Interactive Effects of Salicylic Acid and Nitric Oxide in Enhancing Rice Tolerance to Cadmium Stress
Abstract
:1. Introduction
2. Results
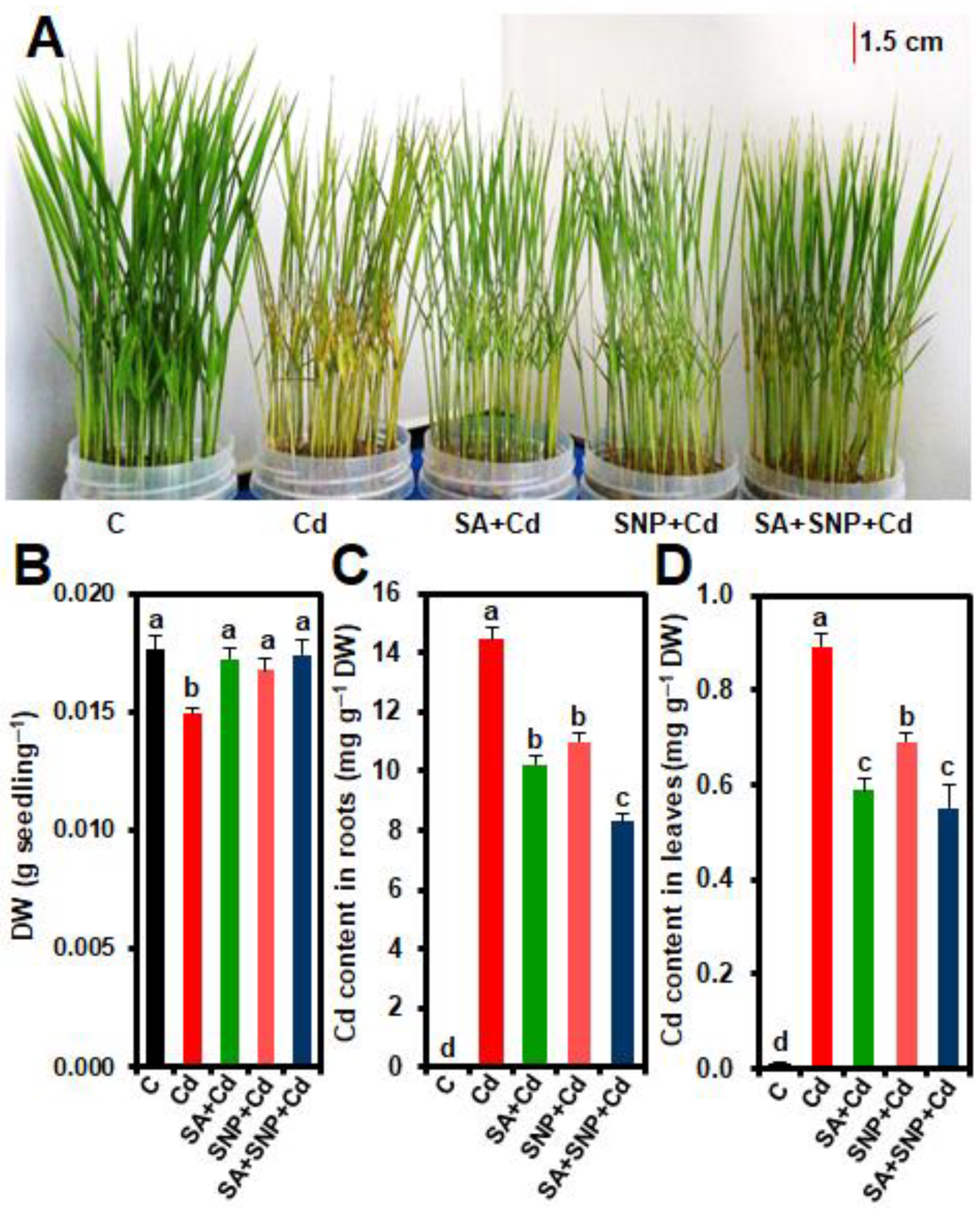
2.1. Effects of SA and/or SNP Treatments on Phenotypic Appearance, Biomass, and Cd Homeostasis
2.2. Effects of SA and/or SNP Treatments on Photosynthetic Pigment Contents, Water Status, and Proline (Pro) Accumulation
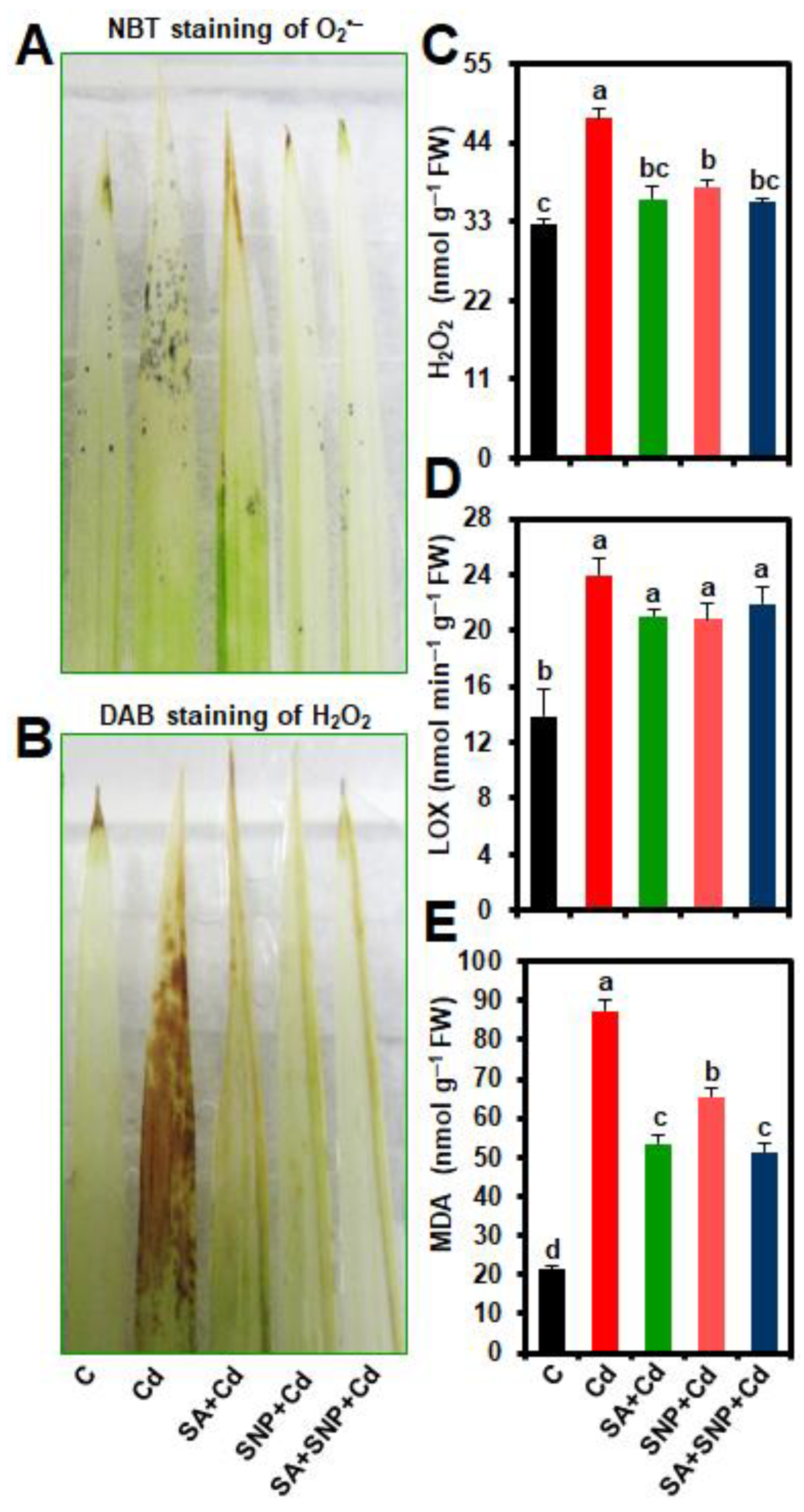
2.3. Effects of SA and/or SNP Treatments on ROS Accumulation, and the Levels of Hydrogen Peroxide (H2O2), Lipoxygenase (LOX) activity, and Malondialdehyde (MDA)
2.4. Effects of SA and/or SNP Treatments on the Levels of Ascorbic Acid (AsA), Dehydroascorbate (DHA), glutathione (GSH) and Oxidized Glutathione (GSSG), and Redox Status
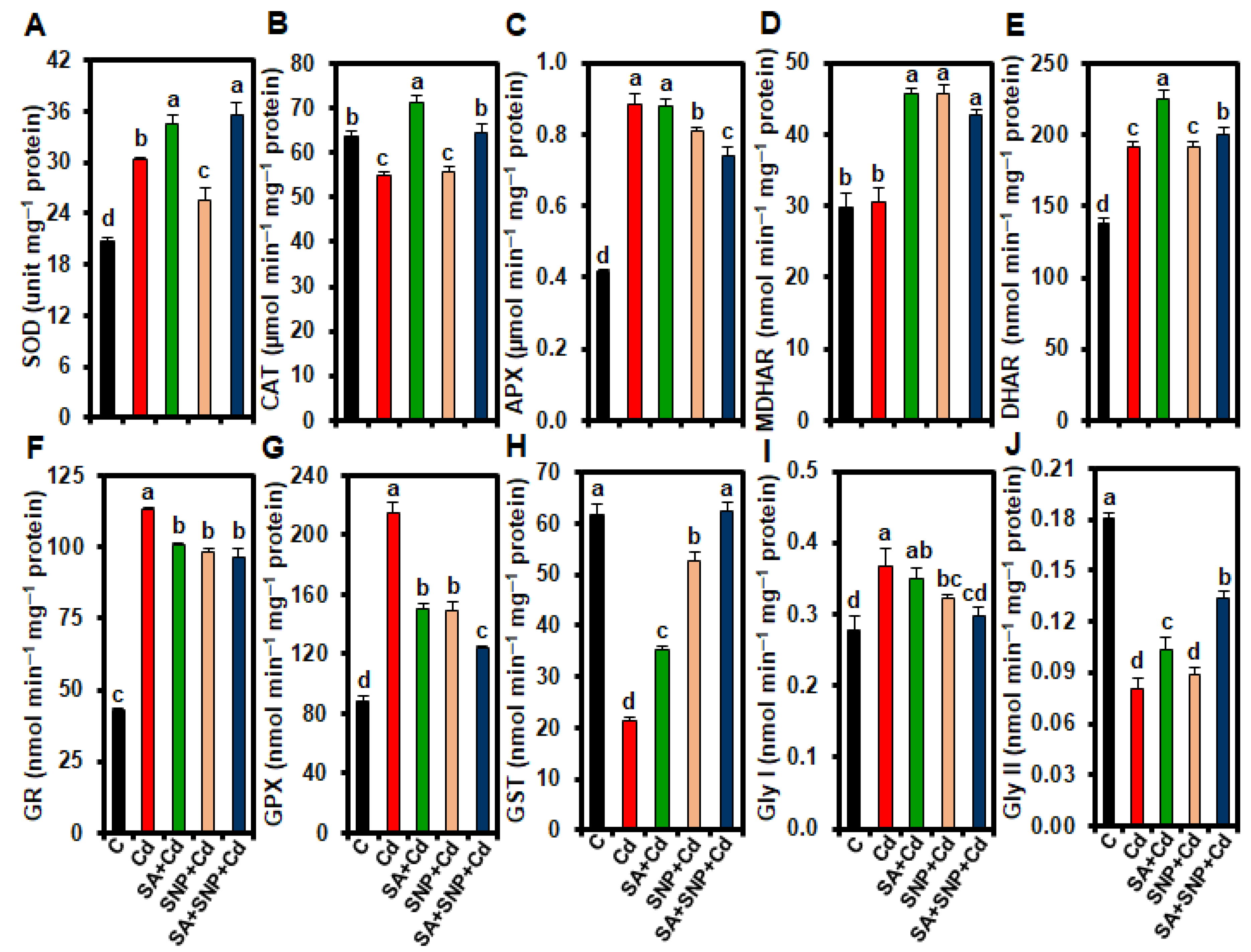
2.5. Effect of SA and/or SNP Treatments on the Activities of Antioxidant Enzymes
3. Discussion
4. Materials and Methods
4.1. Plant Materials, Growth Conditions, and Treatments
4.2. Assessment of Plant Biomass, and Quantification of Cd Contents in Leaves and Roots
4.3. Photosynthetic Pigment Contents, Relative Water Content, and Proline Content
4.4. Hydrogen Peroxide and Malondialdehyde Levels
4.5. Histochemical Analyses of Superoxide and Hydrogen Peroxide
4.6. Extraction and Estimation of Non-Enzymatic Antioxidants
4.7. Enzyme Extraction and Assessments of Enzyme Activities
4.8. Determination of Total Soluble Protein Contents
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AsA | Ascorbic acid |
| APX | Ascorbate peroxidase |
| CAT | Catalase |
| Chl | Chlorophyll |
| DHA | Dehydroascorbic acid |
| DHAR | Dehydroascorbate reductase |
| DW | Dry weight |
| FW | Fresh weight |
| Gly | Glyoxalase |
| GSH | Reduced glutathione |
| GSSG | Oxidized glutathione |
| GST | Glutathione S-transferase |
| GPX | Glutathione peroxidase |
| GR | Glutathione reductase |
| LOX | Lipoxygenase |
| MDA | Malondialdehyde |
| MDHAR | Monodehydroascorbate reductase |
| MG | Methylglyoxal |
| NO | Nitric oxide |
| Pro | Proline |
| ROS | Reactive oxygen species |
| RWC | Relative water content |
| SA | Salicylic acid |
| SNP | Sodium nitroprusside |
| SOD | Superoxide dismutase |
References
- Mishra, S.; Bharagava, R.N.; More, N.; Yadav, A.; Zainith, S.; Mani, S.; Chowdhary, P. Heavy metal contamination: An alarming threat to environment and human health. In Environmental Biotechnology: For Sustainable Future; Sobti, R.C., Arora, N.K., Kothari, R., Eds.; Springer: Singapore, 2019; pp. 103–125. [Google Scholar]
- Khan, M.A.; Khan, S.; Khan, A.; Alam, M. Soil contamination with cadmium, consequences and remediation using organic amendments. Sci. Total Environ. 2017, 601–602, 1591–1605. [Google Scholar] [CrossRef] [PubMed]
- Huybrechts, M.; Cuypers, A.; Deckers, J.; Iven, V.; Vandionant, S.; Jozefczak, M.; Hendrix, S. Cadmium and plant development: An agony from seed to seed. Int. J. Mol. Sci. 2019, 20, 3971. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://bdnews24.com/health/2013/06/22/too-much-cadmium-in-bangla-rice-us-study (accessed on 30 October 2019).
- White, P.J.; Brown, P.H. Plant nutrition for sustainable development and global health. Ann. Bot. 2010, 105, 1073–1080. [Google Scholar] [CrossRef] [PubMed]
- Rizwan, M.; Ali, S.; Adrees, M.; Rizvi, H.; Zia-ur-Rehman, M.; Hannan, F.; Qayyum, M.F.; Hafeez, F.; Ok, Y.S. Cadmium stress in rice: Toxic effects, tolerance mechanisms, and management: A critical review. Environ. Sci. Pollut. Res. 2016, 23, 17859–17879. [Google Scholar] [CrossRef]
- Semane, B.; Cuypers, A.; Smeets, K.; Van Belleghem, F.; Horemans, N.; Schat, H.; Vangronsveld, J. Cadmium responses in Arabidopsis thaliana: Glutathione metabolism and antioxidative defence system. Physiol. Plant. 2007, 129, 519–528. [Google Scholar] [CrossRef]
- Mostofa, M.G.; Rahman, A.; Ansary, M.M.U.; Watanabe, A.; Fujita, M.; Tran, L.-S.P. Hydrogen sulfide modulates cadmium-induced physiological and biochemical responses to alleviate cadmium toxicity in rice. Sci. Rep. 2015, 5, 14078. [Google Scholar] [CrossRef]
- Zhang, F.; Zhang, H.; Wang, G.; Xu, L.; Shen, Z. Cadmium-induced accumulation of hydrogen peroxide in the leaf apoplast of Phaseolus aureus and Vicia sativa and the roles of different antioxidant enzymes. J. Hazard. Mater. 2009, 168, 76–84. [Google Scholar] [CrossRef]
- Gonçalves, J.F.; Tabaldi, L.A.; Cargnelutti, D.; Pereira, L.B.; Maldaner, J.; Becker, A.G.; Rossato, L.V.; Rauber, R.; Bagatini, M.D.; Bisognin, D.A.; et al. Cadmium-induced oxidative stress in two potato cultivars. Biometals 2009, 22, 779–792. [Google Scholar] [CrossRef]
- Das, K.; Roychoudhury, A. Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Front. Environ. Sci. 2014, 2. [Google Scholar] [CrossRef]
- Mostofa, M.G.; Ghosh, A.; Li, Z.-G.; Siddiqui, M.N.; Fujita, M.; Tran, L.-S.P. Methylglyoxal—A signaling molecule in plant abiotic stress responses. Free Radic. Biol. Med. 2018, 122, 96–109. [Google Scholar] [CrossRef]
- Per, T.S.; Masood, A.; Khan, N.A. Nitric oxide improves S-assimilation and GSH production to prevent inhibitory effects of cadmium stress on photosynthesis in mustard (Brassica juncea L.). Nitric Oxide 2017, 68, 111–124. [Google Scholar] [CrossRef] [PubMed]
- Nabi, R.B.S.; Tayade, R.; Hussain, A.; Kulkarni, K.P.; Imran, Q.M.; Mun, B.-G.; Yun, B.W. Nitric oxide regulates plant responses to drought, salinity, and heavy metal stress. Environ. Exp. Bot. 2019, 161, 120–133. [Google Scholar] [CrossRef]
- Mostofa, M.G.; Fujita, M. Salicylic acid alleviates copper toxicity in rice (Oryza sativa L.) seedlings by up-regulating antioxidative and glyoxalase systems. Ecotoxicology 2013, 22, 959–973. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, F.; Singh, A.; Kamal, A. Chapter 23—Salicylic acid–mediated defense mechanisms to abiotic stress tolerance. In Plant Signaling Molecules; Khan, M.I.R., Reddy, P.S., Ferrante, A., Khan, N.A., Eds.; Woodhead Publishing: Sawston, UK, 2019; pp. 355–369. [Google Scholar]
- Khan, M.I.R.; Fatma, M.; Per, T.S.; Anjum, N.A.; Khan, N.A. Salicylic acid-induced abiotic stress tolerance and underlying mechanisms in plants. Front. Plant Sci. 2015, 6, 462. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Zhang, T.; Zhang, W.; Su, C.; Yang, Y.; Hu, D.; Xu, Q. Alleviation of cadmium toxicity in Lemna minor by exogenous salicylic acid. Ecotoxicol. Environ. Saf. 2018, 147, 500–508. [Google Scholar] [CrossRef]
- Rai, K.K.; Pandey, N.; Rai, S.P. Salicylic acid and nitric oxide signaling in plant heat stress. Physiol. Plant. 2019. [Google Scholar] [CrossRef]
- Shao, R.X.; Xin, L.F.; Guo, J.M.; Zheng, H.F.; Mao, J.; Han, X.P.; Jia, L.; Jia, S.J.; Du, C.G.; Song, R.; et al. Salicylic acid-induced photosynthetic adaptability of Zea mays L. to polyethylene glycol-simulated water deficit is associated with nitric oxide signaling. Photosynthetica 2018, 56, 1370–1377. [Google Scholar] [CrossRef]
- Mostofa, M.G.; Fujita, M.; Tran, L.S.P. Nitric oxide mediates hydrogen peroxide- and salicylic acid-induced salt tolerance in rice (Oryza sativa L.) seedlings. Plant Growth Regul. 2015, 77, 265–277. [Google Scholar] [CrossRef]
- Dong, Y.J.; Wang, Z.L.; Zhang, J.W.; Liu, S.; He, Z.L.; He, M.R. Interaction effects of nitric oxide and salicylic acid in alleviating salt stress of Gossypium hirsutum L. J. Soil Sci. Plant Nutr. 2015, 15, 561–573. [Google Scholar]
- Sun, C.; Liu, L.; Lu, L.; Jin, C.; Lin, X. Nitric oxide acts downstream of hydrogen peroxide in regulating aluminum-induced antioxidant defense that enhances aluminum resistance in wheat seedlings. Environ. Exp. Bot. 2018, 145, 95–103. [Google Scholar] [CrossRef]
- Mostofa, M.G.; Seraj, Z.I.; Fujita, M. Exogenous sodium nitroprusside and glutathione alleviate copper toxicity by reducing copper uptake and oxidative damage in rice (Oryza sativa L.) seedlings. Protoplasma 2014, 251, 1373–1386. [Google Scholar] [CrossRef] [PubMed]
- Esim, N.; Atici, Ö. Effects of exogenous nitric oxide and salicylic acid on chilling-induced oxidative stress in wheat (Triticum aestivum). Front. Life Sci. 2015, 8, 124–130. [Google Scholar] [CrossRef]
- Naser Alavi, S.M.; Arvin, M.J.; Manoochehri Kalantari, K. Salicylic acid and nitric oxide alleviate osmotic stress in wheat (Triticum aestivum L.) seedlings. J. Plant Interact. 2014, 9, 683–688. [Google Scholar] [CrossRef]
- Hou, L.L.; Tong, T.; Tian, B.; Xue, D.W. Chapter 1—Crop yield and quality under cadmium stress. In Cadmium Tolerance in Plants; Hasanuzzaman, M., Vara Prasad, M.N., Nahar, K., Eds.; Academic Press: Cambridge, MA, USA, 2019; pp. 1–18. [Google Scholar]
- Asgher, M.; Khan, M.I.R.; Anjum, N.A.; Khan, N.A. Minimising toxicity of cadmium in plants—Role of plant growth regulators. Protoplasma 2015, 252, 399–413. [Google Scholar] [CrossRef]
- Chen, H.; Yang, X.; Wang, P.; Wang, Z.; Li, M.; Zhao, F.-J. Dietary cadmium intake from rice and vegetables and potential health risk: A case study in Xiangtan, southern China. Sci. Total Environ. 2018, 639, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Küpper, H.; Andresen, E. Mechanisms of metal toxicity in plants. Metallomics 2016, 8, 269–285. [Google Scholar] [CrossRef]
- Bączek-Kwinta, R.; Juzoń, K.; Borek, M.; Antonkiewicz, J. Photosynthetic response of cabbage in cadmium-spiked soil. Photosynthetica 2019, 57, 731–739. [Google Scholar] [CrossRef]
- Jan, S.; Alyemeni, M.N.; Wijaya, L.; Alam, P.; Siddique, K.H.; Ahmad, P. Interactive effect of 24-epibrassinolide and silicon alleviates cadmium stress via the modulation of antioxidant defense and glyoxalase systems and macronutrient content in Pisum sativum L. seedlings. BMC Plant Biol. 2018, 18, 146. [Google Scholar] [CrossRef]
- Wu, J.; Mock, H.-P.; Giehl, R.F.H.; Pitann, B.; Mühling, K.H. Silicon decreases cadmium concentrations by modulating root endodermal suberin development in wheat plants. J. Hazard. Mater. 2019, 364, 581–590. [Google Scholar] [CrossRef]
- Gondor, O.K.; Pál, M.; Darkó, É.; Janda, T.; Szalai, G. Salicylic acid and sodium salicylate alleviate cadmium toxicity to different extents in maize (Zea mays L.). PLoS ONE 2016, 11, e0160157. [Google Scholar] [CrossRef]
- Kusznierewicz, B.; Baczek-Kwinta, R.; Bartoszek, A.; Piekarska, A.; Huk, A.; Manikowska, A.; Antonkiewicz, J.; Namie´snik, J.; Konieczka, P. The dose-dependent influence of zinc and cadmium contamination of soil on their uptake and glucosinolate content in white cabbage (Brassica oleracea var. capitata f. alba). Environ. Toxicol. Chem. 2012, 31, 2482–2489. [Google Scholar] [CrossRef] [PubMed]
- Kaya, C.; Ashraf, M.; Alyemeni, M.N.; Ahmad, P. Responses of nitric oxide and hydrogen sulfide in regulating oxidative defence system in wheat plants grown under cadmium stress. Physiol. Plant. 2019. [Google Scholar] [CrossRef] [PubMed]
- Paunov, M.; Koleva, L.; Vassilev, A.; Vangronsveld, J.; Goltsev, V. Effects of different metals on photosynthesis: Cadmium and zinc affect chlorophyll fluorescence in durum wheat. Int. J. Mol. Sci. 2018, 19, 787. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmad, P.; Abd Allah, E.F.; Hashem, A.; Sarwat, M.; Gucel, S. Exogenous application of selenium mitigates cadmium toxicity in Brassica juncea L. (Czern & Cross) by up-regulating antioxidative system and secondary metabolites. J. Plant Growth Regul. 2016, 35, 936–950. [Google Scholar] [CrossRef]
- Kaur, G.; Asthir, B. Proline: A key player in plant abiotic stress tolerance. Biol. Plant 2015, 59, 609–619. [Google Scholar] [CrossRef]
- Metwally, A.; Finkemeier, I.; Georgi, M.; Dietz, K.-J. Salicylic acid alleviates the cadmium toxicity in barley seedlings. Plant Physiol. 2003, 132, 272–281. [Google Scholar] [CrossRef] [Green Version]
- Li, Q.; Wang, G.; Wang, Y.; Dan, Y.; Guan, C.; Ji, J. Foliar application of salicylic acid alleviate the cadmium toxicity by modulation the reactive oxygen species in potato. Ecotoxicol. Environ. Saf. 2019, 172, 317–325. [Google Scholar] [CrossRef]
- Ahmad, P.; Ahanger, M.A.; Alyemeni, M.N.; Wijaya, L.; Alam, P. Exogenous application of nitric oxide modulates osmolyte metabolism, antioxidants, enzymes of ascorbate-glutathione cycle and promotes growth under cadmium stress in tomato. Protoplasma 2018, 255, 79–93. [Google Scholar] [CrossRef]
- Bashir, H.; Ibrahim, M.M.; Bagheri, R.; Ahmad, J.; Arif, I.A.; Baig, M.A.; Qureshi, M.I. Influence of sulfur and cadmium on antioxidants, phytochelatins and growth in Indian mustard. AoB Plants 2015, 7, plv001. [Google Scholar] [CrossRef] [Green Version]
- Lou, L.; Kang, J.; Pang, H.; Li, Q.; Du, X.; Wu, W.; Chen, J.; Lv, J. Sulfur protects pakchoi (Brassica chinensis L.) seedlings against cadmium stress by regulating ascorbate-glutathione metabolism. Int. J. Mol. Sci. 2017, 18, 1628. [Google Scholar] [CrossRef] [Green Version]
- Arnon, D.I. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol. 1949, 24, 1–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lichtenthaler, H.K.; Wellburn, A.R. Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents. Biochem. Soc. Trans. 1983, 11, 591–592. [Google Scholar] [CrossRef] [Green Version]
- Bates, L.; Waldren, R.; Teare, I. Rapid determination of free proline for water-stress studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Heath, R.L.; Packer, L. Photoperoxidation in isolated chloroplasts: I. kinetics and stoichiometry of fatty acid peroxidation. Arch. Biochem. Biophys. 1968, 125, 189–198. [Google Scholar] [CrossRef]
- Mostofa, M.G.; Saegusa, D.; Fujita, M.; Tran, L.-S.P. Hydrogen sulfide regulates salt tolerance in rice by maintaining Na+/K+ balance, mineral homeostasis and oxidative metabolism under excessive salt stress. Front. Plant Sci. 2015, 6, 1055. [Google Scholar] [CrossRef] [Green Version]
- Griffith, O.W. Determination of glutathione and glutathione disulfide using glutathione reductase and 2-vinylpyridine. Anal. Biochem. 1980, 106, 207–212. [Google Scholar] [CrossRef]
- Doderer, A.; Kokkelink, I.; van der Veen, S.; Valk, B.E.; Schram, A.; Douma, A.C. Purification and characterization of two lipoxygenase isoenzymes from germinating barley. Biochim. Biophys. Acta Protein Struct. Mol. Enzymol. 1992, 1120, 97–104. [Google Scholar] [CrossRef]
- Hossain, M.A.; Nakano, Y.; Asada, K. Monodehydroascorbate reductase in spinach chloroplasts and its participation in regeneration of ascorbate for scavenging hydrogen peroxide. Plant Cell Physiol. 1984, 25, 385–395. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
| Treatments | Chl a (mg g−1 FW) | Chl b (mg g−1 FW) | Total Chls (mg g−1 FW) | Carotenoids (mg g−1 FW) | RWC (%) | Pro (µmol g−1 FW) |
|---|---|---|---|---|---|---|
| C | 2.54 ± 0.07 a | 0.70 ± 0.03 a | 3.24 ± 0.09 a | 0.73 ± 0.00 a | 98.78 ± 0.60 a | 0.08 ± 0.00 e |
| Cd | 1.86 ± 0.01 d | 0.27 ± 0.02 c | 2.13 ± 0.01 e | 0.59 ± 0.01 c | 71.88 ± 3.78 b | 0.47 ± 0.01 a |
| SA + Cd | 1.98 ± 0.03 b,c | 0.49 ± 0.01 b | 2.47 ± 0.04 c | 0.69 ± 0.00 b | 97.02 ± 0.52 a | 0.14 ± 0.01 d |
| SNP + Cd | 1.87 ± 0.01 c,d | 0.46 ± 0.02 b | 2.33 ± 0.02 d | 0.60 ± 0.01 c | 75.91 ± 2.83 b | 0.38 ± 0.01 b |
| SA + SNP + Cd | 2.07 ± 0.05 b | 0.68 ± 0.04 a | 2.75 ± 0.02 b | 0.71 ± 0.01 a | 95.37 ± 1.25 a | 0.17 ± 0.01 c |
| Treatments | AsA (nmol g−1 FW) | DHA (nmol g−1 FW) | AsA/DHA Ratio | GSH (nmol g−1 FW) | GSSG (nmol g−1 FW) | GSH/GSSG Ratio |
|---|---|---|---|---|---|---|
| C | 14209.41 ± 157.51 a | 977.65 ± 14.20 b | 14.54 ± 0.35 b | 425.24 ± 18.25 b | 120.63 ± 3.18 a | 3.54 ± 0.25 e |
| Cd | 9127.06 ± 115.11 d | 1646.47 ± 49.19 a | 5.55 ± 0.14 d | 267.55 ± 11.30 d | 36.50 ± 0.36 b | 7.33 ± 0.31 d |
| SA + Cd | 11425.59 ± 59.92 b | 1034.12 ± 9.34 b | 11.05 ± 0.10 c | 314.44 ± 7.22 c | 33.08 ± 1.09 b | 9.53 ± 0.42 c |
| SNP + Cd | 11094.71 ± 101.68 c | 1008.53 ± 13.75 b | 11.01 ± 0.19 c | 299.05 ± 7.37 c | 18.34 ± 0.93 d | 16.41 ± 1.03 b |
| SA + SNP + Cd | 11376.18 ± 220.62 b | 528.53 ± 12.82 c | 21.56 ± 0.84 a | 617.22 ± 9.16 a | 26.79 ± 0.48 c | 23.04 ± 0.07 a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mostofa, M.G.; Rahman, M.M.; Ansary, M.M.U.; Fujita, M.; Tran, L.-S.P. Interactive Effects of Salicylic Acid and Nitric Oxide in Enhancing Rice Tolerance to Cadmium Stress. Int. J. Mol. Sci. 2019, 20, 5798. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms20225798
Mostofa MG, Rahman MM, Ansary MMU, Fujita M, Tran L-SP. Interactive Effects of Salicylic Acid and Nitric Oxide in Enhancing Rice Tolerance to Cadmium Stress. International Journal of Molecular Sciences. 2019; 20(22):5798. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms20225798
Chicago/Turabian StyleMostofa, Mohammad Golam, Md. Mezanur Rahman, Md. Mesbah Uddin Ansary, Masayuki Fujita, and Lam-Son Phan Tran. 2019. "Interactive Effects of Salicylic Acid and Nitric Oxide in Enhancing Rice Tolerance to Cadmium Stress" International Journal of Molecular Sciences 20, no. 22: 5798. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms20225798





