the determination of folates in baker’s yeast by HPLC with ultraviolet and fluorescence detection. J. Agric. Food Chem. 2005, 53, 2406–2411. [Google Scholar] [CrossRef] [PubMed]
- Giusti, M.; Wrolstad, R.E. Characterization and Measurement of Anthocyanins by UV-Visible Spectroscopy. Curr. Protoc. Food Anal. Chem. 2001. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. Ferric reducing ability of plasma (FRAP) as a measure of antioxidant power: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Prymont-Przyminska, A.; Zwolinska, A.; Sarniak, A.; Wlodarczyk, A.; Krol, M.; Nowak, M.; de Graft-Johnson, J.; Padula, G.; Bialasiewicz, P.; Markowski, J.; et al. Consumption of strawberries on a daily basis increases the non-urate 2,2-diphenyl-1-picryl-hydrazyl (DPPH) radical scavenging activity of fasting plasma in healthy subjects. J. Clin. Biochem. Nutr. 2014, 55, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Gasparrini, M.; Forbes-Hernandez, T.Y.; Afrin, S.; Alvarez-Suarez, J.M.; Gonzàlez-Paramàs, A.M.; Santos-Buelga, C.; Bompadre, S.; Quiles, J.L.; Mezzetti, B.; Giampieri, F. A Pilot Study of the Photoprotective Effects of Strawberry-Based Cosmetic Formulations on Human Dermal Fibroblasts. Int. J. Mol. Sci. 2015, 16, 17870–17884. [Google Scholar] [CrossRef] [PubMed]
- Moongkarndi, P.; Kosem, N.; Kaslungka, S.; Luanratana, O.; Pongpan, N.; Neungton, N. Antiproliferation, antioxidation and induction of apoptosis by Garcinia mangostana (mangosteen) on SKBR3 human breast cancer cell line. J. Ethnopharmacol. 2004, 90, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Lyu, S.Y.; Park, W.B. Photoprotective Potential of Anthocyanins Isolated from Acanthopanax divaricatus Var. albeofructus Fruits against UV Irradiation in Human Dermal Fibroblast Cells. Biomol. Ther. 2012, 20, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Ghahary, A.; Shen, Y.J.; Scott, P.G.; Tredget, E.E. Human dermal fibroblasts produce nitric oxide and express both constitutive and inducible nitric oxide synthase isoforms. J. Investig. Dermatol. 1996, 106, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Doktorovová, S.; Santos, D.L.; Costa, I.; Andreani, T.; Souto, E.B.; Silva, A.M. Cationic solid lipid nanoparticles interfere with the activity of antioxidant enzymes in hepatocellular carcinoma cells. Int. J. Pharm. 2014, 471, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Levine, R.L.; Garland, D.; Oliver, C.N.; Amici, A.; Climent, I.; Lenz, A.G.; Ahn, B.W.; Shaltiel, S.; Stadtman, E.R. Determination of carbonyl content in oxidatively modified proteins. Methods Enzymol. 1990, 186, 464–478. [Google Scholar] [PubMed]
- Griffith, O.W. Determination of glutathione and glutathione disulfide using glutathione reductase and 2-vinylpyridine. Anal. Biochem. 1980, 106, 207–212. [Google Scholar] [CrossRef]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the strawberry compounds are not available from the authors.






| Parameter | Quantification |
|---|---|
| TPC (mg GAEq/g FW) | 2.52 ± 0.01 |
| vit C (mg vit C/g FW) | 0.58 ± 0.02 |
| TFC (mg CEq/g FW) | 0.66 ± 0.01 |
| ACYs (mg/100g FW) | |
| Cy-3-glucoside | 3.11 ± 0.03 |
| Pg 3-glucoside | 39.74 ± 0.13 |
| Pg 3-rutinoside | 3.87 ± 0.16 |
| Pg 3-malonylglucoside | 6.69 ± 0.04 |
| Pg 3-acetylglucoside | 0.39 ± 0.01 |
| Folate (µg folate/g FW) | |
| folinic acid calcium salt hydrate | 0.99 ± 0.09 |
| 5-methyltetrahydrofolic acid | 0.06 ± 0.01 |
| TAC (µmol Teq/g FW) | |
| FRAP | 22.85 ± 0.39 |
| TEAC | 22.64 ± 0.49 |
| DPPH | 7.71 ± 0.32 |
| Treatment | Protein Carbonyl Content (nmol/mg Prot) | GSH Level (nmol/mg Prot) | TBARS Level (nmol/100 mg Prot) |
|---|---|---|---|
| No treatment (ctrl) | 65.54 ± 1.63 c,d | 25.89 ± 1.29 c,d | 21.02 ± 2.12 b,c |
| Strawberry 50 µg/mL | 54.97 ± 4.92 d,e | 26.18 ± 1.31 c,d | 18.12 ± 1.39 c,d |
| Strawberry 100 µg/mL | 41.15 ± 8.89 e,f | 37.63 ± 1.88 b | 14.56 ± 1.54 d,e |
| Strawberry 1000 µg/mL | 31.55 ± 7.28 f | 42.84 ± 2.14 a | 11.23 ± 1.47 e |
| LPS | 94.78 ± 4.74 a | 9.72 ± 0.49 f | 33.25 ± 2.89 a |
| Strawberry 50 µg/mL + LPS | 93.28 ± 4.66 a | 14.86 ± 0.74 e | 31.66 ± 3.54 a |
| Strawberry 100 µg/mL + LPS | 74.56 ± 3.72 b,c | 22.42 ± 1.12 d | 23.85 ± 1.58 b |
| Strawberry 1000 µg/mL + LPS | 62.24 ± 3.11 c,d | 31.89 ± 1.59 b,c | 16.23 ± 1.77 d |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gasparrini, M.; Forbes-Hernandez, T.Y.; Giampieri, F.; Afrin, S.; Mezzetti, B.; Quiles, J.L.; Bompadre, S.; Battino, M. Protective Effect of Strawberry Extract against Inflammatory Stress Induced in Human Dermal Fibroblasts. Molecules 2017, 22, 164. https://0-doi-org.brum.beds.ac.uk/10.3390/molecules22010164
Gasparrini M, Forbes-Hernandez TY, Giampieri F, Afrin S, Mezzetti B, Quiles JL, Bompadre S, Battino M. Protective Effect of Strawberry Extract against Inflammatory Stress Induced in Human Dermal Fibroblasts. Molecules. 2017; 22(1):164. https://0-doi-org.brum.beds.ac.uk/10.3390/molecules22010164
Chicago/Turabian StyleGasparrini, Massimiliano, Tamara Y. Forbes-Hernandez, Francesca Giampieri, Sadia Afrin, Bruno Mezzetti, Josè L. Quiles, Stefano Bompadre, and Maurizio Battino. 2017. "Protective Effect of Strawberry Extract against Inflammatory Stress Induced in Human Dermal Fibroblasts" Molecules 22, no. 1: 164. https://0-doi-org.brum.beds.ac.uk/10.3390/molecules22010164