PhSeZnCl in the Synthesis of Steroidal β-Hydroxy-Phenylselenides Having Antibacterial Activity
Abstract
:1. Introduction
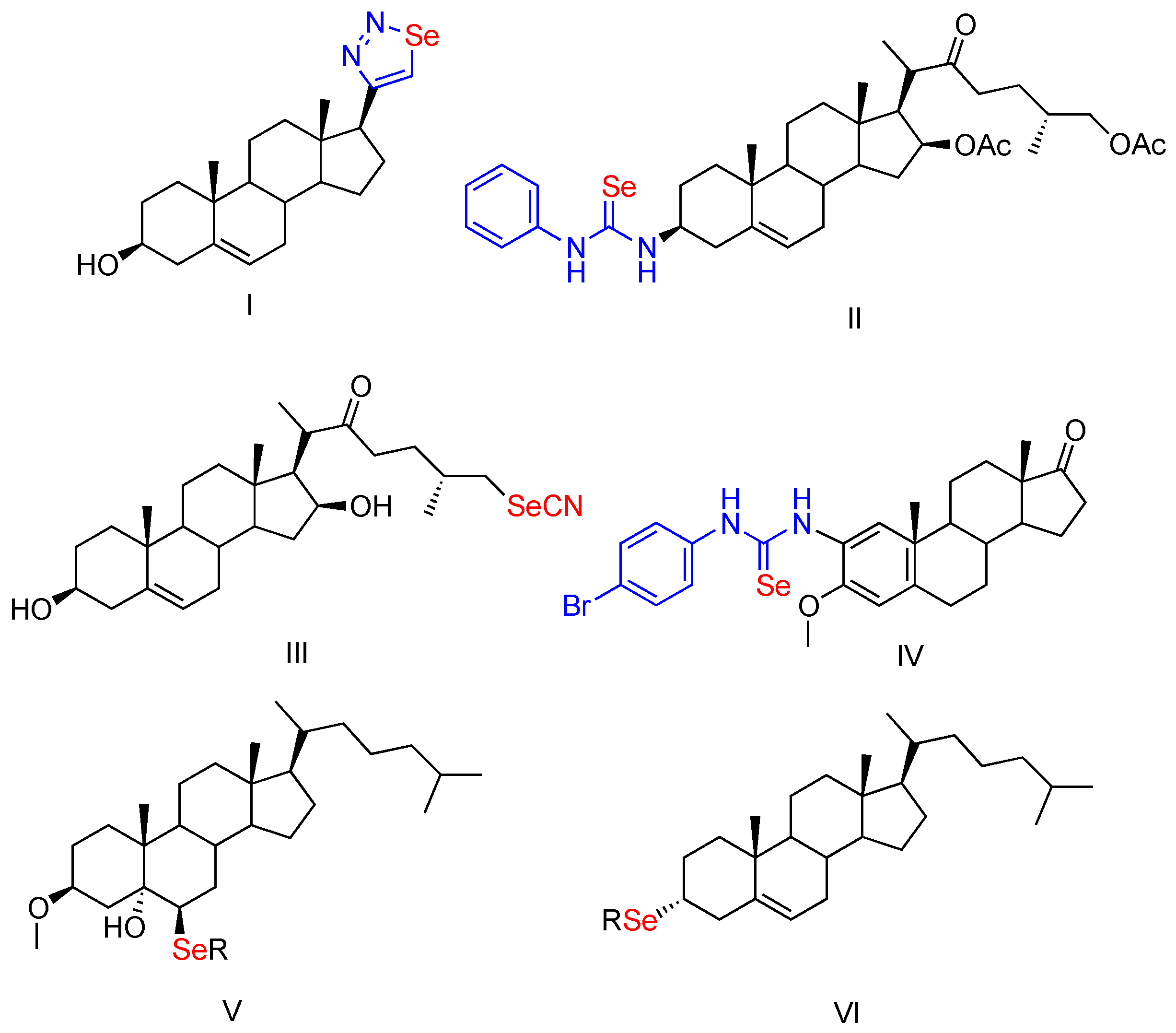
2. Results and Discussion
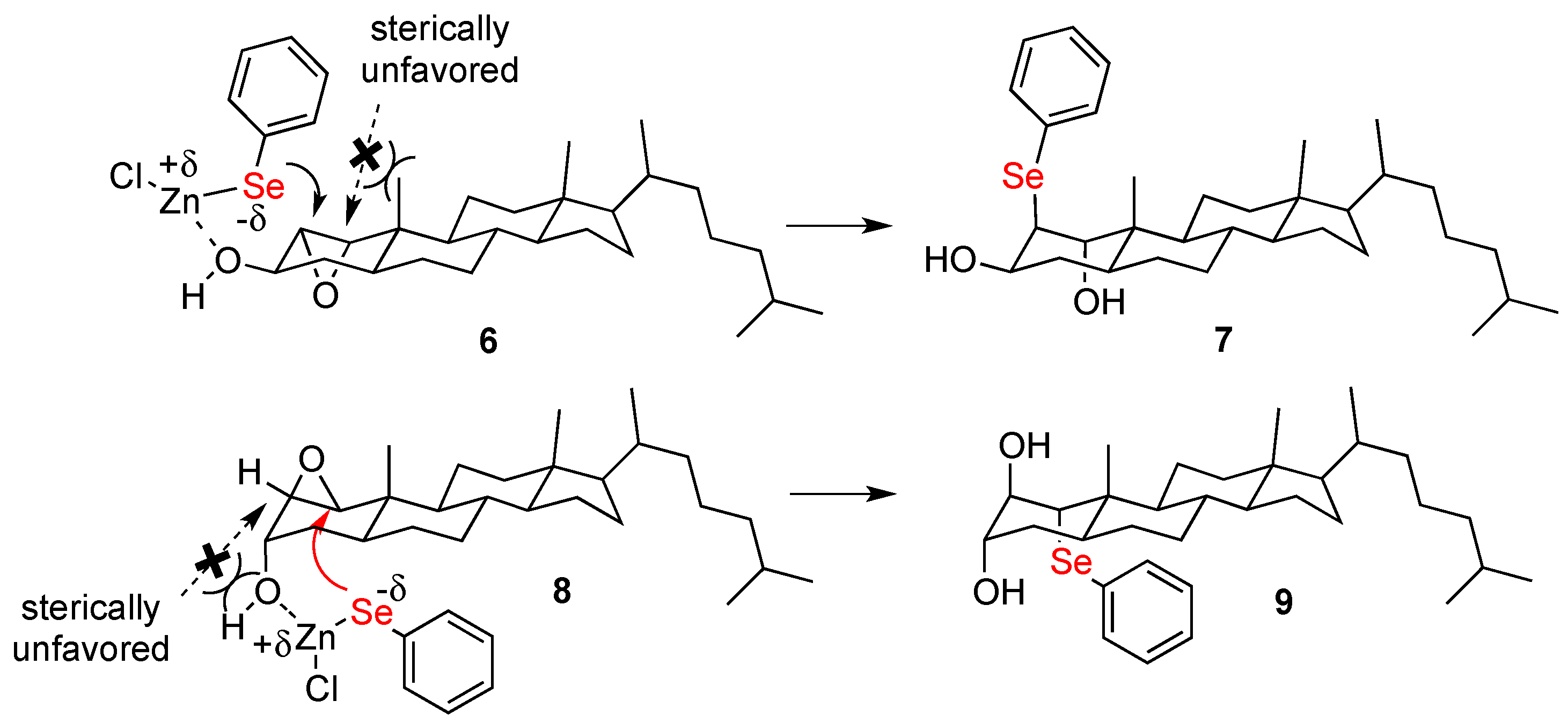
2.1. Chemistry
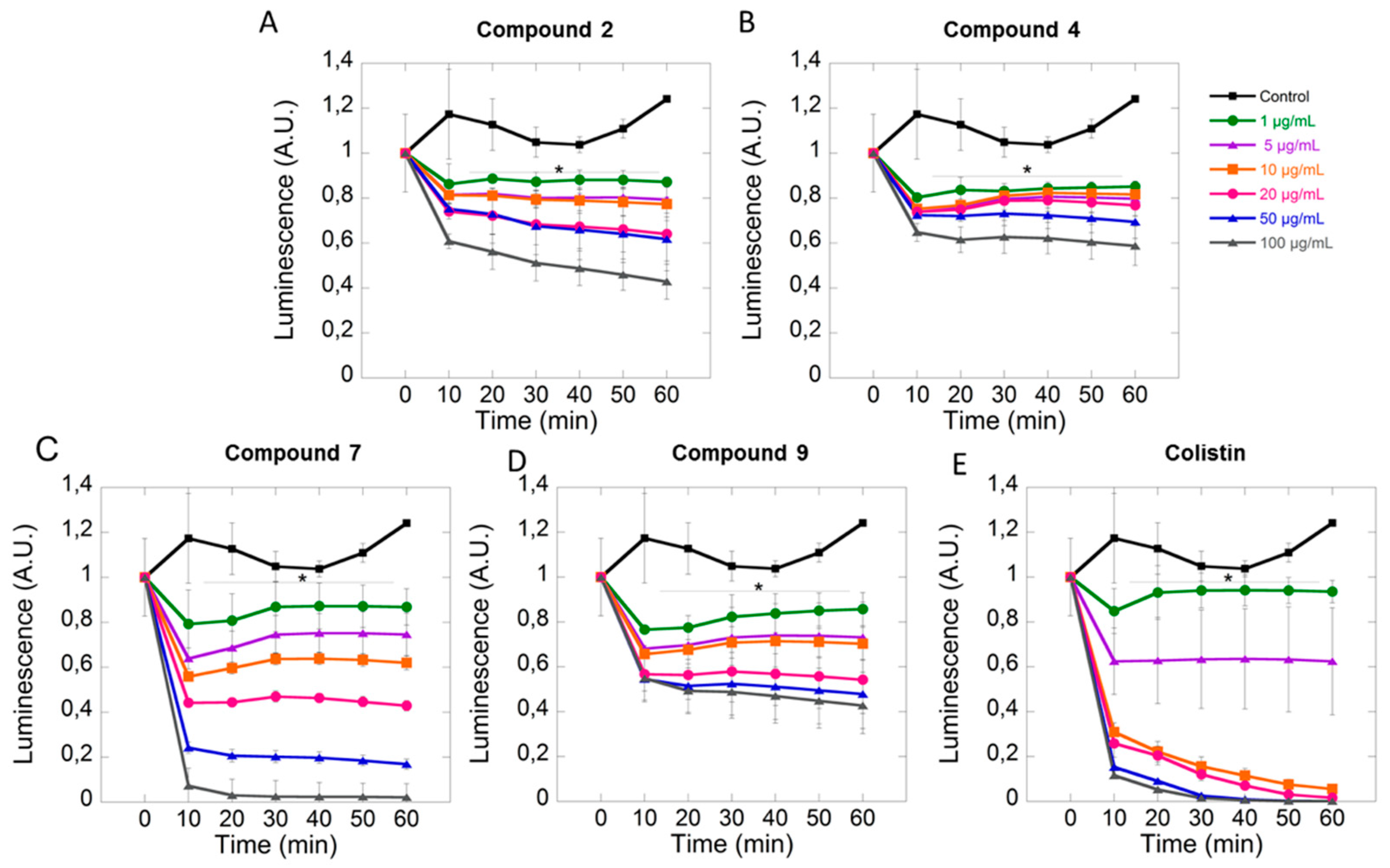
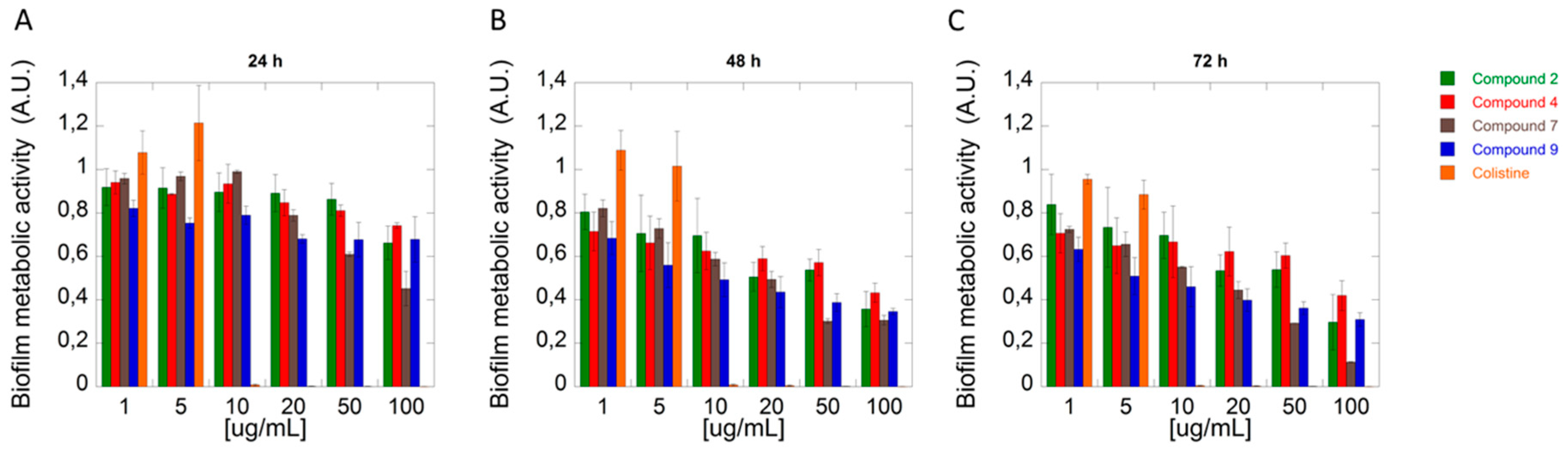
2.2. Biological Activity
3. Materials and Methods
3.1. Chemistry
3.1.1. General Methods
3.1.2. General Procedure
β,5α-Dihydroxy-6β-Phenylselenylcholestane (2)
(25R)-5α-Hydroxy-6β-Phenylselenylspirostan-3β-ol Acetate (4)
1α,3β-Dihydroxy-2β-Phenylselenylcholestane (7)
2β,3α-Dihydroxy-1α-Phenylselenylcholestane (9)
3.2. Biological Activity
3.2.1. Antibacterial Testing
3.2.2. Anti-Biofilm Activity
3.2.3. GPx-Like Activity by NMR
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| COSY | COrrelation SpectrocpY |
| DTT | DiThioTreitol |
| GPx | Glutathione Peroxidase |
| HMBC | Heteronuclear Multiple Bond Correlation |
| HSQC | Heteronuclear Single Quantum Correlation |
| IR | InfraRed |
| MIC | Minimum Inhibitory Concentration |
| MS | Mass |
| NMR | Nuclear Magnetic Resonance |
| SD | Standard Deviation |
| THF | TetraHydroFuran |
| TLC | Thin Layer Chromatography |
References
- Sk, U.H.; Sharma, A.K.; Ghosh, S.; Bhattacharya, S. Synthesis and biological evaluation of novel spiro 6-methoxytetralin-1,3′-pyrrolidine based organoselenocyanates against cadmium-induced oxidative and hepatic damage in mice. Eur. J. Med. Chem. 2010, 45, 3265–3273. [Google Scholar] [CrossRef] [PubMed]
- de Souza, D.; Mariano, D.O.C.; Nedel, F.; Schultze, E.; Campos, V.F.; Seixas, F.; da Silva, R.S.; Munchen, T.S.; Ilha, V.; Dornelles, L.; et al. New Organochalcogen Multitarget Drug: Synthesis and Antioxidant and Antitumoral Activities of Chalcogenozidovudine Derivatives. J. Med. Chem. 2015, 58, 3329–3339. [Google Scholar] [CrossRef]
- Barcellos, A.M.; Abenante, L.; Sarro, M.T.; Leo, I.D.; Lenardao, E.J.; Perin, G.; Santi, C. New Prospective for Redox Modulation Mediated by Organo selenium and Organotellurium Compounds. Curr. Org. Chem. 2017, 21, 2044–2061. [Google Scholar] [CrossRef]
- Brigelius-Flohé, R.; Maiorino, M. Glutathione peroxidases. Biochim. Biophys. Acta Gen. Subj. 2013, 1830, 3289–3303. [Google Scholar] [CrossRef]
- Sancineto, L.; Piccioni, M.; De Marco, S.; Pagiotti, R.; Nascimento, V.; Braga, A.L.; Santi, C.; Pietrella, D. Diphenyl diselenide derivatives inhibit microbial biofilm formation involved in wound infection. BMC Microbiol. 2016, 16, 220. [Google Scholar] [CrossRef] [PubMed]
- Rossato, L.; Loreto, E.S.; Venturini, T.P.; de Azevedo, M.I.; Al-Hatmi, A.M.S.; Santurio, J.M.; Alves, S.H. In vitro combination between antifungals and diphenyl diselenide against Cryptococcus species. Mycoses 2019. [Google Scholar] [CrossRef]
- Sancineto, L.; Mariotti, A.; Bagnoli, L.; Marini, F.; Desantis, J.; Iraci, N.; Santi, C.; Pannecouque, C.; Tabarrini, O. Design and Synthesis of DiselenoBisBenzamides (DISeBAs) as Nucleocapsid Protein 7 (NCp7) Inhibitors with anti-HIV Activity. J. Med. Chem. 2015, 58, 9601–9614. [Google Scholar] [CrossRef]
- Bartolini, D.; Commodi, J.; Piroddi, M.; Incipini, L.; Sancineto, L.; Santi, C.; Galli, F. Glutathione S-transferase pi expression regulates the Nrf2-dependent response to hormetic diselenides. Free Radic. Biol. Med. 2015, 88, 466–480. [Google Scholar] [CrossRef] [PubMed]
- Bartolini, D.; Piroddi, M.; Tidei, C.; Giovagnoli, S.; Pietrella, D.; Manevich, Y.; Tew, K.D.; Giustarini, D.; Rossi, R.; Townsend, D.M.; et al. Reaction kinetics and targeting to cellular glutathione S-transferase of the glutathione peroxidase mimetic PhSeZnCl and its d,l-polylactide microparticle formulation. Free Radic. Biol. Med. 2015, 78, 56–65. [Google Scholar] [CrossRef]
- Bartolini, D.; Sancineto, L.; Fabro de Bem, A.; Tew, K.D.; Santi, C.; Radi, R.; Toquato, P.; Galli, F. Selenocompounds in Cancer Therapy: An Overview. In Advances in Cancer Research; Elsevier: San Diego, CA, USA, 2017; pp. 259–302. ISBN 978-0-12-812016-3. [Google Scholar]
- Tan, S.M.; Sharma, A.; Stefanovic, N.; de Haan, J.B. Late-intervention study with ebselen in an experimental model of type 1 diabetic nephropathy. Free Radic. Res. 2015, 49, 219–227. [Google Scholar]
- He, J.; Li, D.; Xiong, K.; Ge, Y.; Jin, H.; Zhang, G.; Hong, M.; Tian, Y.; Yin, J.; Zeng, H. Inhibition of thioredoxin reductase by a novel series of bis-1,2-benzisoselenazol-3(2H)-ones: Organoselenium compounds for cancer therapy. Bioorg. Med. Chem. 2012, 20, 3816–3827. [Google Scholar]
- Romero-Hernandez, L.L.; Merino-Montiel, P.; Montiel-Smith, S.; Meza-Reyes, S.; Vega-Báez, J.L.; Abasolo, I.; Schwartz, S., Jr.; Lopez, O.; Fernandez-Bolanos, J.G. Diosgenin-based thio (seleno) ureas and triazolyl glycoconjugates as hybrid drugs. Antioxidant and antiproliferative profile. Eur. J. Med. Chem. 2015, 99, 67–81. [Google Scholar] [CrossRef]
- Fuentes-Aguilar, A.; Romero-Hernández, L.L.; Arenas-González, A.; Merino-Montiel, P.; Montiel-Smith, S.; Meza-Reyes, S.; Fernández-Bolaños, J.G. New selenosteroids as antiproliferative agents. Org. Biomol. Chem. 2017, 15, 5041–5054. [Google Scholar] [CrossRef]
- Cui, J.G.; Qi, B.B.; Gan, C.F. Synthesis and in vitro antiproliferative evaluation of some B-norcholesteryl benzimidazole and benzothiazole derivatives. Mar. Drugs 2015, 13, 2488–2504. [Google Scholar] [CrossRef]
- Affeldt, R.F.; Santos, F.P.; da Silva, R.S.; Rodrigues, O.E.D.; Wessjohann, L.A.; Lüdtke, D.S. Stereoselective glycoconjugation of steroids with selenocarbohydrates. RSC Advances 2016, 6, 93905–93914. [Google Scholar] [CrossRef]
- Rodrigues, O.E.D.; de Souza, D.; Soares, L.C.; Dornelles, L.; Burrow, R.A.; Appelt, H.R.; Alves, C.F.; Alves, D.; Braga, A.L. Stereoselective synthesis of selenosteroids. Tetrahedron Letters 2010, 51, 2237–2240. [Google Scholar] [CrossRef]
- Santi, C.; Capoccia, L.; Monti, B. Zinc-Selenium reagents in organic synthesis. Phys. Sci. Rev. 2018, 3. Available online: https://0-www-degruyter-com.brum.beds.ac.uk/view/j/psr.2018.3.issue-12/psr-2017-0129/psr-2017-0129.xml (accessed on 11 August 2018). [CrossRef]
- Santi, C.; Santoro, S.; Battistelli, B.; Testaferri, L.; Tiecco, M. Preparation of the First Bench-Stable Phenyl Selenolate: An Interesting “On Water” Nucleophilic Reagent. Eur. J. Org. Chem. 2008, 2008, 5387–5390. [Google Scholar] [CrossRef]
- Perin, G.; Alves, D.; Jacob, R.G.; Barcellos, A.M.; Soares, L.K.; Lenardão, E.J. Synthesis of Organochalcogen Compounds using Non-Conventional Reaction Media. ChemistrySelect 2016, 1, 205–258. [Google Scholar] [CrossRef]
- Santi, C.; Battistelli, B.; Testaferri, L.; Tiecco, M. On water preparation of phenylselenoesters. Green Chemistry 2012, 14, 1277. [Google Scholar] [CrossRef]
- Battistelli, B.; Lorenzo, T.; Tiecco, M.; Santi, C. “On-Water” Michael-Type Addition Reactions Promoted by PhSeZnCl. Eur. J. Org. Chem. 2011, 2011, 1848–1851. [Google Scholar] [CrossRef]
- Santoro, S.; Battistelli, B.; Testaferri, L.; Tiecco, M.; Santi, C. Vinylic Substitutions Promoted by PhSeZnCl: Synthetic and Theoretical Investigations. Eur. J. Org. Chem. 2009, 2009, 4921–4925. [Google Scholar] [CrossRef]
- Salman, S.; Schwab, R.; Alberto, E.; Vargas, J.; Dornelles, L.; Rodrigues, O.; Braga, A. Efficient Ring Opening of Protected and Unprotected Aziridines Promoted by Stable Zinc Selenolate in Ionic Liquid. Synlett 2011, 2011, 69–72. [Google Scholar] [CrossRef]
- Nagasawa, T.; Shimada, N.; Torihata, M.; Kuwahara, S. Enantioselective total synthesis of idesolide via NaHCO3-promoted dimerization. Tetrahedron 2010, 66, 4965–4969. [Google Scholar] [CrossRef]
- Jiang, H.; Pan, X.; Li, N.; Zhang, Z.; Zhu, J.; Zhu, X. Selenide-containing high refractive index polymer material with adjustable refractive index and Abbe’s number. React. Funct. Polym. 2017, 111, 1–6. [Google Scholar] [CrossRef]
- Paillasse, M.R.; Saffon, N.; Gornitzka, H.; Silvente-Poirot, S.; Poirot, M.; de Medina, P. Surprising unreactivity of cholesterol-5,6-epoxides towards nucleophiles. J. Lipid Res. 2012, 53, 718–725. [Google Scholar] [CrossRef]
- Jastrzebska, I. Chemistry of Steroidal Sapogenins-New Advances in a Classical Field. Curr. Org. Chem. 2012, 16, 353–372. [Google Scholar] [CrossRef]
- Petrow, V.A. 212. Steroids and related compounds. Part III. The constitution of Westphalen’s diol. J. Chem. Soc. (Resumed) 1939, 998. [Google Scholar] [CrossRef]
- Kasal, A.; Budesinsky, M.; Griffiths, W.J. Spectroscopic Methods of Steroid Analysis. In Steroid Analysis, 2nd ed.; Makin, H.L.J., Gower, D.B., Eds.; Springer: London, UK, 2010; pp. 62–106. [Google Scholar]
- Fürst, A.; Plattner, P.A. 2α, 3α-und 2β, 3β-Oxido-chlolestane; Konfiguration der 2-Oxy-cholestane. Helvetica Chimica Acta 1949, 32, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Veesenmeyer, J.L.; Hauser, A.R.; Lisboa, T.; Rello, J. Pseudomonas aeruginosa virulence and therapy: Evolving translational strategies. Crit. Care Med. 2009, 37, 1777–1786. [Google Scholar] [CrossRef] [PubMed]
- Kadurugamuwa, J.L.; Sin, L.; Albert, E.; Yu, J.; Francis, K.; DeBoer, M.; Rubin, M.; Bellinger-Kawahara, C.; Parr, T.R.; Contag, P.R. Direct Continuous Method for Monitoring Biofilm Infection in a Mouse Model. Infect. Immun. 2003, 71, 882–890. [Google Scholar]
- Epand, R.M.; Epand, R.F.; Savage, P.B. Ceragenins (Cationic Steroid Compounds), a novel class of antimicrobial agents. Drug News Perspect. 2008, 21, 307. [Google Scholar] [CrossRef]
- Mori, K.; Nakayama, T.; Sakuma, M. Synthesis of some analogues of blattellastanoside A, the steroidal aggregation pheromone of the German cockroach. Bioorganic Med. Chem. 1996, 4, 401–408. [Google Scholar] [CrossRef]
- Shawakfeh, K.Q.; Al-Said, N.H. Synthesis of new symmetrical bis-steroidal pyrazine analogues from diosgenin. Steroids 2011, 76, 232–237. [Google Scholar] [CrossRef]
- Weissenberg, M.; Lavie, D.; Glotter, E. Studies on epoxides—VI. Tetrahedron 1973, 29, 353–358. [Google Scholar] [CrossRef]
- Weissenberg, M.; Glotter, E. Steric course of reduction with sodium borohydride of steroidal αβ-epoxy-ketones. J. Chem. Soc. Perkin Trans. 1978, 1, 568–571. [Google Scholar] [CrossRef]
- Niemirowicz, K.; Piktel, E.; Wilczewska, A.; Markiewicz, K.; Durnaś, B.; Wątek, M.; Puszkarz, I.; Wróblewska, M.; Niklińska, W.; Savage, P.; et al. Core-shell magnetic nanoparticles display synergistic antibacterial effects against Pseudomonas aeruginosa and Staphylococcus aureus when combined with cathelicidin LL-37 or selected ceragenins. Int. J. Nanomed. 2016, 11, 5443–5455. [Google Scholar] [CrossRef]




| Entry | Substrate | Time (h) | Product | Yield |
|---|---|---|---|---|
| 1 |  | 3 |  | 75% |
| 2 |  | 3 | -- | -- |
| 3 |  | 6 |  | 62 |
| 4 |  | 8 | -- | -- |
| 5 |  | 2 |  | 54 |
| 6 |  | 2 |  | 53 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jastrzebska, I.; Mellea, S.; Salerno, V.; Grzes, P.A.; Siergiejczyk, L.; Niemirowicz-Laskowska, K.; Bucki, R.; Monti, B.; Santi, C. PhSeZnCl in the Synthesis of Steroidal β-Hydroxy-Phenylselenides Having Antibacterial Activity. Int. J. Mol. Sci. 2019, 20, 2121. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms20092121
Jastrzebska I, Mellea S, Salerno V, Grzes PA, Siergiejczyk L, Niemirowicz-Laskowska K, Bucki R, Monti B, Santi C. PhSeZnCl in the Synthesis of Steroidal β-Hydroxy-Phenylselenides Having Antibacterial Activity. International Journal of Molecular Sciences. 2019; 20(9):2121. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms20092121
Chicago/Turabian StyleJastrzebska, Izabella, Stefano Mellea, Valerio Salerno, Pawel Adam Grzes, Leszek Siergiejczyk, Katarzyna Niemirowicz-Laskowska, Robert Bucki, Bonifacio Monti, and Claudio Santi. 2019. "PhSeZnCl in the Synthesis of Steroidal β-Hydroxy-Phenylselenides Having Antibacterial Activity" International Journal of Molecular Sciences 20, no. 9: 2121. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms20092121