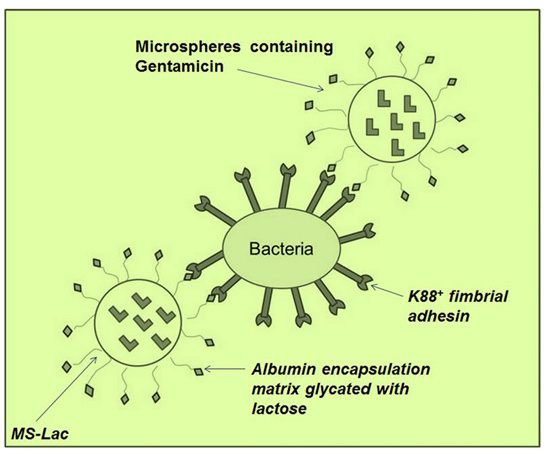
K88 Fimbrial Adhesin Targeting of Microspheres Containing Gentamicin Made with Albumin Glycated with Lactose †
Abstract
:1. Introduction
2. Results
| Formulation | Glutaraldehyde in Toluene (mg/L) | Entrapment Efficiency (%) a | Particle (µm) b | |
|---|---|---|---|---|
| Average Size | Polydispersity | |||
| MS-Lac1 | 4.2 | 89.4 ± 2.0 | 10 | 3–25 |
| MS-Lac2 | 8.2 | 66.3 ± 2.5 | 12 | 3–30 |
| MS-Lac3 | 16.8 | 48.5 ± 6.5 | 17 | 8–35 |
2.1. Entrapment Efficiency
2.2. In Vitro Release Studies

| Formulation | First Order F = 1 − e(−k∙t) | Peppas F = k∙tn | Square root time F = k∙t2 | |||
|---|---|---|---|---|---|---|
| r2 | k | r2 | n | r2 | k | |
| MS-Lac1 | 0.97 | 0.352 | 0.91 | 0.234 | 0.68 | 25.19 |
| MS-Lac2 | 0.97 | 0.297 | 0.95 | 0.273 | 0.74 | 24.87 |
| MS-Lac3 | 0.96 | 0.353 | 0.91 | 0.2341 | 0.61 | 25.19 |
2.3. In Vitro Antimicrobial Activity
| MIC (µg/mL) | ||
|---|---|---|
| Formulation | MS-Lac | Estimated Gentamicin in Microspheres * |
| MS-Lac1 | 2.16 | 0.31 |
| MS-Lac2 | 2.92 | 0.31 |
| MS-Lac3 | 4.00 | 0.31 |
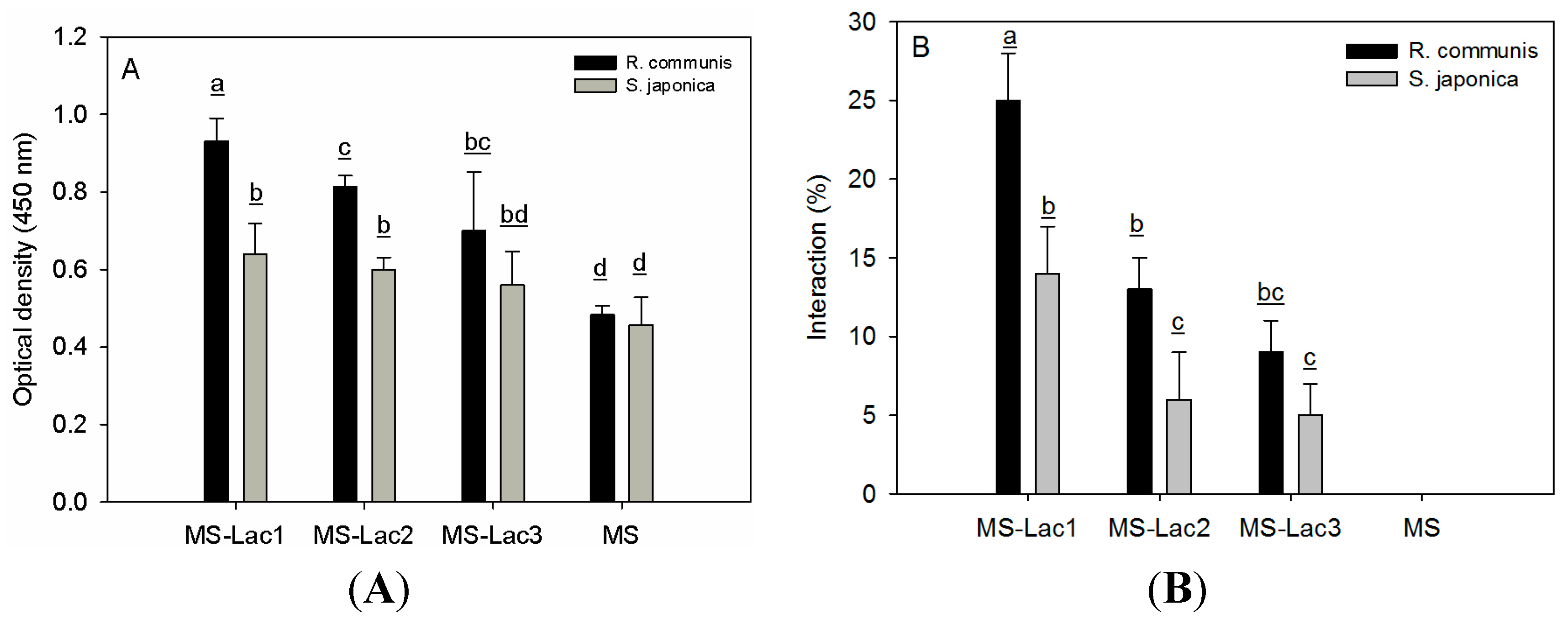
2.4. Interaction Assays

3. Discussion
4. Experimental
4.1. Materials
4.2. Preparation of Microspheres
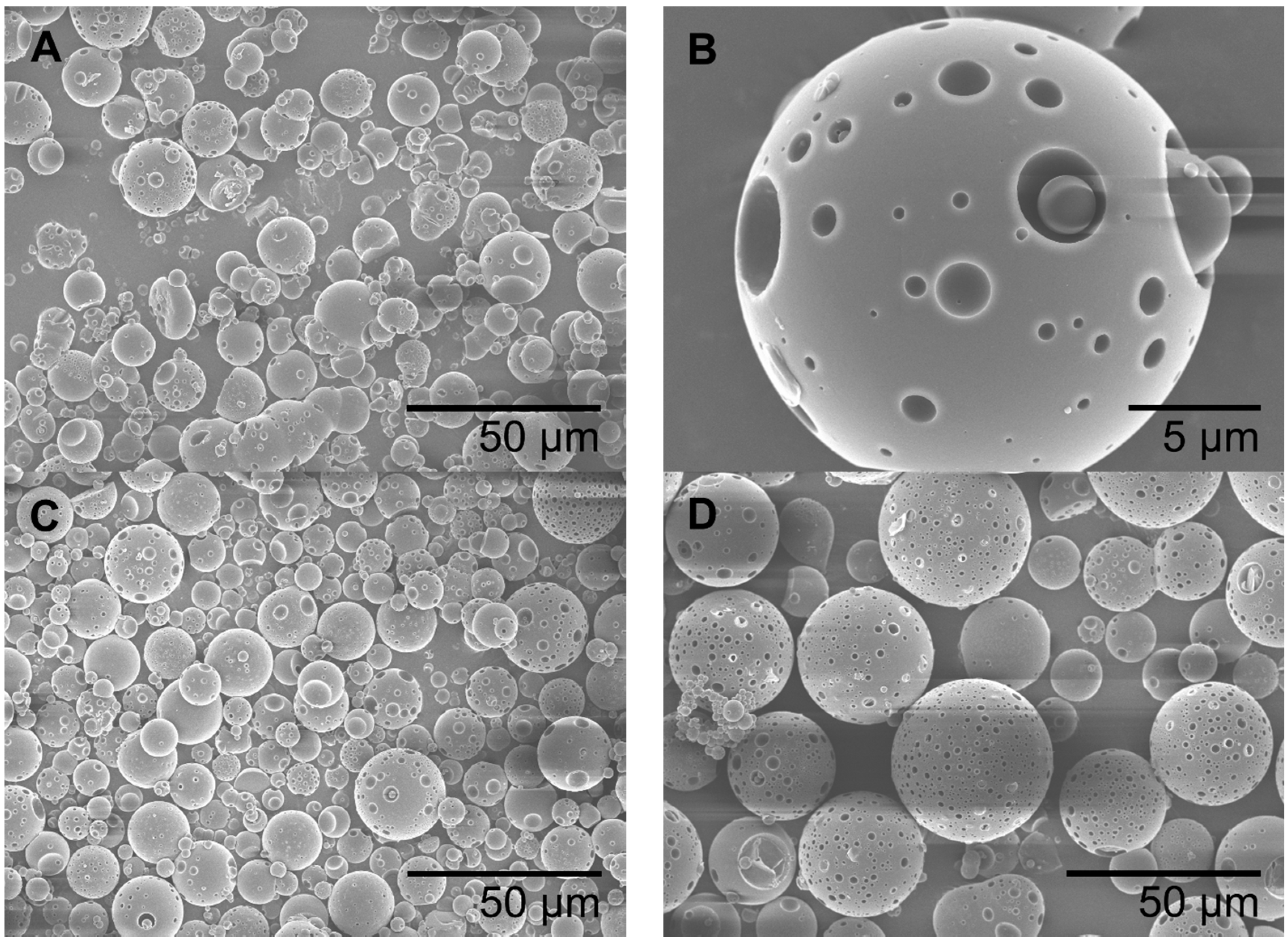
4.3. Morphology Studies
4.4. Entrapment Efficiency
4.5. In Vitro Release Studies
4.6. In Vitro Antimicrobial Activity
4.7. Enzyme-Linked Lectin Recognition Assays
4.8. Enzyme-Linked Adhesin Recognition Assays
4.9. Flow Cytometry
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sinha, V.R.; Aggarwal, A.; Trehan, A. Biodegradable pegylated microspheres and nanospheres. Am. J. Drug Deliv. 2004, 2, 157–171. [Google Scholar] [CrossRef]
- Varde, N.K.; Pack, D.W. Microspheres for controlled release drug delivery. Expert Opin. Biol. Ther. 2004, 4, 35–51. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.Z.; Langer, R.; Farokhzad, O.C. Nanoparticle delivery of cancer drugs. Annu. Rev. Med. 2012, 63, 185–198. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, D.; El-Maraghi, R.H.; Hammel, P.; Heinemann, V.; Kunzmann, V.; Sastre, J.; Scheithauer, W.; Siena, S.; Tabernero, J.; Teixeira, L. Nab-paclitaxel plus gemcitabine for metastatic pancreatic cancer: Long-term survival from a phase iii trial. J. Natl. Cancer Inst. 2015, 107. [Google Scholar] [CrossRef] [PubMed]
- Langer, K.; Anhorn, M.; Steinhauser, I.; Dreis, S.; Celebi, D.; Schrickel, N.; Faust, S.; Vogel, V. Human serum albumin (hsa) nanoparticles: Reproducibility of preparation process and kinetics of enzymatic degradation. Int. J. Pharm. 2008, 347, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Dhar, S.; Kolishetti, N.; Lippard, S.J.; Farokhzad, O.C. Targeted delivery of a cisplatin prodrug for safer and more effective prostate cancer therapy in vivo. Proc. Natl. Acad. Sci. USA 2011, 108, 1850–1855. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.S.; Lee, B.F.; Leibfarth, F.A.; Eisenstein, M.; Robb, M.J.; Lynd, N.A.; Hawker, C.J.; Soh, H.T. Synthetic aptamer-polymer hybrid constructs for programmed drug delivery into specific target cells. J. Am. Chem. Soc. 2014, 136, 15010–15015. [Google Scholar] [CrossRef] [PubMed]
- Arruebo, M.; Valladares, M.; González-Fernández, Á. Antibody-conjugated nanoparticles for biomedical applications. J. Nanomater. 2009, 2009, 37. [Google Scholar] [CrossRef]
- Zhang, C.; Cheng, Y.; Qu, G.; Wu, X.; Ding, Y.; Cheng, Z.; Yu, L.; Ping, Q. Preparation and characterization of galactosylated chitosan coated bsa microspheres containing 5-fluorouracil. Carbohydr. Polym. 2008, 72, 390–397. [Google Scholar] [CrossRef]
- Yang, Y.; Yuan, S.-X.; Zhao, L.-H.; Wang, C.-W.; Ni, J.-S.; Wang, Z.-G.; Lin, C.; Wu, M.-C.; Zhou, W.-P. Ligand-directed stearic acid grafted chitosan micelles to increase therapeutic efficacy in hepatic cancer. Mol. Pharm. 2015, 12, 644–652. [Google Scholar] [CrossRef] [PubMed]
- Ohya, Y.; Takei, T.; Fukushima, H.; Ouchi, T. Preparation of albumin microspheres grafted galactose residues through polyethylene-glycol spacers, release behavior of 5-fluorouracil from them, and their lectin-mediated aggregation. J. Macromol. Sci. Chem. 1991, 28, 743–760. [Google Scholar] [CrossRef]
- Kassab, R.; Parrot-Lopez, H.; Fessi, H.; Menaucourt, J.; Bonaly, R.; Coulon, J. Molecular recognition by kluyveromyces of amphotericin b-loaded, galactose-tagged, poly (lactic acid) microspheres. Bioorg. Med. Chem. 2002, 10, 1767–1775. [Google Scholar] [CrossRef]
- Ledesma-Osuna, A.I.; Ramos-Clamont, G.; Vazquez-Moreno, L. Biorecognition of chemically modified bovine serum albumin with lactose prepared under different conditions. J. Agric. Food Chem. 2009, 57, 9734–9739. [Google Scholar] [CrossRef] [PubMed]
- Sarabia-Sainz, A.-I.; Ramos-Clamont, G.; Winzerling, J.; Vázquez-Moreno, L. Bacterial recognition of thermal glycation products derived from porcine serum albumin with lactose. Acta Biochim. Pol. 2011, 58, 95–100. [Google Scholar] [PubMed]
- Mathew, S.T.; Devi, S.G.; Sandhya, K. Formulation and evaluation of ketorolac tromethamine-loaded albumin microspheres for potential intramuscular administration. Aaps Pharmscitech 2007, 8, E100–E108. [Google Scholar] [CrossRef] [PubMed]
- Zahirul, M.; Khan, I. Dissolution testing for sustained or controlled release oral dosage forms and correlation with in vivo data: Challenges and opportunities. Int. J. Pharm. 1996, 140, 131–143. [Google Scholar] [CrossRef]
- Narasimhan, B.; Mallapragada, S.; Peppas, N. Release kinetics, data interpretation. Encycl. Control. Drug Deliv. 1999, 2, 921–935. [Google Scholar]
- Costa, P.; Sousa Lobo, J.M. Modeling and comparison of dissolution profiles. Eur. J. Pharm. Sci. 2001, 13, 123–133. [Google Scholar] [CrossRef]
- Grange, P.A.; Mouricout, M.l.A.; Levery, S.B.; Francis, D.H.; Erickson, A.K. Evaluation of receptor binding specificity of escherichia coli k88 (f4) fimbrial adhesin variants using porcine serum transferrin and glycosphingolipids as model receptors. Infect. Immun. 2002, 70, 2336–2343. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.; Li, B.; Shen, F.; Fu, Q. Formulation and characterization of albumin microspheres containing norcantharidate for liver tumor targeting. Drug Deliv. 2014, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Sarabia-Sainz, A.-I.; Montfort, G.R.-C.; Lizardi-Mendoza, J.; Sáchez-Saavedra, M.D.P.; Candia-Plata, M.D.C.; Guzman, R.Z.; Lucero-Acuña, A.; Vazquez-Moreno, L. Formulation and characterization of gentamicin-loaded albumin microspheres as a potential drug carrier for the treatment of e. Coli k88 infections. Int. J. Drug Deliv. 2012, 4, 209–218. [Google Scholar]
- Damgé, C.; Maincent, P.; Ubrich, N. Oral delivery of insulin associated to polymeric nanoparticles in diabetic rats. J. Control. Release 2007, 117, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Patil, G.V. Biopolymer albumin for diagnosis and in drug delivery. Drug Dev. Res. 2003, 58, 219–247. [Google Scholar] [CrossRef]
- Leo, E.; Angela Vandelli, M.; Cameroni, R.; Forni, F. Doxorubicin-loaded gelatin nanoparticles stabilized by glutaraldehyde: Involvement of the drug in the cross-linking process. Int. J. Pharm. 1997, 155, 75–82. [Google Scholar] [CrossRef]
- Thakkar, H.P.; Murthy, R.R. Effect of cross-linking agent on the characteristics of celecoxib loaded chitosan microspheres. Asian J. Pharm. 2008, 2, 246. [Google Scholar] [CrossRef]
- Peppas, N. 1. Commentary on an exponential model for the analysis of drug delivery: Original research article: A simple equation for description of solute release: I II. Fickian and non-fickian release from non-swellable devices in the form of slabs, spheres, cylinders or discs, 1987. J. Control. Release: Off. J. Control. Release Soc. 2014, 190, 31. [Google Scholar]
- Theodorou, V.; Fioramonti, J.; Hachet, T.; Bueno, L. Absorptive and motor components of the antidiarrhoeal action of loperamide: An in vivo study in pigs. Gut 1991, 32, 1355–1359. [Google Scholar] [CrossRef] [PubMed]
- Haswani, D.K.; Nettey, H.; Oettinger, C.; D’Souza, M.J. Formulation, characterization and pharmacokinetic evaluation of gentamicin sulphate loaded albumin microspheres. J. Microencapsul. 2006, 23, 875–886. [Google Scholar] [CrossRef] [PubMed]
- Stubbings, W.; Bostock, J.; Ingham, E.; Chopra, I. Mechanisms of the post-antibiotic effects induced by rifampicin and gentamicin in escherichia coli. J. Antimicrob. Chemother. 2006, 58, 444–448. [Google Scholar] [CrossRef] [PubMed]
- Sarabia-Sainz, A.-I.; Ramos-Clamont, G.; Candia-Plata, M.D.C.; Vázquez-Moreno, L. Biorecognition of escherichia coli k88 adhesin for glycated porcine albumin. Int. J. Biol. Macromol. 2009, 44, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Myllyharju, J.; Nokkala, S. Localization and identification of galactose/n-acetylgalactosamine and sialic acid-containing proteins in chinese hamster metaphase chromosomes. Cell Biol. Int. 1998, 22, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Moreno, N.; Chevalier, M.; Ronzon, F.; Manin, C.; Dupuy, M.; Krell, T.; Rieu, J.P. Unbinding forces of single pertussis toxin-antibody complexes measured by atomic force spectroscopy correlate with their dissociation rates determined by surface plasmon resonance. J. Mol. Recognit. 2011, 24, 1105–1114. [Google Scholar] [CrossRef] [PubMed]
- Vandana Grover, S.G.; Chakraborti, Anuradha; Majumdar, Siddhartha; Ganguly, Nirmal Kumar. Galactose-specific fimbrial adhesin of enteroaggregative escherichia coli: A possible aggregative factor. Curr. Microbiol. 2007, 54, 175–179. [Google Scholar] [CrossRef] [PubMed]
- Prior, S.; Gander, B.; Lecároz, C.; Irache, J.M.; Gamazo, C. Gentamicin-loaded microspheres for reducing the intracellular brucella abortus load in infected monocytes. J. Antimicrob. Chemother. 2004, 53, 981–988. [Google Scholar] [CrossRef] [PubMed]
- Fayle, S.N.E.; Healy, J.P.; Brown, P.A.; Reid, E.A.; Gerrard, J.A.; Ames, J.M. Novel approaches to the analysis of the maillard reaction of proteins. Electrophoresis 2001, 22, 1518–1525. [Google Scholar] [CrossRef]
- Shah, M.V.; de Gennaro, M.D.; Suryakasuma, H. An evaluation of albumin microcapsules prepared using a multiple emulsion technique. J. Microencapsul. 1987, 4, 223–238. [Google Scholar] [CrossRef] [PubMed]
- Peppas, N. Analysis of fickian and non-fickian drug release from polymers. Pharm. Acta Helv. 1984, 60, 110–111. [Google Scholar]
- Higuchi, T. Rate of release of medicaments from ointment bases containing drugs in suspension. J. Pharm. Sci. 1961, 50, 874–875. [Google Scholar] [CrossRef] [PubMed]
- Holowachuk, S.A.; Bal’a, M.F.; Buddington, R.K. A kinetic microplate method for quantifying the antibacterial properties of biological fluids. J. Microbiol. Methods 2003, 55, 441–446. [Google Scholar] [CrossRef]
- Lourenço, F.R.; Pinto, T.J.A. Antibiotic microbial assay using kinetic-reading microplate system. Braz. J. Pharm. Sci. 2011, 47, 573–584. [Google Scholar] [CrossRef] [Green Version]
- Mooi, F.; de Graaf, F.; van Embden, J. Cloning mapping and expression of the genetic determinant that encodes for the k88ab antigen. Nucleic Acids Res. 1979, 6, 849–865. [Google Scholar] [CrossRef] [PubMed]
- Keegan, M.E.; Royce, S.M.; Fahmy, T.; Saltzman, W.M. In vitro evaluation of biodegradable microspheres with surface-bound ligands. J. Control. Release 2006, 110, 574–580. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sarabia-Sainz, A.-i.; Sarabia-Sainz, H.M.; Montfort, G.R.-C.; Mata-Haro, V.; Guzman-Partida, A.M.; Guzman, R.; Garcia-Soto, M.; Vazquez-Moreno, L. K88 Fimbrial Adhesin Targeting of Microspheres Containing Gentamicin Made with Albumin Glycated with Lactose. Int. J. Mol. Sci. 2015, 16, 22425-22437. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms160922425
Sarabia-Sainz A-i, Sarabia-Sainz HM, Montfort GR-C, Mata-Haro V, Guzman-Partida AM, Guzman R, Garcia-Soto M, Vazquez-Moreno L. K88 Fimbrial Adhesin Targeting of Microspheres Containing Gentamicin Made with Albumin Glycated with Lactose. International Journal of Molecular Sciences. 2015; 16(9):22425-22437. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms160922425
Chicago/Turabian StyleSarabia-Sainz, Andre-i, Hector Manuel Sarabia-Sainz, Gabriela Ramos-Clamont Montfort, Veronica Mata-Haro, Ana María Guzman-Partida, Roberto Guzman, Mariano Garcia-Soto, and Luz Vazquez-Moreno. 2015. "K88 Fimbrial Adhesin Targeting of Microspheres Containing Gentamicin Made with Albumin Glycated with Lactose" International Journal of Molecular Sciences 16, no. 9: 22425-22437. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms160922425