Nutritional Strategies for the Individualized Treatment of Non-Alcoholic Fatty Liver Disease (NAFLD) Based on the Nutrient-Induced Insulin Output Ratio (NIOR)
Abstract
:1. Introduction
2. Results
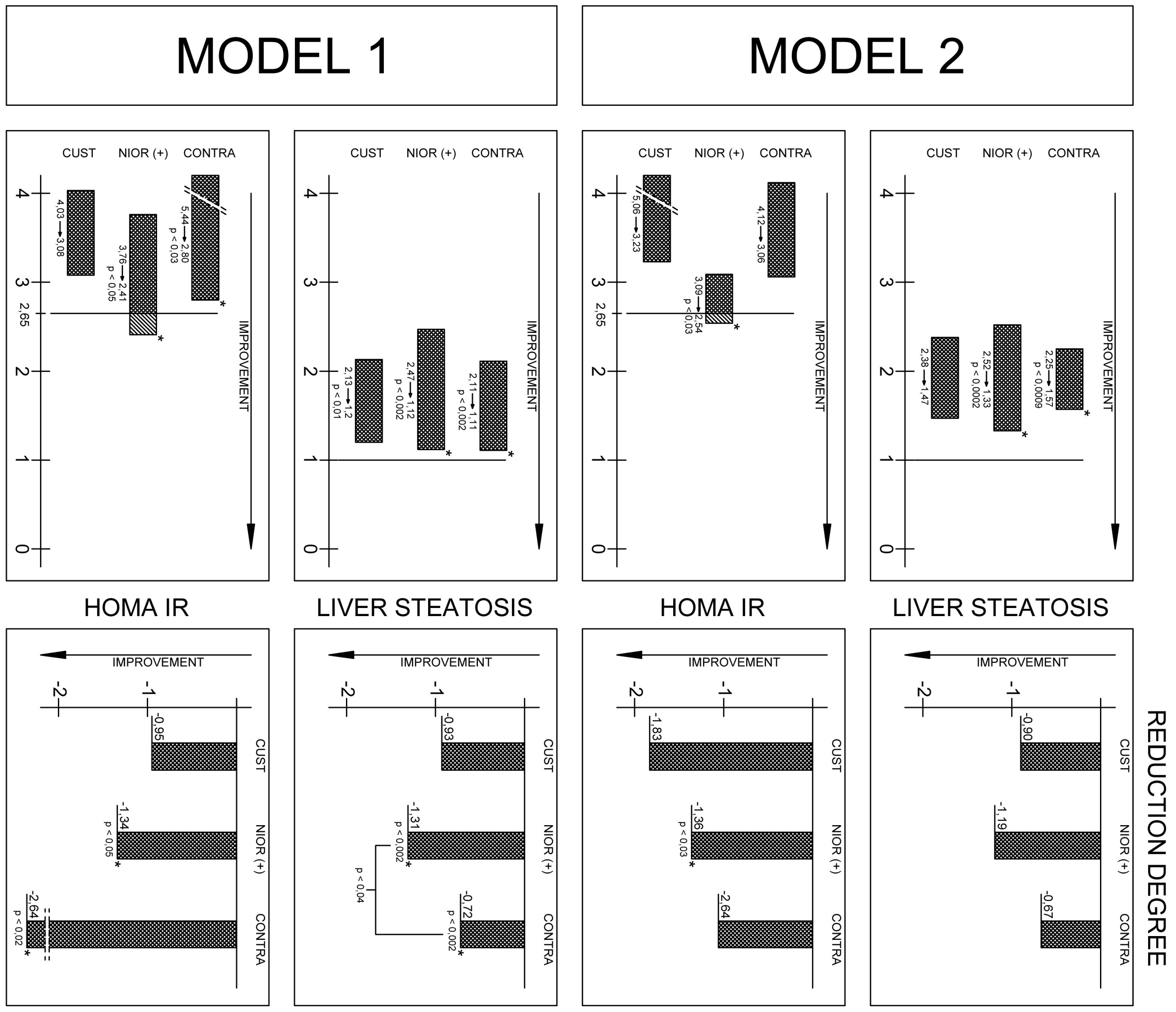
2.1. The Analysis of the Data Using Model 1
2.1.1. Changes in Anthropometric Parameters after Six Months Depending on the Type of Diet
2.1.2. Changes in Biochemical Parameters after Six Months in Model 1
2.1.3. A Significant Reduction in the Degree of Fatty Liver Disease Was Observed in Patients with a Diet Selected According to NIOR
2.2. The Data Analysis in Model 2
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. The Anthropometric Data
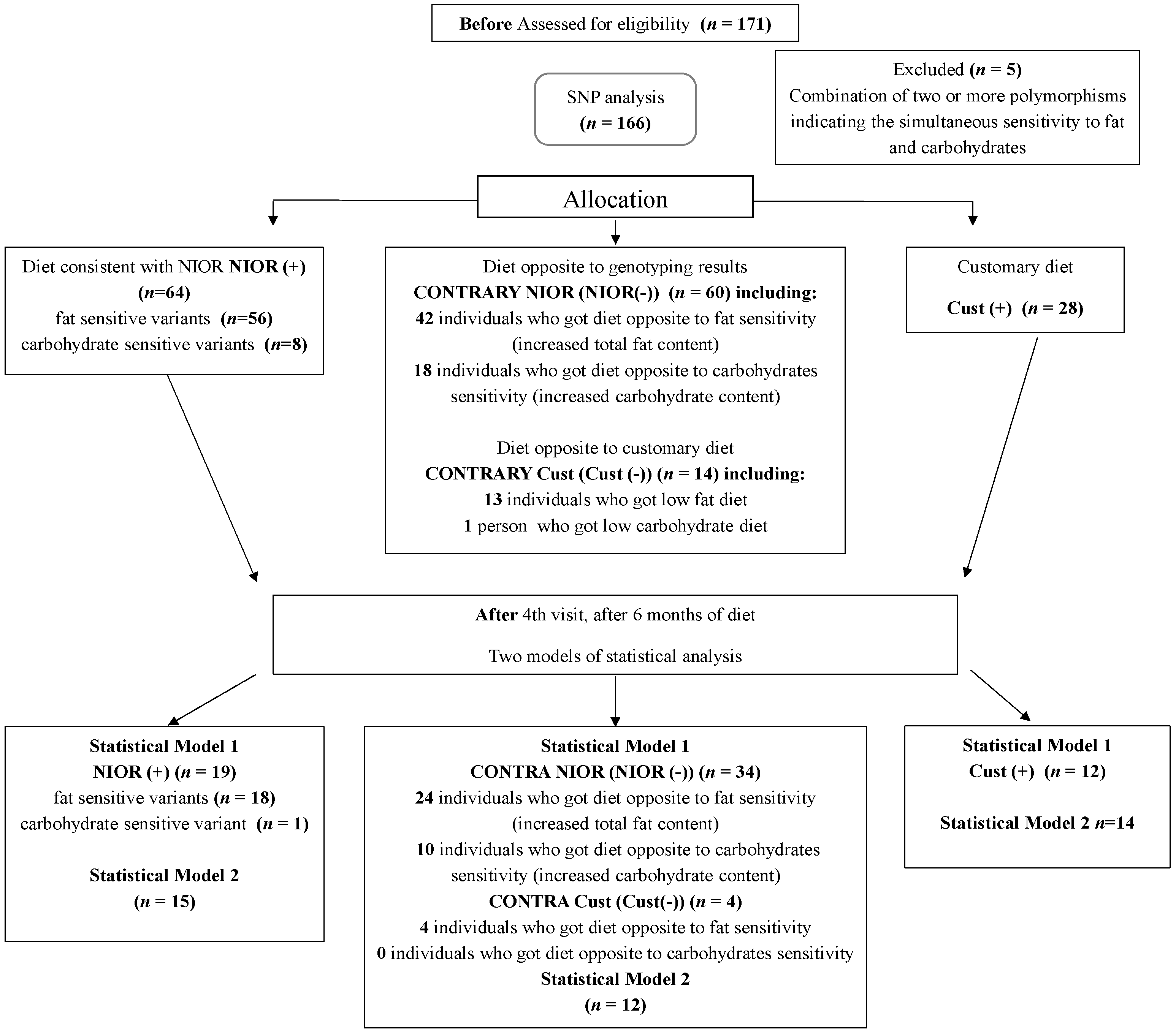
4.3. Methods and Experimental Design
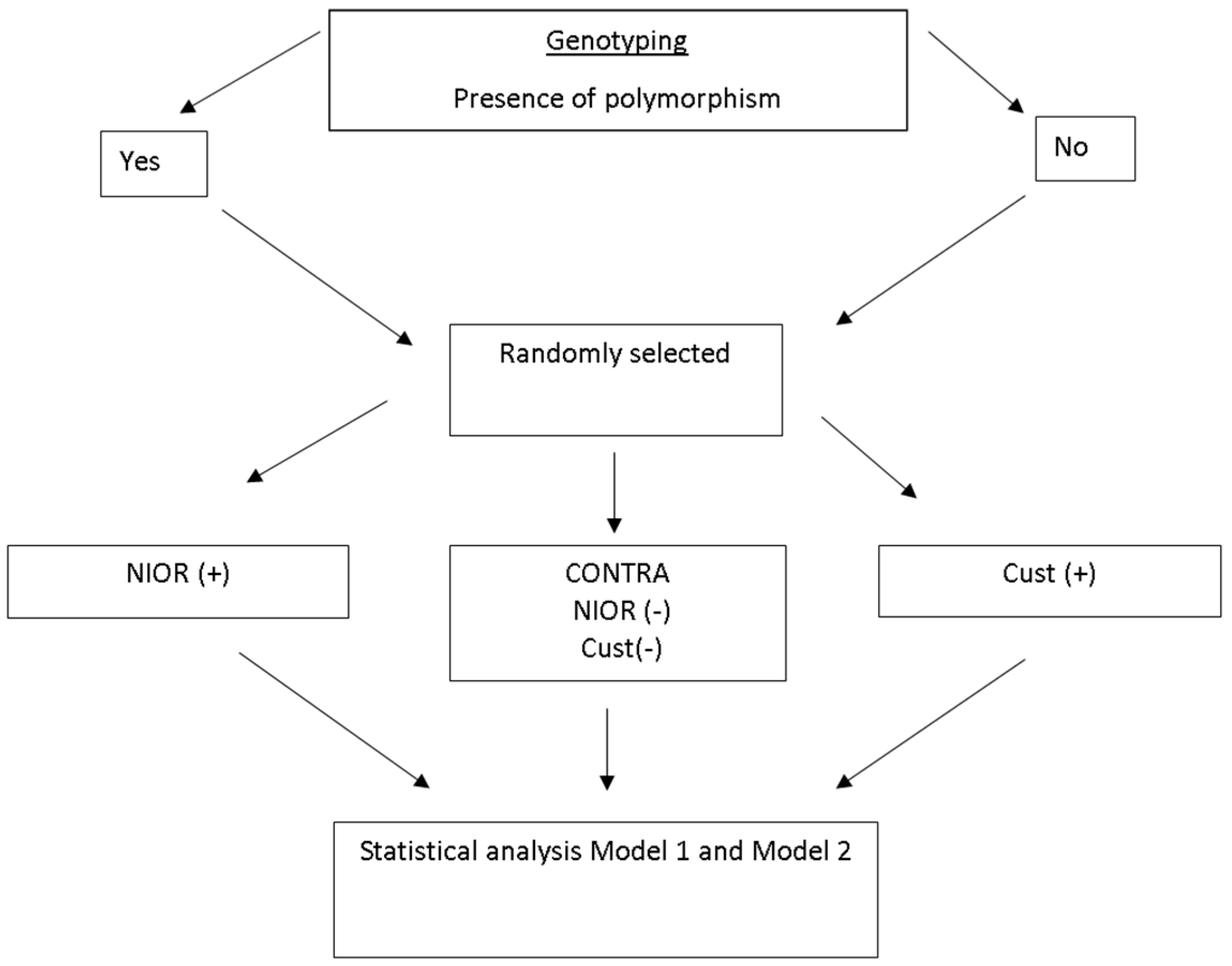
4.4. Allocation to Groups
- (a)
- a single polymorphism indicative of sensitivity to carbohydrates or fats
- (b)
- more than one polymorphism indicative of sensitivity to carbohydrates or to fats (e.g., two polymorphisms indicative of sensitivity to fat)
4.5. Dietary Intervention
4.5.1. General Recommendation
4.5.2. Recommendations Based on the Nutrient-Induced Insulin Output Ratio (NIOR)
- (a)
- NIOR (+) patients received dietary recommendations with a reduced fat content (20% TDEE when NIOR polymorphisms showed sensitivity to fat) or reduced carbohydrate content (55% of TDEE, including <5% of sugars, when the polymorphisms showed sensitivity to simple carbohydrates).
- (b)
- Cust (+) patients received dietary advice with the following nutrient content: fat content at 30% of TDEE and carbohydrates at 55% of TDEE (including 10% of simple carbohydrates).
- (c)
- NIOR (−) patients, when they had “fat-sensitive” gene variants, received dietary recommendations that increased total fat content up to 30% of TDEE.
4.5.3. Dietary Control
4.6. Laboratory Analyses
4.7. Genotyping
4.8. Statistical Analysis
Two Models of Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Vernon, G.; Baranova, A.; Younossi, Z.M. Systematic review: The epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment. Pharmacol. Ther. 2011, 34, 274–285. [Google Scholar] [CrossRef] [PubMed]
- Argo, C.K.; Northup, P.G.; Al-Osaimi, A.M.; Caldwell, S.H. Systematic review of risk factors for fibrosis progression in non-alcoholic steatohepatitis. J. Hepatol. 2009, 51, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Targher, G.; Day, C.P.; Bonora, E. Risk of cardiovascular disease in patients with nonalcoholic fatty liver disease. N. Engl. J. Med. 2010, 363, 1341–1350. [Google Scholar] [CrossRef] [PubMed]
- Promrat, K.; Kleiner, D.E.; Niemeier, H.M.; Jackvony, E.; Kearns, M.; Wands, JR.; Fava, J.L.; Wing, R.R. Randomized controlled trial testing the effects of weight loss on nonalcoholic steatohepatitis. Hepatology 2010, 51, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Yoon, H.J.; Cha, B.S. Pathogenesis and therapeutic approaches for non-alcoholic fatty liver disease. World J. Hepatol. 2014, 6, 800–811. [Google Scholar] [CrossRef] [PubMed]
- Hassan, K.; Bhalla, V.; El Regal, M.E.; A-Kader, H.H. Nonalcoholic fatty liver disease: A comprehensive review of a growing epidemic. World J. Gastroenterol. 2014, 20, 12082–120101. [Google Scholar] [CrossRef] [PubMed]
- Weiß, J.; Rau, M.; Geier, A. Non-alcoholic fatty liver disease: Epidemiology, clinical course, investigation, and treatment. Dtsch. Ärzteblatt Int. 2014, 111, 447–452. [Google Scholar]
- Kargulewicz, A.; Stankowiak-Kulpa, H.; Grzymisławski, M. Dietary recommendations for patients with nonalcoholic fatty liver disease. Prz. Gastroenterol. 2014, 9, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Barrera, F.; George, J. The role of diet and nutritional intervention for the management of patients with NAFLD. Clin. Liver. Dis. 2014, 18, 91–112. [Google Scholar] [CrossRef] [PubMed]
- Keijer, J.; Hoevenaars, F.P.; Nieuwenhuizen, A.; van Schothorst, E.M. Nutrigenomics of body weight regulation: A rationale for careful dissection of individual contributors. Nutrients 2014, 6, 4531–4551. [Google Scholar] [CrossRef] [PubMed]
- Arslan, N. Obesity, fatty liver disease and intestinal microbiota. World J. Gastroenterol. 2014, 20, 16452–16463. [Google Scholar] [CrossRef] [PubMed]
- Hesketh, J.; Wybranska, I.; Dommels, Y.; King, M.; Elliott, R.; Pico, C.; Keijer, J. Nutrient-gene interactions in benefit-risk analysis. Br. J. Nutr. 2006, 95, 1232–1239. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.X. The coming of age of nutrigenetics and nutrigenomics. J. Nutr. Nutr. 2012, 5. [Google Scholar] [CrossRef] [PubMed]
- Wybranska, I.; Malczewska-Malec, M.; Partyka, L.; Kiec-Wilk, B.; Kosno, K.; Leszczynska-Golabek, I.; Zdzienicka, A.; Gruca, A.; Kwasniak, M.; Dembinska-Kiec, A. Evaluation of genetic predisposition to insulin resistance by nutrient-induced insulin output ratio (NIOR). Clin. Chem. Lab. Med. 2007, 45, 1124–1132. [Google Scholar] [CrossRef] [PubMed]
- Zafarmand, M.H.; van der Schouw, Y.T.; Grobbee, D.E.; de Leeuw, P.W.; Bots, M.L. T64A polymorphism in β3-adrenergic receptor gene (ADRB3) and coronary heart disease: A case-cohort study and meta-analysis. J. Intern. Med. 2008, 263, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Miranda, J.L.; Perez-Martinez, P.P.; Marin, C.F.; Fuentes, F.; Delgado, J.; Pérez-Jiménez, F. Dietary fat, genes, and insulin sensitivity. J. Mol. Med. 2007, 85, 213–226. [Google Scholar] [CrossRef] [PubMed]
- Henneman, P.; van der Sman-de Beer, F.; Moghaddam, P.H.; Huijts, P.; Stalenhoef, AF.; Kastelein, J.J.; van Duijn, C.M.; Havekes, L.M.; Frants, R.R.; van Dijk, K.W.; et al. The expression of type III hyperlipoproteinemia: Involvement of lipolysis genes. Eur. J. Hum. Genet. 2009, 54, 3043–3048. [Google Scholar] [CrossRef] [PubMed]
- Hamada, T.; Kotani, K.; Nagai, N. Low-calorie diet-induced reduction in serum HDL cholesterol is ameliorated in obese women with the −3826 G allele in the uncoupling protein-1 gene. Tohoku J. Exp. Med. 2009, 219, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Meirhaeghem, A.; Cottel, D.; Amouyel, P.; Dallongeville, J. Association between peroxisome proliferator-activated receptor γ haplotypes and the metabolic syndrome in French men and women. Diabetes 2005, 54, 3043–3048. [Google Scholar] [CrossRef]
- Marchesini, G.; Pagotto, U.; Bugianesi, E.; De Iasio, R.; Manini, R.; Vanni, E.; Pasquali, R.; Melchionda, N.; Rizzetto, M. Low ghrelin concentrations in nonalcoholic fatty liver disease are related to insulin resistance. J. Clin. Endocrinol. Metab. 2003, 88, 5674–5679. [Google Scholar] [CrossRef] [PubMed]
- Pfützner, A.; Derwahl, M.; Jacob, S.; Hohberg, C.; Blümner, E.; Lehmann, U.; Fuchs, W.; Forst, T. Limitations of the HOMA-B score for assessment of β-cell functionality in interventional trials-results from the PIOglim study. Diabetes. Technol. Ther. 2010, 12, 599–604. [Google Scholar]
- Haffner, S.M.; Kennedy, E.; Gonzalez, C.; Stern, M.P.; Miettinen, H. A prospective analysis of the HOMA model. The Mexico City Diabetes Study. Diabetes Care 1996, 19, 1138–1141. [Google Scholar] [CrossRef] [PubMed]
- Eslamparast, T.; Eghtesad, S.; Poustchi, H.; Hekmatdoost, A. Recent advances in dietary supplementation, in treating non-alcoholic fatty liver disease. World J. Hepatol. 2015, 7, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Zheng, R.D.; Chen, Z.R.; Chen, J.N.; Lu, Y.H.; Chen, J. Role of body mass index, waist-to-height and waist-to-hip ratio in prediction of nonalcoholic fatty liver disease. Gastroenterol. Res. Pract. 2012, 2012, 362147. [Google Scholar] [CrossRef] [PubMed]
- Dudekula, A.; Rachakonda, V.; Shaik, B.; Behari, J. Weight loss in nonalcoholic Fatty liver disease patients in an ambulatory care setting is largely unsuccessful but correlates with frequency of clinic visits. PLoS ONE 2014, 9, e111808. [Google Scholar] [CrossRef]
- Deer, J.; Koska, J.; Ozias, M.; Reaven, P. Dietary models of insulin resistance. Metabolism 2015, 64, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Jia, Q.; Xia, Y.; Zhang, Q.; Wu, H.; Du, H.; Liu, L.; Wang, C.; Shi, H.; Guo, X.; Liu, X.; et al. Dietary patterns are associated with prevalence of fatty liver disease in adults. Eur. J. Clin. Nutr. 2015, 69, 914–921. [Google Scholar] [CrossRef] [PubMed]
- Adams, L.A. Biomarkers of liver fibrosis. J. Gastroenterol. Hepatol. 2011, 26, 802–809. [Google Scholar] [CrossRef] [PubMed]
- Rossi, E.; Adams, L.A.; Ching, H.L.; Bulsara, M.; MacQuillan, G.C.; Jeffrey, G.P. High biological variation of serum hyaluronic acid and Hepascore, a biochemical marker model for the prediction of liver fibrosis. Clin. Chem. Lab. Med. 2013, 51, 1107–1114. [Google Scholar] [CrossRef] [PubMed]
- Van Herpen, N.A.; Schrauwen-Hinderling, V.; Schaart, G.; Mensink, R.P.; Schrauwen, P. Three weeks on a high-fat diet increases intrahepatic lipid accumulation and decreases metabolic flexibility in healthy overweight men. JCEM 2011, 96, E691–E695. [Google Scholar] [CrossRef] [PubMed]
- Marina, A.; von Frankenberg, A.D.; Suvag, S.; Callahan, H.S.; Kratz, M.; Richards, T.L.; Utzschneider, K.M. Effects of dietary fat and saturated fat content on liver fat and markers of oxidative stress in overweight/obese men and women under weight-stable conditions. Nutrients 2014, 6, 4678–4690. [Google Scholar] [CrossRef] [PubMed]
- Westerbacka, J.; Lammi, K.; Hakkinen, A.M.; Rissanen, A.; Salminen, I.; Aro, A.; Yki-Järvinen, H. Dietary fat content modifies liver fat in overweight nondiabetic subjects. J. Clin. Endocrinol. Metab. 2005, 90, 2804–2809. [Google Scholar] [CrossRef] [PubMed]
- Moore, J.B.; Gunn, P.J.; Fielding, B.A. The role of dietary sugars and de novo lipogenesis in non-alcoholic fatty liver disease. Nutrients 2014, 6, 5679–5703. [Google Scholar] [CrossRef] [PubMed]
- Bémeur, C.; Butterworth, R.F. Nutrition in the management of cirrhosis and its neurological complications. J. Clin. Exp. Hepatol. 2014, 4, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Jin, R.; Welsh, J.A.; Le, N.A.; Holzberg, J.; Sharma, P.; Martin, D.R.; Vos, M.B. Dietary fructose reduction improves markers of cardiovascular disease risk in Hispanic-American adolescents with NAFLD. Nutrients 2014, 6, 3187–3201. [Google Scholar] [CrossRef] [PubMed]
- Chiu, S.; Sievenpiper, J.L.; de Souza, R.J.; Cozma, A.I.; Mirrahimi, A.; Carleton, A.J.; Ha, V.; di Buono, M.; Jenkins, A.L.; Leiter, L.A.; et al. Effect of fructose on markers of non-alcoholic fatty liver disease (NAFLD): A systematic review and meta-analysis of controlled feeding trials. Eur. J. Clin. Nutr. 2014, 68, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Chung, M.; Ma, J.; Patel, K.; Berger, S.; Lau, J.; Lichtenstein, A.H. Fructose, high-fructose corn syrup, sucrose, and nonalcoholic fatty liver disease or indexes of liver health: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2014, 100, 833–849. [Google Scholar] [CrossRef] [PubMed]
- Sluik, D.; Engelen, A.I.; Feskens, E.J. Fructose consumption in the Netherlands: The Dutch National Food Consumption Survey 2007–2010. Eur. J. Clin. Nutr. 2015, 69, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Veena, J.; Muragundla, A.; Sidgiddi, S.; Subramaniam, S. Non-alcoholic fatty liver disease: Need for a balanced nutritional source. Br. J. Nutr. 2014, 112, 1858–1872. [Google Scholar] [CrossRef] [PubMed]
- Rinella, M.E.; Sanyal, A.J. Management of NAFLD: A stage-based approach. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Hagströmer, M.; Oja, P.; Sjöström, M. The International Physical Activity Questionnaire (IPAQ): A study of concurrent and construct validity. Public Health Nutr. 2006, 9, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Hamaguchi, M.; Kojima, T.; Itoh, Y.; Harano, Y.; Fujii, K.; Nakajima, T.; Kato, T.; Takeda, N.; Okuda, J.; Ida, K.; et al. The severity of ultrasonographic findings in nonalcoholic fatty liver disease reflects the metabolic syndrome and visceral fat accumulation. Am. J. Gastroenterol. 2007, 102, 2708–2715. [Google Scholar] [CrossRef] [PubMed]
- Wells, J.C. Commentary: The paradox of body mass index in obesity assessment: Not a good index of adiposity, but not a bad index of cardio-metabolic risk. Int. J. Epidemiol. 2014, 43, 672–674. [Google Scholar] [CrossRef] [PubMed]
| Parameters | Baseline | 24W | p Value | ||||
|---|---|---|---|---|---|---|---|
| CUST (+) | NOR (+) | CONTRA CUST (−) and NOR (−) | CUST (+) | NOR (+) | CONTRA CUST (−) and NOR (−) | ||
| Age | 52.12 ± 14.74 | 52.80 ± 12.37 | 51.87 ± 12.11 | 52.12 ± 14.74 | 52.80 ± 12.37 | 51.87 ± 12.11 | |
| Body mass (kg) | 94.70 ± 22.55 a | 89.01 ± 15.26 a | 92.20 ± 19.34 b | 87.59 ± 17.96 a | 82.21 ± 15.35 a,# | 89.63 ± 20.79 b,# | a p < 0.0005 |
| b p < 0.005 | |||||||
| BMI (kg/m2) | 32.10 ± 4.13 | 30.70 ± 3.64 | 32.27 ± 6.59 | 30.01 ± 2.84 | 28.29 ± 15.35 # | 28.29 ± 15.35 # | # p < 0.015 |
| Arm circumference (cm) | 33.30 ± 3.64 a | 31.66 ± 2.87 | 32.33 ± 3.86 | 31.85 ± 2.56 a,& | 30.75 ± 3.46 | 33.38 ± 4.53 & | a p < 0.0005 |
| & p < 0.04 | |||||||
| Waist circumference (cm) | 105.14 ± 14.69 a | 100.12 ± 11.75 b | 106.25 ± 14.45 b | 97.02 ± 10.63 a | 94.00 ± 11.97 b | 102.60 ± 16.02 b | a p < 0.0009 |
| b p < 0.005 | |||||||
| Hip circumference (cm) | 105.14 ± 14.69 a | 100.12 ± 11.75 b | 106.25 ± 15.73 b | 104.20 ± 14.89 a | 94.00 ± 13.33 b | 102.60 ± 17.04 b | a p < 0.0009 |
| b p < 0.005 | |||||||
| Fat mass (%) | 34.71 ± 5.78 b | 35.41 ± 5.67 a | 36.02 ± 14.09 | 31.31 ± 5.78 b,* | 29.74 ± 8.21 a,*,# | 35.09 ± 14.94 # | a p < 0.0006 |
| b p < 0.002 | |||||||
| # p < 0.007 | |||||||
| * p < 0.04 | |||||||
| Fat content (%) | 37.00 ± 6.63 b | 35.61 ± 10.96 b | 38.07 ± 7.79 | 36.34 ± 5.75 b,* | 31.03 ± 13.46 b,*,# | 38.95 ± 7.90 # | b p < 0.005 |
| # p < 0.005 | |||||||
| * p < 0.04 | |||||||
| Lean mass (%) | 60.02 ± 16.47 b | 53.48 ± 10.96 | 55.94 ± 7.80 b | 55.92 ± 14.17 b | 51.03 ± 13.46 | 53.66 ± 9.28 b | b p < 0.002 |
| AST (U/L) | 36.10 ± 25.83 b | 30.70 ± 13.99 b | 32.71 ± 10.46 b | 35.10 ± 29.60 b | 34.20 ± 26.31 b,# | 23.85 ± 5.21 b,# | b p < 0.035 |
| # p < 0.041 | |||||||
| ALT (U/L) | 54.00 ± 36.86 b | 46.70 ± 26.56 a | 47.92 ± 15.85 a | 44.40 ± 27.86 b | 39.40 ± 33.04 a | 32.57 ± 18.40 a | a p < 0.0006 |
| a p < 0.038 | |||||||
| Trigliceride (mg/dL) | 129.30 ± 36.30 b | 123.80 ± 52.82 | 238.42 ± 482.09 | 106.40 ± 56.24 b | 121.80 ± 90.52 | 204.78 ± 319.17 | b p < 0.04 |
| HDL (mg/dL) | 51.00 ± 12.22 b | 54.40 ± 12.83 | 51.57 ± 15.73 | 54.00 ± 16.39 b | 54.30 ± 12.82 | 52.14 ± 14.31 | b p < 0.025 |
| Insulin (mcU/L) | 15.80 ± 9.05 | 14.59 ± 8.20 b | 18.54 ± 19.69 b | 12.07 ± 8.62 | 9.58 ± 6.81 b | 9.63 ± 9.46 b | b p < 0.05 |
| HOMA–IR | 4.03 ± 2.31 | 3.76 ± 1.94 b | 5.44 ± 6.65 b | 3.08 ± 2.08 | 2.41 ± 1.74 b | 2.80 ± 3.00 b | b p < 0.05 |
| Hyaluronic acid (U/L) | 54.56 ± 29.57 | 45.47 ± 25.82b | 50.23 ± 31.76 | 45.93 ± 22.62 | 32.56 ± 16.28 b | 37.42 ± 22.21 | b p < 0.04 |
| Fatty liver Hamaguchi score | 2.13 ± 0.74 b | 2.47 ± 0.94 b | 2.11 ± 0.98 b | 1.2 ± 1.0 b | 1.12 ± 1.08 b,# | 1.11 ± 0.92 b,# | b p < 0.01 |
| # p < 0.04 | |||||||
| Content of Diet | NIOR (+) Variant Sensitive for Fat | NIOR (+) Variant Sensitive for Carbohydrate | Cust (+) | CONTRA NIOR NIOR (−) If Variant Was Sensitive for Fat | CONTRA NIOR NIOR (−) If Variant Was Sensitive for Carbohydrate | CONTRA Cust Cust (−) Randomly Selected to Group with Lower Amount of Fat of CHO * |
|---|---|---|---|---|---|---|
| Energy | Calculated individually | Calculated individually | Calculated individually | Calculated individually | Calculated individually | Calculated individually |
| Fat percent of total caloric in % | 20 | 30 | 30 | 30 | 20 | 20 or 30 * |
| Carbohydrates in % | 65 | 55 | 55 | 55 | 65 | 65 or 55 * |
| Simple carbohydrate in % | ≥10 | <5 | ≥10 | <5 | ≥10 | ≥10 or <5 * |
| Protein (%) | 15 | 15 | 15 | 15 | 15 | 15 |
| Fiber (g/day) | 30–35 | 30–35 | 30–35 | 30–35 | 30–35 | 30–35 |
| Fluid ( mL/kg) | 35 | 35 | 35 | 35 | 35 | 35 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stachowska, E.; Ryterska, K.; Maciejewska, D.; Banaszczak, M.; Milkiewicz, P.; Milkiewicz, M.; Gutowska, I.; Ossowski, P.; Kaczorowska, M.; Jamioł-Milc, D.; et al. Nutritional Strategies for the Individualized Treatment of Non-Alcoholic Fatty Liver Disease (NAFLD) Based on the Nutrient-Induced Insulin Output Ratio (NIOR). Int. J. Mol. Sci. 2016, 17, 1192. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms17071192
Stachowska E, Ryterska K, Maciejewska D, Banaszczak M, Milkiewicz P, Milkiewicz M, Gutowska I, Ossowski P, Kaczorowska M, Jamioł-Milc D, et al. Nutritional Strategies for the Individualized Treatment of Non-Alcoholic Fatty Liver Disease (NAFLD) Based on the Nutrient-Induced Insulin Output Ratio (NIOR). International Journal of Molecular Sciences. 2016; 17(7):1192. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms17071192
Chicago/Turabian StyleStachowska, Ewa, Karina Ryterska, Dominika Maciejewska, Marcin Banaszczak, Piotr Milkiewicz, Małgorzata Milkiewicz, Izabela Gutowska, Piotr Ossowski, Małgorzata Kaczorowska, Dominika Jamioł-Milc, and et al. 2016. "Nutritional Strategies for the Individualized Treatment of Non-Alcoholic Fatty Liver Disease (NAFLD) Based on the Nutrient-Induced Insulin Output Ratio (NIOR)" International Journal of Molecular Sciences 17, no. 7: 1192. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms17071192







