Immunological and Inflammatory Impact of Non-Intubated Lung Metastasectomy
Abstract
:1. Introduction
2. Results
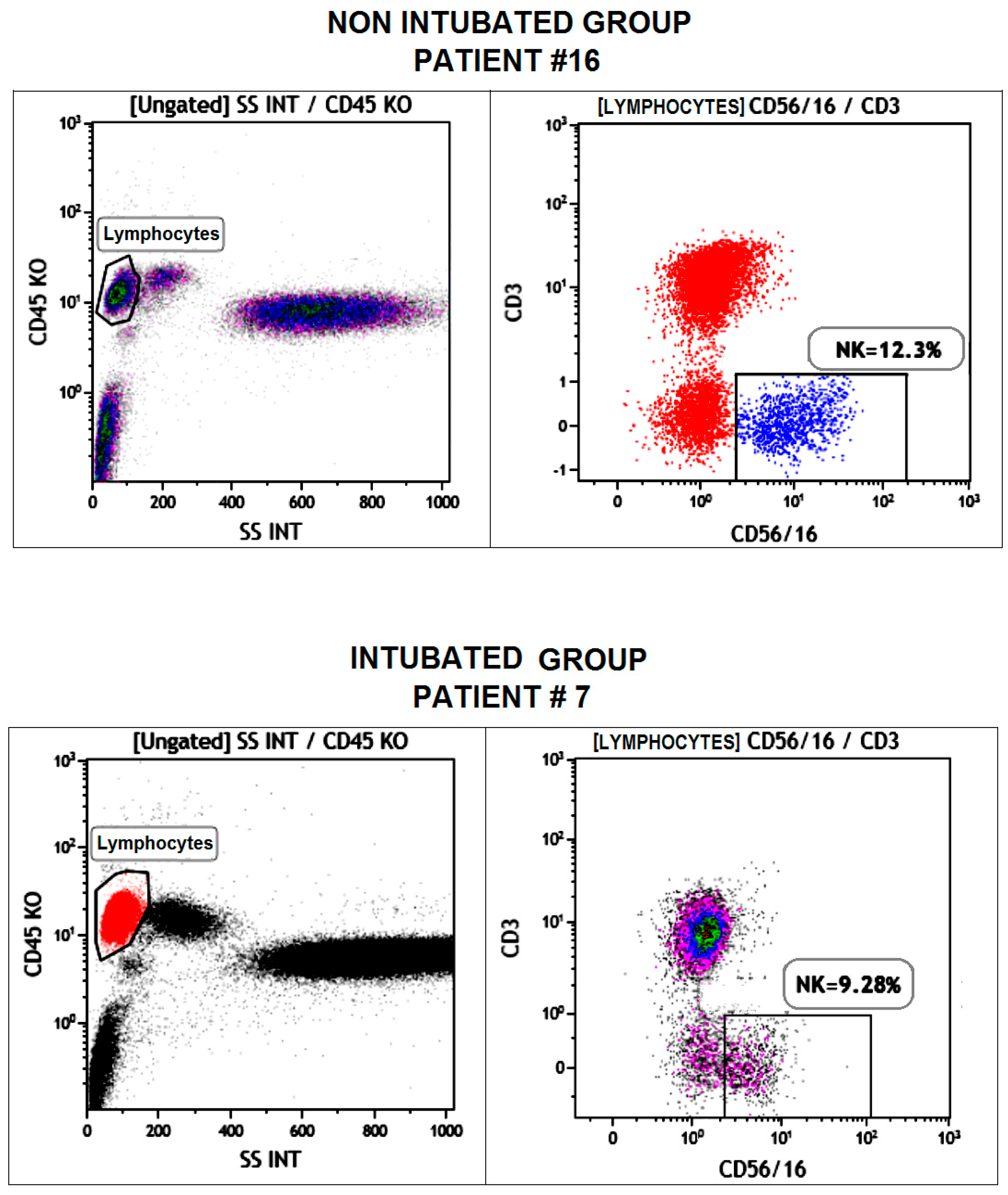
2.1. Immunological Impact
2.2. Inflammatory Impact
2.3. Morbidity
3. Discussion
4. Materials and Methods
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sihoe, A.L. The evolution of minimally invasive thoracic surgery: Implications for the practice of uniportal thoracoscopic surgery. J. Thorac. Dis. 2014, 6, S604–S617. [Google Scholar] [PubMed]
- Ng, C.S.; Gonzalez-Rivas, D.; D’Amico, T.A.; Rocco, G. Uniportal VATS-a new era in lung cancer surgery. J. Thorac. Dis. 2015, 7, 1489–1491. [Google Scholar] [PubMed]
- Rocco, G.; Martucci, N.; La Manna, C.; Jones, D.R.; De Luca, G.; La Rocca, A.; Cuomo, A.; Accardo, R. Ten-year experience on 644 patients undergoing single-port (uniportal) video-assisted thoracoscopic surgery. Ann. Thorac. Surg. 2013, 96, 434–438. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Rivas, D.; Yang, Y.; Ng, C. Advances in uniportal video-assisted thoracoscopic surgery: Pushing the envelope. Thorac. Surg. Clin. 2016, 26, 187–201. [Google Scholar] [CrossRef] [PubMed]
- Mineo, T.C.; Ambrogi, V. Efficacy of awake thoracic surgery. J. Thorac. Cardiovasc. Surg. 2012, 143, 249–250. author reply 250–251. [Google Scholar] [CrossRef] [PubMed]
- Mineo, T.C.; Tacconi, F. From “awake” to “monitored anesthesia care” thoracic surgery: A 15-year evolution. Thoracic. Cancer 2014, 5, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Mineo, T.C.; Ambrogi, V.; Sellitri, F. Non-intubated video-assisted thoracic surgery from multi to uniport approaches: Single centre experience. Eur. Med. J. Respir. 2016, 4, 104–112. [Google Scholar]
- Mineo, T.C.; Tamburrini, A.; Perroni, G.; Ambrogi, V. One thousand cases of tubeless video-assisted thoracic surgery at the Rome Tor Vergata University. Future Oncol. 2016, 12, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Pompeo, E.; Mineo, T.C. Awake pulmonary metastasectomy. J. Thorac. Cardiovasc. Surg. 2007, 133, 960–966. [Google Scholar] [CrossRef] [PubMed]
- Mineo, T.C. Epidural anesthesia in awake thoracic surgery. Eur. J. Cardiothorac. Surg. 2007, 32, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Ambrogi, V.; Sellitri, F.; Perroni, G.; Schillaci, O.; Mineo, T.C. Uniportal video-assisted thoracic surgery colorectal lung metastasectomy in non-intubated anesthesia. J. Thorac. Dis. 2017, 9, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Meakins, J.L. Surgeons, surgery, and immunomodulation. Arch. Surg. 1991, 126, 494–498. [Google Scholar] [CrossRef] [PubMed]
- Wrigge, H.; Zinserling, J.; Stüber, F.; von Spiegel, T.; Hering, R.; Wetegrove, S.; Hoeft, A.; Putensen, C. Effects of mechanical ventilation on release of cytokines into systemic circulation in patients with normal pulmonary function. Anesthesiology 2000, 93, 1413–1417. [Google Scholar] [CrossRef] [PubMed]
- Funakoshi, T.; Ishibe, Y.; Okazaki, N.; Miura, K.; Liu, R.; Nagai, S.; Minami, Y. Effect of re-expansion after short-period lung collapse on pulmonary capillary permeability and pro-inflammatory cytokines gene expression in isolated rabbit lung. Br. J. Anaesth. 2004, 92, 558–563. [Google Scholar] [CrossRef] [PubMed]
- Schilling, T.; Kozian, A.; Huth, C.; Bühling, F.; Kretzschmar, M.; Welte, T.; Hachenberg, T. The pulmonary immune effects of mechanical ventilation in patients undergoing thoracic surgery. Anesth. Analg. 2005, 101, 957–965. [Google Scholar] [CrossRef] [PubMed]
- Gothart, J. Lung injury after thoracic surgery and one-lung ventilation. Curr. Opin. Anaesthesiol. 2006, 19, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Baker, E.A.; El-Gaddal, S.; Williams, L.; Leaper, D.J. Profiles of inflammatory cytokines following colorectal surgery: Relationship with wound healing and outcome. Wound Repair Regen. 2006, 14, 566–572. [Google Scholar] [CrossRef] [PubMed]
- Ghanim, B.; Schweiger, T.; Jedamzik, J.; Glueck, O.; Glogner, C.; Lang, G.; Klepetko, W.; Hoetzenecker, K. Elevated inflammatory parameters and inflammation scores are associated with poor prognosis in patients undergoing pulmonary metastasectomy for colorectal cancer. Interact. Cardiovasc. Thorac. Surg. 2015, 21, 616–623. [Google Scholar] [CrossRef] [PubMed]
- Walsh, K.J.; Tan, K.S.; Zhang, H.; Amar, D. Neutrophil-lymphocyte ratio and risk of atrial fibrillation after thoracic surgery. Interact. Cardiovasc. Thorac. Surg. 2017, 24, 555–559. [Google Scholar] [CrossRef] [PubMed]
- Tacconi, F.; Pompeo, E.; Sellitri, F.; Mineo, T.C. Surgical stress hormones response is reduced after awake videothoracoscopy. Interact. Cardiovasc. Thorac. Surg. 2010, 10, 666–671. [Google Scholar] [CrossRef] [PubMed]
- Mineo, T.C.; Tacconi, F. Role of systemic inflammation scores in pulmonary metastasectomy for colorectal cancer. Thorac. Cancer 2014, 5, 431–437. [Google Scholar] [CrossRef] [PubMed]
- Koltun, W.A.; Bloomer, M.M.; Tilberg, A.F.; Seaton, J.F.; Ilahi, O.; Rung, G.; Gifford, R.M.; Kauffman, G.L., Jr. Awake epidural anesthesia is associated with improved natural killer cell cytotoxicity and a reduced stress response. Am. J. Surg. 1996, 171, 68–72. [Google Scholar] [CrossRef]
- Vanni, G.; Tacconi, F.; Sellitri, F.; Ambrogi, V.; Mineo, T.C.; Pompeo, E. Impact of awake videothoracoscopic surgery on postoperative lymphocyte responses. Ann. Thorac. Surg. 2010, 90, 973–978. [Google Scholar] [CrossRef] [PubMed]
- Yin, K.; Gribbin, E.; Emanuel, S.; Orndoff, R.; Walker, J.; Weese, J.; Fallahnejad, M. Histochemical alterations in one lung ventilation. J. Surg. Res. 2007, 137, 16–20. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, R.; Yasuda, M. Awake thoracoscopic surgery under epidural anesthesia: Is it really safe? Chin. J. Cancer Res. 2014, 26, 368–370. [Google Scholar] [PubMed]
- Whitson, B.A.; D’Cunha, J.; Andrade, R.S.; Kelly, R.F.; Groth, S.S.; Wu, B.; Miller, J.S.; Kratzke, R.A.; Maddaus, M.A. Thoracoscopic versus thoracotomy approaches to lobectomy: Differential impairment of cellular immunity. Ann. Thorac. Surg. 2008, 86, 1735–1744. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.B.; Wang, B.; Wang, X.Y.; Zhang, L. Influence of video-assisted thoracoscopic lobectomy on immunological functions in non-small cell lung cancer patients. Med. Oncol. 2015, 32, 201. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.T.; Zhong, J.H.; Yang, Y.; Mao, N.Q.; Liu, D.S.; Huang, D.M.; Zhao, Y.X.; Zuo, C.T. Comparison of postoperative immune function in patients with thoracic esophageal cancer after video-assisted thoracoscopic surgery or conventional open esophagectomy. Int. J. Surg. 2016, 30, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Baigrie, R.J.; Lamont, P.M.; Dallman, M.; Morris, P.J. The release of interleukin-1 beta (IL-1) precedes that of interleukin 6 (IL-6) in patients undergoing major surgery. Lymphokine Cytokine Res. 1991, 10, 253–256. [Google Scholar] [PubMed]
- Keegan, A.D.; Ryan, J.J.; Paul, W.E. IL-4 regulates growth and differentiation by distinct mechanisms. Immunologist 1996, 4, 194–198. [Google Scholar]
- Biffl, W.L.; Moore, E.E.; Moore, F.A. Interleukin-6 delays neutrophil apoptosis. Arch. Surg. 1996, 131, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Szczensy, T.J.; Slotwinski, R.; Stankiewcz, A.; Szczygiel, B.; Zaleska, M.; Kopacz, M. Interleukin 6 and interleukin 1 receptor antagonist as early markers of complications after lung cancer surgery. Eur. J. Cardiothorac. Surg. 2007, 31, 719–724. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, J.; Zhang, S.J. Effects of anesthesia using propofol and etomidate on T lymphocyte subpopulation of infectious shock patients in perioperative period. J. Biol. Regul. Homeost. Agents 2017, 31, 119–123. [Google Scholar] [PubMed]
- Tønnesen, E.; Höhndorf, K.; Lerbjerg, G.; Christensen, N.J.; Hüttel, M.S.; Andersen, K. Immunological and hormonal responses to lung surgery during one-lung ventilation. Eur. J. Anaesth. 1993, 10, 189–195. [Google Scholar] [CrossRef]
- Kozian, A.; Schilling, T.; Fredén, F.; Maripuu, E.; Rocken, C.; Strang, C.; Hachemberg, T.; Hedenstierna, G. One-lung ventilation induces hyperperfusion and alveolar damage in the ventilated lung: An experimental study. Br. J. Anaesth. 2008, 100, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.J.; Chan, K.C.; Chien, C.T.; Sun, W.Z.; Lin, C.J. Oxidative stress during one-lung ventilation. J. Thorac. Cardiovasc. Surg. 2006, 132, 513–518. [Google Scholar] [CrossRef] [PubMed]
- De Conno, E.; Steurer, M.P.; Wittlinger, M.; Zalunardo, M.P.; Weder, W.; Schneiter, D.; Schimmer, R.C.; Klaghofer, R.; Neff, T.A.; Schmid, E.R.; et al. Anesthetic induced improvement of the inflammatory response to one-lung ventilation. Anesthesiology 2009, 110, 1316–1326. [Google Scholar] [CrossRef] [PubMed]
- Kutza, J.; Gratz, I.; Afshar, M.; Murasko, D.M. The effects of general anesthesia and surgery on basal and interferon stimulated Natural killer cell activity of humans. Anesth. Analg. 1997, 85, 918–923. [Google Scholar] [CrossRef] [PubMed]
- Desborough, J.P. The stress response to trauma and surgery. Br. J. Anaesth. 2000, 85, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Herberman, R.B.; Ortaldo, J.R. Natural Killer cells: Their role in defenses against disease. Science 1981, 241, 24–30. [Google Scholar] [CrossRef]
- Imai, K.; Matsuyama, S.; Miyake, S.; Suga, K.; Nakachi, K. Natural cytotoxic activity of peripheral-blood lymphocytes and cancer incidence: An 11-year follow-up study of a general population. Lancet 2000, 356, 1795–1799. [Google Scholar] [CrossRef]
- Abdelnour-Berchtold, E.; Perentes, J.Y.; Ris, H.B.; Beigelman, C.; Lovis, A.; Peters, S.; Krueger, T.; Gonzalez, M. Survival and Local Recurrence After Video-Assisted Thoracoscopic Lung Metastasectomy. World J. Surg. 2016, 40, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Mineo, T.C.; Ambrogi, V.; Paci, M.; Iavicoli, N.; Pompeo, E.; Nofroni, I. Transxiphoid bilateral palpation in video-assisted thoracoscopic lung metastasectomy. Ann. Thorac. Surg. 1999, 67, 1808–1810. [Google Scholar] [CrossRef]
- Mineo, T.C.; Ambrogi, V.; Tonini, G.; Nofroni, I. Pulmonary metastasectomy: Might type of resection affect the survival? J. Surg. Oncol. 2001, 76, 47–52. [Google Scholar] [CrossRef]
- Mineo, T.C.; Ambrogi, V.; Tacconi, F.; Mineo, D. Multi-reoperations for lung metastases. Future Oncol. 2015, 11, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Treasure, T.; Mineo, T.C.; Ambrogi, V.; Fiorentino, F. Survival is higher after repeat lung metastasectomy than after a first metastasectomy: Too good to be true? J. Thorac. Cardiovasc. Surg. 2015, 149, 1249–1252. [Google Scholar] [CrossRef] [PubMed]
- Mineo, T.C.; Ambrogi, V. Lung metastasectomy: An experience-based therapeutic option. Ann. Transl. Med. 2015, 3, 194. [Google Scholar] [PubMed]
- Kissin, I. Depth of anesthesia and bispectral index monitoring. Anesth. Analg. 2000, 90, 1114–1117. [Google Scholar] [CrossRef] [PubMed]

| Non-Intubated Group (n = 55) | Intubated Group (n = 13) | p-Value | |
|---|---|---|---|
| Age (range), years | 64 (47–74) | 66 (51–73) | 0.1 |
| Sex (m:f) | 30:25 | 7:6 | 0.9 |
| Primitive Histology | |||
| carcinoma:sarcoma | 45:10 | 13:0 | 0.09 |
| Previous Adjuvant Chemotherapy | |||
| yes:no | 37:18 | 9:4 | 0.8 |
| Disease-Free Interval | |||
| <1 year | 28 | 6 | 0.7 |
| >3 years | 16 | 3 | 0.6 |
| Resected metastases (mean) | 1.55 | 1.77 | 0.8 |
| Baseline | Day 1 | Between-Group p-Value | Day 7 | Between-Group p-Value | Day 14 | Between-Group p-Value | |
|---|---|---|---|---|---|---|---|
| Total Leucocytes (n 109/L) | |||||||
| Non-intubated group | 5.45 (4.22–8.44) | 7.51 (5.15–9.02) | 0.08 | 6.04 (5.36–9.32) | 0.06 | 6.17 (5.07–8.73) | 0.3 |
| Intubated group | 5.81 (4.47–8.21) | 8.01 (7.44–9.82) | 7.33 (7.06–9.98) | 6.34 (5.32–8.41) | |||
| Total Lymphocytes (n 109/L) | |||||||
| Non-intubated group | 1.91 (1.67–2.45) | 1.86 (1.52–1.99) | 0.05 | 1.89 (1.53–2.12) | 0.05 | 1.91 (1.03–2.09) | 0.06 |
| Intubated group | 1.90 (1.53–2.39) | 1.69 (1.46–2.05) | 1.69 (1.29–1.91) | 1.71 (1.21–2.04) | |||
| B Lymphocytes (%) | |||||||
| Non-intubated group | 11 (7–15) | 12 (7–16) | 0. 8 | 11 (7–15) | 1 | 9 (7–16) | 0.9 |
| Intubated group | 12 (7–15) | 12 (7–15) | 11 (7–15) | 9 (7–15) | |||
| T Lymphocytes (%) | |||||||
| Non-intubated group | 71 (60 –75) | 70 (64–73) | 0.1 | 73 (65–85) | 0.06 | 73 (61–77) | 0.3 |
| Intubated group | 72 (64–75) | 67 (61–76) | 64 (61–75) | 70 (69–86) | |||
| T Helper/T Suppressor (Ratio) | |||||||
| Non-intubated group | 2.3 (1.3–3.7) | 2.3 (1.3–3.3) | 0.9 | 2.3 (2.0–3.9) | 0.9 | 2.3 (1.7–3.6) | 0.9 |
| Intubated group | 2.3 (1.3–3.5) | 2.3 (1.3–3.1) | 2.3 (1.7–3.8) | 2.1 (1.6–2.8) | |||
| Natural-Killer (%) | |||||||
| Non-intubated group | 12 (7–14) | 12 (7–14) | 0.09 | 12 (10–16) | 0.04 | 11 (10–15) | 0.06 |
| Intubated group | 11 (9–12) | 11 (9–12) | 9 (7–11) | 9 (8–16) | |||
| Baseline | Day 1 | Between-Group p-Value | Day 7 | Between-Group p-Value | Day 14 | Between-Group p-Value | |
|---|---|---|---|---|---|---|---|
| IL–6 (pg/mL) | |||||||
| Non–intubated group | 12.1 (11.1–12.9) | 14.1 (11.9–17.3) | 0.03 | 12.5 (10.4–14.4) | 0.04 | 12.0 (10.9–13.1) | 0.05 |
| Intubated group | 12.3 (10.9–13.1) | 17.2 (13.3–22.1) | 14.5 (12.2–21.1) | 13.7 (10.1–18.3) | |||
| IL–10 (pg/mL) | |||||||
| Non–intubated group | 5.8 (4.3–12.6) | 8.4 (5.6–14.1) | 0.1 | 9.6 (9.1–12.2) | 0.3 | 8.1 (7.3–11.1) | 0.5 |
| Intubated group | 5.8 (3.9–12.9) | 6.9 (4.2–14.7) | 10.9 (7.9–14.2) | 9.3 (7.2–13.7) | |||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mineo, T.C.; Sellitri, F.; Vanni, G.; Gallina, F.T.; Ambrogi, V. Immunological and Inflammatory Impact of Non-Intubated Lung Metastasectomy. Int. J. Mol. Sci. 2017, 18, 1466. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms18071466
Mineo TC, Sellitri F, Vanni G, Gallina FT, Ambrogi V. Immunological and Inflammatory Impact of Non-Intubated Lung Metastasectomy. International Journal of Molecular Sciences. 2017; 18(7):1466. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms18071466
Chicago/Turabian StyleMineo, Tommaso Claudio, Francesco Sellitri, Gianluca Vanni, Filippo Tommaso Gallina, and Vincenzo Ambrogi. 2017. "Immunological and Inflammatory Impact of Non-Intubated Lung Metastasectomy" International Journal of Molecular Sciences 18, no. 7: 1466. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms18071466




