Clock Genes and Altered Sleep–Wake Rhythms: Their Role in the Development of Psychiatric Disorders
Abstract
:1. Introduction: Circadian Rhythms and Their Molecular Mechanisms
1.1. Daily and Circadian Rhythms
1.2. Clock Genes
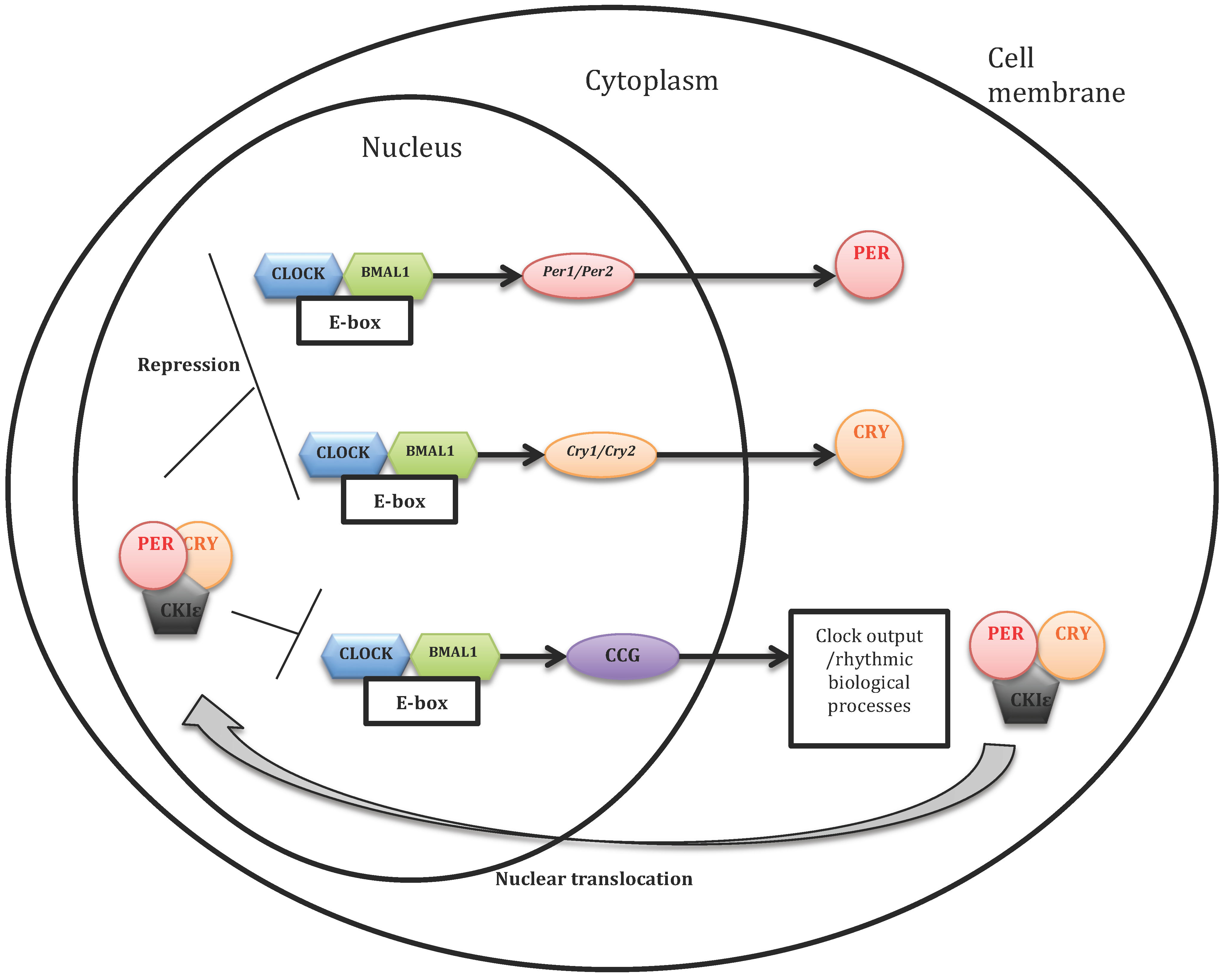
1.3. Molecular Working of the Cellular Circadian Clock
2. Relationships between Circadian Rhythms and Psychiatric Disorders
- (1)
- (2)
- Recent studies in cognitive and developmental psychology have highlighted the importance of rhythmicity and synchrony of motor, emotional, and inter-personal rhythms in early development of social communication; the synchronization of rhythms allows tuning and adaptation to the external environment [83]. Impaired circadian rhythms with an absence of synchronization of the circadian clocks network might alter the functioning of motor, emotional and interpersonal rhythms, leading to social communication impairments and vulnerability to psychiatric disorders with social communication deficit such as ASD or schizophrenia (for a review of literature on the importance of rhythmicity and synchrony of motor, emotional, and inter-personal rhythms in early development of social communication, see [83,84]). In addition, circadian rhythms involve sequences of continuities/discontinuities that might be important for typical fetal and child development in order to provide a secure environment (through stable and predictable regularities) but also variations allowing the individual to adapt to changes. Impaired circadian rhythms with no, little or irregular variability might lead to anxiety and difficulties in adapting to changes associated with restricted and repetitive interests observed in some psychiatric disorders described in this article such as ASD, schizophrenia and anxiety disorder;
- (3)
- Clock genes control critical periods of brain development [85] and therefore, abnormal expression of clock genes might participate to neurodevelopmental disorders such as psychiatric disorders. It is noteworthy that only a few days of circadian rhythm impairments may impact the maturation and specialization of some brain structures at specific developmental periods; these abnormalities can alter the temporal organization of brain maturation and development [86];
- (4)
- Circadian rhythm impairments may alter transcriptional and splicing regulation of Parvalbumin (PV) neurons, knowing that PV knockout mice (PV−/−) or heterozygous (PV+/−) mice showed autism behavioral phenotype [87]. More generally, circadian rhythm impairments may affect gene expression involved in synapse formation and brain maturation;
- (5)
- Also, clock-controlled genes (CCGs) may have pleiotropic effects outside the molecular clock and have therefore more widespread impact on cognition, mood, and reward-related behaviors [88];
- (6)
- Finally, circadian rhythm impairments (provoked or not by sleep problems) may also alter the adaptation of the individual to his/her environment and therefore his/her state of homeostasis. In this perspective, psychiatric disorders might reflect a loss of synchronization between the external environment’s rhythms and the individual’s internal rhythms, leading to major problems of adaptation for the individual and the appearance of psychiatric disorders. Single-nucleotide polymorphisms (SNPs) in core circadian clock genes have been associated with autism spectrum disorder [89], attention deficit hyperactivity disorder [90,91], anxiety disorder [92], major depressive disorder [93,94,95], bipolar disorder [95,96,97] and schizophrenia [98,99,100,101]. However, the causal relationship for these associations remains to be better ascertained. Circadian clock genes may affect specific aspects of psychiatric disorders through circadian control or through distinct regulation of downstream effectors.
2.1. Bipolar Spectrum Disorder
2.2. Anxiety and Depression-Related Disorders
2.2.1. Anxiety Disorder
2.2.2. Major Depressive Disorder (MDD)
2.2.3. Familial Advanced Sleep Phase Syndrome (FASPS)
2.2.4. Seasonal Affective Disorder (SAD) and Delayed Sleep Phase Syndrome (DSPS)
2.3. Attention Deficit Hyperactivity Disorder (ADHD)
2.4. Schizophrenia
2.5. Autism Spectrum Disorder (ASD)
3. Conclusions
Author Contributions
Conflicts of Interest
References
- Czeisler, C.A.; Duffy, J.F.; Shanahan, T.L.; Brown, E.N.; Mitchell, J.F.; Rimmer, D.W.; Ronda, J.M.; Silva, E.J.; Allan, J.S.; Emens, J.S.; et al. Stability, precision, and near-24-hour period of the human circadian pacemaker. Science 1999, 284, 2177–2181. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Zimmerman, A.W. Sleep in Autism Spectrum Disorder and Attention Deficit Hyperactivity Disorder. Semin. Pediatr. Neurol. 2015, 22, 113–125. [Google Scholar] [CrossRef] [PubMed]
- Lai, M.C.; Lombardo, M.V.; Baron-Cohen, S. Autism. Lancet 2014, 383, 896–910. [Google Scholar] [CrossRef]
- Kotagal, S.; Broomall, E. Sleep in children with autism spectrum disorder. Pediatr. Neurol. 2012, 47, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Onore, C.; Careaga, M.; Ashwood, P. The role of immune dysfunction in the pathophysiology of autism. Brain Behav. Immun. 2012, 26, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Kohyama, J. Possible neuronal mechanisms of sleep disturbances in patients with autism spectrum disorders and attention-deficit/hyperactivity disorder. Med. Hypotheses 2016, 97, 131–133. [Google Scholar] [CrossRef] [PubMed]
- Allik, H.; Larsson, J.O.; Smedje, H. Sleep patterns of school-age children with Asperger syndrome or high-functioning autism. J. Autism Dev. Disord. 2006, 36, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Richdale, A.L.; Schreck, K.A. Sleep problems in autism spectrum disorders: Prevalence, nature, and possible biopsychosocial aetiologies. Sleep Med. Rev. 2009, 13, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Polimeni, M.A.; Richdale, A.L.; Francis, A.J. A survey of sleep problems in autism, Asperger’s disorder and typically developing children. J. Intellect. Disabil. Res. 2005, 49, 260–268. [Google Scholar] [CrossRef] [PubMed]
- Doo, S.; Wing, Y.K. Sleep problems of children with pervasive developmental disorders: Correlation with parental stress. Dev. Med. Child. Neurol. 2006, 48, 650–655. [Google Scholar] [CrossRef] [PubMed]
- Giannotti, F.; Cortesi, F.; Cerquiglini, A. An investigation of sleep characteristics, electroencephalogram abnormalities and epilepsy in developmentally regressed and non-regressed children with autism. J. Autism Dev. Disord. 2008, 38, 1888–1897. [Google Scholar] [CrossRef] [PubMed]
- Stoleru, S.; Nottelmann, E.D.; Belmont, B.; Ronsaville, D. Sleep problems in children of affectively ill mothers. J. Child. Psychol. Psychiatry Allied Discip. 1997, 38, 831–841. [Google Scholar] [CrossRef]
- Brown, W.D. Insomnia: Prevalence and daytime consequences. In Sleep: A Comprehensive Handbook; Lee-Chiong, T., Ed.; John Wiley and Sons: Hoboken, NJ, USA, 2006; pp. 93–98. [Google Scholar]
- Ahmed, A.E.; Al-Jahdali, H.; Fatani, A.; Al-Rouqi, K.; Al-Jahdali, F.; Al-Harbi, A.; Baharoon, S.; Ali, Y.Z.; Khan, M.; Rumayyan, A. The effects of age and gender on the prevalence of insomnia in a sample of the Saudi population. Ethn. Health 2016, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Lane, J.M.; Vlasac, I.; Anderson, S.G.; Kyle, S.D.; Dixon, W.G.; Bechtold, D.A.; Gill, S.; Little, M.A.; Luik, A.; Loudon, A.; et al. Genome-wide association analysis identifies novel loci for chronotype in 100,420 individuals from the UK Biobank. Nat. Commun. 2016, 7, 10889. [Google Scholar] [CrossRef] [PubMed]
- Benedetti, F.; Dallaspezia, S.; Fulgosi, M.C.; Lorenzi, C.; Serretti, A.; Barbini, B.; Colombo, C.; Smeraldi, E. Actimetric evidence that CLOCK 3111 T/C SNP influences sleep and activity patterns in patients affected by bipolar depression. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2007, 144B, 631–635. [Google Scholar] [CrossRef] [PubMed]
- Serretti, A.; Benedetti, F.; Mandelli, L.; Lorenzi, C.; Pirovano, A.; Colombo, C.; Smeraldi, E. Genetic dissection of psychopathological symptoms insomnia in mood disorders and CLOCK gene polymorphism. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2003, 121B, 35–38. [Google Scholar] [CrossRef] [PubMed]
- Scott, A.J.; Monk, T.H.; Brink, L.L. Shiftwork as a Risk Factor for Depression: A Pilot Study. Int. J. Occup. Environ. Health 1997, 3, S2–S9. [Google Scholar] [PubMed]
- Bildt, C.; Michelsen, H. Gender differences in the effects from working conditions on mental health: A 4-year follow-up. Int. Arch. Occup. Environ. Health 2002, 75, 252–258. [Google Scholar] [CrossRef] [PubMed]
- Costa, G. Shift work and occupational medicine: An overview. Occup. Med. 2003, 53, 83–88. [Google Scholar] [CrossRef]
- Muecke, S. Effects of rotating night shifts: Literature review. J. Adv. Nurs. 2005, 50, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Bara, A.C.; Arber, S. Working shifts and mental health–findings from the British Household Panel Survey (1995–2005). Scand. J. Work Environ. Health 2009, 35, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Pallesen, S.; Bjorvatn, B.; Mageroy, N.; Saksvik, I.B.; Waage, S. Measures to counteract the negative effects of night work. Scand. J. Work Environ. Health 2010, 36, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Harma, M.; Kecklund, G. Shift work and health—How to proceed? Scand. J. Work Environ. Health 2010, 36, 81–84. [Google Scholar] [CrossRef] [PubMed]
- James, F.O.; Cermakian, N.; Boivin, D.B. Circadian rhythms of melatonin, cortisol, and clock gene expression during simulated night shift work. SLEEP 2007, 30, 11–1427. [Google Scholar] [CrossRef]
- Husse, J.; Hintze, S.C.; Eichele, G.; Lehnert, H.; Oster, H. Circadian Clock Genes Per1 and Per2 Regulate the Response of Metabolism-Associated Transcripts to Sleep Disruption. PLoS ONE 2012, 7, 12–52983. [Google Scholar] [CrossRef] [PubMed]
- Taniyama, Y.; yamauchi, T.; takeuchi, S.; Kuroda, Y. PER1 polymorphism associated with shift work disorder. Sleep Biol. Rhythm. 2015, 13, 342–347. [Google Scholar] [CrossRef]
- Duffy, J.F.; Kronauer, R.E.; Czeisler, C.A. Phase-shifting human circadian rhythms: Influence of sleep timing, social contact and light exposure. J. Physiol. 1996, 495, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Dawson, D.; Lack, L.; Morris, M. Phase resetting of the human circadian pacemaker with use of a single pulse of bright light. Chronobiol. Int. 1993, 10, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Honman, K.; Honman, S.; Nakamura, K.; Sasaki, M.; Endo, T.; Takahashi, T. Differential effects of bright light and social cues on reentrainment of human circadian rhythms. Am. J. Physiol. 1995, 268, R528–R535. [Google Scholar]
- Klerman, E.B.; Rimmer, D.W.; Dijk, D.J.; Kronauer, R.E.; Rizzo, J.F.I.; Czeisler, C.A. Nonphotic entrainment of the human circadian pacemaker. Am. J. Physiol. 1998, 274, R991. [Google Scholar] [PubMed]
- Reinberg, A.; Touitou, Y.; Restoin, A.; Migraine, C.; Levi, F.; Montagner, H. The genetic background of circadian and ultradian rhythm patterns of 17-hydroxycorticosteroids: A cross-twin study. J. Endocrinol. 1985, 105, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Moore, R.Y. Circadian rhythms: Basic neurobiology and clinical applications. Annu. Rev. Med. 1997, 48, 253–266. [Google Scholar] [CrossRef] [PubMed]
- Reppert, S.M.; Weaver, D.R. Coordination of circadian timing in mammals. Nature 2002, 418, 935–941. [Google Scholar] [CrossRef] [PubMed]
- Johnson, C.H. Testing the Adaptive Value of Circadian Systems. Methods Enzymol. 2005, 393, 818–837. [Google Scholar] [PubMed]
- Pevet, P.; Challet, E. Melatonin: Both master clock output and internal time-giver in the circadian clock network. J. Physiol. Paris 2011, 105, 170–182. [Google Scholar] [CrossRef] [PubMed]
- Konopka, R.J.; Benzer, S. Clock mutants of Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 1971, 58, 2112–2116. [Google Scholar] [CrossRef]
- Edery, I.; Rutila, J.E.; Rosbash, M. Phase shifting of the circadian clock by induction of the Drosophila period protein. Science 1994, 263, 237–240. [Google Scholar] [CrossRef] [PubMed]
- Sehgal, A.; Price, J.L.; Man, B.; Young, M.W. Loss of circadian behavioral rhythms and per RNA oscillations in the Drosophila mutant timeless. Science 1994, 263, 1603–1606. [Google Scholar] [CrossRef] [PubMed]
- Gekakis, N.; Saez, L.; Delahaye-Brown, A.M.; Myers, M.P.; Sehgal, A.; Young, M.W.; Weitz, C.J. Isolation of timeless by PER protein interaction: Defection interaction between timeless protein and long-period mutant per1. Science 1995, 270, 811–815. [Google Scholar] [CrossRef] [PubMed]
- Myers, M.; Wager-Smith, K.; Rothenfluh-Hilfiker, A.; Young, M. Light-induced degradation of TIMELESS and entrainment of the Drosophila circadian clock. Science 1996, 271, 1736–1740. [Google Scholar] [CrossRef] [PubMed]
- Vitaterna, M.H.; King, D.P.; Chang, A.M.; Kornhauser, J.M.; Lowrey, J.D. Mutagenesis and mapping of a mouse gene, Clock, essential for circadian behavior. Science 1994, 264, 719–725. [Google Scholar] [CrossRef] [PubMed]
- Pevet, P.; Bothorel, B.; Slotten, H.; Saboureau, M. The chronobiotic properties of melatonin. Cell. Tissue Res. 2002, 309, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Slotten, H.A.; Krekling, S.; Sicard, B.; Pévet, P. Daily infusion of melatonin entrains circadian activity rhythms in the diurnal rodent Arvicanthis ansorgei. Behav. Brain Res. 2002, 133, 11–19. [Google Scholar] [CrossRef]
- Slotten, H.A.; Pitrosky, B.; Krekling, S.; Pévet, P. Entrainment of circadian activity rhythms in rats to melatonin administered at T cycles different from 24 hours. Neurosignals 2002, 11, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Johnston, J.D.; Messager, S.; Barrett, P.; Hazlerigg, D.G. Melatonin action in the pituitary: Neuroendocrine synchronizer and developmental modulator? J. Neuroendocrinol. 2003, 15, 405–408. [Google Scholar] [CrossRef] [PubMed]
- Dardente, H.; Menet, J.S.; Poirel, V.J.; Streincher, D.; Gauer, F.; Vivien-Roels, B.; Klosen, P.; Pévet, P.; Masson-Mévet, M. Melatonin induces Cry1 expression in the pars tuberalis of the rat. Mol. Brain Res. 2003, 114, 101–106. [Google Scholar] [CrossRef]
- Von Gall, C.; Weaver, D.R.; Moek, J.; Jilg, A.; Stehle, J.H.; Korf, H.W. Melatonin plays a crucial role in the regulation of rhythmic clock gene expression in the mouse pars tuberalis. Ann. N. Y. Acad. Sci. 2005, 1040, 508–511. [Google Scholar] [CrossRef] [PubMed]
- Stehle, J.H.; von Gall, C.; Korf, H.W. Organisation of the circadian system in melatonin- proficient C3H and melatonin-deficient C57BL mice: A comparative investigation. Cell. Tissue Res. 2002, 309, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Von Gall, C.; Garabette, M.L.; Kell, C.A.; Frenzel, S.; Dehghani, F.; Schumm-Draeger, P.M.; Weaver, D.R.; Korf, H.W.; Hastings, M.H.; Stehle, J.H. Rhythmic gene expression in pituitary depends on heterologous sensitization by the neurohormone melatonin. Nat. Neurosci. 2002, 5, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Messager, S.; Garabette, M.L.; Hastings, M.H.; Hazlerigg, D.G. Tissue-specific abolition of Per1 expression in the pars tuberalis by pinealectomy in the Syrian hamster. Neuroreport 2001, 12, 579–582. [Google Scholar] [CrossRef] [PubMed]
- Agez, L.; Laurent, V.; Guerrero, H.Y.; Pévet, P.; Masson-Pévet, M.; Gauer, F. Endogenous melatonin provides an effective circadian message to both the suprachiasmatic nuclei and the pars tuberalis of the rat. J. Pineal Res. 2009, 46, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Zeman, M.; Herichova, I. Melatonin and clock genes expression in the cardiovascular system. Front. Biosci. 2013, 5, 743–753. [Google Scholar] [CrossRef]
- Johnston, J.D.; Tournier, B.B.; Andersson, H.; Masson-Pevet, M.; Lincoln, G.A.; Hazlerigg, D.G. Multiple effects of melatonin on rhythmic clock gene expression in the mammalian pars tuberalis. Endocrinology 2006, 147, 959–965. [Google Scholar] [CrossRef] [PubMed]
- Agez, L.; Laurent, V.; Pevet, P.; Gauer, F. Melatonin affects nuclear orphan receptors mRNA in the rat suprachiasmatic nuclei. Neuroscience 2007, 144, 522–530. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Vale, M.I.; Andreotti, S.; Mukai, P.Y.; Borges-Silva, C.D.; Peres, S.B.; Cipolla-Neto, J.; Lima, F.B. Melatonin and the circadian entrainment of metabolic and hormonal activities in primary isolated adipocytes. J. Pineal Res. 2008, 45, 422–429. [Google Scholar] [CrossRef] [PubMed]
- Kennaway, D.J.; Owens, J.A.; Voultsios, A.; Wight, N. Adipokines and adipocyte function in Clock mutant mice that retain melatonin rhythmicity. Obesity 2012, 20, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Delezie, J.; Dumont, S.; Dardente, H.; Oudart, H.; Gréchez-Cassiau, A.; Klosen, P.; Teboul, M.; Delaunay, F.; Pévet, P.; Challet, E. The nuclear receptor REV-ERBα is required for the daily balance of carbohydrate and lipid metabolism. FASEB J. 2012, 26, 3321–3335. [Google Scholar] [CrossRef] [PubMed]
- Xiang, S.; Mao, L.; Duplessis, T.; Yuan, L.; Dauchy, R.; Dauchy, E.; Blask, D.E.; Frasch, T.; Hill, S.M. Oscillation of clock and clock controlled genes induced by serum shock in human breast epithelial and breast cancer cells: Regulation by melatonin. Breast Cancer Epub. 2012, 6, 137–150. [Google Scholar]
- Torres-Farfan, C.; Mendez, N.; Abarzua-Catalan, L.; Vilches, N.; Valenzuela, G.J.; Seron-Ferre, M. A circadian clock entrained by melatonin is ticking in the rat fetal adrenal. Endocrinology 2011, 152, 1891–1900. [Google Scholar] [CrossRef] [PubMed]
- Steeves, T.D.; King, D.P.; Zhao, Y.; Sangoram, A.M.; Du, F.; Bowcock, A.M.; Moore, R.Y.; Takahashi, J.S. Molecular cloning and characterization of the human CLOCK gene: Expression in the suprachiasmatic nuclei. Genomics 1999, 57, 189–200. [Google Scholar] [CrossRef] [PubMed]
- Wisor, J.P.; Pasumarthi, R.; Gerashchenko, D.; Thompson, C.; Pathak, S.; Sancar, A.; Kilduff, T.S. Sleep Deprivation Effects on Circadian Clock Gene Expression in the Cerebral Cortex Parallel Electroencephalographic Differences Among Mouse Strains. J. Neurosci. 2008, 28, 28–7193. [Google Scholar] [CrossRef] [PubMed]
- Cedernaes, J.; Osler, M.E.; Voisin, S.; Broman, J.; Vogel, H.; Dickson, S.L.; Zierath, J.R.; Schiöth, H.B.; Benedict, C. Acute sleep loss induces tissue-specific epigenetic and transcriptional alterations to circadian clock genes in men. J. Clin. Endocrinol. Metab. 2015, 100, 9. [Google Scholar] [CrossRef] [PubMed]
- Franken, P.; Thomason, R.; Heller, H.C.; O’Hara, B.F. A non-circadian role for clock-genes in sleep homeostasis: A strain comparison. BMC Neurosci. 2007, 8, 87. [Google Scholar] [CrossRef] [PubMed]
- Franken, P.; Dijk, D.J. Circadian clock genes and sleep homeostasis. Eur. J. Neurosci. 2009, 29, 1820–1829. [Google Scholar] [CrossRef] [PubMed]
- Panda, S.; Antoch, M.P.; Miller, B.H.; Su, A.I.; Schook, A.B.; Straume, M.; Schultz, P.G.; Kay, S.A.; Takahashi, J.S.; Hogenesch, J.B. Coordinated transcription of key pathways in the mouse by the circadian clock. Cell 2002, 109, 307–320. [Google Scholar] [CrossRef]
- Hardin, P.E. Activating inhibitors and inhibiting activators: A day in the life of a fly. Curr. Opin. Neurobiol. 1998, 8, 642–647. [Google Scholar] [CrossRef]
- Dunlap, J.C. Molecular bases for circadian clocks. Cell 1999, 96, 271–290. [Google Scholar] [CrossRef]
- Lowrey, P.L.; Takahashi, J.S. Genetics of Circadian Rhythms in Mammalian Model Organisms. Adv. Genet. 2011, 74, 175–230. [Google Scholar] [PubMed]
- Lee, C.; Etchegaray, J.P.; Cagampang, F.R.; Loudon, A.S.; Reppert, S.M. Posttranslational mechanisms regulate the mammalian circadian clock. Cell 2001, 107, 855–867. [Google Scholar] [CrossRef]
- Kume, K.; Zylka, M.J.; Sriram, S.; Shearman, L.P.; Weaver, D.R. mCRY1 and mCRY2 are essential components of the negative limb of the circadian clock feedback loop. Cell 1999, 98, 193–205. [Google Scholar] [CrossRef]
- Shearman, L.P.; Sriram, S.; Weaver, D.R.; Maywood, E.S.; Chaves, I. Interacting molecular loops in the mammalian circadian clock. Science 2000, 288, 1013–1019. [Google Scholar] [CrossRef] [PubMed]
- Marcheva, B.; Ramsey, K.M.; Buhr, E.D.; Kobayashi, Y.; Su, H.; Ko, C.H.; Bass, J. Disruption of the clock components CLOCK and BMAL1 leads to hypoinsulinaemia and diabetes. Nature 2010, 466, 627–631. [Google Scholar] [CrossRef] [PubMed]
- Janich, P.; Pascual, G.; Merlos-Suárez, A. The circadian molecular clock creates epidermal stem cell heterogeneity. Nature 2011, 480, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Paschos, G.K.; Ibrahim, S.; Song, W.; Kunieda, T.; Grant, G.; Reyes, T.M.; Fitzgerald, G.A. Obesity in mice with adipocyte-specific deletion of clock component Arntl. Nat. Med. 2012, 18, 12–1768. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, J.S.; Hong, H.K.; Ko, C.H.; McDearmon, E.L. The genetics of mammalian circadian order and disorder: Implications for physiology and disease. Nat. Rev. Genet. 2008, 9, 764–775. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.; Van Dyke, R.D.; Zhang, J.; Li, F.; Gozal, D.; Shen, X. Effect of chronic sleep restriction on sleepiness and working memory in adolescents and young adults. J. Clin. Exp. Neuropsychol. 2011, 33, 892–900. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Grone, B.; Colas, D.; Appelbaum, L.; Mourrain, P. Synaptic plasticity in sleep: Learning, homeostasis and disease. Trends Neurosci. 2011, 34, 452–463. [Google Scholar] [CrossRef] [PubMed]
- Berger, R.H.; Miller, A.L.; Seifer, R.; Cares, S.R.; Le Bourgeois, M.K. Acute sleep restriction effects on emotion responses in 30- to 36-month-old children. J. Sleep Res. 2012, 21, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Deliens, G.; Gilson, M.; Peigneux, P. Sleep and the processing of emotions. Exp. Brain Res. 2014, 232, 5–1403. [Google Scholar] [CrossRef] [PubMed]
- Coogan, A.N.; Baird, A.L.; Popa-Wagner, A.; Thome, J. Circadian rhythms and attention deficit hyperactivity disorder: The what, the when and the why. Prog. Neuropsychopharmacol. Biol. Psychiatry. 2016, 67, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Seegers, V.; Touchette, E.; Dionne, G.; Petit, D.; Seguin, J.R.; Montplaisir, J.; Vitaro, F.; Falissard, B.; Boivin, M.; Tremblay, R.E. Short persistent sleep duration is associated with poor receptive vocabulary performance in middle childhood. J. Sleep Res. 2016, 25, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Tordjman, S.; Davlantis, K.S.; Georgieff, N.; Geoffray, M.M.; Speranza, M.; Anderson, G.M.; Xavier, J.; Botbol, M.; Oriol, C.; Bellissant, E.; et al. Autism as a disorder of biological and behavioral rhythms: Toward new therapeutic perspectives. Front. Pediatr. 2015, 3, 1. [Google Scholar] [CrossRef] [PubMed]
- Tordjman, S.; Najjar, I.; Bellissant, E.; Anderson, G.M.; Barburoth, M.; Cohen, D.; Jaafari, N.; Schischmanoff, O.; Fagard, O.; Lagdas, I.; et al. Advances in the research of melatonin in autism spectrum disorders: Literature review and new perspectives. Int. J. Mol. Sci. 2013, 14, 20508–20542. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y.; Ye, Z.; Hensch, T.K. Clock genes control cortical critical period timing. Neuron 2015, 86, 264–275. [Google Scholar] [CrossRef] [PubMed]
- Geoffray, M.M.; Nicolas, A.; Speranza, M.; Georgieff, N. Are circadian rhythms new pathways to understand Autism spectrum disorder? J. Neuropsychol 2016, in press. [Google Scholar]
- Wöhr, M.; Orduz, D.; Gregory, P.; Moreno, H.; Khan, U.; Vörckel, K.J.; Wolfer, D.P.; Welzl, H.; Gall, D.; Schiffmann, S.N.; et al. Lack of parvalbumin in mice leads to behavioral deficits relevant to all human autism core symptoms and related neural. Transl. Psychiatry 2015, 5, 525. [Google Scholar] [CrossRef] [PubMed]
- Wulff, K.; Dijk, D.J.; Middleton, B.; Foster, R.G.; Joyce, E.M. Sleep and circadian rhythm disruption in schizophrenia. Br. J. Psychiatry J. Ment. Sci. 2012, 200, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Nicholas, B.; Rudrasingham, V.; Nash, S.; Kirov, G.; Owen, M.J.; Wimpory, D.C. Association of Per1 and Npas2 with autistic disorder: Support for the clock genes/social timing hypothesis. Mol. Psychiatry 2007, 12, 581–592. [Google Scholar] [CrossRef] [PubMed]
- Kissling, C.; Retz, W.; Wiemann, S.; Coogan, AN.; Clement, RM.; Hunnerkopf, R.; Conner, A.C.; Freitag, C.M.; Rösler, M.; Thome, J. A polymorphism at the 3′-untranslated region of the CLOCK gene is associated with adult attention-deficit hyperactivity disorder. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2008, 147, 333–338. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Breen, G.; Chen, C.K.; Huang, Y.S.; Wu, Y.Y.; Asherson, P. Association study between a polymorphism at the 3′-untranslated region of CLOCK gene and attention deficit hyperactivity disorder. Behav. Brain Funct. 2010, 6, 48. [Google Scholar] [CrossRef] [PubMed]
- Sipila, T.; Kananen, L.; Greco, D.; Donner, J.; Silander, K.; Terwilliger, J.D. An association analysis of circadian genes in anxiety disorders. Biol. Psychiatry 2010, 67, 1163–1170. [Google Scholar] [CrossRef] [PubMed]
- Partonen, T.; Treutlein, J.; Alpman, A.; Frank, J.; Johansson, C.; Depner, M.; Aron, L.; Rietschel, M.; Wellek, S.; Soronen, P. Three circadian clock genes Per2, Arntl, and Npas2 contribute to winter depression. Ann. Med. 2007, 39, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Lavebratt, C.; Sjöholm, L.K.; Soronen, P.; Paunio, T.; Vawter, M.P.; Bunney, W.E.; Adolfsson, R.; Forsell, Y.; Wu, J.C.; Kelsoe, J.R.; et al. CRY2 Is Associated with Depression. PLoS ONE 2010, 5, e9407. [Google Scholar] [CrossRef] [PubMed]
- Soria, V.; Martinez-Amoros, E.; Escaramis, G.; Valero, J.; Perez-Egea, R.; Garcia, C. Differential association of circadian genes with mood disorders: CRY1 and NPAS2 are associated with unipolar major depression and CLOCK and VIP with bipolar disorder. Neuropsychopharmacology 2010, 35, 1279–1289. [Google Scholar] [CrossRef] [PubMed]
- Nievergelt, C.M.; Kripke, D.F.; Barrett, T.B.; Burg, E.; Remick, R.A.; Sadovnick, A.D. Suggestive evidence for association of the circadian genes PERIOD3 and ARNTL with bipolar disorder. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2006, 141B, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Wittke-Thompson, J.K.; Badner, J.A.; Hattori, E.; Potash, J.B.; Willour, V.L. Clock genes may influence bipolar disorder susceptibility and dysfunctional circadian rhythm. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2008, 147B, 1047–1055. [Google Scholar] [CrossRef] [PubMed]
- Aston, C.; Jiang, L.; Sokolov, B.P. Microarray analysis of postmortem temporal cortex from patients with schizophrenia. J. Neurosci. Res. 2004, 77, 858–866. [Google Scholar] [CrossRef] [PubMed]
- Mansour, H.A.; Wood, J.; Logue, T.; Chowdari, K.V.; Dayal, M.; Kupfer, D.J.; Monk, T.H.; Devlin, B.; Nimgaonkar, V.L. Association study of eight circadian genes with bipolar I disorder, schizoaffective disorder and schizophrenia. Genes Brain Behav. 2006, 5, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.Q.; Li, S.X.; Chen, F.B.; Zhang, Y.; Li, P.; Jin, M.; Sun, Y.; Wang, F.; Mi, W.F.; Shi, L.; et al. Diurnal neurobiological alterations after exposure to clozapine in first-episode schizophrenia patients. Psychoneuroendocrinology 2016, 64, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Johansson, A.S.; Owe-Larsson, B.; Hetta, J.; Lundkvist, G.B. Altered circadian clock gene expression in patients with schizophrenia. Schizophr. Res. 2016, 174, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Buhr, E.D.; Takahashi, J.S. Molecular components of the Mammalian circadian clock. Handb. Exp. Pharmacol. 2013, 217, 3–27. [Google Scholar]
- Oster, H.; Damerow, S.; Kiessling, S.; Jakubcakova, V.; Abraham, D.; Tian, J.; Hoffmann, M.W.; Eichele, G. The circadian rhythm of glucocorticoids is regulated by a gating mechanism residing in the adrenal cortical clock. Cell. Metab. 2006, 4, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Son, G.H.; Chung, S.; Choe, H.K.; Kim, H.D.; Baik, S.M.; Lee, H.; Lee, H.W.; Choi, S.; Sun, W.; Kim, H.; et al. Adrenal peripheral clock controls the autonomous circadian rhythm of glucocorticoid by causing rhythmic steroid production. Proc. Natl. Acad. Sci. USA 2008, 105, 20970–20975. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T.; Nakahata, Y.; Tanaka, M.; Yoshida, M.; Soma, H.; Shinohara, K.; Yasuda, A.; Mamine, T.; Takumi, T. Acute physical stress elevates mouse PERIOD1 mRNA expression in mouse peripheral tissues via a glucocorticoid-responsive element. J. Biol. Chem. 2005, 280, 42036–42043. [Google Scholar] [CrossRef] [PubMed]
- Dulcis, D.; Jamshidi, P.; Leutgeb, S.; Spitzer, N.C. Neurotransmitter switching in the adult brain regulates behavior. Science 2013, 340, 449–453. [Google Scholar] [CrossRef] [PubMed]
- Jackson, A.; Cavanagh, J.; Scott, J. A systematic review of manic and depressive prodromes. J. Affect. Disord. 2003, 74, 209–217. [Google Scholar] [CrossRef]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2015. [Google Scholar]
- Millar, A.; Espie, C.A.; Scott, J. The sleep of remitted bipolar outpatients: A controlled naturalistic study using actigraphy. J. Affect. Disord. 2004, 80, 145–153. [Google Scholar] [CrossRef]
- Mansour, H.A.; Wood, J.; Chowdari, K.V.; Dayal, M.; Thase, M.E.; Kupfer, D.J.; Monk, T.H.; Devlin, B.; Nimgaonkar, V.L. Circadian phase variation in bipolar I disorder. Chronobiol. Int. 2005, 22, 571–584. [Google Scholar] [CrossRef] [PubMed]
- McClung, C.A. Role for the Clock gene in bipolar disorder. Cold Spring Harb. Symp. Quant. Biol. 2007, 72, 637–644. [Google Scholar] [CrossRef] [PubMed]
- Klemfuss, H. Rhythms and the pharmacology of lithium. Pharmacol. Ther. 1992, 56, 53–78. [Google Scholar] [CrossRef]
- Abe, M.; Herzog, E.D.; Block, G.D. Lithium lengthens the circadian period of individual suprachiasmatic nucleus neurons. Neuroreport 2000, 11, 3261–3264. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Wang, J.; Klein, P.S.; Lazar, M.A. Nuclear receptor REV-ERBα is a critical lithium-sensitive component of the circadian clock. Science 2006, 311, 1002–1005. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Lu, W.Q.; Beesley, S.; Loudon, A.S.; Meng, Q.J. Lithium impacts on the amplitude and period of the molecular circadian clockwork. PLoS ONE 2012, 7, 33292. [Google Scholar] [CrossRef] [PubMed]
- Kaladchibachi, S.A.; Doble, B.; Anthopoulos, N.; Woodgett, J.R.; Manoukian, A.S. Glycogen synthase kinase 3, circadian rythms, and bipolar disorder: A molecular link in the therapeutic action of lithium. J. Circadian Rythm. 2007, 5, 3. [Google Scholar] [CrossRef] [PubMed]
- Gould, T.D.; Manji, H.K. Glycogen synthase kinase-3: A putative molecular target for lithium mimetic drugs. Neuropsychopharmacology 2005, 30, 1223–1237. [Google Scholar] [CrossRef] [PubMed]
- Iitaka, C.; Miyazaki, K.; Akaike, T.; Ishida, N. A role for glycogen synthase kinase-3β in the mammalian circadian clock. J. Biol. Chem. 2005, 280, 29397–29402. [Google Scholar] [CrossRef] [PubMed]
- Serretti, A.; Cusin, C.; Benedetti, F.; Mandelli, L.; Pirovano, A.; Zanardi, R.; Colombo, C.; Smeraldi, E. Insomnia improvement during antidepressant treatment and CLOCK gene polymorphism. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2005, 137B, 36–39. [Google Scholar] [CrossRef] [PubMed]
- Karthikeyan, K.; Marimuthu, G.; Ramasubramanian, C.; Arunachal, G.; Bahammam, A.; Spence, D.W.; Cardinali, D.P.; Brown, G.M.; Pandi-Perumal, S.R. Association of Per3 length polymorphism with bipolar disorder and schizophrenia. Neuropsychiatr. Dis. Treat. 2014, 10, 2325–2330. [Google Scholar] [PubMed]
- Sjöholm, L.; Backlund, L.; Cheteh, E.H.; Ek, I.R.; Frisén, L.; Schalling, M.; Ösby, U.; Lavebratt, C.; Nikamo, P. CRY2 is associated with rapid cycling in bipolar disorder patients. PLoS ONE 2010, 5, e12632. [Google Scholar] [CrossRef] [PubMed]
- McGrath, C.L.; Glatt, S.L.; Sklar, P.; Le-Niculescu, H.; Kuczenski, R.; Doyle, A.E.; Biederman, J.; Mick, E.; Faraone, S.V.; Niculescu, A.B.; et al. Evidence for genetic association of RORB with bipolar disorder. BMC Psychiatry 2009, 9, 70. [Google Scholar] [CrossRef] [PubMed]
- Kato, T. Molecular genetics of bipolar disorder and depression. Psychiatry Clin. Neurosci. 2007, 61, 3–19. [Google Scholar] [CrossRef] [PubMed]
- Kato, T.; Kakiuchi, C.; Iwamoto, K. Comprehensive gene expression analysis in bipolar disorder. Can. J. Psychiatry 2007, 52, 763–771. [Google Scholar] [PubMed]
- McClung, C.A. Circadian genes, rhythm and biology of mood disorder. Pharmacol. Ther. 2007, 114, 222–232. [Google Scholar] [CrossRef] [PubMed]
- DeBundel, D.; Gangarossa, G.; Biever, A.; Bonnefont, X.; Valjent, E. Cognitive dysfunction, elevated anxiety, and reduced cocaine response in circadian clock-deficient cryptochrome knockout mice. Front. Behav. Neurosci. 2013, 7, 152. [Google Scholar]
- Li, J.Z.; Bunney, B.G.; Meng, F.; Hagenauer, M.H.; Walsh, D.M.; Vawter, M.P.; Evans, S.J.; Choudary, P.V.; Cartagena, P.; Barchas, J.D.; et al. Circadian patterns of gene expression in the human brain and disruption in major depressive disorder. Proc. Natl. Acad. Sci. USA 2013, 110, 9950–9955. [Google Scholar] [CrossRef] [PubMed]
- Kripke, D.F.; Nievergelt, C.M.; Joo, E.; Shekhtman, T.; Kelsoe, J.R. Circadian polymorphisms associated with affective disorders. J. Circadian Rhythm. 2009, 7, 2. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Nuccio, S.R.; Yang, J.Y.; Wu, X.; Bogoni, A.; Willner, A.E. High-speed addition/subtraction/complement/doubling of quaternary numbers using optical nonlinearities and DQPSK signals. Opt. Lett. 2012, 37, 1139–1141. [Google Scholar] [CrossRef] [PubMed]
- Kovanen, L.; Kaunisto, M.; Donner, K.; Saarikoski, S.T.; Partonen, T. CRY2 genetic variants associate with dysthymia. PLoS ONE 2013, 8, 71450. [Google Scholar] [CrossRef] [PubMed]
- Hua, P.; Liu, W.; Chen, D.; Zhao, Y.; Chen, L.; Zhang, N.; Wang, C.; Guo, S.; Wang, L.; Xiao, H.; et al. Cry1 and Tef gene polymorphisms are associated with major depressive disorder in the Chinese population. J. Affect. Disord. 2014, 157, 100–103. [Google Scholar] [CrossRef] [PubMed]
- Shi, S.Q.; White, M.J.; Borsetti, H.M.; Pendergast, J.S.; Hida, A.; Ciarleglio, C.M.; de Verteuil, P.A.; Cadar, A.G.; Cala, C.; McMahon, D.G.; et al. Molecular analyses of circadian gene variants reveal sex-dependent links between depression and clocks. Transl. Psychiatry 2016, 6, e748. [Google Scholar] [CrossRef] [PubMed]
- Pirovano, A.; Lorenzi, C.; Serretti, A.; Ploia, C.; Landoni, S.; Catalano, M.; Smeraldi, E. Two new rare variants in the circadian “clock” gene may influence sleep pattern. Genet. Med. 2005, 7, 455–457. [Google Scholar] [CrossRef] [PubMed]
- Desan, P.H.; Oren, D.A.; Malison, R.; Price, L.H.; Rosenbaum, J.; Smoller, J.; Charney, D.S.; Gelernter, J. Genetic polymorphism at the CLOCK gene locus and major depression. Am. J. Med. Genet. 2000, 96, 418–421. [Google Scholar] [CrossRef]
- Serretti, A.; Gaspar-Barba, E.; Calati, R.; Cruz-Fuentes, C.S.; Gomez-Sanchez, A.; Perez-Molina, A.; de Ronchi, D. 3111T/C clock gene polymorphism is not associated with sleep disturbances in untreated depressed patients. Chronobiol. Int. 2010, 27, 265–277. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.R.; Campbell, S.S.; Zone, S.E.; Cooper, F.; DeSano, A.; Murphy, P.J.; Jones, B.; Czajkowski, L.; Ptácek, L.J. Familial advanced sleep-phase syndrome: A short-period circadian rhythm variant in humans. Nat. Med. 1999, 5, 1062–1065. [Google Scholar] [PubMed]
- Toh, K.L.; Jones, C.R.; He, Y.; Eide, E.J.; Hinz, W.A.; Virshup, D.M.; Ptácek, L.J.; Fu, Y.H. An hPer2 phosphorylation site mutation in familial advanced sleep-phase syndrome. Science 2001, 291, 1040–1043. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Padiath, Q.S.; Shapiro, R.E.; Jones, C.R.; Wu, S.C.; Saigoh, N.; Saigoh, K.; Ptacek, L.J.; Fu, Y.H. Functional consequences of a CKIδ mutation causing familial advanced sleep phase syndrome. Nature 2005, 434, 640–644. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.-J.; Logunova, L.; Maywood, E.S.; Gallego, M.; Lebiecki, J.; Brown, T.M.; Loudon, A.S.I. Setting clock speed in mammals: The CK1ε tau mutation in mice accelerates the circadian pacemaker by selectively destabilizing PERIOD proteins. Neuron 2008, 58, 78–88. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Toh, K.L.; Jones, C.R.; Shin, J.Y.; Fu, Y.H. Modeling of a human circadian mutation yields insights into clock regulation by PER2. Cell 2007, 128, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Hirano, A.; Shi, G.; Jones, C.R.; Lipzen, A.; Pennacchio, L.A.; Xu, Y.; Hallows, W.C.; McMahon, T.; Yamazaki, M.; Ptáček, L.J.; et al. A Cryptochrome 2 mutation yields advanced sleep phase in humans. Hum. Biol. Med. Neurosci. 2016, 10, 7554. [Google Scholar] [CrossRef] [PubMed]
- Wehr, T.A. A circadian signal of change of season in patients with seasonal affective disorder. Arch. Gen. Psychiatry 2001, 58, 1108–1114. [Google Scholar] [CrossRef] [PubMed]
- Lewy, A.J.; Bauer, V.K.; Cutler, N.L.; Sack, R.L.; Ahmed, S.; Thomas, K.H.; Blood, M.L.; Jackson, J.M. Morning versus eveninglight treatment of patients with winter depression. Arch. Gen. Psychiatry 1998, 55, 890–896. [Google Scholar] [CrossRef] [PubMed]
- Johansson, C.; Willeit, M.; Smedh, C.; Ekholm, J.; Paunio, T.; Kieseppa, T.; Lichtermann, D.; Praschak-Rieder, N.; Neumeister, A.; Nilsson, L.G. Circadian clock-related polymorphisms in seasonal affective disorder and their relevance to diurnal preference. Neuropsychopharmacology 2003, 28, 734–739. [Google Scholar] [CrossRef] [PubMed]
- Iwase, T.; Kajimura, N.; Uchiyama, M.; Ebisawa, T.; Yoshimura, K.; Kamei, Y.; Shibui, K.; Kim, K.; Kudo, Y.; Katoh, M.; et al. Mutation screening of the human Clock gene in circadian rhythm sleep disorders. Psychiatry Res. 2002, 109, 121–128. [Google Scholar] [CrossRef]
- Takimoto, M.; Hamada, A.; Tomoda, A.; Ohdo, S.; Ohmura, T.; Sakato, H.; Kawatani, J.; Jodoi, T.; Nakagawa, H.; Terazono, H.; et al. Daily expression of clock genes in whole blood cells in healthy subjects and a patient with circadian rhythm sleep disorder. Am. J. Physiol. 2005, 289, R1273–R1279. [Google Scholar] [CrossRef] [PubMed]
- Utge, S.J.; Soronen, P.; Loukola, A.; Kronholm, E.; Ollila, H.M.; Pirkola, S.; Porkka-Heiskanen, T.; Partonen, T.; Paunio, T. Systematic analysis of circadian genes in a population-based sample reveals association of TIMELESS with depression and sleep disturbance. PLoS ONE 2010, 5, e9259. [Google Scholar] [CrossRef] [PubMed]
- Konofal, E.; Lecendreux, M.; Mouren-Siméoni, M.C. Mise au point des études cliniques sur le rapport veille-sommeil dans le trouble déficit de l’attention/hyperactivité de l’enfant. Ann. Medico-Psychol. 2002, 2, 105–117. [Google Scholar] [CrossRef]
- Dickerson Mayes, S.; Calhoun, S.L.; Bixler, E.O.; Vgontzas, A.N.; Mahr, F.; Hillwig-Garcia, J.; Elamir, B.; Edhere-Ekezie, L.; Parvin, M. ADHD Subtypes and Comorbid Anxiety, Depression, and Oppositional-Defiant Disorder: Differences in Sleep Problems. J. Pediatr. Psychol. 2009, 34, 328–337. [Google Scholar] [CrossRef] [PubMed]
- Monti, J.M.; BaHammam, A.S.; Pandi-Perumal, S.R.; Bromundt, V.; Spence, D.W.; Cardinali, D.P.; Brown, G.M. Sleep and circadian rhythm dysregulation in schizophrenia. Prog. Neuro Psychopharmacol. Biol. Psychiatry 2013, 43, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Chouinard, S.; Poulin, J.; Stip, E.; Godbout, R. Sleep in untreated patients with schizophrenia: A meta-analysis. Schizophr. Bull. 2004, 30, 957–967. [Google Scholar] [CrossRef] [PubMed]
- Afonso, P.; Figueira, M.L.; Paiva, T. Sleep-promoting action of the endogenous melatonin in schizophrenia compared to healthy controls. Int. J. Psychiatry Clin. Pract. 2011, 15, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Bromundt, V.; Koster, M.; Georgiev-Kill, A.; Opwis, K.; Wirz-Justice, A.; Stoppe, G.; Cajochen, C. Sleep-wake cycles and cognitive functioning in schizophrenia. Br. J. Psychiatry J. Ment. Sci. 2011, 198, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Morgan, R.; Cheadle, A.J. Circadian body temperature in chronic schizophrenia. Br. J. Psychiatry J. Ment. Sci. 1976, 129, 350–354. [Google Scholar] [CrossRef]
- Rao, M.L.; Gross, G.; Strebel, B.; Halaris, A.; Huber, G.; Braunig, P.; Marler, M. Circadian rhythm of tryptophan, serotonin, melatonin, and pituitary hormones in schizophrenia. Biol. Psychiatry 1994, 35, 151–163. [Google Scholar] [CrossRef]
- Lamont, E.W.; Coutu, D.L.; Cermakian, N.; Boivin, D.B. Circadian rhythms and clock genes in psychotic disorders. Isr. J. Psychiatry Relat. Sci. 2010, 47, 27–35. [Google Scholar] [PubMed]
- Takao, T.; Tachikawa, H.; Kawanishi, Y.; Mizukami, K.; Asada, T. CLOCK gene T3111C polymorphism is associated with Japanese schizophrenics: A preliminary study. Eur. Neuropsychopharmacol. 2007, 17, 273–276. [Google Scholar] [CrossRef] [PubMed]
- Rivto, E.R.; Ritvo, R.; Yuwiler, A.; Brothers, A.; Freeman, B.J.; Plotkin, S. Elevated daytime melatonin concentrations in autism: A pilot study. Eur. Child Adolesc. Psychiatry 1993, 2, 75. [Google Scholar]
- Nir, I.; Meir, D.; Zilber, N.; Knobler, H.; Hadjez, J.; Lerner, Y. Brief report: Circadian melatonin, thyroid-stimulating hormone, prolactin, and cortisol levels in serum of young adults with autism. J. Autism Dev. Disord. 1995, 25, 641. [Google Scholar] [CrossRef] [PubMed]
- Kulman, G.; Lissoni, P.; Rovelli, F.; Roselli, M.G.; Brivio, F.; Sequeri, P. Evidence of pineal endocrine hypofunction in autistic children. Neuroendocrinol. Lett. 2000, 21, 31. [Google Scholar] [PubMed]
- Tordjman, S.; Anderson, G.M.; Pichard, N.; Charbuy, H.; Touitou, Y. Nocturnal excretion of 6-sulphatoxymelatonin in children and adolescents with autistic disorder. Biol. Psychiatry 2005, 57, 134. [Google Scholar] [CrossRef] [PubMed]
- Tordjman, S.; Anderson, G.M.; Bellissant, E.; Botbol, M.; Charbuy, H.; Camus, F.; Graignic, R.; Kermarrec, S.; Fougerou, C.; Cohen, D.; et al. Day and nighttime excretion of 6-sulphatoxymelatonin in adolescents and young adults with autistic disorder. Psychoneuroendocrinology 2012, 37, 1990–1997. [Google Scholar] [CrossRef] [PubMed]
- Melke, J.; Goubran-Botros, H.; Chaste, P.; Betancur, C.; Nygren, G.; Anckarsäter, H.; Rastam, M.; Ståhlberg, O.; Gillberg, I.C.; Delorme, R.; et al. Abnormal melatonin synthesis in autism spectrum disorders. Mol. Psychiatry 2007, 13, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Freitag, C.M. The genetics of autistic disorders and its clinical relevance: A review of the literature. Mol. Psychiatry 2007, 12, 2. [Google Scholar] [CrossRef] [PubMed]
- Sarowar, T.; Chhabra, R.; Vilella, A.; Boeckers, T.M.; Zoli, M.; Grabrucker, A.M. Activity and circadian rhythm influence synaptic Shank3 protein levels in mice. J. Neurochem. 2016, 138, 887–895. [Google Scholar] [CrossRef] [PubMed]
- Bourgeron, T. The Possible Interplay of Synaptic and Clock Genes in Autism Spectrum Disorders. Cold Spring Harb. Symp. Quant. Biol. 2007, 72, 645–654. [Google Scholar] [CrossRef] [PubMed]
- Wimpory, D.; Nicholas, B.; Nash, S. Social timing, clock genes and autism: A new hypothesis. J. Intellect. Disabil. Res. 2002, 46, 352. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Matsumoto, A.; Nakayama, K.; Jimbo, E.F.; Kojima, K.; Nagata, K.; Iwamoto, S.; Yamagata, T. Circadian-relevant genes are highly polymorphic in autism spectrum disorder patients. Brain Dev. 2016, 38, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Roubertoux, P.L.; Tordjman, S. The autism spectrum disorders (ASD): From the clinics to the molecular analysis. In Organism Models of Autism Spectrum Disorders; Roubertoux, P.L., Ed.; Springer: New York, NY, USA, 2015; pp. 29–66. [Google Scholar]
| Studies | Measure | Individuals with Psychiatric Disorder and/or Organisms Models (n) | Controls (n) | Results |
|---|---|---|---|---|
| Kaladchibachi et al. [116] | Cyclical expression of clock genes (Per2) | Mouse embryonic fibroblasts (MEFs) | - | Genetic depletion of glycogen synthase kinase 3 (GSK3) activity results in a significant delay in the cycling period of Per2. |
| McGrath et al. [122] | Genotyping and analysis of 312 single-nucleotide polymorphisms (SNPs) in RORA and 43 SNPs in RORB | Bipolar disorder (BD) children (n = 305) | Healthy parents (n = 306) Healthy individuals (n = 140) | Four intronic RORB SNPs showed positive associations with the pediatric bipolar phenotype. |
| Lavebratt et al. [94] | Assessment of Cry2 gene expression before and after one night of sleep deprivation | BD individuals (n = 13) | Healthy individuals (n = 8) | Cry2 mRNA levels are reduced and unresponsive to sleep deprivation in depressed patients with bipolar disorder. |
| Sjöholm et al. [121] | Analysis of four Cry2 single-nucleotide polymorphisms | BD individuals in Sweden (n = 577); BD type I (n = 497); BD type II (n = 60); BD with rapid cycling (n = 155) | Healthy individuals (n = 1044) | Association between the circadian gene Cry2 and rapid cycling in bipolar disorder. |
| Karthikeyan et al. [120] | Genotyping and analysis of Per3 in blood samples | Bipolar type I disorder individuals in South India (n = 311) | Healthy individuals (n = 346) | The occurrence of the five repeat allele of Per3 may be a risk factor for bipolar type I disorder onset in this ethnic group. |
| Studies | Measure | Individuals with Psychiatric Disorder (n) | Controls (n) | Results |
|---|---|---|---|---|
| Takimoto et al. [146] | Daily variation of melatonin and cortisol, and daily expression of clock genes (Per, Bmal1 and Clock) in whole blood cells | Individuals with circadian rhythm sleep disorder (n = 1) | Healthy male individuals (n = 12) | The peak phase of Per1, Per2, and Per3 appeared in the early morning, whereas that of Bmal1 and Clock appeared in the midnight hours in healthy male individuals. |
| Partonen et al. [93] | Analysis of sequence variations (single-nucleotide polymorphisms) in three core clock genes: Per2, Bmal, and Npas2 | Depressed individuals (n = 189) | Healthy individuals (n = 189) | Variations in the three circadian clock genes Per2, Bmal, and Npas2 are associated with winter depression. |
| Utge et al. [147] | Analysis of 113 single-nucleotide polymorphisms of 18 genes of the circadian system | Depressed individuals (n = 384) | Healthy individuals (n = 1270) | Significant association between Tim variants and depression with fatigue in females, and association to depression with early morning awakening in males. |
| Lavebratt et al. [94] | Genotyping of single nucleotide polymorphism of the Cry2 gene | Depressed individuals with bipolar disorder (n = 204) | Healthy individuals (n = 2017) | The Cry2 gene was significantly associated with winter depression in both samples. |
| Kovanen et al. [130] | Genotyping of 48 single-nucleotide polymorphisms in Cry1 and Cry2 gene | Individuals with dysthymia (n = 136) | Healthy individuals (n = 3871) | Four Cry2 genetic variants (rs10838524, rs7121611, rs7945565, rs1401419) are significantly associated with dysthymia. |
| Hua et al. [131] | Genotyping of single nucleotide polymorphisms (SNPs) of Cry1 rs2287161, Cry2 rs10838524 and Tef (thyrotroph embryonic factor) rs738499 | Chinese individuals with major depressive disorder (MDD) (n = 105) | Chinese healthy individuals (n = 485) | The polymorphisms of Cry1 rs2287161and Tef rs738499 are associated to major depressive disorder. |
| Shi et al. [132] | Genotyping of 32 genetic variants from eight clock genes | Major depressive disorder individuals (n = 592) | Healthy individuals (n = 776) | Genetic polymorphisms in circadian genes, especially Clock and Per3, influence risk of developing depression in a sex- and stress-dependent manner. |
| Studies | Measure | Individuals with Psychiatric Disorder (n) | Controls (n) | Results |
|---|---|---|---|---|
| Kissling et al. [90] | Analysis of polymorphism (rs1801260) at the 3’-untranslated region of the Clock gene | ADHD individuals (n = 143) | Healthy individuals (n = 143) | Significant association (p < 0.001) between genotype and ADHD-scores of the adult ADHD assessments, and the rs1801260 polymorphism with at least one T-mutation is the risk allele. |
| Xu et al. [91] | Analysis of polymorphism (rs1801260) at the 3’-untranslated region of the Clock gene in ADHD using within-family transmission disequilibrium test | Two clinical ADHD samples: United Kingdom (UK) sample: (n = 180); Taiwan sample: (n = 212) | Both parents or mother alone or father alone UK sample: (n = 296); Taiwan sample: (n = 326) | Increased transmission of the T allele of the rs1801260 polymorphism in Tawainese samples. |
| Studies | Measure | Individuals with Psychiatric Disorder (n) | Controls (n) | Results |
|---|---|---|---|---|
| Takao et al. [157] | Analysis of 3111C single nucleotide polymorphism of the Clock gene | Individuals with schizophrenia (n = 145) | Healthy individuals (n = 128) | Individuals with schizophrenia had a significantly higher frequency of the C allele compared to controls. |
| Sun et al. [100] | Relative expression of clock gene mRNA: Per1, Per2 and Per3 in blood samples | Individuals with schizophrenia (n = 13) | Healthy controls (n = 15) | Individuals with schizophrenia presented disruptions in diurnal rhythms of the expression of Per1, Per3, and Npas2 compared with healthy controls, accompanied by a delayed phase in the expression of Per2 and by a decrease in Per3 and Npas2 expression. |
| Johansson et al. [101] | Analysis of Clock, Bmal1, Per1, Per2, Cry1, Cry2, REV-ERBα and Dbp in fibroblasts from skin samples | Individuals with chronic schizophrenia under neuroleptic medication (n = 11) | Healthy individuals (n = 11) | Loss of rhythmic expression of Cry1 and Per2 in fibroblasts from individuals with schizophrenia compared to cells from healthy controls. |
| Studies | Measure | Individuals with Psychiatric Disorder (n) | Controls (n) | Results |
|---|---|---|---|---|
| Nicholas et al. [89] | Screening of eleven clock/clock-related genes | High-functioning ASD individuals (n = 110) | Healthy parents (n = 220) | Significant association for two single-nucleotide polymorphisms in Per1 and in Npas2. |
| Yang et al. [168] | Direct sequencing analysis of the coding regions of 18 canonical clock genes and clock-controlled genes | ASD individuals with sleep disorders (n = 14); ASD individuals without sleep disorders (n = 14) | Healthy individuals (n = 23) | Mutations in circadian-relevant genes (specifically Per1, Per2, Per3, Clock, Npas2, Bmal1, Tim, Cry1, Cry2, Dbp and Ck1ε) affecting gene function are more frequent in individuals with ASD than in controls. |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Charrier, A.; Olliac, B.; Roubertoux, P.; Tordjman, S. Clock Genes and Altered Sleep–Wake Rhythms: Their Role in the Development of Psychiatric Disorders. Int. J. Mol. Sci. 2017, 18, 938. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms18050938
Charrier A, Olliac B, Roubertoux P, Tordjman S. Clock Genes and Altered Sleep–Wake Rhythms: Their Role in the Development of Psychiatric Disorders. International Journal of Molecular Sciences. 2017; 18(5):938. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms18050938
Chicago/Turabian StyleCharrier, Annaëlle, Bertrand Olliac, Pierre Roubertoux, and Sylvie Tordjman. 2017. "Clock Genes and Altered Sleep–Wake Rhythms: Their Role in the Development of Psychiatric Disorders" International Journal of Molecular Sciences 18, no. 5: 938. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms18050938





