Endophytic Bacteria Potentially Promote Plant Growth by Synthesizing Different Metabolites and their Phenotypic/Physiological Profiles in the Biolog GEN III MicroPlateTM Test
Abstract
:1. Introduction
2. Results
2.1. Phenotypic Profiling and Identification of Endophytic Bacteria Using BiologTM GEN III MicroPlates
2.2. In Vitro Screening of Bacterial Isolates for Potential Plant Growth Promoting (PGP) Activities
2.2.1. Colorimetric Analysis of Indole-3-Acetic Acid (IAA)-like Compounds Production
2.2.2. Qualitative and Quantitative Production of Siderophores
2.2.3. Phosphate Solubilization
2.2.4. Oligonitrotrophic and Nitrogen-Fixation Screening
2.3. Statistical Analysis
3. Discussion
3.1. Phenotypic Profiling and Identification of Endophytic Bacteria Using BiologTM GEN III MicroPlates
3.2. In Vitro Screening of Bacterial Isolates for Potential PGP Activity
3.3. Principal Component Analysis (PCA) of Isolates’ Features
4. Materials and Methods
4.1. Sample Collection
4.2. Phenotypic Profile and Identification of Endophytic Bacteria Using BiologTM GEN III MicroPlates
4.3. In Vitro Screening of Bacterial Isolates for their Potential PGP Activities
4.3.1. Colorimetric Analysis of IAA-like Compounds Production
4.3.2. Qualitative and Quantitative Production of Siderophores
4.3.3. Phosphate-Solubilization
4.3.4. Oligonitrotrophic and Nitrogen-Fixation Screening
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| PGPE | Plant Growth-Promoting Endophytes |
| PGPB | Plant Growth-Promoting Bacteria |
| PGP activities | Plant Growth-Promoting activities |
| AWCD | Average well color development |
| H′ | Shannon diversity |
| E | Shannon evenness |
| LB medium | Luria-Bertani medium |
| ACC | Aminocyclopropane-1-carboxylic acid |
| HCN | Hydrogen cyanide |
| ISR | Induced systemic resistance |
| Nfa | Nitrogen-free agar |
| BTB | Bromothymol blue |
| IAA | Indole-3-acetic acid |
| Trp | L-tryptophan |
| CAS | Chrome Azurol S |
| PSU | Percent siderophores unit |
| HTS | Hydroxamate-type siderophores |
| CTS | Catechol-type siderophores |
| SI | Phosphate solubilization index |
| PCA | Principal component analysis |
| ROS | Reactive oxygen species |
| SOS | Salt overly sensitive |
| ABA | Abscisic acid |
References
- Yadav, A.N.; Verma, P.; Singh, B.; Chauhan, V.S.; Suman, A.; Saxena, A.K. Plant growth promoting bacteria: Biodiversity and multifunctional attributes for sustainable agriculture. Adv. Biotechnol. Microbiol. 2017, 5, 1–16. [Google Scholar] [CrossRef]
- Baez-Rogelio, A.; Morales-García, Y.E.; Quintero-Hernández, V.; Muñoz-Rojas, J. Next generation of microbial inoculants for agriculture and bioremediation. Microb. Biotechnol. 2017, 10, 19–21. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization of the United Nations Rome. The Future of Food and Agriculture: Trends and Challenges. 2017. Available online: http://www.fao.org/3/a-i6583e.pdf (accessed on 10 August 2019).
- Timmusk, S.; Behers, L.; Muthoni, J.; Muraya, A.; Aronsson, A.-C. Perspectives and challenges of microbial application for crop improvement. Front. Plant. Sci. 2017, 8, 49. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Carvalhais, L.C.; Crawford, M.; Singh, E.; Dennis, P.G.; Pieterse, C.M.J.; Schenk, P.M. Inner plant values: Diversity, colonization and benefits from endophytic bacteria. Front. Microbiol. 2017, 19, 2552. [Google Scholar] [CrossRef] [PubMed]
- Calvo, P.; Nelson, L.; Kloepper, J.W. Agricultural uses of plant biostiumulants. Plant. Soil. 2014, 383, 3. [Google Scholar] [CrossRef]
- Hardoim, P.R.; Van Overbeek, L.S.; Berg, G.; Pirttilä, A.M.; Compant, S.; Campisano, A.; Döring, M.; Sessitsch, A. The hidden world within plants: Ecological and evolutionary considerations for defining functioning of microbial endophytes. Microbiol. Mol. Biol. Rev. 2015, 79, 293–320. [Google Scholar] [CrossRef] [PubMed]
- Le Cocq, K.; Gurr, S.J.; Hirsch, P.R.; Mauchline, T.H. Exploitation of endophytes for sustainable agricultural intensification. Mol. Plant. Pathol. 2016, 18, 469–473. [Google Scholar] [CrossRef] [Green Version]
- van Overbeek, L.S.; Saikkonen, K. Impact of bacterial–fungal interactions on the colonization of the endosphere. Trends Plant. Sci. 2016, 21, 230–242. [Google Scholar] [CrossRef]
- Wdowiak-Wróbel, S.; Marek-Kozaczuk, M.; Kalita, M.; Karaś, M.; Wójcik, M.; Małek, W. Diversity and plant growth promoting properties of rhizobia isolated from root nodules of Ononis arvensis. Antonie van Leeuwenhoek 2017, 110, 1087–1103. [Google Scholar] [CrossRef] [Green Version]
- Mauchline, W.S.; Keevil, C.W. Development of the BIOLOG substrate utilization system for identification of Legionella spp. Appl. Environ. Microbiol. 1991, 57, 3345–3349. [Google Scholar]
- LaCroce, S.J.; Wilson, M.N.; Romanowski, J.E.; Newman, J.D.; Jhanji, V.; Shanks, R.M.; Kowalski, R.P. Moraxella nonliquefaciens and M. osloensis Are Important Moraxella Species That Cause Ocular Infections. Microorganisms 2019, 7, 163. [Google Scholar] [CrossRef] [PubMed]
- Chojniak, J.; Jałowiecki, Ł.; Dorgeloh, E.; Hegedusova, B.; Ejhed, H.; Magnér, J.; Płaza, G. Application of the BIOLOG system for characterization of Serratia marcescens ss marcescens isolated from onsite wastewater technology (OSWT). Acta Biochim. Pol. 2015, 62. [Google Scholar] [CrossRef] [PubMed]
- Souza, R.D.; Ambrosini, A.; Passaglia, L.M. Plant growth-promoting bacteria as inoculants in agricultural soils. Genet. Mol. Biol. 2015, 38, 401–419. [Google Scholar] [CrossRef] [PubMed]
- Patle, P.N.; Navnage, N.P.; Ramteke, P.R. Endophytes in plant system: Roles in growth promotion, mechanism and their potentiality in achieving agriculture sustainability. Int. J. Chem. Stud. 2018, 6, 270–274. [Google Scholar]
- Krawczyk, K.; Zwolińska, A.; Kamasa, J.; Maćkowiak-Sochacka, A.; Przemieniecki, S. Identyfikacja i charakterystyka bakterii endofitycznych wykazujących cechy promujące wzrost roślin. Prog. Plant. Prot. 2016, 56, 100–109. [Google Scholar] [CrossRef]
- Khan, Z.; Doty, S.L. Characterization of bacterial endophytes of sweet potato plants. Plant. Soil 2009, 322, 197–207. [Google Scholar] [CrossRef]
- Patel, J.K.; Archana, G. Diverse culturable diazotrophic endophytic bacteria from Poaceae plants show cross-colonization and plant growth promotion in wheat. Plant. Soil 2017, 417, 99–116. [Google Scholar] [CrossRef]
- Woźniak, M.; Gałązka, A.; Grządziel, J.; Głodowska, M. The identification and genetic diversity of endophytic bacteria isolated from selected crops. J. Agri. Sci. 2018, 156, 547–556. [Google Scholar] [CrossRef]
- Gaiero, J.R.; McCall, C.A.; Thompson, K.A.; Day, N.J.; Best, A.S.; Dunfield, K.E. Inside the root microbiome: Bacterial root endophytes and plant growth promotion. Am. J. Bot. 2013, 100, 1738–1750. [Google Scholar] [CrossRef] [Green Version]
- Łyszcz, M.; Gałązka, A. Selected molecular methods used in assessing the biodiversity of soil organisms. Post. Mikrobiol. 2016, 55, 309–319. [Google Scholar]
- Łyszcz, M.; Gałązka, A. Genetic differentiation methods of microorganisms in the soil-plant system. Post. Mikrobiol. 2017, 56, 341–352. [Google Scholar]
- Arnow, L.E. Colorimetric determination of the components of 3,4-dihydroxyphenylalanine tyrosine mixtures. J. Biol. Chem. 1937, 118, 531–537. [Google Scholar]
- Csáky, T.Z. On the estimation of bound hydroxylamines in biological materials. Act. Chem. Scand. 1948, 2, 450–454. [Google Scholar] [CrossRef]
- Berraquero, F.R.; Baya, B.; Cormenzana, A.R. Estabelecimiento de índices para el estudio de la solubilización de fosfatos por bacterias del suelo. Ars. Pharm. 1976, 17, 399–406. [Google Scholar]
- Figueiredo, M.D.V.B.; Seldin, L.; de Araujo, F.F.; Mariano, R.D.L.R. Plant growth promoting rhizobacteria: Fundamentals and applications. In Plant Growth and Health Promoting Bacteria; Maheshwari, D.K., Ed.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 21–43. [Google Scholar]
- Szilagyi-Zecchin, V.J.; Ikeda, A.C.; Hungria, M.; Adamoski, D.; Kava-Cordeiro, V.; Glienke, C.; Galli-Terasawa, L.V. Identification and characterization of endophytic bacteria from corn (Zea mays L.) roots with biotechnological potential in agriculture. AMB Express. 2014, 4, 26. [Google Scholar] [CrossRef] [PubMed]
- Hacquard, S.; Garrido-Oter, R.; Gonz-Alez, A.; Spaepen, S.; Ackermann, G.; Lebeis, S.; McHardy, A.C.; Dangl, J.L.; Knight, R.; Ley, R.; et al. Microbiota and host nutrition across plant and animal kingdoms. Cell Host Microbe. 2015, 17, 603–616. [Google Scholar] [CrossRef]
- Szymańska, S.; Płociniczak, T.; Piotrowska-Seget, Z.; Hrynkiewicz, K. Endophytic and rhizosphere bacteria associated with the roots of the halophyte Salicornia europaea L.—Community structure and metabolic potential. Microbiol. Res. 2016, 192, 37–51. [Google Scholar] [CrossRef]
- Liotti, R.G.; da Silva Figueiredo, M.T.; da Silva, G.F.; Furtado de Mendonça, E.A.; Soares, M.A. Diversity of cultivable bacterial endophytes in Paullinia cupana and their potential for plant growth promotion and phytopathogen control. Microbiol. Res. 2018, 207, 8–18. [Google Scholar] [CrossRef]
- Partida-Martínez, L.P.; Martin Heil, M. The microbe-free plant: Fact or artifact? Front. Plant. Sci. 2011, 100, 1–16. [Google Scholar] [CrossRef]
- Huang, Y.-H. Comparison of rhizosphere and endophytic microbial communities of Chinese leek through high-throughput 16S rRNA gene Illumina sequencing. J. Integr. Agric. 2018, 17, 359–367. [Google Scholar] [CrossRef] [Green Version]
- Petti, C.A.; Polage, C.R.; Schreckenberger, P. The role of 16S rRNA gene sequencing in identification of microorganisms misidentified by conventional methods. J. Clin. Microbiol. 2005, 43, 6123–6125. [Google Scholar] [CrossRef] [PubMed]
- Morgan, M.C.; Boyette, M.; Goforth, C.; Sperry, K.V.; Greene, S.R. Comparison of the Biolog OmniLog identification system and 16S ribosomal RNA gene sequencing for accuracy in identification of atypical bacteria of clinical origin. J. Microbiol. Meth. 2009, 79, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Tuteja, N. Mechanisms of high salinity tolerance in plants. Methods Enzymol. 2007, 428, 419–438. [Google Scholar] [CrossRef] [PubMed]
- Numan, M.; Bashir, S.; Khan, Y.; Mumtaz, R.; Shinwari, Z.K.; Khan, A.L.; Al-Harrasi, A. Plant growth promoting bacteria as an alternative strategy for salt tolerance in plants: A review. Microbiol. Res. 2018, 209, 21–32. [Google Scholar] [CrossRef] [PubMed]
- Li, O.; Xiao, R.; Sun, L.; Guan, C.; Kong, D.; Hu, X. Bacterial and diazotrophic diversities of endophytes in Dendrobium catenatum determined through barcoded pyrosequencing. PLoS ONE 2017, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Banach, A.; Kuźniar, A.; Mencfel, R.; Wolińska, A. The study on the cultivable microbiome of the aquatic fern Azolla Filiculoides, L. as new source of beneficial microorganisms. Appl. Sci. 2019, 9, 2143. [Google Scholar] [CrossRef]
- Brana, V.; Cadige, C.; Morel, M.A. The sustainable use of Delfia in agriculture, bioremediation, and bioproducts synthesis. In Microbial Models: From Environmental to Industrial Sustainability Part II Microbes in Envirinmental Sustainability; Castro-Sowinski, S., Ed.; Springer: Singapore, Singapore, 2016; pp. 227–251. [Google Scholar] [CrossRef]
- Morel, M.A.; Ubalde, M.C.; Braña, V.; Castro-Sowinski, S. Delftia sp. JD2: A potential Cr(VI)-reducing agent with plant growth-promoting activity. Arch. Microbiol. 2011, 193, 63–68. [Google Scholar] [CrossRef]
- Morel, M.A.; Cagide, C.; Minteguiaga, M.A.; Dardanelli, M.S.; Castro-Sowinski, S. The pattern of secreted molecules during the co-inoculation of alfalfa plants with Sinorhizobium meliloti and Delftia sp. strain JD2: An interaction that improves plant yield. Mol. Plant-Microbe Interact. 2015, 28, 134–142. [Google Scholar] [CrossRef]
- Ubalde, M.C.; Braña, V.; Sueiro, F.; Morel, M.A.; Martínez-Rosales, C.; Marquez, C.; Castro-Sowinski, S. The versatility of Delftia sp. isolates as tools for bioremediation and biofertilization technologies. Curr. Microbiol. 2012, 64, 597–603. [Google Scholar] [CrossRef]
- Han, J.; Sun, L.; Dong, X.; Cai, Z.; Sun, X.; Yang, H.; Wang, T.; Song, W. Characterization of a novel plant growth-promoting bacteria strain Delftia tsuruhatensis HR4 both as a diazotroph and a potential biocontrol agent against various plant pathogens. Syst. Appl. Microbiol. 2005, 28, 66–76. [Google Scholar] [CrossRef]
- Janahiraman, V.; Anandham, R.; Kwon, S.W.; Sundaram, S.; Karthik Pandi, V.; Krishnamoorthy, R.; Kim, K.; Samaddar, S.; Sa, T. Control of wilt and rot pathogens of tomato by antagonistic pink pigmented facultative methylotrophic Delftia lacustris and Bacillus spp. Front. Plant Sci. 2016, 7, 1626. [Google Scholar] [CrossRef] [PubMed]
- Ambawade, M.S.; Pathade, G.R. Production of indole acetic acid (IAA) by Stenotrophomonas maltophilia BE25 isolated from roots of banana (Musa spp). Int. J. Sci. Res. 2013, 4, 2644–2650. [Google Scholar]
- Brigido, C.; Singh, S.; Menendez, E.; Tavares, M.J.; Glick, B.R.; Felix, M.R.; Oliveira, S.; Carvalho, M. Diversity and functionality of culturable endophytic bacterial communities in chickpea plants. Plants 2019, 8, 42. [Google Scholar] [CrossRef]
- Shi, Y.; Lou, K.; Li, C. Promotion of plant growth by phytohormone-producing endophytic microbes of sugar beet. Biol. Fertil. Soils 2009, 45, 645–653. [Google Scholar] [CrossRef]
- Chun, J.; Lee, J.; Bae, J.; Kim, M.; Lee, J.G.; Shin, S.Y.; Kim, Y.R.; Lee, K.H. Delftia acidovorans isolated from the drainage in an immunocompetent patient with empyema. Tuberc. Respir. Dis. 2009, 67, 239–243. [Google Scholar] [CrossRef]
- Trifonova, A.; Strateva, T. Stenotrophomonas maltophilia—A low-grade pathogen with numerous virulence factors. Infectious Diseases 2019, 51, 168–178. [Google Scholar] [CrossRef]
- Sarwar, M.; Arshad, M.; Martens, D.A.; Frankenberger, W.T., Jr. Tryptophan-dependent biosynthesis of auxins in soil. Plant. Soil 1992, 147, 207–215. [Google Scholar] [CrossRef]
- Mohite, B. Isolation and characterization of indole acetic acid (IAA) producing bacteria from rhizospheric soil and its effect on plant growth. J. Soil Sci. Plant. Nutr. 2013, 13, 638–649. [Google Scholar] [CrossRef]
- Singh, R.; Prasad, M.P. Isolation and screening of rice rhizosphere soil microorganisms for the production of IAA. Int. J. Curr. Microbiol. App. Sci. 2014, 3, 993–998. [Google Scholar]
- Ozimek, E.; Jaroszuk-Ściseł, J.; Bohacz, J.; Korniłłowicz-Kowalska, T.; Tyśkiewicz, R.; Słomka, A.; Nowak, A.; Hanaka, A. Synthesis of indoleacetic acid, gibberellic acid and ACC-deaminase by Mortierella strains promote winter wheat seedling growth under different conditions. Int. J. Mol. Sci. 2018, 19, 3218. [Google Scholar] [CrossRef]
- Tanimoto, E. Regulation of root growth by plant hormones—Roles for auxin and gibberellin. Crit. Rev. Plant. Sci. 2005, 24, 249–265. [Google Scholar] [CrossRef]
- Baldi, B.G.; Maher, B.R.; Slovin, J.P.; Cohen, J.D. Stableisotope labeling, in vitro, of D- and L-tryptophan pools in Lemna gibba and the low incorporation of label into IAA. Plant. Physiol. 1991, 95, 1203–1208. [Google Scholar] [CrossRef] [PubMed]
- Wright, A.D.; Sampson, M.B.; Neuffer, M.G.; Michalczuk, L.; Slovin, J.P.; Cohen, J.D. Indole-3-acetic acid biosynthesis in the mutant maize orange pericarp, a tryptophan auxotroph. Science 1991, 254, 998–1000. [Google Scholar] [CrossRef]
- Normanly, J.; Cohen, J.D.; Fink, G.R. Arabidopsis thaliana auxotrophs reveal a tryptophan-independent biosynthetic pathway for indole-3-acetic acid. Proc. Natl. Acad. Sci. USA 1993, 90, 10355–10359. [Google Scholar] [CrossRef] [PubMed]
- Michalczuk, L.; Ribnicky, D.M.; Cooke, T.J.; Cohen, J.D. Regulation of indole-3-acetic acid biosynthetic pathways in carrot cell cultures. Plant. Physiol. 1992, 100, 1346–1353. [Google Scholar] [CrossRef]
- Olatunji, D.; Danny Geelen, D.; Verstraeten, I. Control of Endogenous Auxin Levels in Plant Root Development. Int. J. Mol. Sci. 2017, 18, 2587. [Google Scholar] [CrossRef]
- Ribnicky, D.M.; Ilić, N.; Cohen, J.D.; Cooke, T.J. The Effects of Exogenous Auxins on Endogenous Indole-3-Acetic Acid Metabolism (The lmplications for Carrot Somatic Embryogenesis). Plant. Physiol. 1996, 11, 549–558. [Google Scholar] [CrossRef]
- Hermosa, R.; Viterbo, A.; Chet, I.; Monte, E. Plant-beneficial effects of Trichoderma and of its genes. Microbiology 2012, 158, 17–25. [Google Scholar] [CrossRef]
- Liaqat, F.; Eltem, R. Identification and characterization of endophytic bacteria isolated from in vitro cultures of peach and pear rootstocks. Biotech 2016, 6, 120. [Google Scholar] [CrossRef]
- Gothwal, R.K.; Nigam, V.K.; Mohan, M.K.; Sasmal, D.; Ghosh, P. Screening of nitrogen fixers from rhizospheric bacterial isolates associated with important desert plants. Appl. Ecol. Env. Res. 2007, 6, 101–109. [Google Scholar] [CrossRef]
- Kilfe, M.H.; Laing, M.D. Isolation and screening of bacteria for their diazotrophic potential and their influence on growth promotion of maize seedlings in greenhouses. Front. Plant. Sci. 2016, 6, 1225. [Google Scholar] [CrossRef]
- Roesch, L.F.W.; de Quadros, P.S.; Camargo, F.A.O.; Triplett, E.W. Screening of diazotrophic bacteria Azopirillum spp. for nitrogen fixation and auxin production in multiple field sites in southern Brazil. World J. Microbiol. Biotechnol. 2007, 23, 1377–1383. [Google Scholar] [CrossRef]
- Romera, F.J.; García, M.J.; Lucena, C.; Martínez-Medina, A.; Aparicio, M.A.; José Ramos, J.; Alcántara, E.; Angulo, M.; Pérez-Vicente, R. Induced systemic resistance (ISR) and Fe deficiency responses in dicot plants. Front. Plant. Sci. 2019, 10, 287. [Google Scholar] [CrossRef] [PubMed]
- Briat, J.F.; Rouached, H.; Tissot, N.; Gaymard, F.; Dubos, C. Integration of P, S, Fe, and Zn nutrition signals in Arabidopsis thaliana: Potential involvement of PHOSPHATE STARVATION RESPONSE 1 (PHR1). Front. Plant Sci. 2015, 6, 290. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Nishizawa, N.K. Iron uptake, translocation, and regulation in higher plants. Annu. Rev. Plant. Biol. 2012, 63, 131–152. [Google Scholar] [CrossRef] [PubMed]
- Aznar, A.; Chen, N.W.G.; Thomine, S.; Dellagi, A. Immunity to plant pathogens and iron homeostasis. Plant. Sci. 2015, 240, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Verbon, E.H.; Trapet, P.L.; Stringlis, I.A.; Kruijs, S.; Bakker, P.A.H.M.; Pieterse, C.M.J. Iron and immunity. Annu. Rev. Phytopathol. 2017, 55, 355–375. [Google Scholar] [CrossRef]
- Ahemad, M.; Kibret, M. Mechanisms and applications of plant growth promoting rhizobacteria: Current perspective. J. King. Saud. Univ. Sci. 2014, 26, 1–20. [Google Scholar] [CrossRef] [Green Version]
- Arora, N.K.; Verma, M. Modified microplate method for rapid and efficient estimation of siderophore produced by bacteria. 3 Biotech. 2017, 7, 381. [Google Scholar] [CrossRef]
- Ghosh, S.K.; Pal, S.; Chakraborty, N. The qualitative and quantitative assay of siderophore production by some microorganisms and effect of different media on its production. Int. J. Chem. 2015, 13, 1621–1629. [Google Scholar]
- Jacoby, R.; Peukert, M.; Succurro, A.; Koprivova, A.; Kopriva, S. The role of soil microorganisms in plant mineral nutrition—Current knowledge and future directions. Front. Plant. Sci. 2017, 8, 1617. [Google Scholar] [CrossRef] [PubMed]
- Gouda, S.; Kerry, R.G.; Das, G.; Paramithiotis, S.; Shin, H.S.; Patra, J.K. Revitalization of plant growth promoting rhizobacteria for sustainable development in agriculture. Microbiol. Res. 2018, 206, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Santoyoa, G.; Moreno-Hagelsieb, G.; Orozco-Mosqueda, M.C.; Glick, B.R. Plant growth-promoting bacterial endophytes. Microbiol. Res. 2016, 183, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Müller, D.B.; Srinivas, G.; Garrido-Oter, R.; Potthoff, E.; Rott, M.; Dombrowski, N.; Münch, P.C.; Spaepen, S.; Remus-Emsermann, M.; et al. Functional overlap of the Arabidopsis leaf and root microbiota. Nature 2015, 528, 364. [Google Scholar] [CrossRef]
- Emenike, C.U.; Agamuthu, P.; Fauziah, S.H. Blending Bacillus sp., Lysinibacillus sp. and Rhodococcus sp. for optimal reduction of heavy metals in leachate contaminated soil. Environ. Earth Sci. 2016, 75, 26. [Google Scholar] [CrossRef]
- Glickmann, E.; Dessaux, Y. A critical examination of the specificity of the Salkowski reagent for indolic compounds produced by phytopathogenic bacteria. Appl. Environ. Microbiol. 1995, 61, 793–796. [Google Scholar]
- Pilet, P.E.; Chollet, R. Colorimetric determination of indole-3-acetic acid. Cr. Acad. Sci. Ser. D 1970, 271, 1675–1678. [Google Scholar]
- Salkowski, E. Ueber das verhalten der skatolcarbonsaüre im organismus. Z. Physiol. Chem. 1885, 9, 23–33. [Google Scholar]
- Jaroszuk-Ściseł, J.; Kurek, E.; Trytek, M. Efficiency of indoleacetic acid, gibberellic acid and ethylene synthesized in vitro by Fusarium culmorum strains with different effects on cereal growth. Biologia 2014, 69, 281–292. [Google Scholar] [CrossRef]
- Schwyn, B.; Neilands, J.B. Universal chemical assay for the detection and determination of siderophores. Anal. Biochem. 1987, 160, 47–56. [Google Scholar] [CrossRef]
- Louden, B.C.; Haarmann, D.; Lynne, A.M. Use of blue agar CAS assay for siderophore detection. J. Microbiol. Biol. Educ. 2011, 12, 51–53. [Google Scholar] [CrossRef] [PubMed]
- Payne, S.M. Iron acquisition in microbial pathogenesis. Trends Microbiol. 1993, 1, 66–69. [Google Scholar] [CrossRef]
- Pikovskaya, R.I. Mobilization of phosphorus in soil in connection with the vital activity of some microbial species. Mikrobiologial 1948, 17, 362–370. [Google Scholar]
- Premono, M.E.; Moawad, A.M.; Vlek, P.L.G. Effect of phosphate-solubilizing Pseudomonas putida on the growth of maize and its survival in the rhizosphere. Indones. J. Crop. Sci. 1996, 11, 13–23. [Google Scholar]
- Pande, A.; Pandey, P.; Mehra, S.; Singh, M.; Kaushik, S. Phenotypic and genotypic characterization of phosphate solubilizing bacteria and their efficiency on the growth of maize. J. Genet. Eng. Biotechnol. 2017, 15, 379–391. [Google Scholar] [CrossRef]
- Subba Rao, N.S. Soil Microorganisms and Plant Growth. Oxford and IBH Publishing Co. Pvt. Ltd.: New Delhi, India, 1977; pp. 63–65. [Google Scholar]
- Döbereiner, J. Isolation and identification of root associated diazotrophs. Plant. Soil 1988, 110, 207–212. [Google Scholar] [CrossRef]
- Ueda, T.; Suga, Y.; Yahiro, N.; Matsuguchi, T. Remarkable N2-fixing bacterial diversity detected in rice roots by molecular evolutionary analysis of nifH gene sequences. J. Bacteriol. 1995, 177, 1414–1417. [Google Scholar] [CrossRef]
- Kirk, P.L. Kjeldahl method for total nitrogen. Anal. Chem. 1950, 22, 354–358. [Google Scholar] [CrossRef]
- Din, M.; Nelofer, R.; Salman, M.; Khan, F.H.; Khan, A.; Ahmad, M.; Khan, M. Production of nitrogen fixing Azotobacter (SR-4) and phosphorus solubilizing Aspergillus niger and their evaluation on Lagenaria siceraria and Abelmoschus esculentus. Biotechnol. Rep. 2019, 22, e00323. [Google Scholar] [CrossRef]
- Shen, H.; He, X.; Liu, Y.; Chen, Y.; Tang, J.; Guo, T. A complex inoculant of N2-fixing, P-and K-solubilizing bacteria from a purple soil improves the growth of kiwifruit (Actinidia chinensis) plantlets. Front. Microbiol. 2016, 7, 841. [Google Scholar] [CrossRef]
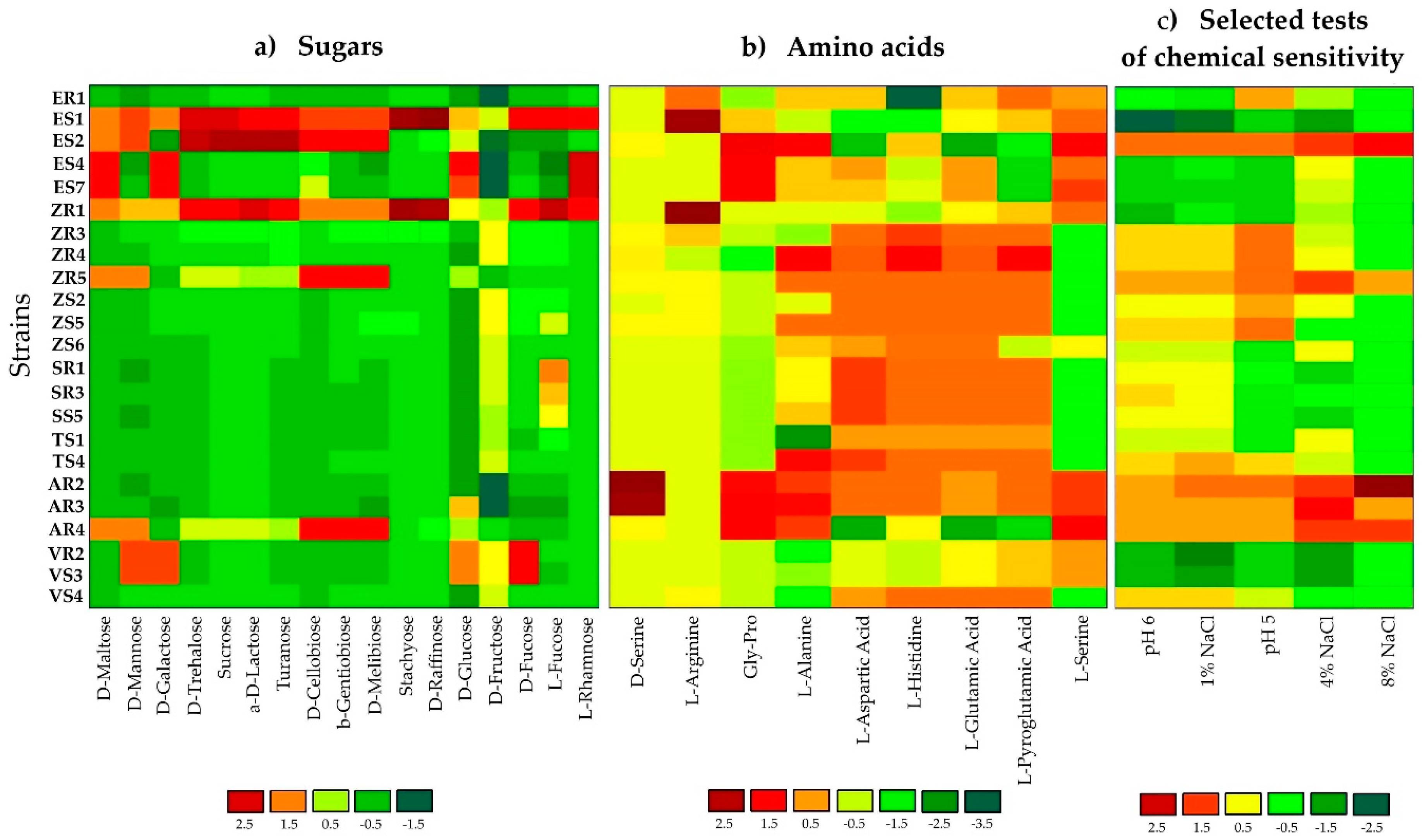
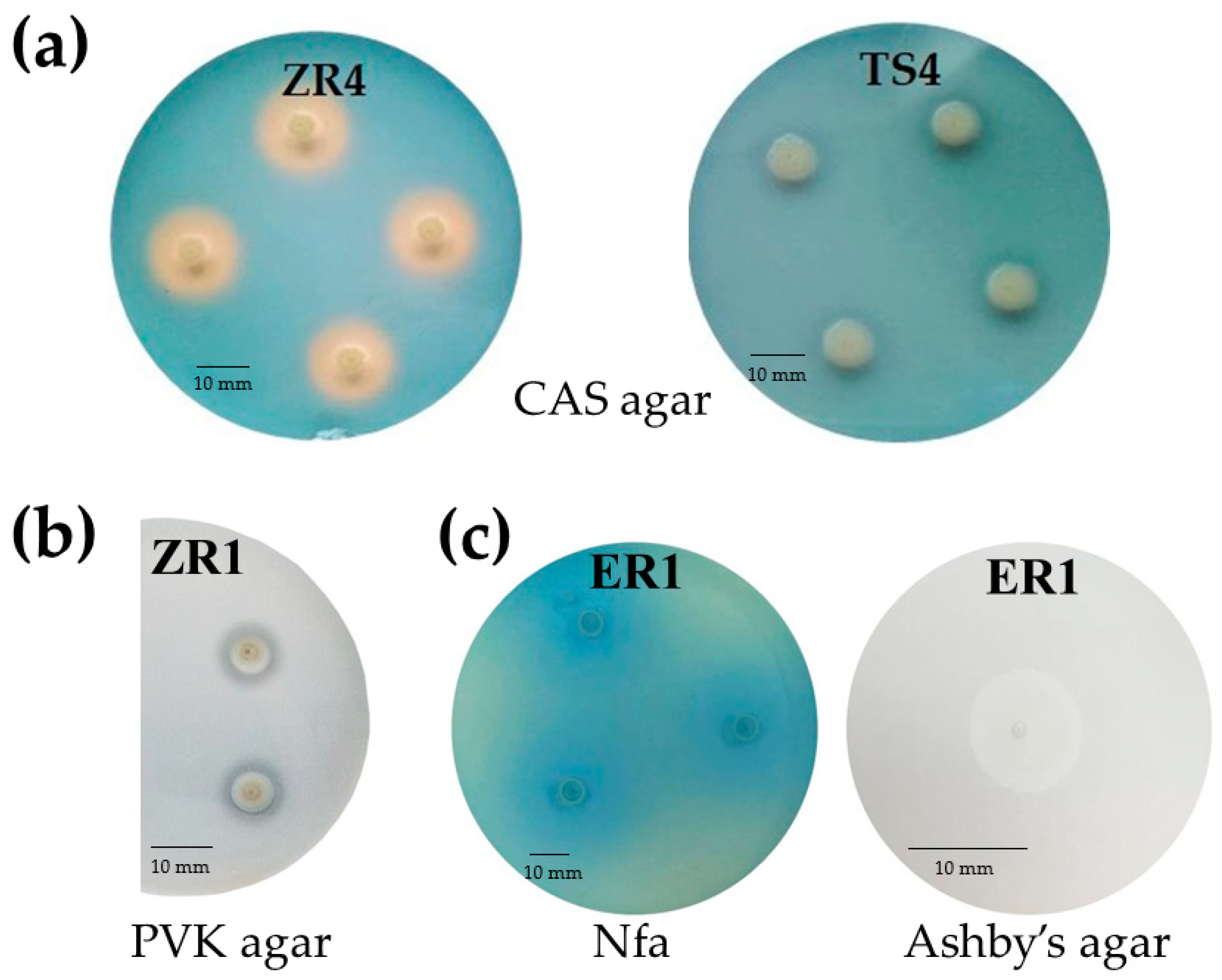
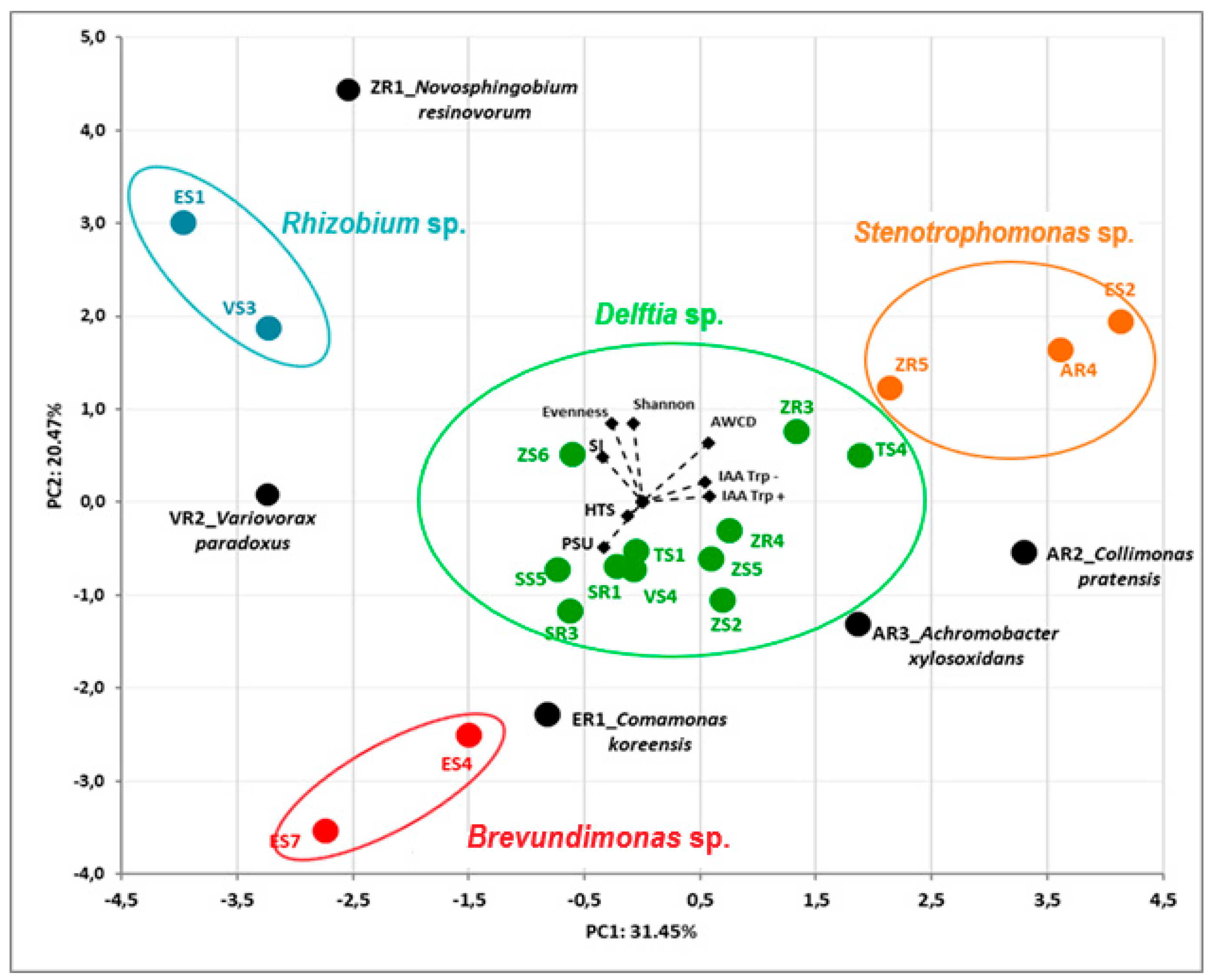
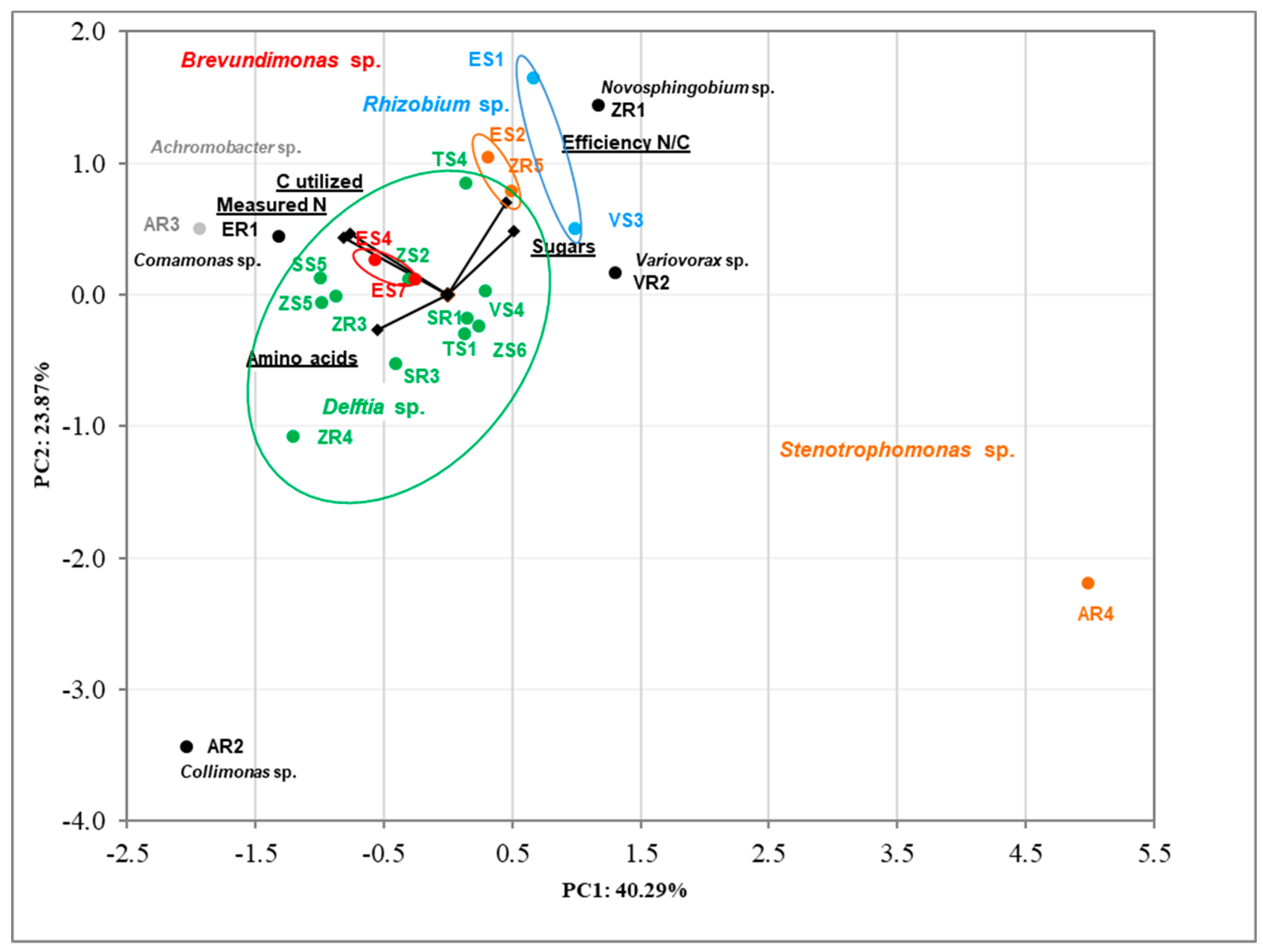
| Isolated from | Identification and Functional Diversity Indices | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Phylum | Species | Strain | GenBank Accession no. | 16S rRNA | Biolog GEN III | |||||
| Closest known Relative | Identification Based on Biolog GEN III | Similarity | AWCD | Shannon Diversity (H′) | Shannon Evenness (E) | |||||
| Monilophyta | Equisetum arvense | ER1 | KY486814 | Comamonas koreensis | Bordetella trematum | 0.68 ± 0.03 | 92.58 ± 0.81 | 3.93 ± 0.01 | 0.92 ± 0.00 | |
| ES1 | KY486815 | Rhizobium sp. | Rhizobium radiobacter | 0.54 ± 0.03 | 113.88 ± 0.07 | 4.30 ± 0.00 | 0.97 ± 0.00 | |||
| ES2 | KY486848 | Stenotrophomonas maltophilia | Stenotrophomonas maltophilia | 0.65 ± 0.07 | 139.85 ± 0.30 | 4.08 ± 0.00 | 0.93 ± 0.00 | |||
| ES4 | KY486828 | Brevundimonas sp. | Brevundimonas vesicularis | 0.58 ± 0.03 | 77.60 ± 0.51 | 3.75 ± 0.01 | 0.90 ± 0.01 | |||
| ES7 | KY486816 | Brevundimonas sp. | Brevundimonas vesicularis | 0.56 ± 0.04 | 81.86 ± 0.22 | 3.88 ± 0.00 | 0.90 ± 0.00 | |||
| Spermatophyta | Monocots | Zea mays | ZR1 | KY486807 | Novosphingobium resinovorum | Rhizobium radiobacter | 0.53 ± 0.00 | 132.17 ± 0.05 | 4.34 ± 0.00 | 0.98 ± 0.00 |
| ZR3 | KY486832 | Delftia acidovorans | Delftia acidovorans | 0.57 ± 0.03 | 130.32 ± 1.00 | 4.18 ± 0.01 | 0.92 ± 0.00 | |||
| ZR4 | KY486833 | Delftia acidovorans | Delftia acidovorans | 0.71 ± 0.02 | 130.27 ± 0.11 | 4.11 ± 0.00 | 0.93 ± 0.00 | |||
| ZR5 | KY486808 | Stenotrophomonas sp. | Stenotrophomonas maltophilia | 0.66 ± 0.05 | 126.79 ± 0.95 | 4.09 ± 0.01 | 0.94 ± 0.00 | |||
| ZS2 | KY486834 | Delftia acidovorans | Delftia acidovorans | 0.76 ± 0.04 | 120.73 ± 0.47 | 4.07 ± 0.00 | 0.92 ± 0.00 | |||
| ZS5 | KY486835 | Delftia acidovorans | Delftia acidovorans | 0.54 ± 0.01 | 126.73 ± 0.12 | 4.12 ± 0.00 | 0.92 ± 0.00 | |||
| ZS6 | KY486831 | Delftia sp. | Delftia acidovorans | 0.71 ± 0.02 | 115.90 ± 0.94 | 4.05 ± 0.00 | 0.93 ± 0.00 | |||
| Secale cereale | SR1 | KY486822 | Delftia sp. | Delftia acidovorans | 0.65 ± 0.01 | 112.13 ± 0.14 | 3.97 ± 0.00 | 0.94 ± 0.00 | ||
| SR3 | KY486810 | Delftia acidovorans | Delftia acidovorans | 0.65 ± 0.00 | 113.33 ± 0.54 | 3.98 ± 0.00 | 0.94 ± 0.00 | |||
| SS5 | KY486813 | Delftia acidovorans | Delftia acidovorans | 0.67 ± 0.03 | 110.89 ± 0.25 | 3.96 ± 0.00 | 0.94 ± 0.00 | |||
| Triticum aestivum | TS1 | KY486817 | Delftia acidovorans | Delftia acidovorans | 0.62 ± 0.05 | 101.44 ± 0.17 | 3.93 ± 0.00 | 0.94 ± 0.00 | ||
| TS4 | KY486820 | Delftia acidovorans | Delftia acidovorans | 0.74 ± 0.02 | 120.32 ± 0.02 | 4.04 ± 0.00 | 0.95 ± 0.00 | |||
| Eudicots | Arctium lappa | AR2 | KY486811 | Collimonas pratensis | Achromobacter denitrificans/ruhlandii | 0.72 ± 0.01 | 118.07 ± 0.47 | 3.97 ± 0.00 | 0.93 ± 0.00 | |
| AR3 | KY486824 | Achromobacter xylosoxidans | Achromobacter ruhlandii/denitrificans | 0.72 ± 0.01 | 118.10 ± 0.59 | 3.96 ± 0.00 | 0.93 ± 0.00 | |||
| AR4 | KY486847 | Stenotrophomonas maltophilia | Stenotrophomonas maltophilia | 0.74 ± 0.02 | 126.72 ± 1.15 | 4.09 ± 0.01 | 0.94 ±0.00 | |||
| Vicia faba | VR2 | KY486805 | Variovorax paradoxus | Acidovorax cattleyae | 0.57 ± 0.03 | 102.86 ± 0.87 | 4.05 ± 0.00 | 0.95 ± 0.00 | ||
| VS3 | KY486825 | Rhizobium sp. | Acidovorax cattleyae | 0.57 ± 0.03 | 103.61 ± 0.65 | 4.07 ± 0.00 | 0.96 ± 0.00 | |||
| VS4 | KY486829 | Delftia acidovorans | Delftia acidovorans | 0.64 ± 0.02 | 119.92 ± 0.15 | 4.08 ± 0.00 | 0.92 ± 0.00 | |||
| Strain | IAA-like Compounds Production (µg/mL) | Siderophores Production | SI | |||||
|---|---|---|---|---|---|---|---|---|
| Trp+ | Trp- | Sid-CAS | Sid-CAS (psu) | HTS | CTS | |||
| ER1 | C. koreensis | 8.72 ± 0.22 | 1.69 ± 0.04 | +++ | 72.52 ± 0.22 | 1.45 ± 0.41 | 0.58 ± 0.09 | 0.00 |
| ES1 | Rhizobium sp. | 0.75 ± 0.03 | 0.67 ± 0.04 | + | 34.57 ± 0.22 | 2.02 ± 0.07 | 0.01 ± 0.00 | 0.00 |
| ES2 | S. maltophilia | 19.18 ± 0.20 | 4.26 ± 0.15 | - | 6.09 ± 0.08 | 0.38 ± 0.047 | 0.00 | 0.00 |
| ES4 | Brevundimonas sp. | 6.51 ± 0.10 | 0.83 ± 0.03 | + | 25.10 ± 0.06 | 0.68 ± 0.01 | 0.00 | 0.00 |
| ES7 | Brevundimonas sp. | 1.70 ± 0.03 | 0.35 ± 0.04 | ++ | 55.70 ± 0.08 | 0.38 ± 0.03 | 1.93 ± 0.03 | 0.00 |
| ZR1 | N. resinovorum | 0.22 ± 0.03 | 0.17 ± 0.01 | + | 23.69 ± 0.10 | 0.33 ± 0.51 | 0.12 ± 0.01 | 2.82 ± 0.11 |
| ZR3 | D. acidovorans | 2.65 ± 0.13 | 0.94 ± 0.07 | − | 00.00 | 00.00 | 00.00 | 0.00 |
| ZR4 | D. acidovorans | 1.73 ± 0.17 | 0.73 ± 0.03 | ++ | 60.26 ± 0.15 | 2.04 ± 0.17 | 0.19 ± 0.02 | 0.00 |
| ZR5 | Stenotrophomonas sp. | 8.27 ± 0.15 | 0.78 ± 0.02 | − | 0.00 | 0.00 | 0.00 | 0.00 |
| ZS2 | D. acidovorans | 10.43 ± 0.31 | 0.77 ± 0.04 | +++ | 81.62 ± 0.23 | 0.51 ± 0.20 | 0.49 ± 0.05 | 0.00 |
| ZS5 | D. acidovorans | 5.58 ± 0.17 | 0.92 ± 0.02 | +++ | 77.92 ± 0.07 | 1.02 ± 0.32 | 0.28 ± 0.02 | 0.00 |
| ZS6 | Delftia sp. | 2.66 ± 0.27 | 1.07 ± 0.05 | - | 36.84 ± 0.13 | 0.21 ± 0.04 | 0.00 | 4.1 ± 0.11 |
| SR1 | Delftia sp. | 14.53 ± 0.31 | 1.39 ± 0.05 | ++ | 69.87 ± 0.10 | 0.000 | 0.11 ± 0.01 | 0.00 |
| SR3 | D. acidovorans | 2.41 ± 0.08 | 1.27 ± 0.03 | ++ | 44.74 ± 0.16 | 0.23 ± 0.06 | 1.00 ± 0.04 | 0.00 |
| SS5 | D. acidovorans | 5.87 ± 0.09 | 0.95 ± 0.04 | ++ | 43.31 ± 0.19 | 1.24 ± 0.11 | 0.27 ± 0.06 | 0.00 |
| TS1 | D. acidovorans | 9.62 ± 0.14 | 0.94 ± 0.02 | − | 0.00 | 0.00 | 0.00 | 0.00 |
| TS4 | D. acidovorans | 22.51 ± 0.48 | 1.23 ± 0.02 | − | 0.00 | 0.00 | 0.00 | 0.00 |
| AR2 | C. pratensis | 2.68 ± 0.08 | 0.46 ± 0.02 | − | 3.45 ± 0.20 | 0.26 ± 0.06 | 0.00 | 0.00 |
| AR3 | A. xylosoxidans | 0.13 ± 0.01 | 0.00 | ++ | 40.31 ± 0.20 | 2.11 ± 0.40 | 0.00 | 0.00 |
| AR4 | S. maltophilia | 20.60 ± 0.37 | 4.35 ± 0.08 | − | 0.00 | 0.00 | 0.00 | 0.00 |
| VR2 | V. paradoxus | 1.26 ± 0.07 | 0.44 ± 0.03 | ++ | 59.64 ± 0.21 | 0.00 | 0.19 ± 0.03 | 0.00 |
| VS3 | Rhizobium sp. | 0.56 ± 0.01 | 0.75 ± 0.03 | − | 0.00 | 0.00 | 0.00 | 2.18 ± 0.06 |
| VS4 | D. acidovorans | 1.02 ± 0.76 | 0.76 ± 0.03 | +++ | 71.83 ± 0.15 | 0.00 | 0.11 ± 0.01 | 0.00 |
| Strain No. | Growth | nifH Gene | N Concentrations and C Utilization; Efficiency Factor Obtained by N/C | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 24 h | 48 h | 72 h | ||||||||||
| Nfa | Ashby agar | Measured N | C utilized | Efficiency N/C | Measured N | C utilized | Efficiency N/C | Measured N | C utilized | Efficieny N/C | ||
| ER1 | +++ | ++ | + | 1.84 ± 0.15 | 0.13 ± 0.01 | 14.15 | 2.85 ± 0.05 | 0.15 ± 0.01 | 19.00 | 3.44 ± 0.23 | 0.15 ± 0.03 | 22.93 |
| ES1 | +++ | +++ | + | 2.51 ± 0.15 | 0.14 ± 0.01 | 17.93 | 3.65 ± 0.08 | 0.15 ± 0 | 24.33 | 4.80 ± 0.21 | 0.16 ± 0 | 30.00 |
| ES2 | +++ | ++ | + | 2.74 ± 0.15 | 0.15 ± 0.01 | 18.27 | 4.14 ± 0.13 | 0.16 ± 0 | 25.88 | 4.34 ± 0.07 | 0.16 ± 0 | 27.13 |
| ES4 | ++ | + | + | 2.28 ± 0.07 | 0.15 ± 0.01 | 15.20 | 2.61 ± 0.19 | 0.15 ± 0.01 | 17.40 | 3.81 ± 0.16 | 0.16 ± 0.01 | 23.81 |
| ES7 | ++ | + | + | 2.26 ± 0.08 | 0.14 ± 0.02 | 16.14 | 3.23 ± 0.10 | 0.15 ± 0.02 | 21.53 | 4.01 ± 0.19 | 0.16 ± 0.01 | 25.06 |
| ZR1 | +++ | +++ | + | 3.53 ± 0.14 | 0.12 ± 0.02 | 29.42 | 4.26 ± 0.03 | 0.13 ± 0.02 | 32.77 | 5.16 ± 0.17 | 0.15 ± 0 | 34.40 |
| ZR3 | ++ | ++ | + | 1.93 ± 0.08 | 0.12 ± 0.02 | 16.08 | 2.63 ± 0.25 | 0.14 ± 0.03 | 18.79 | 3.28 ± 0.21 | 0.15 ± 0.02 | 21.87 |
| ZR4 | + | + | + | 0.00 | 0.00 | 0.00 | 1.37 ± 0.14 | 0.14 ± 0.01 | 9.79 | 1.85 ± 0.07 | 0.16 ± 0.01 | 11.56 |
| ZR5 | +++ | ++ | + | 2.35 ± 0.09 | 0.13 ± 0.02 | 18.08 | 3.17 ± 0.08 | 0.13 ± 0.02 | 24.38 | 3.69 ± 0.33 | 0.13 ± 0.02 | 28.38 |
| ZS2 | +++ | ++ | + | 2.96 ± 0.09 | 0.14 ± 0.01 | 21.14 | 3.11 ± 0.01 | 0.14 ± 0 | 22.21 | 3.85 ± 0.15 | 0.14 ± 0 | 27.50 |
| ZS5 | ++ | ++ | + | 2.61 ± 0.07 | 0.14 ± 0.01 | 19.64 | 2.88 ± 0.09 | 0.14 ± 0.01 | 20.57 | 3.39 ± 0.25 | 0.15 ± 0.01 | 22.60 |
| ZS6 | +++ | ++ | + | 2.53 ± 0.09 | 0.12 ± 0.01 | 21.08 | 3.27 ± 0.09 | 0.14 ± 0.01 | 23.36 | 4.01 ± 0.09 | 0.15 ± 0.01 | 26.73 |
| SR1 | +++ | ++ | + | 3.25 ± 0.15 | 0.15 ± 0.01 | 21.67 | 3.99 ± 0.15 | 0.16 ± 0.01 | 24.94 | 4.73 ± 0.15 | 0.17 ± 0 | 27.82 |
| SR3 | ++ | + | + | 1.82 ± 0.07 | 0.14 ± 0.02 | 13.00 | 2.56 ± 0.07 | 0.15 ± 0.01 | 17.07 | 3.30 ± 0.07 | 0.16 ± 0.01 | 20.63 |
| SS5 | ++ | + | + | 2.39 ± 0.04 | 0.13 ± 0.01 | 18.38 | 3.27 ± 0.24 | 0.15 ± 0.01 | 21.80 | 3.69 ± 0.38 | 0.16 ± 0.01 | 23.06 |
| TS1 | ++ | + | + | 2.12 ± 0.07 | 0.13 ± 0.02 | 16.31 | 2.99 ± 0.2 | 0.14 ± 0.01 | 21.36 | 3.33 ± 0.21 | 0.14 ± 0.01 | 23.79 |
| TS4 | +++ | +++ | + | 4.53 ± 0.13 | 0.13 ± 0.02 | 34.85 | 5.43 ± 0.12 | 0.14 ± 0.01 | 38.79 | 5.91 ± 0.07 | 0.15 ± 0.01 | 39.40 |
| AR2 | + | + | - | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| AR3 | +++ | +++ | + | 3.34 ± 0.09 | 0.13 ± 0.01 | 25.69 | 5.02 ± 0.08 | 0.15 ± 0.02 | 33.47 | 5.99 ± 0.08 | 0.16 ± 0 | 37.44 |
| AR4 | +++ | ++ | + | 3.94 ± 0.05 | 0.14 ± 0.01 | 28.14 | 4.30 ± 0.08 | 0.15 ± 0.01 | 28.66 | 4.86 ± 0.10 | 0.15 ± 0.01 | 32.40 |
| VR2 | +++ | +++ | + | 3.22 ± 0.05 | 0.13 ± 0.02 | 24.77 | 3.81 ± 0.05 | 0.14 ± 0 | 27.21 | 4.68 ± 0.05 | 0.15 ± 0 | 31.20 |
| VS3 | +++ | +++ | + | 3.48± 0.14 | 0.13 ± 0.01 | 26.77 | 4.07 ± 0.14 | 0.13 ± 0.01 | 31.31 | 4.94 ± 0.014 | 0.15 ± 0 | 32.93 |
| VS4 | +++ | +++ | + | 2.77 ± 0.06 | 0.13 ± 0.01 | 21.31 | 3.40 ± 0.06 | 0.13 ± 0.01 | 26.15 | 4.30 ± 0.06 | 0.15 ± 0 | 28.67 |
| Parameters | AWCD | E | H′ | IAA Trp- | IAA Trp+ | SI | psu | HTS | CTS | pH 6 | pH 5 | 1% NaCl | 4% NaCl | 8% NaCl | Sugars | Amino Acids | Efficiency N/C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AWCD | 0.11 | 0.80 * | 0.05 | 0.07 | 0.07 | −0.17 | −0.13 | −0.22 | 0.61 * | 0.66 * | 0.67 * | 0.37 | 0.52 * | 0.3 | 0.22 | 0.08 | |
| E | 0.11 | 0.31 | −0.11 | −0.15 | 0.29 | −0.46 | −0.29 | −0.26 | −0.14 | −0.30 | −0.13 | −0.36 | −0.39 | 0.27 | −0.13 | 0.48 | |
| H′ | 0.80 * | 0.31 | −0.07 | −0.18 | 0.23 | −0.10 | −0.18 | −0.07 | 0.14 | 0.30 | 0.18 | −0.11 | 0.13 | 0.59 | 0.09 | 0.17 | |
| IAA Trp- | 0.05 | −0.11 | −0.07 | 0.78 * | −0.16 | −0.10 | −0.24 | −0.10 | 0.38 | 0.28 | 0.27 | 0.02 | 0.18 | −0.14 | −0.47 | −0.18 | |
| IAA Trp+ | 0.07 | −0.15 | −0.18 | 0.78 * | −0.37 | −0.13 | −0.17 | −0.18 | 0.44 | 0.36 | 0.39 | 0.33 | 0.36 | −0.17 | −0.37 | −0.15 | |
| SI | 0.07 | 0.29 | 0.23 | −0.16 | −0.37 | −0.21 | −0.20 | −0.19 | −0.40 | −0.34 | −0.34 | −0.174 | −0.45 | 0.16 | −0.03 | 0.32 | |
| psu | −0.17 | −0.46 | −0.10 | −0.10 | −0.13 | −0.21 | 0.39 | 0.81 * | −0.25 | 0.01 | −0.25 | −0.29 | −0.12 | −0.20 | 0.16 | −0.17 | |
| HTS | −0.13 | −0.29 | −0.18 | −0.24 | −0.17 | −0.20 | 0.39 | 0.29 | −0.17 | 0.03 | −0.14 | 0.19 | 0.01 | −0.17 | 0.49 | −0.31 | |
| CTS | −0.22 | −0.26 | −0.07 | −0.1 | −0.18 | −0.19 | 0.81 * | 0.29 | −0.36 | −0.22 | −0.37 | −0.45 | −0.21 | −0.02 | 0.10 | −0.23 | |
| pH 6 | 0.61 * | −0.14 | 0.14 | 0.38 | 0.44 | −0.40 | −0.25 | −0.17 | −0.36 | 0.80 * | 0.97 * | 0.66 * | 0.81 * | −0.19 | 0.05 | −0.16 | |
| pH 5 | 0.66 * | −0.30 | 0.30 | 0.28 | 0.36 | −0.34 | 0.01 | 0.03 | −0.22 | 0.80 * | 0.80 * | 0.54 | 0.78 * | −0.22 | 0.04 | −0.30 | |
| 1% NaCl | 0.67 * | −0.13 | 0.18 | 0.27 | 0.39 | −0.34 | −0.25 | −0.14 | −0.37 | 0.97 * | 0.80 * | 0.68 * | 0.82 * | −0.224 | 0.13 | −0.10 | |
| 4% NaCl | 0.37 | −0.36 | −0.11 | 0.02 | 0.33 | −0.17 | −0.29 | 0.19 | −0.45 | 0.66 * | 0.54 | 0.68 * | 0.73 * | −0.14 | 0.14 | −0.14 | |
| 8% NaCl | 0.52 | −0.39 | 0.13 | 0.18 | 0.36 | −0.45 | −0.12 | 0.01 | −0.21 | 0.81 * | 0.78 * | 0.82 * | 0.73 * | −0.08 | 0.124 | −0.18 | |
| Sugars | 0.30 | 0.27 | 0.59 | −0.14 | −0.17 | 0.16 | −0.20 | −0.17 | −0.02 | −0.19 | −0.22 | −0.22 | −0.14 | −0.08 | −0.07 | 0.29 | |
| Amino acids | 0.22 | −0.13 | 0.09 | −0.47 | −0.37 | −0.03 | 0.16 | 0.49 | 0.10 | 0.05 | 0.04 | 0.13 | 0.14 | 0.12 | −0.07 | −0.10 | |
| Efficiency N/C | 0.08 | 0.48 | 0.17 | −0.18 | −0.15 | 0.32 | −0.17 | −0.31 | −0.23 | −0.16 | −0.30 | −0.10 | −0.14 | −0.18 | 0.29 | −0.10 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Woźniak, M.; Gałązka, A.; Tyśkiewicz, R.; Jaroszuk-Ściseł, J. Endophytic Bacteria Potentially Promote Plant Growth by Synthesizing Different Metabolites and their Phenotypic/Physiological Profiles in the Biolog GEN III MicroPlateTM Test. Int. J. Mol. Sci. 2019, 20, 5283. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms20215283
Woźniak M, Gałązka A, Tyśkiewicz R, Jaroszuk-Ściseł J. Endophytic Bacteria Potentially Promote Plant Growth by Synthesizing Different Metabolites and their Phenotypic/Physiological Profiles in the Biolog GEN III MicroPlateTM Test. International Journal of Molecular Sciences. 2019; 20(21):5283. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms20215283
Chicago/Turabian StyleWoźniak, Małgorzata, Anna Gałązka, Renata Tyśkiewicz, and Jolanta Jaroszuk-Ściseł. 2019. "Endophytic Bacteria Potentially Promote Plant Growth by Synthesizing Different Metabolites and their Phenotypic/Physiological Profiles in the Biolog GEN III MicroPlateTM Test" International Journal of Molecular Sciences 20, no. 21: 5283. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms20215283






