C/EBP β Mediates Endoplasmic Reticulum Stress Regulated Inflammatory Response and Extracellular Matrix Degradation in LPS-Stimulated Human Periodontal Ligament Cells
Abstract
:1. Introduction
2. Results
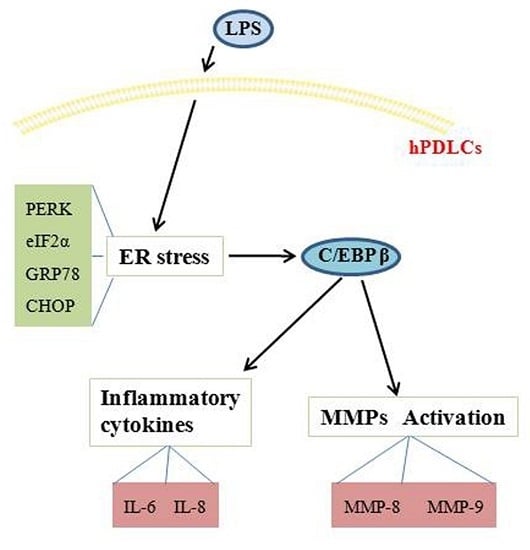
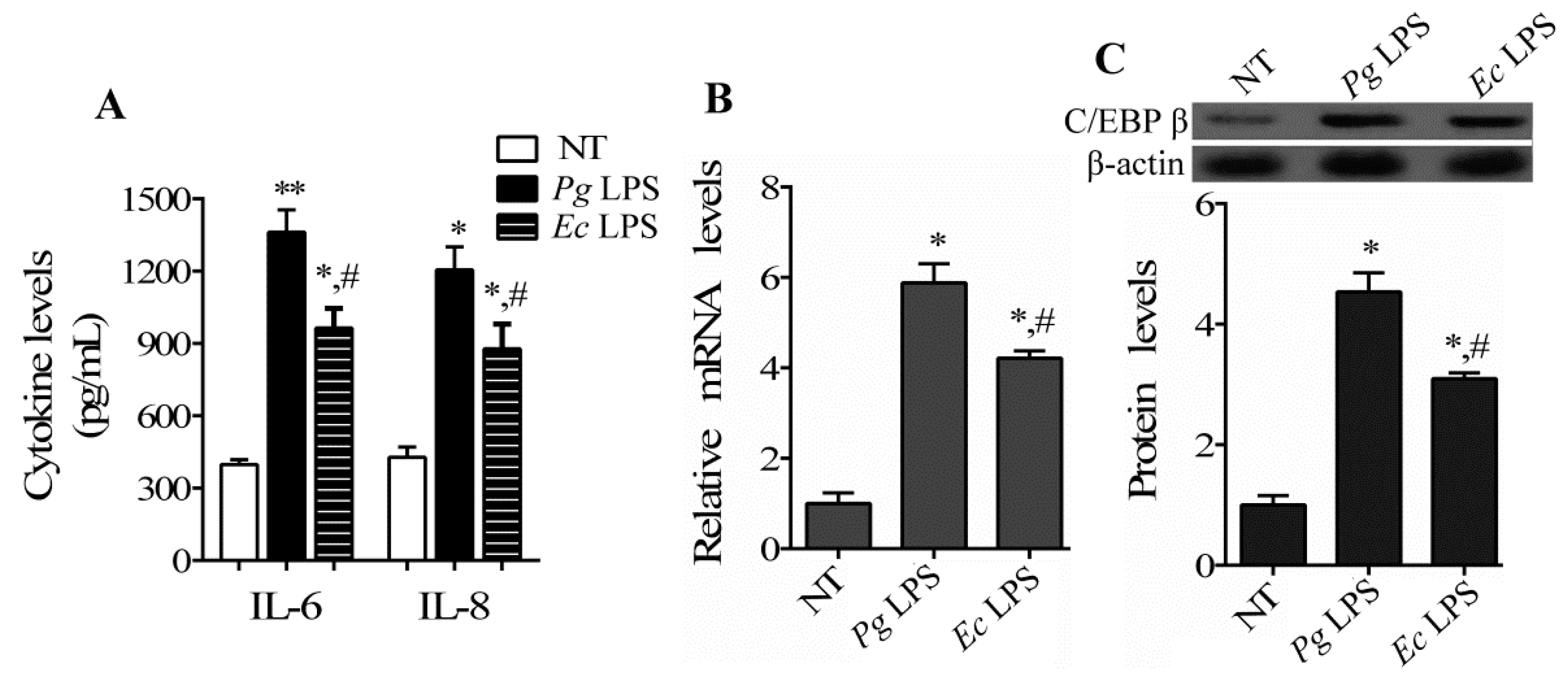
2.1. C/EBP β Gene Expression Is Increased in Human Periodontal Ligament Cells (hPDLCs) Exposed to Lipopolysaccharide (LPS)
2.2. C/EBP β Induces the Production of Pro-Inflammatory Cytokines and Enhances the Activation of Matrix Metalloproteinases (MMPs) in hPDLCs Stimulated by LPS
2.3. Endoplasmic Reticulum (ER) Stress Regulates Inflammatory Response and Extracellular Matrix Degradation Induced by LPS in hPDLCs
2.4. C/EBP β Mediates the Regulatory Role of ER Stress in hPDLCs
3. Discussion
4. Experimental Section
4.1. Cell Culture
4.2. LPS Preparation and Stimulation
4.3. Cell Transfection
4.4. Quantitative RT-PCR
4.5. Western Blotting
4.6. ELISA
4.7. Gelatin Zymography
4.8. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hajishengallis, G. Periodontitis: From microbial immune subversion to systemic inflammation. Nat. Rev. Immunol. 2015, 15, 30–44. [Google Scholar] [CrossRef] [PubMed]
- Darveau, R.P.; Tanner, A.; Page, R.C. The microbial challenge in periodontitis. Periodontology 1997, 14, 12–32. [Google Scholar] [CrossRef]
- Pink, C.; Kocher, T.; Meisel, P.; Dorr, M.; Markus, M.R.; Jablonowski, L.; Grotevendt, A.; Nauck, M.; Holtfreter, B. Longitudinal effects of systemic inflammation markers on periodontitis. J. Clin. Periodontol. 2015, 42, 988–997. [Google Scholar] [CrossRef] [PubMed]
- Kezhong, Z.; Kaufman, R.J. From endoplasmic-reticulum stress to the inflammatory response. Nature 2008, 454, 455–462. [Google Scholar]
- Uehara, Y.; Hirose, J.; Yamabe, S.; Okamoto, N.; Okada, T.; Oyadomari, S.; Mizuta, H. Endoplasmic reticulum stress-induced apoptosis contributes to articular cartilage degeneration via C/EBP homologous protein. Osteoarthr. Cartil. 2014, 22, 1007–1017. [Google Scholar] [CrossRef] [PubMed]
- Yamada, H.; Nakajima, T.; Domon, H.; Honda, T.; Yamazaki, K. Endoplasmic reticulum stress response and bone loss in experimental periodontitis in mice. J. Periodontal Res. 2015, 50, 500–508. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Xiong, M.; Huang, B.; Chen, H. Advanced glycation end products upregulate the endoplasmic reticulum stress in human periodontal ligament cells. J. Periodontol. 2015, 86, 440–447. [Google Scholar] [CrossRef] [PubMed]
- Ko, C.Y.; Chang, W.C.; Wang, J.M. Biological roles of CCAAT/enhancer-binding protein δ during inflammation. J. Biomed. Sci. 2015, 22, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Poli, V. The role of C/EBP isoforms in the control of inflammatory and native immunity functions. J. Biol. Chem. 1998, 273, 29279–29282. [Google Scholar] [CrossRef] [PubMed]
- Chiribau, C.B.; Gaccioli, F.; Huang, C.C.; Yuan, C.L.; Hatzoglou, M. Molecular symbiosis of CHOP and C/EBP β isoform LIP contributes to endoplasmic reticulum stress-induced apoptosis. Mol. Cell. Biol. 2010, 30, 3722–3731. [Google Scholar] [CrossRef] [PubMed]
- Arensdorf, A.M.; Rutkowski, D.T. Endoplasmic reticulum stress impairs IL-4/IL-13 signaling through C/EBP β-mediated transcriptional suppression. J. Cell Sci. 2013, 126, 4026–4036. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.I.; Kang, K.L.; Shin, S.I.; Herr, Y.; Lee, Y.M.; Kim, E.C. Endoplasmic reticulum stress modulates nicotine-induced extracellular matrix degradation in human periodontal ligament cells. J. Periodontal Res. 2012, 47, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Beklen, A.; Ainola, M.; Hukkanen, M.; Gurgan, C.; Sorsa, T.; Konttinen, Y.T. MMPs, IL-1, and TNF are regulated by IL-17 in periodontitis. J. Dent. Res. 2007, 86, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Meir, O.; Dvash, E.; Werman, A.; Rubinstein, M. C/EBP-β regulates endoplasmic reticulum stress-triggered cell death in mouse and human models. PLoS ONE 2010, 5, e9516. [Google Scholar] [CrossRef]
- Andrukhov, O.; Andrukhova, O.; Hulan, U.; Tang, Y.; Bantleon, H.P.; Rausch-Fan, X. Both 25-hydroxyvitamin-D3 and 1,25-dihydroxyvitamin-D3 reduces inflammatory response in human periodontal ligament cells. PLoS ONE 2014, 9, e90301. [Google Scholar] [CrossRef] [PubMed]
- Ozdemir, B.; Shi, B.; Bantleon, H.P.; Moritz, A.; Rausch-Fan, X.; Andrukhov, O. Endocannabinoids and inflammatory response in periodontal ligament cells. PLoS ONE 2014, 9, e107407. [Google Scholar] [CrossRef] [PubMed]
- Soder, B.; Airila Mansson, S.; Soder, P. O.; Kari, K.; Meurman, J. Levels of matrix metalloproteinases-8 and -9 with simultaneous presence of periodontal pathogens in gingival crevicular fluid as well as matrix metalloproteinase-9 and cholesterol in blood. J. Periodontal Res. 2006, 41, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Mouzakiti, E.; Pepelassi, E.; Fanourakis, G.; Markopoulou, C.; Tseleni-Balafouta, S.; Vrotsos, I. Expression of MMPs and TIMP-1 in smoker and nonsmoker chronic periodontitis patients before and after periodontal treatment. J. Periodontal Res. 2012, 47, 532–542. [Google Scholar] [CrossRef] [PubMed]
- Kasamatsu, A.; Uzawa, K.; Shimada, K.; Shiiba, M.; Otsuka, Y.; Seki, N.; Abiko, Y.; Tanzawa, H. Elevation of galectin-9 as an inflammatory response in the periodontal ligament cells exposed to Porphylomonas gingivalis lipopolysaccharide in vitro and in vivo. Int. J. Biochem. Cell Biol. 2005, 37, 397–408. [Google Scholar] [CrossRef] [PubMed]
- Anand, S.; Ebner, J.; Warren, C.B.; Raam, M.S.; Piliang, M.; Billings, S.D.; Maytin, E.V. C/EBP transcription factors in human squamous cell carcinoma: Selective changes in expression of isoforms correlate with the neoplastic state. PLoS ONE 2014, 9, e112073. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, K.; van Phi, V.D.; Brase, A.; Phi-van, L. Inhibition of serotonin transporter expression by C/EBPβ in LPS-activated macrophage cells (HD11). Innate Immun. 2015, 21, 406–415. [Google Scholar] [CrossRef] [PubMed]
- Gerlach, J.C.; Over, P.; Foka, H.G.; Turner, M.E.; Thompson, R.L.; Gridelli, B.; Schmelzer, E. Role of transcription factor CCAAT/enhancer-binding protein α in human fetal liver cell types in vitro. Hepatol. Res. 2015, 45, 919–932. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S.M.; Schroeder-Gloeckler, J.M.; Janssen, R.C.; Jiang, H.; Qadri, I.; Maclean, K.N.; Friedman, J.E. CCAAT/enhancing binding protein β deletion in mice attenuates inflammation, endoplasmic reticulum stress, and lipid accumulation in diet-induced nonalcoholic steatohepatitis. Hepatology 2007, 45, 1108–1117. [Google Scholar] [CrossRef] [PubMed]
- Xia, P.; Zhang, R.; Ge, G. C/EBPβ Mediates TNF-α-induced cancer cell migration by inducing MMP expression dependent on p38 MAPK. J. Cell Biochem. 2015, 116, 2766–2777. [Google Scholar] [CrossRef] [PubMed]
- Savage, T.; Bennett, T.; Huang, Y.F.; Kelly, P.L.; Durant, N.E.; Adams, D.J.; Mina, M.; Harrison, J.R. Mandibular phenotype of p20C/EBPβ transgenic mice: Reduced alveolar bone mass and site-specific dentin dysplasia. Bone 2006, 39, 552–564. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.; Guochun, Z.; Liang, H.; Mengrui, W.; Hongliang, C.; Yi-Ping, L. C/EBPα regulates osteoclast lineage commitment. Proc. Natl. Acad. Sci. USA 2013, 110, 7294–7299. [Google Scholar]
- Domon, H.; Takahashi, N.; Honda, T.; Nakajima, T.; Tabeta, K.; Abiko, Y.; Yamazaki, K. Up-regulation of the endoplasmic reticulum stress-response in periodontal disease. Clin. Chim. Acta 2009, 401, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Dudenhausen, E.E.; Pan, Y.X.; Zhong, C.; Kilberg, M.S. Human CCAAT/enhancer-binding protein β gene expression is activated by endoplasmic reticulum stress through an unfolded protein response element downstream of the protein coding sequence. J. Biol. Chem. 2004, 279, 27948–27956. [Google Scholar] [CrossRef] [PubMed]
- Fiorentini, C.; Bodei, S.; Bedussi, F.; Fragni, M.; Bonini, S.A.; Simeone, C.; Zani, D.; Berruti, A.; Missale, C.; Memo, M. GPNMB/OA protein increases the invasiveness of human metastatic prostate cancer cell lines DU145 and PC3 through MMP-2 and MMP-9 activity. Exp. Cell Res. 2014, 323, 100–111. [Google Scholar] [CrossRef] [PubMed]



© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bai, Y.; Wei, Y.; Wu, L.; Wei, J.; Wang, X.; Bai, Y. C/EBP β Mediates Endoplasmic Reticulum Stress Regulated Inflammatory Response and Extracellular Matrix Degradation in LPS-Stimulated Human Periodontal Ligament Cells. Int. J. Mol. Sci. 2016, 17, 385. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms17030385
Bai Y, Wei Y, Wu L, Wei J, Wang X, Bai Y. C/EBP β Mediates Endoplasmic Reticulum Stress Regulated Inflammatory Response and Extracellular Matrix Degradation in LPS-Stimulated Human Periodontal Ligament Cells. International Journal of Molecular Sciences. 2016; 17(3):385. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms17030385
Chicago/Turabian StyleBai, Yudi, Yi Wei, Lian Wu, Jianhua Wei, Xiaojing Wang, and Yuxiang Bai. 2016. "C/EBP β Mediates Endoplasmic Reticulum Stress Regulated Inflammatory Response and Extracellular Matrix Degradation in LPS-Stimulated Human Periodontal Ligament Cells" International Journal of Molecular Sciences 17, no. 3: 385. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms17030385