Antibacterial Activity of Ciprofloxacin-Encapsulated Cockle Shells Calcium Carbonate (Aragonite) Nanoparticles and Its Biocompatability in Macrophage J774A.1
Abstract
:1. Introduction
2. Results and Discussion
2.1. Preparation of Nanoparticles
2.2. Ciprofloxacin-Loading and Encapsulation Efficiency
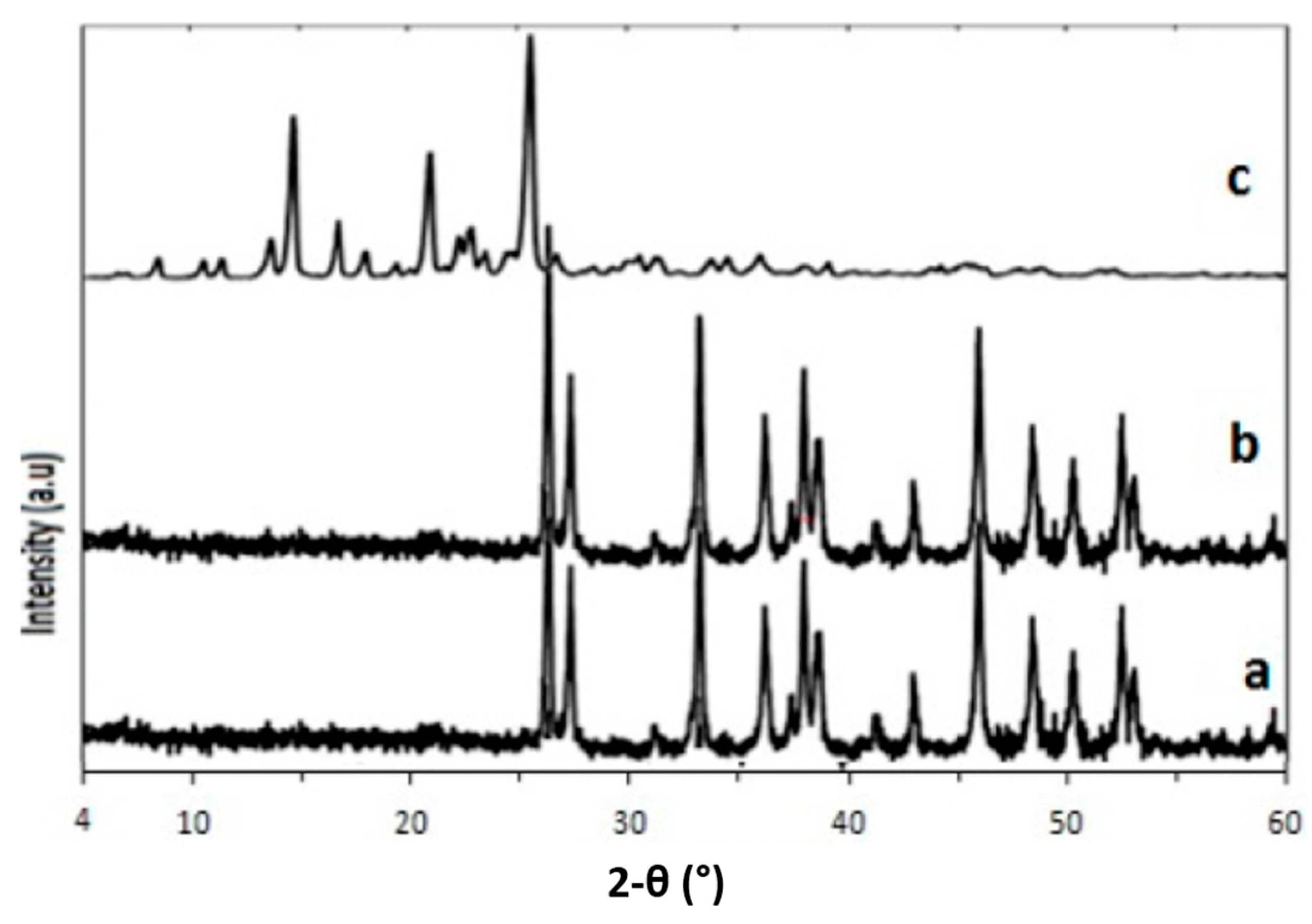
2.3. Characterization and Stability of Ciprofloxacin–Nanoparticles
2.4. Biocompatibility Evaluation
2.4.1. Immunogenicity Assessment
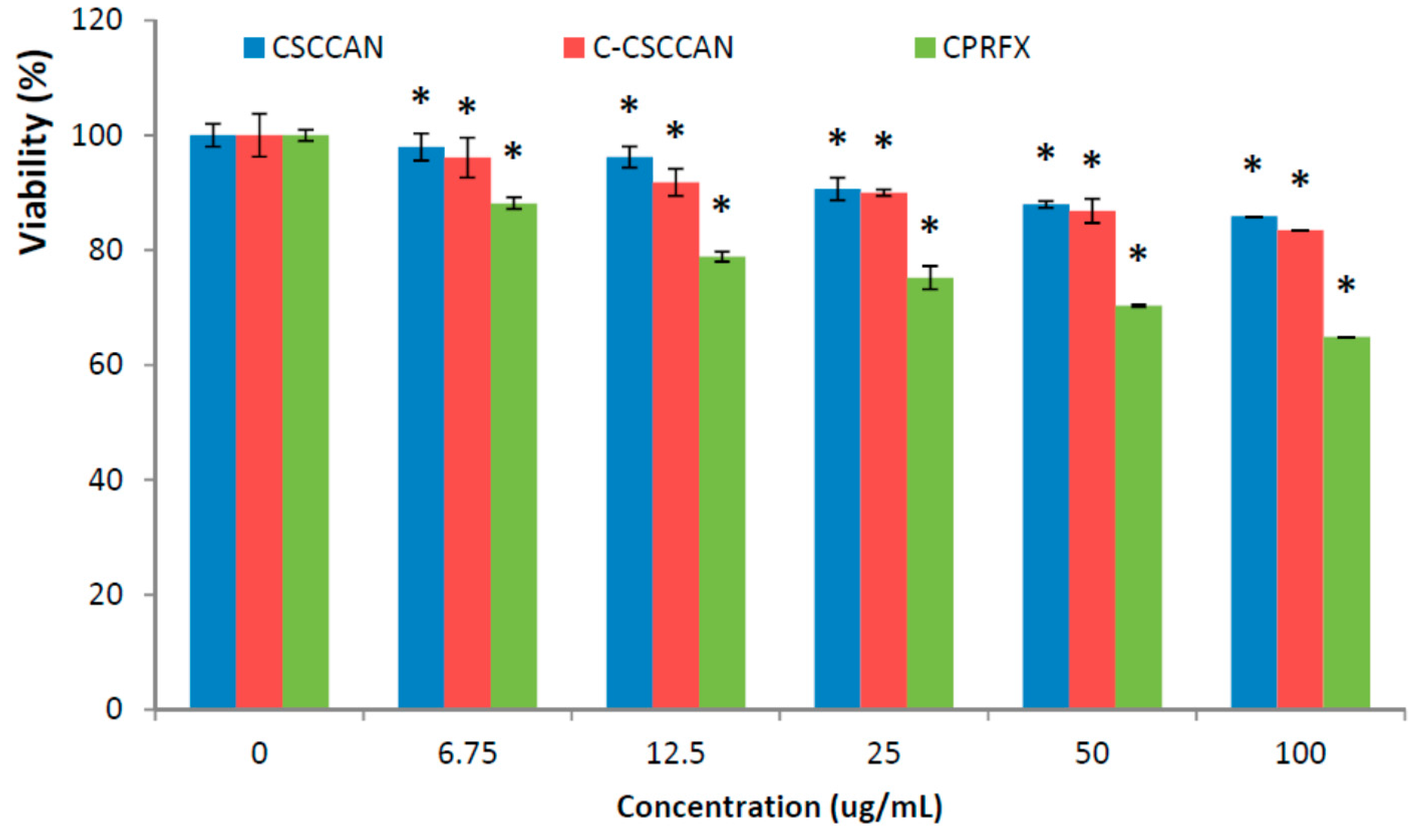
2.4.2. Viability Test
2.4.3. Genotoxicity Test
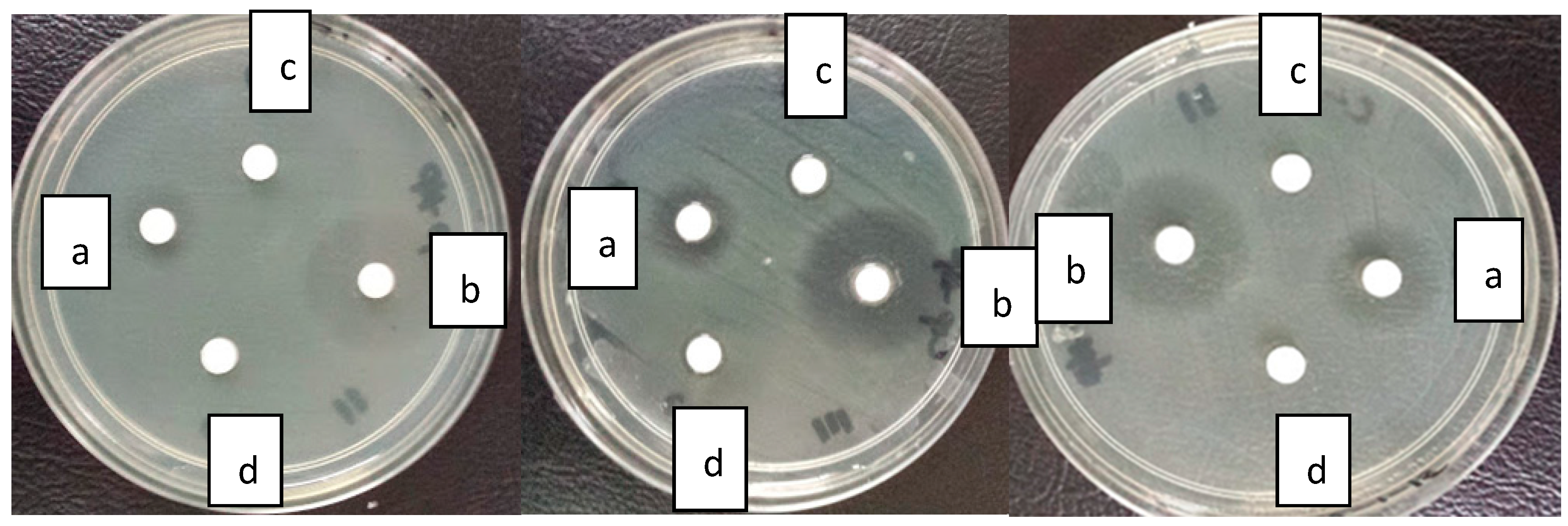
2.5. Antibacterial Activity
3. Materials and Methods
3.1. Reagents, Chemicals and Media
3.2. Bacterial Strain
3.3. Top-Down Synthesis of Cockle Shells Calcium Carbonate Nanoparticles
3.4. Drug Loading and Encapsulation
3.5. Transmission Electron Microscopy
3.6. Field Emission Scanning Electron Microscope
3.7. ζ Potential
3.8. X-ray Powder Diffraction
3.9. In Vitro Biocompatibility Assays
3.9.1. Cell Culture
3.9.2. RNA Extraction and RT-PCR
3.9.3. MTT—Viability Assay
3.9.4. BrdU (ELISA) Genotoxicity Assay
3.10. In Vitro Antibacterial Susceptibility Test
3.10.1. Preparation of Drugs Stock Solutions
3.10.2. Disc Diffusion Susceptibility Assay
3.11. Statistical Analysis
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Hajipour, M.J.; Fromm, K.M.; Ashkarran, A.A.; Jimenez de Aberasturi, D.; de Larramendi, I.R.; Rojo, T. Antibacterial properties of nanoparticles. Trends Biotechnol. 2012, 30, 499–511. [Google Scholar] [CrossRef] [PubMed]
- Pinto-Alphandary, H.; Andremont, A.; Couvreur, P. Targeted delivery of antibiotics using liposomes and nanoparticles: Research and applications. Int. J. Antimicrob. Agents 2000, 13, 155–168. [Google Scholar] [CrossRef]
- Pestova, E.; Millichap, J.J.; Noskin, G.A.; Peterson, L.R. Intracellular targets of moxifloxacin: A comparison with other fluoroquinolones. J. Antimicrob. Chemother. 2000, 45, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Bambeke, F.V.; Balzi, E.; Tulkens, M. Antibiotic efflux pumps franc. Biochem. Pharmacol. 2000, 60, 457–470. [Google Scholar] [CrossRef]
- Singh, M.; Jadaun, G.P.S.; Ramdas, S.K.; Chauhan, V.; Mishra, R. Effect of efflux pump inhibitors on drug susceptibility of ofloxacin resistant Mycobacterium tuberculosis isolates. Indian J. Med. Res. 2011, 133, 535–540. [Google Scholar] [PubMed]
- Ahmed, A.; Ibrahim, M.; Abu-sef, F.A.; Hassan, Z.; Mohamed, S.; Mohamed, N. Contribution of different mechanisms to the resistance to fluoroquinolones in clinical isolates of Salmonella enterica. Braz. J. Infect. Dis. 2013, 7, 431–437. [Google Scholar]
- Carryn, S.; Chanteux, H.; Seral, C.; Mingeot-Leclercq, M.P.; van Bambeke, F.; Tulkens, P.M. Intracellular pharmacodynamics of antibiotics. Infect. Dis. Clin. N. Am. 2003, 17, 615–634. [Google Scholar] [CrossRef]
- Jacoby, G.A. Mechanisms of resistance to quinolones. Clin. Infect. Dis. 2005, 41, S120–S126. [Google Scholar] [CrossRef] [PubMed]
- Briones, E.; Colino, C.I.; Lanao, J.M. Delivery systems to increase the selectivity of antibiotics in phagocytic cells. J. Control. Release 2008, 125, 210–227. [Google Scholar] [CrossRef] [PubMed]
- Webb, M.S.; Boman, N.L.; Wiseman, D.J.; Saxon, D.; Sutton, K.; Wong, K.F. Antibacterial efficacy against an in vivo Salmonella typhimurium infection model and pharmacokinetics of a liposomal ciprofloxacin formulation. Antimicrob. Agents Chemother. 1998, 42, 45–52. [Google Scholar] [PubMed]
- Bhalerao, S.R.; Rote, A.R. Application of UV spectrophotometric methods for estimation of ciprofloxacin and tinidazole in combined tablet dosage form. Int. J. Pharmacol. Pharm. Sci. 2012, 4, 4–7. [Google Scholar]
- George, R.S.; Adrienne, L.W. Delmar Nurse’s Drug Handbook 2012 Edition, 21st ed.; West Virginia University, Cengage Learning: Clifton Park, NY, USA, 2011; Volume 2012, p. 22. [Google Scholar]
- Imbuluzqueta, E.; Gamazo, C.; Ariza, J.; Blanco-Prieto, M.J. Drug delivery systems for potential treatment of intracellular bacterial infections. Front. Biosci. 2010, 15, 397–417. [Google Scholar] [CrossRef]
- Piddock, L.J.V.; Garvey, M.I.; Rahman, M.M.; Gibbons, S. Natural and synthetic compounds such as trimethoprim behave as inhibitors of efflux in Gram-negative bacteria. J. Antimicrob. Chemother. 2010, 65, 1215–1223. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Zhuo, R.X.; Cheng, S.X. Modification of calcium carbonate based gene and drug delivery systems by a cell-penetrating peptide. Mol. BioSyst. 2012, 8, 3288–3294. [Google Scholar] [CrossRef] [PubMed]
- Pelgrift, R.Y.; Friedman, A.J. Nanotechnology as a therapeutic tool to combat microbial resistance. Adv. Drug Deliv. Rev. 2013, 65, 1803–1815. [Google Scholar] [CrossRef] [PubMed]
- Plapied, L.; Duhem, N.; des Rieux, A.; Préat, V. Fate of polymeric nanocarriers for oral drug delivery. Curr. Opin. Colloid Interface Sci. 2011, 16, 228–237. [Google Scholar] [CrossRef]
- Blecher, K.; Nasir, A.; Friedman, A. The growing role of nanotechnology in combating infectious disease. Virulence 2011, 2, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Huh, A.J.; Kwon, Y.J. “Nanoantibiotics”: A new paradigm for treating infectious diseases using nanomaterials in the antibiotics resistant era. J. Control. Release 2011, 156, 128–145. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.M.; Chen, C.H.; Pornpattananangkul, D.; Zhang, L.; Chan, M.; Hsieh, M.F. Eradication of drug resistant Staphylococcus aureus by liposomal oleic acids. Biomaterials 2011, 32, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Akbari, V.; Abedi, D.; Pardakhty, A.; Sadeghi-Aliabadi, H. Ciprofloxacin nano-niosomes for targeting intracellular infections: An in vitro evaluation. J. Nanopart. Res. 2013, 15. [Google Scholar] [CrossRef]
- Hono, S.C.; Anino, T.T.; Eki, T.S.; Orimoto, K.M. Pharmacokinetic and pharmacodynamic efficacy of intrapulmonary administration of ciprofloxacin for the treatment of respiratory infections. Drug Metab. Pharmacokinet. 2007, 22, 88–95. [Google Scholar]
- Ong, H.X.; Traini, D.; Cipolla, D.; Gonda, I.; Bebawy, M.; Agus, H. Liposomal nanoparticles control the uptake of ciprofloxacin across respiratory epithelia. J. Pharm. Res. 2012, 29, 3335–3346. [Google Scholar] [CrossRef] [PubMed]
- Banoee, M.; Seif, S.; Nazari, Z.E.; Jafari-Fesharaki, P.; Shahverdi, H.R.; Moballegh, A. ZnO nanoparticles enhanced antibacterial activity of ciprofloxacin against Staphylococcus aureus and Escherichia coli. J. Biomed. Mater. Res. Part B Appl. Biomater. 2010, 93, 557–561. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, M.; Yousuf, S.; Maitra, S. Decomposition study of calcium carbonate in cockle shell. J. Eng. Sci. Technol. 2012, 7, 1–10. [Google Scholar]
- Rodríguez-Ruiz, I.; Delgado-López, J.M.; Durán-Olivencia, M.A.; Iafisco, M.; Tampieri, A.; Colangelo, D. PH-responsive delivery of doxorubicin from citrate-apatite nanocrystals with tailored carbonate content. Langmuir 2013, 29, 8213–8221. [Google Scholar] [CrossRef] [PubMed]
- Kamba, A.S.; Ismail, M.; Azmi, T.; Ibrahim, T.; Abubakar, Z.Z. Synthesis and characterisation of calcium carbonate aragonite nanocrystals from cockle shell powder (Anadara granosa). J. Nanomater. 2013, 2013. [Google Scholar] [CrossRef]
- Lucas-Girot, A.; Verdier, M.C.; Tribut, O.; Sangleboeuf, J.C.; Allain, H.; Oudadesse, H. Gentamicin-loaded calcium carbonate materials: Comparison of two drug-loading modes. J. Biomed. Mater. Res. Part B Appl. Biomater. 2005, 73, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Kroll, A.; Pillukat, M.H.; Hahn, D.; Schnekenburger, J. Interference of engineered nanoparticles with in vitro toxicity assays. Arch. Toxicol. 2012, 86, 1123–1136. [Google Scholar] [CrossRef] [PubMed]
- Samberg, M.E.; Oldenburg, S.J.; Monteiro-Riviere, N.A. Evaluation of silver nanoparticle toxicity in skin in vivo and keratinocytes in vitro. Environ. Health Perspect. 2010, 118, 407–413. [Google Scholar] [CrossRef] [PubMed]
- McNeil, S.E. Immunological properties of engineered nanomaterials. Nat. Nanotechnol. 2007, 2, 469–478. [Google Scholar]
- Yang, E.; Jang, J.; Lim, D.; Choi, I. Enzyme-linked immunosorbent assay of IL-8 production in response to silver nanoparticles. Totowa Natl J. Humana Press 2012, 926, 131–139. [Google Scholar]
- Dong, Y.; Ying, C.; Li-Hong, D.; Yu-Jiang, Z. Controllable synthesis of CaCO3 micro/anocrystals with different morphologies in microemulsion. Chem. Res. Chin. Univ. 2010, 26, 678–682. [Google Scholar]
- Wada, N.; Seiichi, S.; Kiyoshi, K.; Takao, U. Formation of thin calcium carbonate films with aragonite and vaterite forms coexisting with polyacrylic acids and chitosan membranes. J. Colloid Interface Sci. 2004, 279, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Zou, W.; Liu, C.; Chen, Z.; Zhang, N. Preparation and characterization of cationic PLA-PEG nanoparticles for delivery of plasmid DNA. Nanoscale Res. Lett. 2009, 4, 982–992. [Google Scholar] [CrossRef] [PubMed]
- Velázquez-Castillo, R.; Reyes-Gasga, J.; García-Gutierrez, D.I.; Jose-Yacaman, M. Nanoscale characterization of nautilus shell structure: An example of natural self-assembly. J. Mater. Res. 2006, 21, 1484–1489. [Google Scholar] [CrossRef]
- Das, S.; Banerjee, R.; Bellare, J. Aspirin loaded albumin nanoparticles by coacervation: Implications in drug delivery. Trends Biomater. Artif. Organs. 2005, 18, 203–212. [Google Scholar]
- Mao, Z.; Ma, L.; Gao, C.; Shen, J. Preformed microcapsules for loading and sustained release of ciprofloxacin hydrochloride. J. Control. Release 2005, 104, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Hua, A.; Steven, A.; Jones de Villiers, M.M.; Yuri, M.L. Nano-encapsulation of furosemide microcrystals for controlled drug release. J. Control. Release 2003, 86, 59–68. [Google Scholar]
- Balabushevitch, N.G.; Sukhorukov, G.B.; Moroz, N.A.; Volodkin, D.V.; Larionova, N.I.; Donath, E. Encapsulation of proteins by layer-by-layer adsorption of polyelectrolytes onto protein aggregates: Factors regulating the protein release. Biotechnol. Bioeng. 2001, 76, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Shchukin, D.G.; Patel, A.A.; Sukhorukov, G.B.; Lvov, Y.M. Nanoassembly of biodegradable microcapsules for DNA encasing. J. Am. Chem. Soc. 2004, 126, 3374–3375. [Google Scholar] [CrossRef] [PubMed]
- Jeong, Y.-II.; Na, H.-S.; Seo, D.-H.; Kim, D.-G.; Lee, H.-C.; Jang, M.-K.; Nah, J.W. Ciprofloxacin-encapsulated poly(dl-lactide-co-glycolide) nanoparticles and its antibacterial activity. Int. J. Pharm. 2008, 352, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Tuck-Yun, C.; Sheng-Ming, W.; Zuo-Jun, H.; Zhou-Hao, X.; Guang-Qi, C.; Chen, Y.; Yong, L.; Hui, Z.; An-Wu, X. Calcium carbonate/CaIP6 nanocomposite particles as gene delivery vehicles for human vascular smooth muscle cells. J. Mater. Chem. 2010, 20. [Google Scholar] [CrossRef]
- Abreu, F.O.M.S.; Forte, M.M.C.; Kist, T.B.L.; Honaiser, L.P. Effect of the preparation method on the drug loading of alginate-chitosan microspheres. Express Polym. Lett. 2010, 4, 456–464. [Google Scholar] [CrossRef]
- Ren, G.; Hu, D.; Cheng, E.W.C.; Vargas-Reus, M.A.; Reip, P.; Allaker, R.P. Characterisation of copper oxide nanoparticles for antimicrobial applications. Int. J. Antimicrob. Agents 2009, 33, 587–590. [Google Scholar] [CrossRef] [PubMed]
- Mu, L.; Feng, S.S. PLGA/TPGS nanoparticles for controlled release of paclitaxel: Effects of the emulsifier and drug loading ratio. Pharm. Res. 2003, 20, 1864–1872. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Reineke, T.M. Hydroxyl stereochemistry and amine number within poly(glycoamidoamine)s affect intracellular DNA delivery. J. Am. Chem. Soc. 2005, 127, 3004–3015. [Google Scholar] [CrossRef] [PubMed]
- Shang, L.; Karin, N.; Gerd, U.N. Engineered nanoparticles interacting with cells: Size matters. J. Nanobiotechnol. 2014, 12. [Google Scholar] [CrossRef] [PubMed]
- Ahsan, F.; Rivas, I.P.; Khan, M.A.; Torres Suárez, A.I. Targeting to macrophages: Role of physicochemical properties of particulate carriers—Liposomes and microspheres—On the phagocytosis by macrophages. J. Control. Release 2002, 79, 29–40. [Google Scholar] [CrossRef]
- Kamba, S.A.; Ismail, M.; Hussein-al-ali, S.H.; Azmi, T.; Ibrahim, T.; Abubakar, Z.Z. In vitro delivery and controlled release of doxorubicin for targeting osteosarcoma bone cancer. Molecules 2013, 18, 10580–10598. [Google Scholar] [CrossRef] [PubMed]
- Jain, D.; Banerjee, R. Comparison of ciprofloxacin hydrochloride-loaded protein, lipid, and chitosan nanoparticles for drug delivery. J. Biomed. Mater. Res. Part B Appl. Biomater. 2008, 86, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Voort, J.; van den Mooter, G.; Verheyden, L.; Ludwig, A. Factorial design, physicochemical characterisation and activity of ciprofloxacin-PLGA nanoparticles. Int. J. Pharm. 2004, 275, 171–187. [Google Scholar]
- Surajit, D.; Wai, K.N.; Parijat, K.; Sanggu, K.; Reginald, B.H.T. Formulation design, preparation and physicochemical characterizations of solid lipid nanoparticles containing a hydrophobic drug: Effects of process variables. Colloids Surfaces B Biointerfaces 2011, 88, 483–489. [Google Scholar]
- Dinarello, C. Impact of basic research on tomorrow’s medicine. Proinflammatory cytokines. CHEST J. 2000, 118, 503–508. [Google Scholar] [CrossRef]
- De Groot, A.S.; Scott, D.W. Immunogenicity of protein therapeutics. Trends Immunol. 2007, 28, 482–490. [Google Scholar] [CrossRef] [PubMed]
- Zolnik, B.S.; González, F.Á.; Sadrieh, N.; Dobrovolskaia, M. Minireview: Nanoparticles and the immune system. Endocrinology 2010, 151, 458–465. [Google Scholar] [CrossRef] [PubMed]
- Veranth, J.M.; Kaser, E.G.; Veranth, M.M.; Koch, M.; Yost, G.S. Cytokine responses of human lung cells (BEAS-2B) treated with micron-sized and nanoparticles of metal oxides compared to soil dusts. Part Fibre Toxicol. 2007, 4. [Google Scholar] [CrossRef] [PubMed]
- Lewinski, N.; Colvin, V.; Drezek, R. Cytotoxicity of nanopartides. Small 2008, 4, 26–49. [Google Scholar] [CrossRef] [PubMed]
- Fiona, R.; Chapman, J.; Sullivan, T. Nanoparticles in Anti-Microbial Materials: Use and Characterisation; Royal Society of Chemistry: Cambridge, UK, 2012. [Google Scholar] [CrossRef]
- Jun, Y.; Kim, H.; Song, M.; Lim, J.; Lee, D.; Han, K.; Choi, J. In vitro effects of ciprofloxacin and roxithromycin on apoptosis of Jurkat T lymphocytes. Am. Soc. Microbiol. 2003, 47, 1161–1164. [Google Scholar] [CrossRef]
- Dalhoff, A; Shalit, I. Immunomodulatory effects of quinolones. Lancet Infect. Dis. 2003, 3, 359–371. [Google Scholar]
- Garcia-Käufer, M.; Haddad, T.; Bergheim, M.; Gminski, R.; Gupta, P.; Mathur, N. Genotoxic effect of ciprofloxacin during photolytic decomposition monitored by the in vitro micronucleus test (MNvit) in HepG2 cells. Environ. Sci. Pollut. Res. 2012, 19, 1719–1727. [Google Scholar] [CrossRef] [PubMed]
- Gürbay, A.; Garrel, C.; Osman, M.; Richard, M.J.; Favier, A.; Hincal, F. Cytotoxicity in ciprofloxacin-treated human fibroblast cells and protection by vitamin E. Hum. Exp. Toxicol. 2002, 21, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, J.W.; Claire, D.C.; Weissig, V.; Rowe, T.C. Delayed cytotoxicity and cleavage of mitochondrial DNA in ciprofloxacin-treated mammalian cells. Mol. Pharm. 1996, 50, 1178–1188. [Google Scholar]
- Kean, T.; Roth, S.; Maya, T. Trimethylated chitosans as non-viral gene delivery vectors: Cytotoxicity and transfection efficiency gene delivery. J. Control. Release 2005, 103, 643–653. [Google Scholar] [CrossRef] [PubMed]
- Battal, D.; Celik, A.; Güler, G.; Aktaş, A.; Yildirimcan, S.; Ocakoglu, K.; Çömelekoǧlu, Ü. SiO2 nanoparticule-induced size-dependent genotoxicity—An in vitro study using sister chromatid exchange, micronucleus and comet assay. Drug Chem. Toxicol. 2015, 38, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Ortiz, M.; Acosta-Torres, L.S.; Hernández-Padrón, G.; Mendieta, A.I.; Bernal, R.; Cruz-Vázquez, C. Biocompatibility of crystalline opal nanoparticles. Biomed. Eng. Online 2012, 11. [Google Scholar] [CrossRef] [PubMed]
- Ruparelia, J.P.; Chatterjee, A.K.; Duttagupta, S.P.; Mukherji, S. Strain specificity in antimicrobial activity of silver and copper nanoparticles. Acta Biomater. 2008, 4, 707–716. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Zhu, B.; Jia, Y.; Hou, W.; Su, C. Preparation of biocompatible carboxymethyl chitosan nanoparticles for delivery of antibiotic drug. BioMed Res. Int. 2013, 2013, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Abed, N.; Couvreur, P. Nanocarriers for antibiotics: A promising solution to treat intracellular bacterial infections. Int. J. Antimicrob. Agents 2014, 43, 485–496. [Google Scholar] [CrossRef] [PubMed]
- Shegokar, R.; Müller, R.H. Nanocrystals: Industrially feasible multifunctional formulation technology for poorly soluble actives. Int. J. Pharm. 2010, 399, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Mitri, K.; Shegokar, R.; Gohla, S.; Anselmi, C.; Müller, R.H. Lutein nanocrystals as antioxidant formulation for oral and dermal delivery. Int. J. Pharm. 2011, 420, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Georgieva, A.; Bogdanov, B.; Stefanov, Z.; Koleva, D. Microemulsion water-in-oil (W/O)—Microreactor for synthesis of ultrafine carbonate nanostructures. Univ. Ruse Union Sci.-Ruse 2011, 50, 34–38. [Google Scholar]
- Zaki, N.M.; Hafez, M.M. Enhanced antibacterial effect of ceftriaxone sodium-loaded chitosan nanoparticles against intracellular Salmonella typhimurium. AAPS Pharm. Sci. Technol. 2012, 13, 411–421. [Google Scholar] [CrossRef] [PubMed]
- Yaod, Z.R.G.W.; Sheng, B.; Yao, D.; Wei, Y.H. Pharmacopoeia of the People’s Republic of China/compiled by the State Pharmacopoeia Commission of the People’s Republic of China; Chemical Industry Press: Beijing, China, 2000. [Google Scholar]
- Islam, K.N.; Zuki, A.Z.; Ali, M.E.; Zobir, M.; Hussein, B.; Noordin, M.M. Facile synthesis of calcium carbonate nanoparticles from cockle shells. J. Nanomater. 2012, 2012, 1–5. [Google Scholar] [CrossRef]
- Cockerill, F.R.; Wikler, M.A.; Alder, J.; Dudley, M.N.; Eliopoulos, G.M.; Ferraro, M.J.; Hardy, D.J.; Hecht, D.W.; Hindler, J.A.; Patel, J.B.; et al. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically, Approved Standard–Ninth Edition; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2012. [Google Scholar]
- Roy, S.A. Effect of nano-titanium dioxide with different antibiotics against methicillin-resistants Staphylococcus aureus. J. Biomater. Nanobiotechnol. 2010, 1, 37–41. [Google Scholar] [CrossRef]




| Formulations | C-CSCCAN | Ciprofloxacin | CSCCAN |
|---|---|---|---|
| Tested Bact. | - | - | - |
| S. Typhimurium | 18.6 ± 0.5 | 11.7 ± 0.9 | N/I |
| Gene Name | IL-1β |
| Forward Primer Sequences | GCTGCTTCCAAACCTTTGAC |
| Reverse Primer Sequence | GCTTGTGCTCTGCTTGTGAG |
| Gene Name | β-Actin |
| Forward Primer Sequences | CATGAGGCTTATATCCTTGC |
| Reverse Primer Sequence | TAAAAGGCACTTTGTCCACT |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Isa, T.; Zakaria, Z.A.B.; Rukayadi, Y.; Mohd Hezmee, M.N.; Jaji, A.Z.; Imam, M.U.; Hammadi, N.I.; Mahmood, S.K. Antibacterial Activity of Ciprofloxacin-Encapsulated Cockle Shells Calcium Carbonate (Aragonite) Nanoparticles and Its Biocompatability in Macrophage J774A.1. Int. J. Mol. Sci. 2016, 17, 713. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms17050713
Isa T, Zakaria ZAB, Rukayadi Y, Mohd Hezmee MN, Jaji AZ, Imam MU, Hammadi NI, Mahmood SK. Antibacterial Activity of Ciprofloxacin-Encapsulated Cockle Shells Calcium Carbonate (Aragonite) Nanoparticles and Its Biocompatability in Macrophage J774A.1. International Journal of Molecular Sciences. 2016; 17(5):713. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms17050713
Chicago/Turabian StyleIsa, Tijani, Zuki Abu Bakar Zakaria, Yaya Rukayadi, Mohd Noor Mohd Hezmee, Alhaji Zubair Jaji, Mustapha Umar Imam, Nahidah Ibrahim Hammadi, and Saffanah Khuder Mahmood. 2016. "Antibacterial Activity of Ciprofloxacin-Encapsulated Cockle Shells Calcium Carbonate (Aragonite) Nanoparticles and Its Biocompatability in Macrophage J774A.1" International Journal of Molecular Sciences 17, no. 5: 713. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms17050713