Bacterial Bio-Resources for Remediation of Hexachlorocyclohexane
Abstract
:1. Introduction
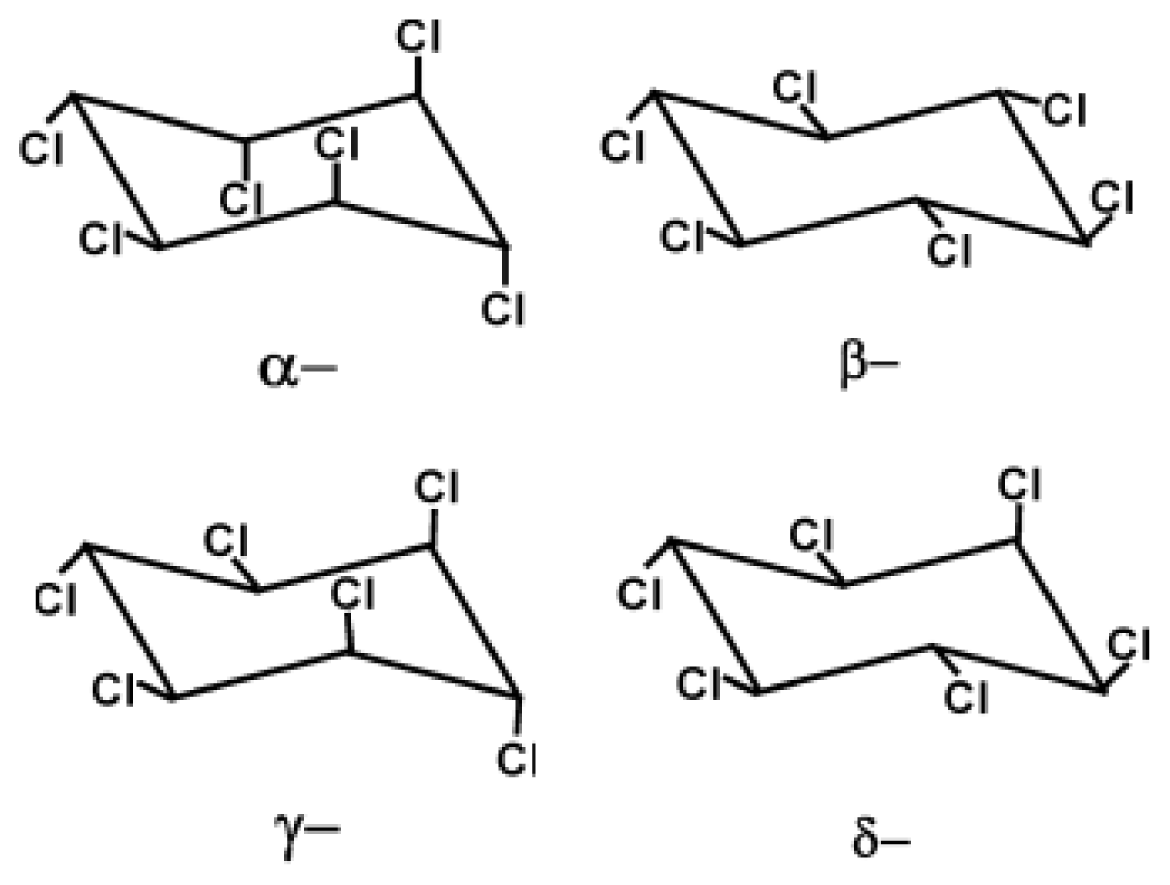
2. Physicochemical Properties of Hexachlorocyclohexane: Persistence and Deleterious Effects
3. Biodegradation Pathways of Hexachlorocyclohexane
3.1. Aerobic Hexachlorocyclohexane Degradation in Gram-Negative Microorganisms
3.2. Aerobic Hexachlorocyclohexane Degradation in Gram-Positive Microorganisms
3.2.1. Indirect Assay to Detect Hexachlorocyclohexane Degradation
4. γ-Hexachlorocyclohexane Removal by Actinobacteria
4.1. Culture Condition for γ-Hexachlorocyclohexane Removal in Culture Medium
4.2. Removal of γ-Hexachlorocyclohexane in Soil
5. Hexachlorocyclohexane Removal by Plants Interacting with Microorganisms
6. Conclusions
Acknowledgments
- Conflict of InterestThe authors declare no conflict of interest.
References
- Wania, F.; Mackay, D. Global fractionation and cold condensation of low volatility organochlorine compounds in polar regions. Ambio 1993, 22, 10–18. [Google Scholar]
- Diez, M.C. Biological aspects involved in the degradation of organic pollutants. J. Soil Sci. Plant Nut 2010, 10, 244–267. [Google Scholar]
- Ntow, W.J. Pesticide residues in Volta Lake, Ghana, Lakes Reservoirs. Res. Manage 2005, 10, 243–248. [Google Scholar]
- Xue, N.; Zhang, D.; Xu, X. Organochlorinated pesticide multiresidues in surface sediments from Beijing Guanting reservoir. Water Res 2006, 40, 183–194. [Google Scholar]
- Fuentes, M.S.; Benimeli, C.S.; Cuozzo, S.A.; Amoroso, M.J. Isolation of pesticide-degrading actinomycetes from a contaminated site: Bacterial growth, removal and dechlorination of organochlorine pesticides. Int. Biodeterior. Biodegrad 2010, 64, 434–441. [Google Scholar]
- Yang, R.; Ji, G.; Zhoe, Q.; Yaun, C.; Shi, J. Occurrence and distribution of organochlorine pesticides (HCH and DDT) in sediments collected from East China Sea. Environ. Int 2005, 31, 799–804. [Google Scholar]
- Fatoki, O.S.; Awofolu, R.O. Methods for selective determination of persistent organochlorine pesticide residues in water and sediments by capillary gas chromatography and electron-capture detection. J. Chromatogr 2003, A983, 225–236. [Google Scholar]
- Pazou, E.Y.; Boko, M.; van Gestel, C.A.; Ahissou, H.; Lalèyè, P.; Akpona, S.; van Hattum, B.; Swart, K.; van Straalen, N.I. Organochlorine and organophosphorous pesticide residues in the Quémé river catchment in the Republic of Bénin. Environ. Int 2006, 32, 616–623. [Google Scholar]
- Vijgen, J.; Abhilash, P.C.; Li, Y.F.; Lal, R.; Forter, M.; Torres, J.; Singh, N.; Yunus, M.; Tian, C.; Schäffer, A.; et al. HCH as new Stockholm Convention POPs, a global perspective on the management of lindane and its waste isomers. Env. Sci. Pollut. Res 2011, 18, 152–162. [Google Scholar]
- Barra, R.; Colombo, J.C.; Eguren, G.; Gamboa, N.; Jardim, W.F.; Mendoza, G. Persistent Organic Pollutants (POPs) in Eastern and Western South American Countries. Rev. Environ. Contam. Toxicol 2006, 185, 1–33. [Google Scholar]
- Manickam, N.; Mau, M.; Schlömann, M. Characterization of the novel HCH-degrading strain, Microbacterium sp. ITRC1. Appl. Microbiol. Biotechnol 2006, 69, 580–588. [Google Scholar]
- Willet, K.L.; Ulrich, E.M.; Hites, R.A. Differential toxicity and environmental fates of hexachlorocyclohexane isomers. Environ. Sci. Technol 1998, 32, 2197–2207. [Google Scholar]
- Lal, R.; Pandey, G.; Sharma, P.; Kumari, K.; Malhotra, S.; Pandey, R.; Raina, V.; Hans, P.E.; Kohler, C.H.; Jackson, C.; et al. Biochemistry of microbial degradation of hexachlorocyclohexane and prospects for bioremediation. Microbiol. Mol. Biol. Rev 2010, 74, 58–80. [Google Scholar]
- Li, J.; Zhu, T.; Wang, F.; Qiu, X.H.; Lin, W.L. Observation of organochlorine pesticides in the air of the Mt. Everest region. Ecotoxicol. Environ. Saf 2006, 63, 33–41. [Google Scholar]
- Popp, P.; Bruggemann, L.; Keil, P.; Thuss, U.; Weiss, H. Chloro-benzenes and hexachlorocyclohexanes (HCHs) in the atmosphere of Bitterfeld and Leipzig (In Germany). Chemosphere 2000, 41, 849–855. [Google Scholar]
- Bakore, N.; John, P.J.; Bhatnagar, P. Organochlorine pesticide residues in wheat and drinking water samples from Jaipur, Rajasthan, India. Environ. Monit. Assess 2004, 98, 381–389. [Google Scholar]
- Kumari, B.; Madan, V.K.; Kathpal, T.S. Status of insecticide contamination of soil and water in Haryana, India. Environ. Monit. Assess 2007, 136, 239–244. [Google Scholar]
- Concha-Graña, E.; Turnes-Carou, M.I.; Muniategui-Lorenzo, S.; López-Mahia, P.; Prada-Rodriguez, D.; Fernández-Fernández, E. Evaluation of HCH isomers and metabolites in soils, leachates, river water and sediments of a highly contaminated area. Chemosphere 2006, 64, 588–595. [Google Scholar]
- Gong, Z.M.; Xu, F.L.; Dawson, R.; Cao, J.; Liu, W.X.; Li, B.G.; Shen, W.R.; Zhang, W.J.; Qin, B.; Sun, R.; et al. Residues of hexachlorocyclohexane isomers and their distribution characteristics in soils in the Tianjin area, China. Arch. Environ. Contam. Toxicol 2004, 46, 432–437. [Google Scholar]
- Raina, V.; Suar, M.; Singh, A.; Prakash, O.; Dadhwal, M.; Gupta, S.K.; Dogra, C.; Lawlor, K.; Lal, S.; van der Meer, J.R.; et al. Enhanced biodegradation of hexachlorocyclohexane (HCH) in contaminated soils via inoculation with Sphingobium indicum B90A. Biodegradation 2008, 19, 27–40. [Google Scholar]
- Bajpai, A.; Shukla, P.; Dixit, B.S.; Banerji, R. Concentrations of organochlorine insecticides in edible oils from different regions of India. Chemosphere 2007, 67, 1403–1407. [Google Scholar]
- Blasco, C.; Lino, C.M.; Pico, Y.; Pena, A.; Font, G.; Silveira, M.I. Determination of organochlorine pesticide residues in honey from the central zone of Portugal and the Valencian community of Spain. J. Chromatogr 2004, A1049, 155–160. [Google Scholar]
- Waliszewski, S.M.; Villalobos-Pietrini, R.; Gomez-Arroyo, S.; Infanzon, R.M. Persistent organochlorine pesticides in Mexican butter. Food Addit. Contam 2003, 20, 361–367. [Google Scholar]
- Sanghi, R.; Pillai, M.K.; Jayalekshmi, T.R.; Nair, A. Organochlorine and organophosphorus pesticide residues in breast milk from Bhopal, Madhya Pradesh, India. Hum. Exp. Toxicol 2003, 22, 73–76. [Google Scholar]
- Zhao, G.; Xu, Y.; Li, W.; Han, G.; Ling, B. PCBs and OCPs in human milk and selected foods from Luqiao and Pingqiao in Zhejiang, China. Sci. Total Environ 2007, 378, 281–292. [Google Scholar]
- Bhatnagar, V.K.; Kashyap, R.; Zaidi, S.S.; Kulkarni, P.K.; Saiyed, H.N. Levels of DDT, HCH, and HCB residues in human blood in Ahmedabad, India. Bull. Environ. Contam. Toxicol 2004, 72, 261–265. [Google Scholar]
- Kutz, F.W.; Wood, P.H.; Bottimore, D.P. Organochlorine pesticides and polychlorinated biphenyls in human adipose tissue. Rev. Environ. Contam. Toxicol 1991, 120, 1–82. [Google Scholar]
- Walker, K.; Vallero, D.A.; Lewis, R.G. Factors influencing the distribution of lindane and other hexachlorocyclohexanes in the environment. Environ. Sci. Technol 1999, 33, 4373–4378. [Google Scholar]
- McGuinness, M.; Dowling, D. Plant-associated bacterial degradation of toxic organic compounds in soil. Int. J. Environ. Res. Public Health 2009, 6, 2226–2247. [Google Scholar]
- De Lorenzo, V. Systems biology approaches to bioremediation. Curr. Opin. Biotechnol 2008, 19, 579–589. [Google Scholar]
- De Schrijver, A.; de Mot, R. Degradation of pesticides by actinomycetes. Crit. Rev. Microbiol 1999, 25, 85–119. [Google Scholar]
- Siripattanakul, S.; Wirojanagud, W.; McEvoy, J.; Limpiyakorn, T.; Khan, E. Atrazine degradation by stable mixed cultures enriched from agricultural soil and their characterization. J. Appl. Microbiol 2009, 106, 986–999. [Google Scholar]
- Yang, C.; Li, Y.; Zhang, K.; Wang, X.; Ma, C.; Tang, H.; Xu, P. Atrazine degradation by a simple consortium of Klebsiella sp. A1 and Comamonas sp. A2 in nitrogen enriched medium. Biodegradation 2010, 21, 97–105. [Google Scholar]
- Saez, J.M.; Benimeli, C.S.; Amoroso, M.J. Lindane removal by pure and mixed cultures of immobilized actinobacteria. Chemosphere 2012, 89, 982–987. [Google Scholar]
- Böltner, D.; Moreno-Morillas, S.; Ramos, J.L. 16S rDNA phylogeny and distribution of lin genes in novel hexachlorocyclohexane-degrading Sphingomonas strains. Environ. Microbiol 2005, 7, 1329–1338. [Google Scholar]
- Mohn, W.W.; Mertens, B.; Neufeld, J.D.; Verstraete, W.; de Lorenzo, V. Distribution and phylogeny of hexachlorocyclohexane degrading bacteria in soils from Spain. Environ. Microbiol 2006, 8, 60–68. [Google Scholar]
- Phillips, T.M.; Seech, A.G.; Lee, H.; Trevors, J.T. Biodegradation of hexachlorocyclohexane (HCH) by microorganisms. Biodegradation 2005, 16, 363–392. [Google Scholar]
- Kidd, P.; Prieto-Fernández, A.; Monterroso, C.; Acea, M.J. Rhizosphere microbial community and hexachlorocyclohexane degradative potential in contrasting plant species. Plant Soil 2008, 302, 233–247. [Google Scholar]
- Polti, M.A.; Amoroso, M.J.; Abate, C.M. Chromium (VI) resistance and removal by actinomycete strains isolated from sediments. Chemosphere 2007, 67, 660–667. [Google Scholar]
- Siñeriz, M.L.; Kothe, E.; Abate, C.M. Cadmium biosorption by Streptomyces sp. F4 isolated from former uranium mine. J. Basic Microbiol 2009, 49, 55–62. [Google Scholar]
- Benimeli, C.S.; Amoroso, M.J.; Chaile, A.; Castro, G. Isolation of four aquatic streptomycetes strains capable of growth on organochlorine pesticides. Bioresour. Technol 2003, 89, 133–138. [Google Scholar]
- Benimeli, C.S.; Castro, G.R.; Chaile, A.P.; Amoroso, M.J. Lindane removal induction by Streptomyces sp. M7. J. Basic Microbiol 2006, 46, 348–357. [Google Scholar]
- Benimeli, C.S.; Castro, G.; Chaile, A.; Amoroso, M.J. Lindane uptake and degradation by aquatic Streptomyces sp. strain M7. Int. Biodeterior. Biodegrad 2007, 59, 148–155. [Google Scholar]
- Benimeli, C.S.; González, A.J.; Chaile, A.P.; Amoroso, M.J. Temperature and pH effect on lindane removal by Streptomyces sp. M7 in soil extract. J. Basic Microbiol 2007, 47, 468–473. [Google Scholar]
- Benimeli, C.S.; Fuentes, M.S.; Abate, C.M.; Amoroso, M.J. Bioremediation of lindane contaminated soil by Streptomyces sp. M7 and its effects on Zea mays growth. Int. Biodeterior. Biodegrad 2008, 61, 233–239. [Google Scholar]
- Cuozzo, S.A.; Rollán, G.C.; Abate, C.M.; Amoroso, M.J. Specific dechlorinase activity in lindane degradation by Streptomyces sp. M7. World J. Microbiol. Biotechnol 2009, 25, 1539–1546. [Google Scholar]
- Cuozzo, S.A.; Fuentes, M.S.; Bourgignon, N.; Benimeli, C.S.; Amoroso, M.J. Chlordane biodegradation under aerobic conditions by indigenous Streptomyces strains. Int. Biodeterior. Biodegrad 2012, 66, 19–24. [Google Scholar]
- Fuentes, M.S.; Sáez, J.M.; Benimeli, C.S.; Amoroso, M.J. Lindane biodegradation by defined consortiaof indigenous Streptomyces strains. Water Air Soil Pollut 2011, 222, 217–231. [Google Scholar]
- Bending, G.; Friloux, D.M.; Walker, A. Degradation of contrasting pesticides by White rot fungi and its relationship with ligninolytic potential. FEMS Microbiol. Lett 2002, 212, 59–63. [Google Scholar]
- Jauregui, J.; Valderrama, B.; Albores, A.; Vazquez-Duhalt, R. Microsomal transformation of organophosphorous pesticides by White rot fungi. Biodegradation 2003, 14, 397–406. [Google Scholar]
- Quintero, J.C.; Lú-Chau, T.A.; Moreira, M.T.; Feijoo, G.; Lema, J.M. Bioremediation of HCH present in soil by the white-rot fungus Bjerkandera adusta in a slurry bacth bioreactor. Int. Biodeter. Biodegr 2007, 60, 319–326. [Google Scholar]
- Rubilar, O.; Feijoo, G.; Diez, M.C.; Lu-Chau, T.A.; Moreira, M.T.; Lema, J.M. Biodegradation of pentachlorophenol in soil slurry cultures by Bjerkandera adusta and Anthracophyllum discolor. Indust. Engin. Chem. Res 2007, 46, 744–751. [Google Scholar]
- Van Aken, B.; Correa, P.; Schnoor, J. Phytoremediation of polychlorinated biphenyls: New trends and promises. Environ. Sci. Technol 2010, 44, 2767–2776. [Google Scholar]
- Alvarez, A.; Yañez, M.L.; Benimeli, C.S.; Amoroso, M.J. Maize plants (Zea mays) root exudates enhance lindane removal by native Streptomyces strains. Int. Biodeterior. Biodegrad 2012, 66, 14–18. [Google Scholar]
- Becerra-Castro, C.; Prieto-Fernández, A.; Kidd, P.S.; Weyens, N.; Rodríguez-Garrido, B.; Touceda-González, M.; Acea, M.J.; Vangronsveld, J. Improving performance of Cytisus striatus on substrates contaminated with hexachlorocyclohexane (HCH) isomers using bacterial inoculants: Developing a phytoremediation strategy. Plant Soil 2012. [Google Scholar] [CrossRef]
- Andreu, V.; Picó, Y. Determination of pesticides and their degradation products in soil: Critical review and comparison of methods. Trend. Anal. Chem 2004, 23, 772–789. [Google Scholar]
- Li, S.; Wania, F. Compilation, evaluation, and selection of physical-chemical property data for organochlorine pesticides. J. Chem. Eng. Data 2005, 50, 742–768. [Google Scholar]
- Muir, D.C.; Howard, P.H. Are there other persistent organic pollutants? A challenge for environmental chemists. Environ. Sci. Technol 2006, 40, 7157–7166. [Google Scholar]
- Bidleman, T.F. Atmospheric processes. Environ. Sci. Technol 1988, 22, 361–367. [Google Scholar]
- Brubaker, W.W., Jr; Hites, R.A. Polychlorinated dibenzo-p-dioxins and dibenzofurans: Gas-phase hydroxyl radical reactions and related atmospheric removal. Environ. Sci. Technol. 1997, 31, 1805–1810. [Google Scholar]
- Office of Environmental Health Hazard Assessment California Environmental Protection Agency, Public Health Goal for Methoxychlor in Drinking Water; Office of Environmental Health Hazard Assessment California Environmental Protection Agency: Sacramento, CA, USA, 1999.
- Yule, W.; Chiba, M.; Morley, H. Fate of insecticide residues. Decomposition of lindane in soil. J. Agric. Food Chem 1967, 15, 1000–1004. [Google Scholar]
- Tu, C.M. Utilization and degradation of lindane by soil microorganisms. Arch. Microbiol 1976, 108, 259–263. [Google Scholar]
- Ma, A.Z.; Wu, J.; Zhang, G.S.; Wang, T.; Li, S.P. Isolation and characterization of a HCH degradation Sphingomanas sp. stain BHC-A. Acta Microbiol. Sin 2005, 45, 728–732. [Google Scholar]
- Nalin, R.; Simonet, P.; Vogel, T.M.; Normand, P. Rhodanobacter lindaniclasticus gen. nov., sp. nov., a lindane-degrading bacterium. Int. J. Syst. Bacteriol 1999, 49, 19–23. [Google Scholar]
- Thomas, J.C.; Berger, F.; Jacquier, M.; Bernillon, D.; Baud-Grasset, F.; Truffaut, N.; Normand, P.; Vogel, T.M.; Simonet, P. Isolation and characterization of a novel gamma-hexachlorocyclohexane-degrading bacterium. J. Bacteriol 1996, 178, 6049–6055. [Google Scholar]
- Cérémonie, H.; Boubakri, H.; Mavingui, P.; Simonet, P.; Vogel, T.M. Plasmid-encoded γ-hexachlorocyclohexane degradation genes and insertion sequences in Sphingobium francense (ex-Sphingomonas paucimobilis Sp+). FEMS Microbiol. Lett 2006, 257, 243–252. [Google Scholar]
- Gupta, A.; Kaushik, C.P.; Kaushik, A. Degradation of hexachlorocyclohexane isomers by two strains of Alcaligenes faecalis isolated from a contaminated site. Bull. Environ. Contam. Toxicol 2001, 66, 794–800. [Google Scholar]
- Datta, J.; Maiti, A.K.; Modak, D.P.; Chakrabartty, P.K.; Bhattacharyya, P.; Ray, P.K. Metabolism of &-hexachlorocyclohexane by Arthrobacter citreus strain BI-100: Identification of metabolites. J. Gen. App. Microbiol 2000, 46, 59–67. [Google Scholar]
- Lodha, B.; Bhat, P.; Kumar, M.S.; Vaidya, A.N.; Mudliar, S.; Killedar, D.J.; Chakrabarti, T. Bioisomerization kinetics of γ-HCH and biokinetics of Pseudomonas aeruginosa degrading technical HCH. Biochem. Eng. J 2007, 35, 12–19. [Google Scholar]
- Kumar, M.; Chaudhary, P.; Dwivedi, M.; Kumar, R.; Paul, D.; Jain, R.K.; Garg, S.K.; Kumar, A. Enhanced biodegradation of β- and δ-hexachlorocyclohexane in the presence of α- and γ-isomers in contaminated soils. Environ. Sci. Technol 2005, 39, 4005–4011. [Google Scholar]
- Nawab, A.; Aleem, A.; Malik, A. Determination of organochlorine pesticides in agricultural soil with special reference to γ-HCH degradation by Pseudomonas strains. Bioresour. Technol 2003, 88, 41–46. [Google Scholar]
- Dadhwal, M.; Jit, S.; Kumari, H.; Lal, R. Sphingobium chinhatense sp. nov., a hexachlorocyclohexane (HCH)-degrading bacterium isolated from an HCH dumpsite. Int. J. Syst. Evol. Microbiol 2009, 59, 3140–3144. [Google Scholar]
- Sahu, S.K.; Patnaik, K.K.; Sharmila, M.; Sethunathan, N. Degradation of alpha-, beta-, and gamma-hexachlorocyclohexane by a soil bacterium under aerobic conditions. Appl. Environ. Microbiol 1990, 56, 3620–3622. [Google Scholar]
- Bala, K.; Sharma, P.; Lal, R. Sphingobium quisquiliarum sp. nov., a hexachlorocyclohexane (HCH)-degrading bacterium isolated from an HCH-contaminated soil. Int. J. Syst. Evol. Microbiol 2010, 60, 429–433. [Google Scholar]
- Singh, A.; Lal, R. Sphingobium ummariense sp. nov., a hexachlorocyclohexane (HCH)-degrading bacterium, isolated from HCH-contaminated soil. Int. J. Syst. Evol. Microbiol 2009, 59, 162–166. [Google Scholar]
- Dadhwal, M.; Singh, A.; Prakash, O.; Gupta, S.K.; Kumari, K.; Sharma, P.; Jit, S.; Verma, M.; Holliger, C.; Lal, R. Proposal of biostimulation for hexachlorocyclohexane (HCH)-decontamination and characterization of culturable bacterial community from high-dose point HCH-contaminated soils. J. Appl. Microbiol 2009, 106, 381–392. [Google Scholar]
- Manickam, N.; Misra, R.; Mayilraj, S. A novel pathway for the biodegradation of γ-hexachlorocyclohexane by a Xanthomonas sp. strain ICH12. J. Appl. Microbiol 2007, 102, 1468–1478. [Google Scholar]
- Senoo, K.; Wada, H. Isolation and identification of an aerobic γ-HCH-decomposing bacterium from soil. Soil Sci. Plant Nutr 1989, 35, 79–87. [Google Scholar]
- Ito, M.; Prokop, Z.; Klvaňa, M.; Otsubo, Y.; Tsuda, M.; Damborský, J.; Nagata, Y. Degradation of β-hexachlorocyclohexane by haloalkane dehalogenase LinB from γ-hexachlorocyclohexane-utilizing bacterium Sphingobium sp. MI1205. Arch. Microbiol 2007, 188, 313–325. [Google Scholar]
- Yamamoto, S.; Otsuka, S.; Murakami, Y.; Nishiyama, M.; Senoo, K. Genetic diversity of gamma-hexachlorocyclohexane-degrading sphingomonads isolated from a single experimental field. Lett. Appl. Microbiol 2009, 49, 472–477. [Google Scholar]
- Francis, A.J.; Spanggord, R.J.; Ouchi, G.I. Degradation of lindane by Escherichia coli. Appl. Microbiol 1975, 29, 567–568. [Google Scholar]
- Benezet, H.J.; Matsumura, F. Isomerization of γ-BHC to α-BHC in the Environment. Nature 1973, 243, 480–481. [Google Scholar]
- Lal, R.; Dogra, C.; Malhotra, S.; Sharma, P.; Pal, R. Diversity, distribution and divergence of lin genes in hexachlorocyclohexane-degrading sphingomonads. Trends Biotechnol 2006, 24, 121–130. [Google Scholar]
- Lal, R.; Dadhwal, M.; Kumari, K.; Sharma, P.; Singh, A.; Kumari, H.; Jit, S.; Gupta, S.K.; Nigam, A.; Lal, D.; et al. Pseudomonas sp. to Sphingobium indicum: A journey of microbial degradation and bioremediation of hexachlorocyclohexane. Indian J. Microbiol 2008, 48, 3–18. [Google Scholar]
- Nagata, Y.; Endo, R.; Ito, M.; Ohtsubo, Y.; Tsuda, M. Aerobic degradation of lindane (gamma-hexachlorocyclohexane) in bacteria and its biochemical and molecular basis. Appl. Microbiol. Biotechnol 2007, 76, 741–752. [Google Scholar]
- Nagata, Y.; Miyauchi, K.; Takagi, M. Complete analysis of genes and enzymes for γ-hexachlorocyclohexane degradation in Sphingomonas paucimobilis UT26. J. Ind. Microbiol. Biotechnol 1999, 23, 380–390. [Google Scholar]
- Wu, J.; Hong, Q.; Han, P.; He, J.; Li, S. A gene linB2 responsible for the conversion of HCH and 2,3,4,5,6-pentachlorocyclohexanol in Sphingomonas sp. BHC-A. Appl. Microbiol. Biotechnol 2007, 73, 1097–1105. [Google Scholar]
- Imai, R.; Nagata, Y.; Fukuda, M.; Takagi, M.; Yano, K. Molecular cloning of a Pseudomonas paucimobilis gene encoding a 17-kilodalton polypeptide that eliminates HCl molecules from γ-hexachlorocyclohexane. J. Bacteriol 1991, 173, 6811–6819. [Google Scholar]
- Nagata, Y.; Nariya, T.; Ohtomo, R.; Fukuda, M.; Yano, K.; Takagi, M. Cloning and sequencing of a dehalogenase gene encoding an enzyme with hydrolase activity involved in the degradation of hexachlorocyclohexane in Pseudomonas paucimobilis. J. Bacteriol 1993, 175, 6403–6410. [Google Scholar]
- Nagata, Y.; Ohtomo, R.; Miyauchi, K.; Fukuda, M.; Yano, K.; Takagi, M. Cloning and sequencing of a 2,5-dichloro-2,5-cyclohexadiene-1,4-diol dehydrogenase gene involved in the degradation of hexachlorocyclohexane in Pseudomonas paucimobilis. J. Bacteriol 1994, 176, 3117–3125. [Google Scholar]
- Miyauchi, K.; Suh, S.K.; Nagata, Y.; Takagi, M. Cloning and sequencing of a 2,5-dichlorohydroquinone reductive dehalogenase gene whose product is involved in degradation of hexachlorocyclohexane by Sphingomonas paucimobilis. J. Bacteriol 1998, 180, 1354–1359. [Google Scholar]
- Endo, R.; Kamakura, M.; Miyauchi, K.; Fukuda, M.; Ohtsubo, Y.; Tsuda, M.; Nagata, Y. Identification and characterization of genes involved in the downstream degradation pathway of γ-hexachlorocyclohexane in Sphingomonas paucimobilis UT26. J. Bacteriol 2005, 187, 847–853. [Google Scholar]
- Miyauchi, K.; Lee, H.S.; Fukuda, M.; Takagi, M.; Nagata, Y. Cloning and characterization of linR, involved in regulation of the downstream pathway for γ-hexachlorocyclohexane degradation in Sphingomonas paucimobilis UT26. Appl. Environ. Microbiol 2002, 68, 1803–1807. [Google Scholar]
- Larkin, M.J.; Kulakov, L.A.; Allen, C.C. Biodegradation and Rhodococcus—Masters of catabolic versatility. Curr. Opin. Biotechnol. 2005, 16, 282–290. [Google Scholar]
- Phillips, T.M.; Seech, A.G.; Lee, H.; Trevors, J.T. Colorimetric assay for lindane dechlorination by bacteria. J. Microbiol. Methods 2001, 47, 181–188. [Google Scholar]
- Andrade, J.M.; Arraiano, C.M. PNPase is a key player in the regulation of small RNAs that control the expression of outer membrane proteins. RNA 2008, 14, 543–551. [Google Scholar]
- Normand, P.; Lapierre, P.; Tisa, L.S.; Gogarten, J.P.; Alloisio, N.; Bagnarol, E.; Bassi, C.A.; Berry, A.M.; Bickhart, D.M.; Choisne, N.; et al. Genome characteristics of facultatively symbiotic Frankia sp. strains reflect host range and host plant biogeography. Genome Res 2007, 17, 7–15. [Google Scholar]
- Benimeli, C.S. Biodegradation of organochlorine pesticides by aquatic actinomycetes. Ph.D. Thesis, Universidad Nacional de Tucumán, Tucumán, Argentina, 2004. [Google Scholar]
- Isaza, J.P.; Duque, C.; Gomez, V.; Robledo, J.; Barrera, L.F.; Alzate, J.F. Whole genome shotgun sequencing of one Colombian clinical isolate of Mycobacterium tuberculosis reveals DosR regulon gene deletions. FEMS Microbiol. Lett 2012, 330, 113–120. [Google Scholar]
- Manickam, N.; Reddy, M.K.; Saini, H.S.; Shanker, R. Isolation of hexachlorocyclohexane-degrading Sphingomonas sp. by dehalogenase assay and characterization of genes involved in γ-HCH degradation. J. Appl. Microbiol 2008, 104, 952–960. [Google Scholar]
- Camacho-Pérez, B.; Ríos-Leal, E.; Esparza-García, F.; Barrera-Cortés, J.; Fava, F.; Poggi-Varaldo, H.M. Bioremediation of an agricultural soil polluted with lindane in triphasic, sequential methanogenic-sulfate reducing slurry bioreactors. J. Biotechnol 2010, 150, 561–562. [Google Scholar]
- Camacho-Pérez, B.; Ríos-Leal, E.; Rinderknecht-Seijas, N.; Poggi-Varaldo, H.M. Enzymes involved in the biodegradation of hexachlorocyclohexane: A mini review. J. Environ. Manag 2012, 95, S306–S318. [Google Scholar]
- Kothe, E.; Dimkpa, C.; Haferburg, G.; Schmidt, A.; Schmidt, A.; Schütze, E. Streptomycete Heavy Metal Resistance: Extracellular and Intracellular Mechanisms. In Soil Heavy Metals; Sherameti, I., Varma, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 225–235. [Google Scholar]
- Speedie, M.K.; Pogell, B.M.; MacDonald, M.J.; Kline, R.; Huang, Y.I. Potential usefulness of Streptomyces for the detoxification of recalcitrant organochlorines and other pollutants. Actinomycetes 1987, 20, 315–335. [Google Scholar]
- Scherr, N.; Nguyen, L. Mycobacterium versus Streptomyces—We are different, we are the same. Curr. Opin. Microbiol 2009, 12, 699–707. [Google Scholar]
- Shelton, D.R.; Khader, S.; Karns, J.S.; Pogell, B.M. Metabolism of twelve herbicides by Streptomyces. Biodegradation 1996, 7, 129–136. [Google Scholar]
- Arisoy, M.; Kolankaya, N. Biodegradation of lindane by Pleurotus sajor-caju and toxic effects of lindane and its metabolites on mice. Bull. Environ. Contam. Toxicol 1997, 59, 352–359. [Google Scholar]
- Bachmann, A.; Wijnen, P.; DeBruin, W.; Huntjens, J.L.M.; Roelofsen, W.; Zehnder, A.J.B. Biodegradation of alpha- and beta-hexachlorocyclohexane in a soil slurry and under different redox conditions. Appl. Environ. Microbiol 1988, 54, 143–149. [Google Scholar]
- Manonmani, H.K.; Chandrashekariah, D.H.; Sreedhar Reddy, N.; Elecy, C.D.; Kunhi, A.A.M. Isolation and acclimation of a microbial consortium for improved aerobic degradation of α-hexachlorocyclohexane. J. Agric. Food Chem 2000, 48, 4341–4351. [Google Scholar]
- Siddique, T.; Okeke, B.C.; Arshad, M.; Frankerberger, W.T., Jr. Temperature and pH effects on biodegradation of hexachlorocyclohexane isomers in water and soil slurry. J. Agric. Food Chem. 2002, 50, 5070–5076. [Google Scholar]
- Zheng, G.; Selvam, A.; Wong, J.W.C. Rapid degradation of lindane (γ-hexachlorocyclohexane) at low temperature by Sphingobium strains. Int. Biodeterior. Biodegrad 2011, 65, 612–618. [Google Scholar]
- Antizar-Ladislao, B. Bioremediation—Bacterial Alchemists. Elements 2010, 6, 389–394. [Google Scholar]
- Rodriguez, R.A.; Toranzos, G.A. Stability of bacterial populations in tropical soil upon exposure to Lindane. Int. Microbiol 2003, 6, 253–258. [Google Scholar]
- Gerhardt, K.E.; Huang, X.D.; Glick, B.R.; Greenberg, B.M. Phytoremediation and rhizoremediation of organic soil contaminants: Potential and challenges. Plant Sci 2009, 176, 20–30. [Google Scholar]
- Kuiper, I.; Lagendijk, E.L.; Bloemberg, G.V.; Lugtenberg, B.J. Rhizoremediation: A beneficial plant-microbe interaction. Mol. Plant Microbe Interact 2004, 17, 6–15. [Google Scholar]
- Gao, Y.; Ren, L.; Ling, W.; Gong, S.; Sun, B.; Zhang, Y. Desorption of phenanthrene and pyrene in soils by root exudates. Bioresour. Technol 2010, 101, 1159–1165. [Google Scholar]
- Barriada-Pereira, M.; Gonzalez-Castro, M.J.; Muniategui-Lorenzo, S.; López-Mahía, P.; Prada-Rodríguez, D.; Fernández-Fernández, E. Organochlorine pesticides accumulation and degradation products in vegetation samples of a contaminated area in Galicia (NW Spain). Chemosphere 2005, 58, 1571–1578. [Google Scholar]
- Magee, K.; Michael, A.; Ulla, H.; Dutta, S.K. Dechlorination of PCB in the presence of plant nitrate reductase. Environ. Toxicol. Pharm 2008, 25, 144–147. [Google Scholar]
- Huang, X.D.; El-Alawi, Y.; Penrose, D.M.; Glick, B.R.; Greenberg, B.M. Responses of three grass species to creosote during phytoremediation. Environ. Pollut 2004, 130, 453–463. [Google Scholar]
- Weyens, N.; Truyens, S.; Dupae, J.; Newman, L.; Taghavi, S.; van der Lelie, D.; Carleer, R.; Vangronsveld, J. Potential of the TCE-degrading endophyte Pseudomonas putida W619-TCE to improve plant growth and reduce TCE phytotoxicity and evapotranspiration in poplar cuttings. Environ. Pollut 2010, 158, 2915–2919. [Google Scholar]
- Lin, Q.; Shen, K.L.; Zhao, H.M.; Li, W.H. Growth response of Zea mays L. in pyrene-copper co-contaminated soil and the fate of pollutants. J. Hazard. Mater 2008, 150, 515–521. [Google Scholar]
- Pereira, C.R.; Camps-Arbestain, M.; Garrido, B.R.; Macías, F.; Monterroso, C. Behaviour of α-, β-, γ-, and δ-hexachlorocyclohexane in the soil—Plant system of a contaminated site. Environ. Pollut 2006, 144, 210–217. [Google Scholar]
- Germaine, K.J.; Keogh, E.; Ryan, D.; Dowling, D.N. Bacterial endophyte-mediated naphthalene phytoprotection and phytoremediation. FEMS Microbiol. Lett 2009, 296, 226–234. [Google Scholar]
- Rentz, J.; Alvarez, P.; Schnoor, J. Repression of Pseudomonas putida phenanthrene-degrading activity by plant root extracts and exudates. Environ. Microbiol 2004, 6, 574–583. [Google Scholar]
- Louvel, B.; Cébron, A.; Leyval, C. Root exudates affect phenanthrene biodegradation, bacterial community and functional gene expression in sand microcosms. Int. Biodeter. Biodegr 2011, 65, 947–953. [Google Scholar]
- Sáinz, M.J.; González-Penalta, B.; Vilariño, A. Effects of hexachlorocyclohexane on rhizosphere fungal propopagules and root colonization by arbuscular mycorrhizal fungi in Plantago lanceolata. Eur. J. Soil Sci 2006, 57, 83–90. [Google Scholar]
| Actinobacteria strain | Isolation source | GenBank access number | Action on γ-HCH |
|---|---|---|---|
| Micromonospora sp. A10 | a OPs contaminated soil | GQ867054 [5] | c Grow, remove and degrade [5,48] |
| Streptomyces sp. A1 | OPs contaminated soil | GU085102 [5] | c Grow, remove and degrade [5,48] |
| Streptomyces sp. A2 | OPs contaminated soil | GU085103 [5] | c Grow, remove and degrade [5,48] |
| Streptomyces sp. A3 | OPs contaminated soil | GU085104 [5] | c Grow, remove and degrade [5,48] |
| Streptomyces sp. A5 | OPs contaminated soil | GQ867050 [5] | c Grow, remove and degrade [5,48] |
| Streptomyces sp. A6 | OPs contaminated soil | GQ867051 [5] | c Grow, remove and degrade [5,48] |
| Streptomyces sp. A7 | OPs contaminated soil | GQ867052 [5] | c Grow, remove and degrade [5,48] |
| Streptomyces sp. A8 | OPs contaminated soil | GQ867053 [5] | c Grow, remove and degrade [5,48] |
| Streptomyces sp. A11 | OPs contaminated soil | GQ867055 [5] | c Grow, remove and degrade [5,48] |
| Streptomyces sp. A12 | OPs contaminated soil | GQ867056 [5] | c Grow, remove and degrade [5,48] |
| Streptomyces sp. A13 | OPs contaminated soil | GQ867057 [5] | Grow and remove [5] |
| Streptomyces sp. A14 | OPs contaminated soil | GU085105 [5] | c Grow, remove and degrade [5,48] |
| Streptomyces sp. C39 | Non-contaminated water | AY741282 [39] | Grow and remove [5] |
| Streptomyces sp. MC1 | Sugar cane | AY741287 [39] | Grow (Unpublished data) |
| Streptomyces sp. M7 | b Co-contaminated wastewater sediment | AY459531 [43] | d Grow, remove and degrade [5,41–46,48,99] |
| Streptomyces sp. M15 | Co-contaminated wastewater sediment | GQ867058 [5] | Grow and remove [5,41] |
| Streptomyces sp. M30 | Co-contaminated wastewater sediment | GU085106 [5] | Grow and remove [5] |
| Streptomyces sp. M50 | Co-contaminated wastewater sediment | GQ867059 [5] | c Grow, remove and degrade [5] |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Alvarez, A.; Benimeli, C.S.; Saez, J.M.; Fuentes, M.S.; Cuozzo, S.A.; Polti, M.A.; Amoroso, M.J. Bacterial Bio-Resources for Remediation of Hexachlorocyclohexane. Int. J. Mol. Sci. 2012, 13, 15086-15106. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms131115086
Alvarez A, Benimeli CS, Saez JM, Fuentes MS, Cuozzo SA, Polti MA, Amoroso MJ. Bacterial Bio-Resources for Remediation of Hexachlorocyclohexane. International Journal of Molecular Sciences. 2012; 13(11):15086-15106. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms131115086
Chicago/Turabian StyleAlvarez, Analía, Claudia S. Benimeli, Juliana M. Saez, María S. Fuentes, Sergio A. Cuozzo, Marta A. Polti, and María J. Amoroso. 2012. "Bacterial Bio-Resources for Remediation of Hexachlorocyclohexane" International Journal of Molecular Sciences 13, no. 11: 15086-15106. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms131115086




