Intravenous Immunoglobulin Therapy Eliminates Candida albicans and Maintains Intestinal Homeostasis in a Murine Model of Dextran Sulfate Sodium-Induced Colitis
Abstract
:1. Introduction
2. Results
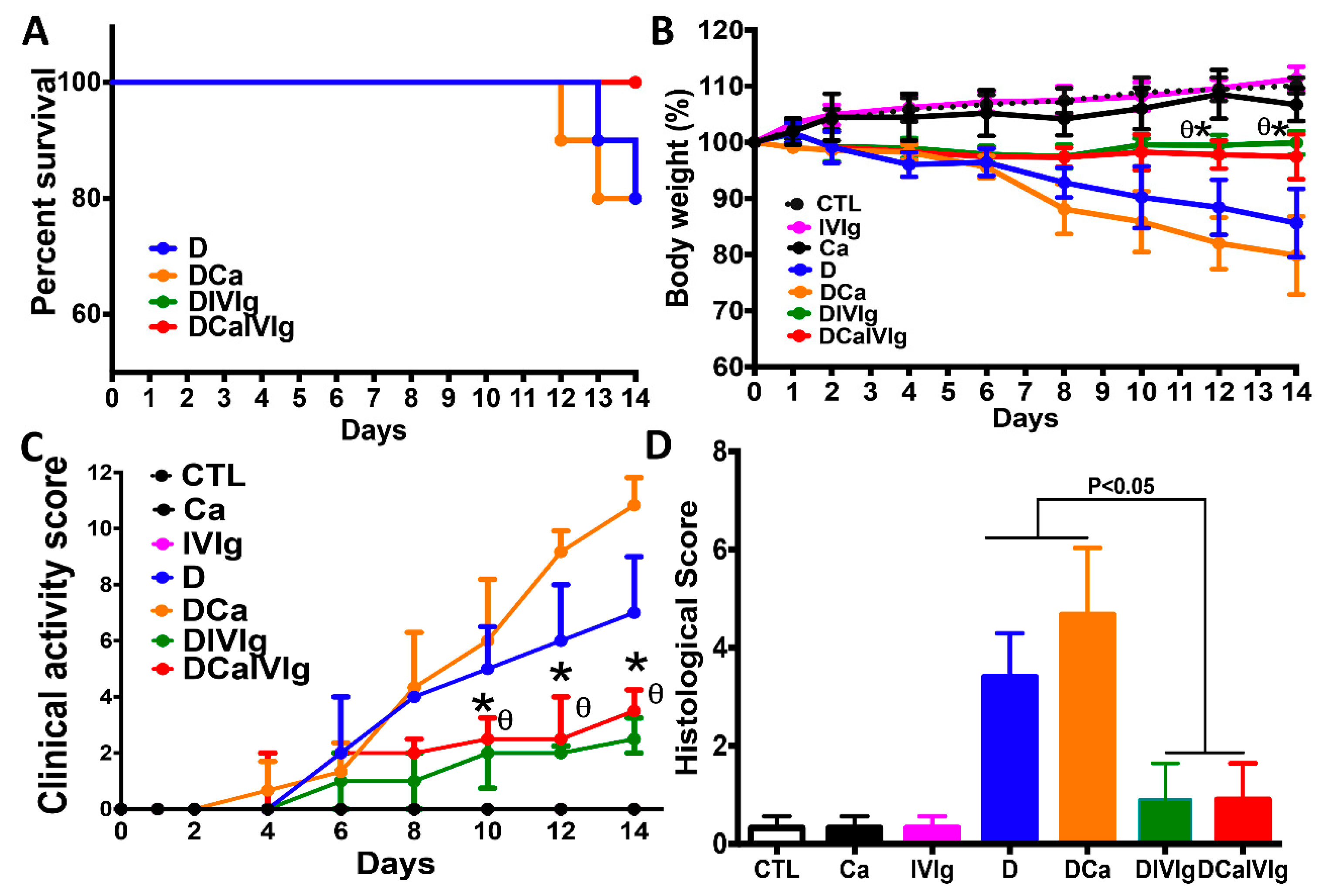
2.1. IVIg Treatment Reduces the Severity of Intestinal Inflammation in C. albicans Colonized Mice with DSS-Induced Colitis
2.2. IVIg Treatment Decreases the Escherichia coli, Enterococcus faecalis, and C. albicans Populations in Mice with DSS-Induced Colitis
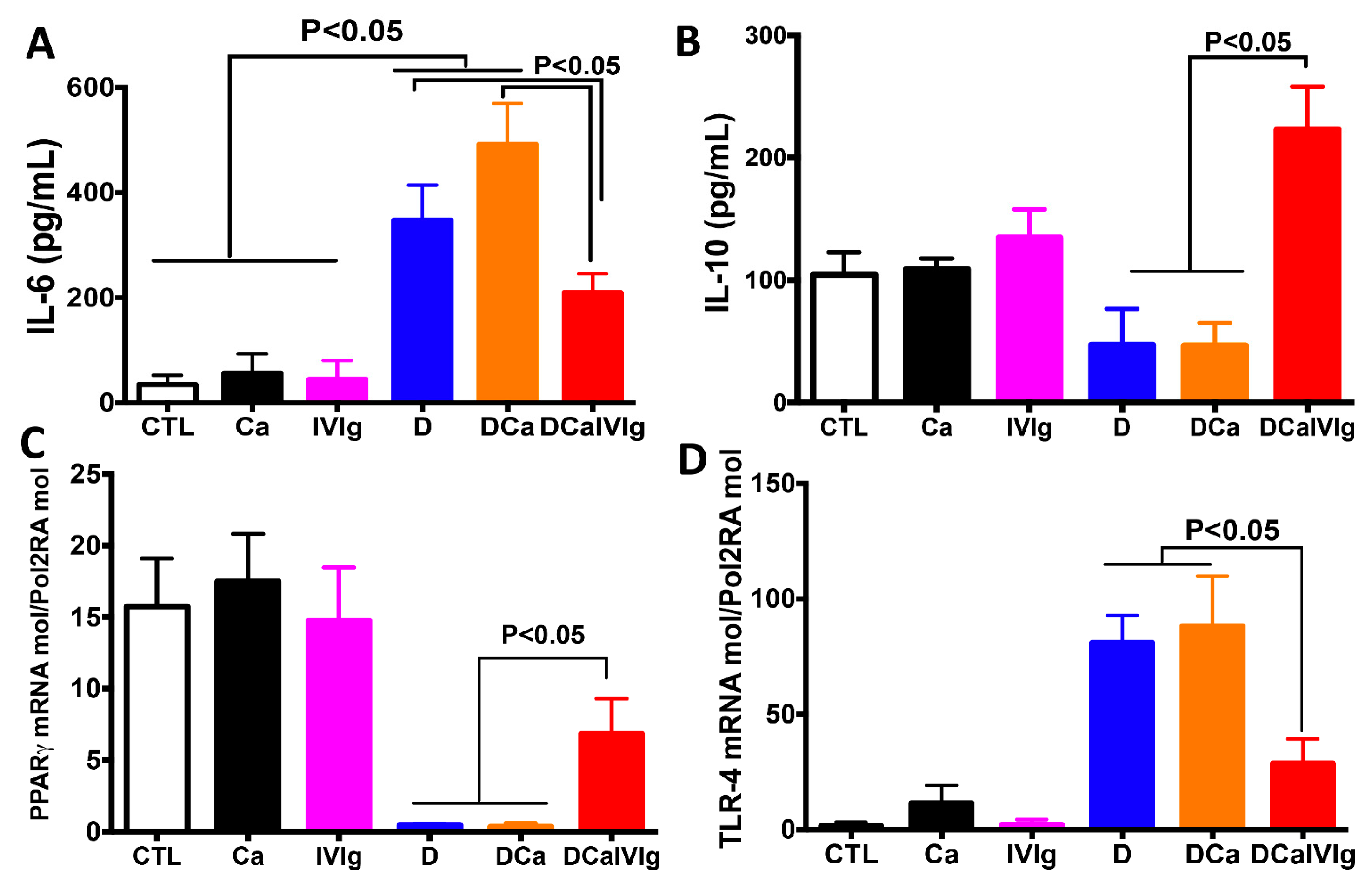
2.3. IVIg Treatment Reduces Inflammatory Cytokines and Enhances IL-10 in the Colon
3. Discussion
4. Methods
4.1. Animals
4.2. Yeast Strain, Inoculum Preparation, and Induction of Colitis
4.3. Assessment of Clinical and Histological Scores
4.4. Real-Time mRNA Quantification of Innate Immune Receptors
4.5. Quantification of Cytokines
4.6. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Poulain, D. Candida albicans, plasticity and pathogenesis. Crit. Rev. Microbiol. 2015, 41, 208–217. [Google Scholar] [CrossRef]
- Kullberg, B.J.; Arendrup, M.C. Invasive candidiasis. N. Engl. J. Med. 2015, 373, 1445–1456. [Google Scholar] [CrossRef] [PubMed]
- Gow, N.A.; van de Veerdonk, F.L.; Brown, A.J.; Netea, M.G. Candida albicans morphogenesis and host defence: Discriminating invasion from colonization. Nat. Rev. Microbiol. 2012, 10, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Sendid, B.; Dotan, N.; Nseir, S.; Savaux, C.; Vandewalle, P.; Standaert, A.; Zerimech, F.; Guery, B.P.; Dukler, A.; Colombel, J.F.; et al. Antibodies against glucan, chitin, and saccharomyces cerevisiae mannan as new biomarkers of candida albicans infection that complement tests based on c. Albicans mannan. Clin. Vaccine Immunol. 2008, 15, 1868–1877. [Google Scholar] [CrossRef] [PubMed]
- Frank, D.N.; St Amand, A.L.; Feldman, R.A.; Boedeker, E.C.; Harpaz, N.; Pace, N.R. Molecular-phylogenetic characterization of microbial community imbalances in human inflammatory bowel diseases. Proc. Natl. Acad. Sci. USA 2007, 104, 13780–13785. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Standaert-Vitse, A.; Sendid, B.; Joossens, M.; Francois, N.; Vandewalle-El Khoury, P.; Branche, J.; Van Kruiningen, H.; Jouault, T.; Rutgeerts, P.; Gower-Rousseau, C.; et al. Candida albicans colonization and asca in familial crohn’s disease. Am. J. Gastroenterol. 2009, 104, 1745–1753. [Google Scholar] [CrossRef] [PubMed]
- Jawhara, S.; Poulain, D. Saccharomyces boulardii decreases inflammation and intestinal colonization by candida albicans in a mouse model of chemically-induced colitis. Med. Mycol. 2007, 45, 691–700. [Google Scholar] [CrossRef] [PubMed]
- Jawhara, S.; Thuru, X.; Standaert-Vitse, A.; Jouault, T.; Mordon, S.; Sendid, B.; Desreumaux, P.; Poulain, D. Colonization of mice by candida albicans is promoted by chemically induced colitis and augments inflammatory responses through galectin-3. J. Infect. Dis. 2008, 197, 972–980. [Google Scholar] [CrossRef]
- Jawhara, S.; Mogensen, E.; Maggiotto, F.; Fradin, C.; Sarazin, A.; Dubuquoy, L.; Maes, E.; Guerardel, Y.; Janbon, G.; Poulain, D. Murine model of dextran sulfate sodium-induced colitis reveals candida glabrata virulence and contribution of beta-mannosyltransferases. J. Biol. Chem. 2012, 287, 11313–11324. [Google Scholar] [CrossRef]
- Perez, E.E.; Orange, J.S.; Bonilla, F.; Chinen, J.; Chinn, I.K.; Dorsey, M.; El-Gamal, Y.; Harville, T.O.; Hossny, E.; Mazer, B.; et al. Update on the use of immunoglobulin in human disease: A review of evidence. J. Allergy Clin. Immunol. 2017, 139, S1–S46. [Google Scholar] [CrossRef] [PubMed]
- Gilardin, L.; Bayry, J.; Kaveri, S.V. Intravenous immunoglobulin as clinical immune-modulating therapy. CMAJ 2015, 187, 257–264. [Google Scholar] [CrossRef] [Green Version]
- Lunemann, J.D.; Nimmerjahn, F.; Dalakas, M.C. Intravenous immunoglobulin in neurology--mode of action and clinical efficacy. Nat. Rev. Neurol. 2015, 11, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Galeotti, C.; Kaveri, S.V.; Bayry, J. Ivig-mediated effector functions in autoimmune and inflammatory diseases. Int. Immunol. 2017, 29, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Bruckner, C.; Lehmann, C.; Dudziak, D.; Nimmerjahn, F. Sweet signs: Igg glycosylation leads the way in ivig-mediated resolution of inflammation. Int. Immunol. 2017, 29, 499–509. [Google Scholar] [CrossRef] [PubMed]
- Rogosnitzky, M.; Danks, R.; Holt, D. Intravenous immunoglobulin for the treatment of crohn’s disease. Autoimmun. Rev. 2012, 12, 275–280. [Google Scholar] [CrossRef]
- Wang, M.; Liang, C.; Hu, H.; Zhou, L.; Xu, B.; Wang, X.; Han, Y.; Nie, Y.; Jia, S.; Liang, J.; et al. Intraperitoneal injection (ip), intravenous injection (iv) or anal injection (ai)? Best way for mesenchymal stem cells transplantation for colitis. Sci. Rep. 2016, 6, 30696. [Google Scholar] [CrossRef]
- Castelo-Branco, M.T.; Soares, I.D.; Lopes, D.V.; Buongusto, F.; Martinusso, C.A.; do Rosario, A., Jr.; Souza, S.A.; Gutfilen, B.; Fonseca, L.M.; Elia, C.; et al. Intraperitoneal but not intravenous cryopreserved mesenchymal stromal cells home to the inflamed colon and ameliorate experimental colitis. PLoS ONE 2012, 7, e33360. [Google Scholar] [CrossRef]
- Bayry, J.; Lacroix-Desmazes, S.; Kazatchkine, M.D.; Kaveri, S.V. Intravenous immunoglobulin for infectious diseases: Back to the pre-antibiotic and passive prophylaxis era? Trends Pharm. Sci. 2004, 25, 306–310. [Google Scholar] [CrossRef] [PubMed]
- Diep, B.A.; Le, V.T.; Badiou, C.; Le, H.N.; Pinheiro, M.G.; Duong, A.H.; Wang, X.; Dip, E.C.; Aguiar-Alves, F.; Basuino, L.; et al. Ivig-mediated protection against necrotizing pneumonia caused by mrsa. Sci. Transl. Med. 2016, 8, 357ra124. [Google Scholar] [CrossRef] [PubMed]
- Shopsin, B.; Kaveri, S.V.; Bayry, J. Tackling difficult staphylococcus aureus infections: Antibodies show the way. Cell Host Microbe 2016, 20, 555–557. [Google Scholar] [CrossRef]
- Ben-Nathan, D.; Lustig, S.; Tam, G.; Robinzon, S.; Segal, S.; Rager-Zisman, B. Prophylactic and therapeutic efficacy of human intravenous immunoglobulin in treating west nile virus infection in mice. J. Infect. Dis. 2003, 188, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Gauduchon, V.; Cozon, G.; Vandenesch, F.; Genestier, A.L.; Eyssade, N.; Peyrol, S.; Etienne, J.; Lina, G. Neutralization of staphylococcus aureus panton valentine leukocidin by intravenous immunoglobulin in vitro. J. Infect. Dis. 2004, 189, 346–353. [Google Scholar] [CrossRef]
- Krause, I.; Wu, R.; Sherer, Y.; Patanik, M.; Peter, J.B.; Shoenfeld, Y. In vitro antiviral and antibacterial activity of commercial intravenous immunoglobulin preparations—A potential role for adjuvant intravenous immunoglobulin therapy in infectious diseases. Transfus Med. 2002, 12, 133–139. [Google Scholar] [CrossRef]
- Srivastava, R.; Ramakrishna, C.; Cantin, E. Anti-inflammatory activity of intravenous immunoglobulins protects against west nile virus encephalitis. J. Gen. Virol. 2015, 96, 1347–1357. [Google Scholar] [CrossRef]
- Ramakrishna, C.; Newo, A.N.; Shen, Y.W.; Cantin, E. Passively administered pooled human immunoglobulins exert il-10 dependent anti-inflammatory effects that protect against fatal hsv encephalitis. PLoS Pathog. 2011, 7, e1002071. [Google Scholar] [CrossRef]
- Elluru, S.R.; Kaveri, S.V.; Bayry, J. The protective role of immunoglobulins in fungal infections and inflammation. Semin Immunopathol. 2015, 37, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Casadevall, A.; Pirofski, L.A. Immunoglobulins in defense, pathogenesis, and therapy of fungal diseases. Cell Host Microbe 2012, 11, 447–456. [Google Scholar] [CrossRef] [PubMed]
- Stratta, R.J.; Shaefer, M.S.; Cushing, K.A.; Markin, R.S.; Reed, E.C.; Langnas, A.N.; Pillen, T.J.; Shaw, B.W., Jr. A randomized prospective trial of acyclovir and immune globulin prophylaxis in liver transplant recipients receiving okt3 therapy. Arch. Surg. 1992, 127, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Mimouni, D.; Gdalevich, M.; Mimouni, F.B.; Grotto, I.; Eldad, A.; Shpilberg, O. Does immune serum globulin confer protection against skin diseases? Int. J. Derm. 2000, 39, 628–631. [Google Scholar] [CrossRef] [PubMed]
- Tollemar, J.; Gross, N.; Dolgiras, N.; Jarstrand, C.; Ringden, O.; Hammarstrom, L. Fungal prophylaxis by reduction of fungal colonization by oral administration of bovine anti-candida antibodies in bone marrow transplant recipients. Bone Marrow Transplant. 1999, 23, 283–290. [Google Scholar] [CrossRef]
- Bozza, S.; Kasermann, F.; Kaveri, S.V.; Romani, L.; Bayry, J. Intravenous immunoglobulin protects from experimental allergic bronchopulmonary aspergillosis via a sialylation-dependent mechanism. Eur. J. Immunol. 2019, 49, 195–198. [Google Scholar] [CrossRef] [PubMed]
- Maddur, M.S.; Kaveri, S.V.; Bayry, J. Circulating normal igg as stimulator of regulatory t cells: Lessons from intravenous immunoglobulin. Trends Immunol. 2017, 38, 789–792. [Google Scholar] [CrossRef] [PubMed]
- Othy, S.; Topcu, S.; Saha, C.; Kothapalli, P.; Lacroix-Desmazes, S.; Kasermann, F.; Miescher, S.; Bayry, J.; Kaveri, S.V. Sialylation may be dispensable for reciprocal modulation of helper t cells by intravenous immunoglobulin. Eur. J. Immunol. 2014, 44, 2059–2063. [Google Scholar] [CrossRef] [PubMed]
- Neely, A.N.; Holder, I.A. Effects of immunoglobulin g and low-dose amphotericin b on candida albicans infections in burned mice. Antimicrob Agents Chemother. 1992, 36, 643–646. [Google Scholar] [CrossRef]
- Darfeuille-Michaud, A.; Neut, C.; Barnich, N.; Lederman, E.; Di Martino, P.; Desreumaux, P.; Gambiez, L.; Joly, B.; Cortot, A.; Colombel, J.F. Presence of adherent escherichia coli strains in ileal mucosa of patients with crohn’s disease. Gastroenterology 1998, 115, 1405–1413. [Google Scholar] [CrossRef]
- Kim, S.C.; Tonkonogy, S.L.; Karrasch, T.; Jobin, C.; Sartor, R.B. Dual-association of gnotobiotic il-10-/- mice with 2 nonpathogenic commensal bacteria induces aggressive pancolitis. Inflamm. Bowel Dis. 2007, 13, 1457–1466. [Google Scholar] [CrossRef]
- Hoarau, G.; Mukherjee, P.K.; Gower-Rousseau, C.; Hager, C.; Chandra, J.; Retuerto, M.A.; Neut, C.; Vermeire, S.; Clemente, J.; Colombel, J.F.; et al. Bacteriome and mycobiome interactions underscore microbial dysbiosis in familial crohn’s disease. MBio 2016, 7, e01250-16. [Google Scholar] [CrossRef] [PubMed]
- Croasdell, A.; Duffney, P.F.; Kim, N.; Lacy, S.H.; Sime, P.J.; Phipps, R.P. Ppargamma and the innate immune system mediate the resolution of inflammation. Ppar Res. 2015, 2015, 549691. [Google Scholar] [CrossRef]
- Ruiz de Souza, V.; Carreno, M.P.; Kaveri, S.V.; Ledur, A.; Sadeghi, H.; Cavaillon, J.M.; Kazatchkine, M.D.; Haeffner-Cavaillon, N. Selective induction of interleukin-1 receptor antagonist and interleukin-8 in human monocytes by normal polyspecific igg (intravenous immunoglobulin). Eur. J. Immunol. 1995, 25, 1267–1273. [Google Scholar] [CrossRef] [PubMed]
- Kozicky, L.K.; Zhao, Z.Y.; Menzies, S.C.; Fidanza, M.; Reid, G.S.; Wilhelmsen, K.; Hellman, J.; Hotte, N.; Madsen, K.L.; Sly, L.M. Intravenous immunoglobulin skews macrophages to an anti-inflammatory, il-10-producing activation state. J. Leukoc Biol. 2015, 98, 983–994. [Google Scholar] [CrossRef]
- Galeotti, C.; Hegde, P.; Das, M.; Stephen-Victor, E.; Canale, F.; Munoz, M.; Sharma, V.K.; Dimitrov, J.D.; Kaveri, S.V.; Bayry, J. Heme oxygenase-1 is dispensable for the anti-inflammatory activity of intravenous immunoglobulin. Sci. Rep. 2016, 6, 19592. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Casulli, S.; Topcu, S.; Fattoum, L.; von Gunten, S.; Simon, H.U.; Teillaud, J.L.; Bayry, J.; Kaveri, S.V.; Elbim, C. A differential concentration-dependent effect of ivig on neutrophil functions: Relevance for anti-microbial and anti-inflammatory mechanisms. PLoS ONE 2011, 6, e26469. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.; Wicki, S.; Graeter, S.; Timcheva, T.M.; Keller, C.W.; Quast, I.; Leontyev, D.; Djoumerska-Alexieva, I.K.; Kasermann, F.; Jakob, S.M.; et al. Ivig regulates the survival of human but not mouse neutrophils. Sci. Rep. 2017, 7, 1296. [Google Scholar] [CrossRef] [PubMed]
- Vos, A.C.; Wildenberg, M.E.; Duijvestein, M.; Verhaar, A.P.; van den Brink, G.R.; Hommes, D.W. Anti-tumor necrosis factor-alpha antibodies induce regulatory macrophages in an fc region-dependent manner. Gastroenterology 2011, 140, 221–230. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Liu, H.; Liu, F.; Wang, H.; Liu, Y.; Duan, S. Inhibitory effect of ppar-gamma activator on il-6 and mpges protein expression in pbmc induced by homocysteine. Hemodial Int. 2005, 9 (Suppl. 1), S15–S20. [Google Scholar] [CrossRef] [PubMed]
- Fukata, M.; Chen, A.; Vamadevan, A.S.; Cohen, J.; Breglio, K.; Krishnareddy, S.; Hsu, D.; Xu, R.; Harpaz, N.; Dannenberg, A.J.; et al. Toll-like receptor-4 promotes the development of colitis-associated colorectal tumors. Gastroenterology 2007, 133, 1869–1881. [Google Scholar] [CrossRef] [PubMed]
- Charlet, R.; Pruvost, Y.; Tumba, G.; Istel, F.; Poulain, D.; Kuchler, K.; Sendid, B.; Jawhara, S. Remodeling of the candida glabrata cell wall in the gastrointestinal tract affects the gut microbiota and the immune response. Sci. Rep. 2018, 8, 3316. [Google Scholar] [CrossRef] [PubMed]
- Bortolus, C.; Billamboz, M.; Charlet, R.; Lecointe, K.; Sendid, B.; Ghinet, A.; Jawhara, S. A small aromatic compound has antifungal properties and potential anti-inflammatory effects against intestinal inflammation. Int. J. Mol. Sci. 2019, 20, 321. [Google Scholar] [CrossRef]
- Jawhara, S.; Habib, K.; Maggiotto, F.; Pignede, G.; Vandekerckove, P.; Maes, E.; Dubuquoy, L.; Fontaine, T.; Guerardel, Y.; Poulain, D. Modulation of intestinal inflammation by yeasts and cell wall extracts: Strain dependence and unexpected anti-inflammatory role of glucan fractions. PLoS ONE 2012, 7, e40648. [Google Scholar] [CrossRef]
- Choteau, L.; Parny, M.; Francois, N.; Bertin, B.; Fumery, M.; Dubuquoy, L.; Takahashi, K.; Colombel, J.F.; Jouault, T.; Poulain, D.; et al. Role of mannose-binding lectin in intestinal homeostasis and fungal elimination. Mucosal Immunol. 2016, 9, 767–776. [Google Scholar] [CrossRef]
- Charlet, R.; Bortolus, C.; Barbet, M.; Sendid, B.; Jawhara, S. A decrease in anaerobic bacteria promotes candida glabrata overgrowth while beta-glucan treatment restores the gut microbiota and attenuates colitis. Gut Pathog. 2018, 10, 50. [Google Scholar] [CrossRef] [PubMed]



© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Charlet, R.; Sendid, B.; Kaveri, S.V.; Poulain, D.; Bayry, J.; Jawhara, S. Intravenous Immunoglobulin Therapy Eliminates Candida albicans and Maintains Intestinal Homeostasis in a Murine Model of Dextran Sulfate Sodium-Induced Colitis. Int. J. Mol. Sci. 2019, 20, 1473. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms20061473
Charlet R, Sendid B, Kaveri SV, Poulain D, Bayry J, Jawhara S. Intravenous Immunoglobulin Therapy Eliminates Candida albicans and Maintains Intestinal Homeostasis in a Murine Model of Dextran Sulfate Sodium-Induced Colitis. International Journal of Molecular Sciences. 2019; 20(6):1473. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms20061473
Chicago/Turabian StyleCharlet, Rogatien, Boualem Sendid, Srini V. Kaveri, Daniel Poulain, Jagadeesh Bayry, and Samir Jawhara. 2019. "Intravenous Immunoglobulin Therapy Eliminates Candida albicans and Maintains Intestinal Homeostasis in a Murine Model of Dextran Sulfate Sodium-Induced Colitis" International Journal of Molecular Sciences 20, no. 6: 1473. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms20061473






