Early Life Oxidative Stress and Long-Lasting Cardiovascular Effects on Offspring Conceived by Assisted Reproductive Technologies: A Review
Abstract
:1. Introduction
2. Origins of OS
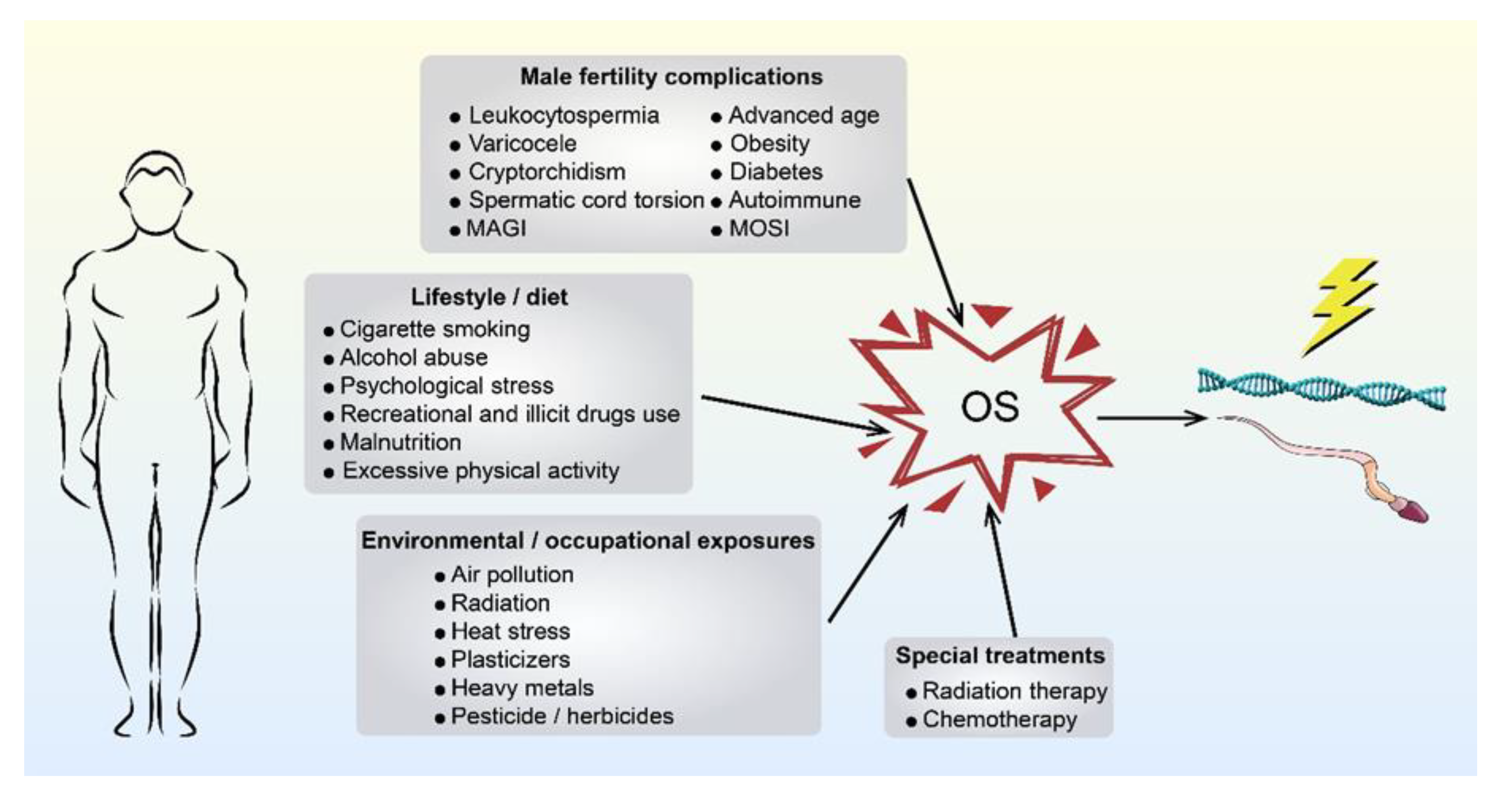
2.1. Paternally Derived OS
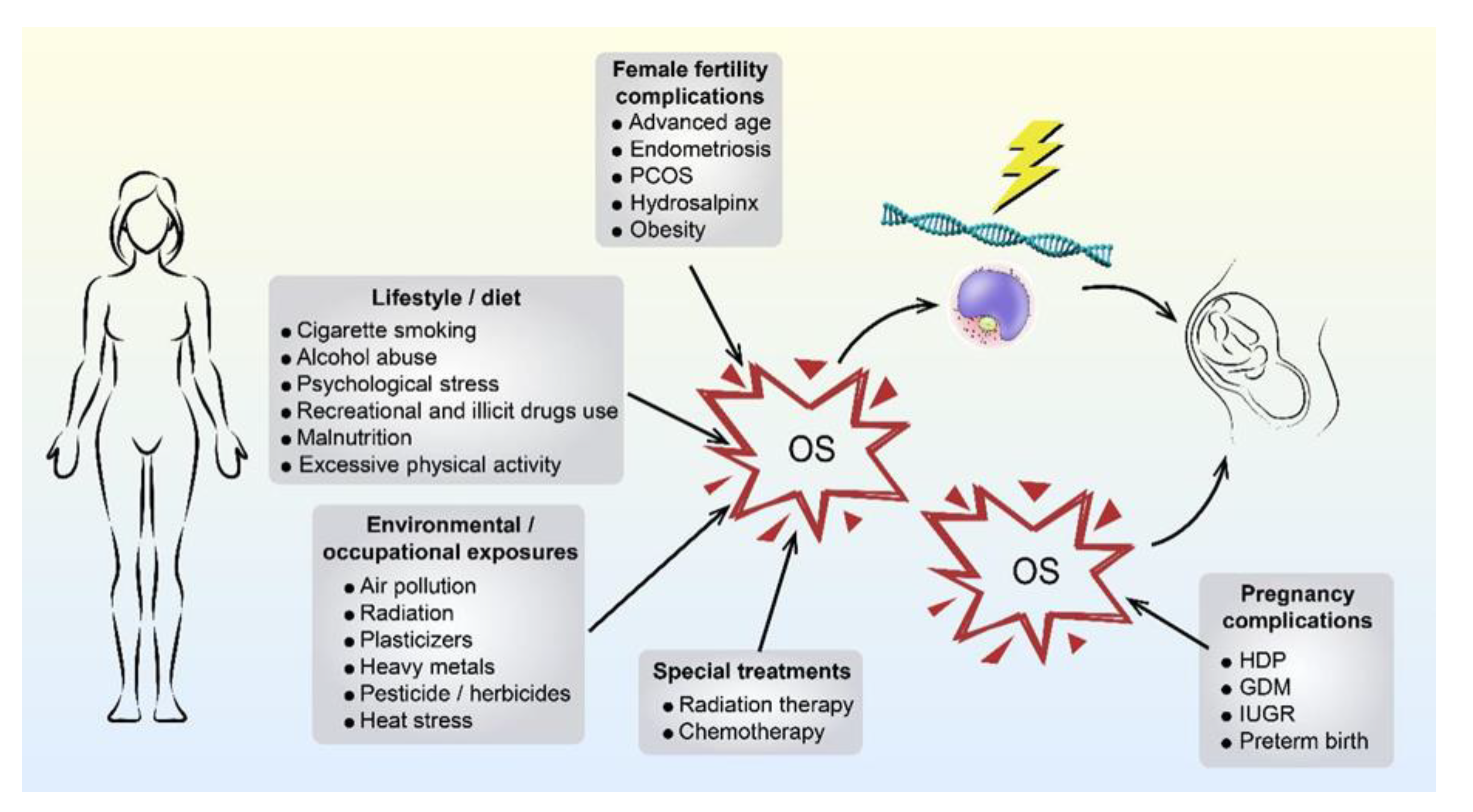
2.2. Maternally Derived OS
2.3. ART-Derived OS
3. OS-Associated Mechanisms in ART
3.1. Formation of OS
3.2. Epigenetic Modifications Resulting from OS
3.3. Nrf2-Mediated Anti-OS Signaling Pathway
4. Long-Lasting Cardiovascular Effects
5. Limitations and Prospects of Current Studies
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ART | Assisted reproductive technology |
| IUI | Intra-uterine insemination |
| IVF | In vitro fertilisation |
| ICSI | Intracytoplasmic sperm injection |
| COH | Controlled ovarian hyperstimulation |
| PGD | Pre-implantation genetic diagnosis |
| PGS | Pre-implantation genetic screening |
| SSR | Surgical sperm retrieval |
| DOHaD | Developmental Origins of Adult Disease |
| NCDs | Non-communicable diseases |
| CVDs | Cardiovascular diseases |
| T2DM | Type 2 diabetes mellitus |
| OS | Oxidative stress |
| ROS | Reactive oxygen species |
| MAGI | Male accessory gland infections |
| MOSI | Male oxidative stress infertility |
| PUFAs | Polyunsaturated fatty acids |
| SDF | Sperm DNA fragmentation |
| PCOS | Polycystic ovary syndrome |
| HDP | Hypertensive disorders of pregnancy |
| GDM | Gestational diabetes mellitus |
| IUGR | Intrauterine growth restriction |
| O2 | Oxygen |
| O2•− | Superoxide |
| HO2• | Hydroperoxyl |
| OH• | Hydroxyl |
| RO2• | Peroxyl radicals |
| H2O2 | Hydrogen peroxide |
| NADPH | Nicotinamide adenine dinucleotide phosphate |
| OXPHOS | Oxidative phosphorylation |
| 5-mC | 5-methylcytosine |
| 5-hmC | 5-hydroxymethylcytosine |
| DNMT1 | DNA methyltransferase 1 |
| H3K4 | Histone H3 lysine 4 |
| H3K27 | Histone H3 lysine 27 |
| H3K9 | Histone H3 lysine 9 |
| H3K4me3 | H3K4 trimethylation |
| TET | Ten-eleven-translocation |
| Nrf2 | Nuclear factor erythroid 2-related factor |
| ARE | Antioxidant response element |
| PEA-OXA | N-palmitoylethanolamine-oxazoline |
| LIN | Linarin |
| RES | Resveratrol |
| Keap1 | Kelch-like ECH-associated protein 1 |
| Maf | Musculoaponeurotic fibrosarcoma |
| AMA | Advanced maternal age |
| LDL-C | Low-density lipoprotein cholesterol |
| hESC | Human embryonic stem cells |
References
- Dyer, S.; Chambers, G.M.; De Mouzon, J.; Nygren, K.G.; Zegers-Hochschild, F.; Mansour, R.; Ishihara, O.; Banker, M.; Adamson, G.D. International committee for monitoring assisted reproductive technologies world report: Assisted reproductive technology 2008, 2009 and 2010. Hum. Reprod. 2016, 31, 1588–1609. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calhaz-Jorge, C.; De Geyter, C. h; Kupka, M.S.; Wyns, C.; Mocanu, E.; Motrenko, T.; Scaravelli, G.; Smeenk, J.; Vidakovic, S.; Goossens, V. Survey on ART and IUI: Legislation, regulation, funding and registries in European countries. Hum. Reprod. Open 2020, 2020, hoz044. [Google Scholar] [CrossRef] [PubMed]
- Farquhar, C.; Marjoribanks, J. Assisted reproductive technology: An overview of Cochrane Reviews. Cochrane Database Syst. Rev. 2018, 8, CD010537. [Google Scholar] [CrossRef] [PubMed]
- Kushnir, V.A.; Barad, D.H.; Albertini, D.F.; Darmon, S.K.; Gleicher, N. Systematic review of worldwide trends in assisted reproductive technology 2004–2013. Reprod. Biol. Endocrinol. 2017, 15, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barker, D.J. The developmental origins of chronic adult disease. Acta Paediatr. Suppl. 2004, 93, 26–33. [Google Scholar] [CrossRef]
- Eriksson, J.G.; Forsén, T.J.; Kajantie, E.; Osmond, C.; Barker, D.J.P. Childhood growth and hypertension in later life. Hypertension 2007, 49, 1415–1421. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barker, D.J.P.; Osmond, C.; Forsén, T.J.; Kajantie, E.; Eriksson, J.G. Trajectories of growth among children who have coronary events as adults. N. Engl. J. Med. 2005, 353, 1802–1809. [Google Scholar] [CrossRef]
- Hales, C.N.; Barker, D.J.P.; Clark, P.M.S.; Cox, L.J.; Fall, C.; Osmond, C.; Winter, P.D. Fetal and infant growth and impaired glucose tolerance at age 64. Br. Med. J. 1991, 303, 1019–1022. [Google Scholar] [CrossRef] [Green Version]
- Yu, H.; Yang, Q.; Sun, X.; Chen, G.; Qian, N.; Cai, R.; Guo, H.; Wang, C. Association of birth defects with the mode of assisted reproductive technology in a Chinese data-linkage cohort. Fertil. Steril. 2018, 109, 849–856. [Google Scholar] [CrossRef]
- Uk, A.; Collardeau-Frachon, S.; Scanvion, Q.; Michon, L.; Amar, E. Assisted Reproductive Technologies and imprinting disorders: Results of a study from a French congenital malformations registry. Eur. J. Med. Genet. 2018, 61, 518–523. [Google Scholar] [CrossRef]
- Jiang, Z.; Wang, Y.; Lin, J.; Xu, J.; Ding, G.; Huang, H. Genetic and epigenetic risks of assisted reproduction. Best Pract. Res. Clin. Obstet. Gynaecol. 2017, 44, 90–104. [Google Scholar] [CrossRef] [PubMed]
- Wainstock, T.; Walfisch, A.; Shoham-Vardi, I.; Segal, I.; Harlev, A.; Sergienko, R.; Landau, D.; Sheiner, E. Fertility treatments and pediatric neoplasms of the offspring: Results of a population-based cohort with a median follow-up of 10 years. Am. J. Obstet. Gynecol. 2017, 216, 314. [Google Scholar] [CrossRef] [PubMed]
- Reigstad, M.M.; Larsen, I.K.; Myklebust, T.Å.; Robsahm, T.E.; Oldereid, N.B.; Brinton, L.A.; Storeng, R. Risk of cancer in children conceived by assisted reproductive technology. Pediatrics 2016, 137, e20152061. [Google Scholar] [CrossRef] [Green Version]
- Fleming, T.P.; Watkins, A.J.; Velazquez, M.A.; Mathers, J.C.; Prentice, A.M.; Stephenson, J.; Barker, M.; Saffery, R.; Yajnik, C.S.; Eckert, J.J.; et al. Origins of lifetime health around the time of conception: Causes and consequences. Lancet 2018, 391, 1842–1852. [Google Scholar] [CrossRef]
- Cadenas, E.; Packer, L.; Traber, M.G. Antioxidants, oxidants, and redox impacts on cell function—A tribute to Helmut Sies. Arch. Biochem. Biophys. 2016, 595, 94–99. [Google Scholar] [CrossRef]
- Covarrubias, L.; Hernández-García, D.; Schnabel, D.; Salas-Vidal, E.; Castro-Obregón, S. Function of reactive oxygen species during animal development: Passive or active? Dev. Biol. 2008, 320, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aitken, R.J.; De Iuliis, G.N.; Drevet, J.R. Oxidants, Antioxidants and Impact of the Oxidative Status in Male Reproduction; Academic Press: Cambridge, MA, USA, 2019; pp. 91–100. [Google Scholar]
- Tremellen, K. Oxidants, Antioxidants and Impact of the Oxidative Status in Male Reproduction; Academic Press: Cambridge, MA, USA, 2019; pp. 225–235. [Google Scholar]
- Aitken, R.J.; Muscio, L.; Whiting, S.; Connaughton, H.S.; Fraser, B.A.; Nixon, B.; Smith, N.D.; De Iuliis, G.N. Analysis of the effects of polyphenols on human spermatozoa reveals unexpected impacts on mitochondrial membrane potential, oxidative stress and DNA integrity; implications for assisted reproductive technology. Biochem. Pharmacol. 2016, 121, 78–96. [Google Scholar] [CrossRef]
- Agarwal, A.; Said, TM.; Bedaiwy, M.A.; Banerjee, J.; Alvarez, J.G. Oxidative stress in an assisted reproductive techniques setting. Fertil. Steril. 2006, 86, 503–512. [Google Scholar] [CrossRef]
- Gupta, S.; Sekhon, L.; Kim, Y.; Agarwal, A. The Role of Oxidative Stress and Antioxidants in Assisted Reproduction. Curr. Women’s Health Rev. 2010, 6, 227–238. [Google Scholar] [CrossRef]
- Sartori, C.; Rimoldi, S.F.; Rexhaj, E.; Allemann, Y.; Scherrer, U. Hypoxia; Springer: New York, NY, USA, 2016; pp. 55–62. [Google Scholar]
- Agarwal, A.; Gupta, S.; Sharma, R.K. Role of oxidative stress in female reproduction. Reprod. Biol. Endocrinol. 2005, 3, 28. [Google Scholar] [CrossRef] [Green Version]
- Muoio, D.M.; Newgard, C.B. Mechanisms of disease: Molecular and metabolic mechanisms of insulin resistance and β-cell failure in type 2 diabetes. Nat. Rev. Mol. Cell Biol. 2008, 9, 193–205. [Google Scholar] [CrossRef]
- Ho, E.; Karimi Galougahi, K.; Liu, C.C.; Bhindi, R.; Figtree, G.A. Biological markers of oxidative stress: Applications to cardiovascular research and practice. Redox Biol. 2013, 1, 483–491. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, M.T.; Beal, M.F. Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 2006, 443, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Jacob, K.D.; Noren Hooten, N.; Trzeciak, A.R.; Evans, M.K. Markers of oxidant stress that are clinically relevant in aging and age-related disease. Mech. Ageing Dev. 2013, 134, 139–157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mahfouz, R.; Sharma, R.; Thiyagarajan, A.; Kale, V.; Gupta, S.; Sabanegh, E.; Agarwal, A. Semen characteristics and sperm DNA fragmentation in infertile men with low and high levels of seminal reactive oxygen species. Fertil. Steril. 2010, 94, 2141–2146. [Google Scholar] [CrossRef] [PubMed]
- Allamaneni, S.S.R.; Naughton, C.K.; Sharma, R.K.; Thomas, A.J.; Agarwal, A. Increased seminal reactive oxygen species levels in patients with varicoceles correlate with varicocele grade but not with testis size. Fertil. Steril. 2004, 82, 1684–1686. [Google Scholar] [CrossRef]
- Biçer, Ş.; Gürsul, C.; Sayar, İ.; Akman, O.; Çakarlı, S.; Aydın, M. Role of ozone therapy in preventing testicular damage in an experimental cryptorchid rat model. Med. Sci. Monit. 2018, 24, 5832–5839. [Google Scholar] [CrossRef]
- Guimarães, S.B.; Aragão, A.A.; Santos, J.M.V.; Kimura, O.D.S.; Barbosa, P.H.U.; De Vasconcelos, P.R.L. Oxidative stress induced by torsion of the spermatic cord in young rats. Acta Cir. Bras. 2007, 22, 30–33. [Google Scholar] [CrossRef] [Green Version]
- La Vignera, S.; Condorelli, R.; D’Agata, R.; Vicari, E.; Calogero, A.E. Semen alterations and flow-citometry evaluation in patients with male accessory gland infections. J. Endocrinol. Investig. 2012, 35, 219–223. [Google Scholar] [CrossRef]
- Tremellen, K. Oxidative stress and male infertility—A clinical perspective. Hum. Reprod. Update 2008, 14, 243–258. [Google Scholar] [CrossRef]
- Pearce, K.L.; Hill, A.; Tremellen, K.P. Obesity related metabolic endotoxemia is associated with oxidative stress and impaired sperm DNA integrity. Basic Clin. Androl. 2019, 29, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Pereira, R.; Sá, R.; Barros, A.; Sousa, M. Major regulatory mechanisms involved in sperm motility. Asian J. Androl. 2017, 19, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Pasqualotto, F.F.; Sharma, R.K.; Pasqualotto, E.B.; Agarwal, A. Poor semen quality and ROS-TAC scores in patients with idiopathic infertility. Urol. Int. 2008, 81, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Sikka, S.C. Oxidative stress and role of antioxidants in normal and abnormal sperm function. Front. Biosci. 1996, 1, e78–e86. [Google Scholar] [CrossRef] [Green Version]
- Agarwal, A.; Parekh, N.; Selvam, M.K.P.; Henkel, R.; Shah, R.; Homa, S.T.; Ramasamy, R.; Ko, E.; Tremellen, K.; Esteves, S.; et al. Male Oxidative Stress Infertility (MOSI): Proposed Terminology and Clinical Practice Guidelines for Management of Idiopathic Male Infertility. World J. Mens Health 2019, 37, 296–312. [Google Scholar] [CrossRef]
- Marchetti, C. Study of mitochondrial membrane potential, reactive oxygen species, DNA fragmentation and cell viability by flow cytometry in human sperm. Hum. Reprod. 2002, 17, 1257–1265. [Google Scholar] [CrossRef] [Green Version]
- Plante, M.; De Lamirande, E.; Gagnon, C. Reactive oxygen species released by activated neutrophils, but not by deficient spermatozoa, are sufficient to affect normal sperm motility. Fertil. Steril. 1994, 62, 387–393. [Google Scholar] [CrossRef]
- Sela, S.; Mazor, R.; Amsalam, M.; Yagil, C.; Yagil, Y.; Kristal, B. Primed polymorphonuclear leukocytes, oxidative stress, and inflammation antecede hypertension in the Sabra rat. Hypertension 2004, 44, 764–769. [Google Scholar] [CrossRef] [Green Version]
- Alvarez, J.G.; Storey, B.T. Differential incorporation of fatty acids into and peroxidative loss of fatty acids from phospholipids of human spermatozoa. Mol. Reprod. Dev. 1995, 42, 334–346. [Google Scholar] [CrossRef]
- Cannarella, R.; Calogero, A.E.; Condorelli, R.A.; Giacone, F.; Mongioi, L.M.; La Vignera, S. Non-hormonal treatment for male infertility: The potential role of Serenoa repens, selenium and lycopene. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 3112–3120. [Google Scholar] [CrossRef]
- Agarwal, A.; Plessis, S.S.D.; Durairajanayagam, D.; Virk, G. Strategies to Ameliorate Oxidative Stress during Assisted Reproduction; Springer: Berlin/Heidelberg, Germany, 2014; p. 7. [Google Scholar]
- Bisht, S.; Faiq, M.; Tolahunase, M.; Dada, R. Oxidative stress and male infertility. Nat. Rev. Urol. 2017, 14, 470–485. [Google Scholar] [CrossRef] [PubMed]
- Benchaib, M.; Braun, V.; Lornage, J.; Hadj, S.; Salle, B.; Lejeune, H.; Guérin, J.F. Sperm DNA fragentation decreases the pregnancy rate in an assisted reproductive technique. Hum. Reprod. 2003, 18, 1023–1028. [Google Scholar] [CrossRef] [PubMed]
- Saleh, R.A.; Agarwal, A.; Sharma, R.K.; Nelson, D.R.; Thomas, A.J. Effect of cigarette smoking on levels of seminal oxidative stress in infertile men: A prospective study. Fertil. Steril. 2002, 78, 491–499. [Google Scholar] [CrossRef]
- Aboulmaouahib, S.; Madkour, A.; Kaarouch, I.; Sefrioui, O.; Saadani, B.; Copin, H.; Benkhalifa, M.; Louanjli, N.; Cadi, R. Impact of alcohol and cigarette smoking consumption in male fertility potential: Looks at lipid peroxidation, enzymatic antioxidant activities and sperm DNA damage. Andrologia 2018, 50, e12926. [Google Scholar] [CrossRef] [PubMed]
- Eskiocak, S.; Gozen, A.S.; Taskiran, A.; Kilic, A.S.; Eskiocak, M.; Gulen, S. Effect of psychological stress on the L-arginine-nitric oxide pathway and semen quality. Braz. J. Med. Biol. Res. 2006, 39, 581–588. [Google Scholar] [CrossRef] [Green Version]
- Nudell, D.M.; Monoski, M.M.; Lipshultz, L.I. Common medications and drugs: How they affect male fertility. Urol. Clin. N. Am. 2002, 29, 965–973. [Google Scholar] [CrossRef]
- Ames, B.N. Micronutrient deficiencies. A major cause of DNA damage. Ann. N. Y. Acad. Sci. 1999, 889, 87–106. [Google Scholar] [CrossRef]
- Anderson, K.; Nisenblat, V.; Norman, R. Lifestyle factors in people seeking infertility treatment—A review. Aust. N. Z. J. Obstet. Gynaecol. 2010, 50, 8–20. [Google Scholar] [CrossRef]
- Rubes, J.; Selevan, S.G.; Evenson, D.P.; Zudova, D.; Vozdova, M.; Zudova, Z.; Robbins, W.A.; Perreault, S.D. Episodic air pollution is associated with increased DNA fragmentation in human sperm without other changes in semen quality. Hum. Reprod. 2005, 20, 2776–2783. [Google Scholar] [CrossRef] [Green Version]
- Zhou, D.D.; Hao, J.L.; Guo, K.M.; Lu, C.W.; Liu, X.D. Sperm quality and DNA damage in men from Jilin Province, China, who are occupationally exposed to ionizing radiation. Genet. Mol. Res. 2016, 15, 1–7. [Google Scholar] [CrossRef]
- Pérez-Crespo, M.; Pintado, B.; Gutiérrez-Adán, A. Scrotal heat stress effects on sperm viability, sperm DNA integrity, and the offspring sex ratio in mice. Mol. Reprod. Dev. 2008, 75, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.X.; Zeng, Q.; Sun, Y.; You, L.; Wang, P.; Li, M.; Yang, P.; Li, J.; Huang, Z.; Wang, C.; et al. Phthalate exposure in association with serum hormone levels, sperm DNA damage and spermatozoa apoptosis: A cross-sectional study in China. Environ. Res. 2016, 150, 557–565. [Google Scholar] [CrossRef]
- Xu, D.X.; Shen, H.M.; Zhu, Q.X.; Chua, L.; Wang, Q.N.; Chia, S.E.; Ong, C.N. The associations among semen quality, oxidative DNA damage in human spermatozoa and concentrations of cadmium, lead and selenium in seminal plasma. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2003, 534, 155–163. [Google Scholar] [CrossRef]
- Paoli, D.; Pallotti, F.; Lenzi, A.; Lombardo, F. Fatherhood and sperm DNA damage in testicular cancer patients. Front. Endocrinol. (Lausanne) 2018, 9, 506. [Google Scholar] [CrossRef] [PubMed]
- Chibber, S.; Farhan, M.; Hassan, I.; Naseem, I. White light-mediated Cu (II)-5FU interaction augments the chemotherapeutic potential of 5-FU: An in vitro study. Tumor Biol. 2011, 32, 881–892. [Google Scholar] [CrossRef] [PubMed]
- Pasqualotto, E.B.; Agarwal, A.; Sharma, R.K.; Izzo, V.M.; Pinotti, J.A.; Joshi, N.J.; Rose, B.I. Effect of oxidative stress in follicular fluid on the outcome of assisted reproductive procedures. Fertil. Steril. 2004, 81, 973–976. [Google Scholar] [CrossRef]
- Bedaiwy, M.A.; Falcone, T.; Mohamed, M.S.; Aleem, A.A.N.; Sharma, R.K.; Worley, S.E.; Thornton, J.; Agarwal, A. Differential growth of human embryos in vitro: Role of reactive oxygen species. Fertil. Steril. 2004, 82, 593–600. [Google Scholar] [CrossRef]
- Guérin, P.; El Mouatassim, S.; Ménézo, Y. Oxidative stress and protection against reactive oxygen species in the pre-implantation embryo and its surroundings. Hum. Reprod. Update 2001, 7, 175–189. [Google Scholar] [CrossRef]
- Oral, O.; Kutlu, T.; Aksoy, E.; Fıçıcıoğlu, C.; Uslu, H.; Tuğrul, S. The effects of oxidative stress on outcomes of assisted reproductive techniques. J. Assist. Reprod. Genet. 2006, 23, 81–85. [Google Scholar] [CrossRef]
- Chattopadhayay, R.; Ganesh, A.; Samanta, J.; Jana, S.K.; Chakravarty, B.N.; Chaudhury, K. Effect of follicular fluid oxidative stress on meiotic spindle formation in infertile women with polycystic ovarian syndrome. Gynecol. Obstet. Investig. 2010, 69, 197–202. [Google Scholar] [CrossRef]
- Máté, G.; Bernstein, L.R.; Török, A.L. Endometriosis Is a Cause of Infertility. Does Reactive Oxygen Damage to Gametes and Embryos Play a Key Role in the Pathogenesis of Infertility Caused by Endometriosis? Front. Endocrinol. (Lausanne) 2018, 9, 725. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, D.; Luo, W.Y.; Liao, H.; Wang, C.F.; Sun, Y. The effects of oxidative stress to PCOS. Sichuan Da Xue Xue Bao 2008, 39, 421–423. [Google Scholar] [PubMed]
- Bedaiwy, M.A.; Goldberg, J.M.; Falcone, T.; Singh, M.; Nelson, D.; Azab, H.; Wang, X.; Sharma, R. Relationship between oxidative stress and embryotoxicity of hydrosalpingeal fluid. Hum. Reprod. 2002, 17, 601–604. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qin, J.; Liu, X.; Sheng, X.; Wang, H.; Gao, S. Assisted reproductive technology and the risk of pregnancy-related complications and adverse pregnancy outcomes in singleton pregnancies: A meta-analysis of cohort studies. Fertil. Steril. 2016, 105, 73–85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suhag, A.; Berghella, V. Intrauterine Growth Restriction (IUGR): Etiology and Diagnosis. Curr. Obstet. Gynecol. Rep. 2013, 2, 102–111. [Google Scholar] [CrossRef] [Green Version]
- Agarwal, A.; Aponte-Mellado, A.; Premkumar, B.J.; Shaman, A.; Gupta, S. The effects of oxidative stress on female reproduction: A review. Reprod. Biol. Endocrinol. 2012, 10, 49. [Google Scholar] [CrossRef] [Green Version]
- Mihalas, B.P.; De Iuliis, G.N.; Redgrove, K.A.; McLaughlin, E.A.; Nixon, B. The lipid peroxidation product 4-hydroxynonenal contributes to oxidative stress-mediated deterioration of the ageing oocyte. Sci. Rep. 2017, 7, 6247. [Google Scholar] [CrossRef]
- Prasad, S.; Tiwari, M.; Pandey, A.N.; Shrivastav, T.G.; Chaube, S.K. Impact of stress on oocyte quality and reproductive outcome. J. Biomed. Sci. 2016, 23, 36. [Google Scholar] [CrossRef] [Green Version]
- Cecchino, G.N.; Seli, E.; Alves da Motta, E.L.; García-Velasco, J.A. The role of mitochondrial activity in female fertility and assisted reproductive technologies: Overview and current insights. Reprod. Biomed. Online 2018, 36, 686–697. [Google Scholar] [CrossRef] [Green Version]
- Gudmundsdottir, S.L.; Flanders, W.D.; Augestad, L.B. Physical activity and fertility in women: The North-Trøndelag Health Study. Hum. Reprod. 2009, 24, 3196–3204. [Google Scholar] [CrossRef] [Green Version]
- Abir, R.; Nitke, S.; Ben-Haroush, A.; Fisch, B. In vitro maturation of human primordial ovarian follicles: Clinical significance, progress in mammals, and methods for growth evaluation. Histol. Histopathol. 2006, 21, 887–898. [Google Scholar] [CrossRef] [PubMed]
- Conklin, K.A. Chemotherapy-associated oxidative stress: Impact on chemotherapeutic effectiveness. Integr. Cancer Ther. 2004, 3, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Conforti, A.; Mascia, M.; Cioffi, G.; De Angelis, C.; Coppola, G.; De Rosa, P.; Pivonello, R.; Alviggi, C.; De Placido, G. Air pollution and female fertility: A systematic review of literature. Reprod. Biol. Endocrinol. 2018, 16, 117. [Google Scholar] [CrossRef] [PubMed]
- Ruder, E.H.; Hartman, T.J.; Blumberg, J.; Goldman, M.B. Oxidative stress and antioxidants: Exposure and impact on female fertility. Hum. Reprod. Update 2008, 14, 345–357. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zribi, N.; Feki Chakroun, N.; El Euch, H.; Gargouri, J.; Bahloul, A.; Ammar Keskes, L. Effects of cryopreservation on human sperm deoxyribonucleic acid integrity. Fertil. Steril. 2010, 93, 159–166. [Google Scholar] [CrossRef]
- Gualtieri, R.; Iaccarino, M.; Mollo, V.; Prisco, M.; Iaccarino, S.; Talevi, R. Slow cooling of human oocytes: Ultrastructural injuries and apoptotic status. Fertil. Steril. 2009, 91, 1023–1034. [Google Scholar] [CrossRef]
- Saeed, Z.; Ali, T.; Hadi, H. Amending in vitro culture condition to overcome oxidative stress in assisted reproduction techniques (ART). J. Paramed. Sci. 2015, 6, 2. [Google Scholar] [CrossRef]
- Shahar, S.; Wiser, A.; Ickowicz, D.; Lubart, R.; Shulman, A.; Breitbart, H. Light-mediated activation reveals a key role for protein kinase A and sarcoma protein kinase in the development of sperm hyper-activated motility. Hum. Reprod. 2011, 26, 2274–2282. [Google Scholar] [CrossRef] [Green Version]
- Will, M.A.; Clark, N.A.; Swain, J.E. Biological pH buffers in IVF: Help or hindrance to success. J. Assist. Reprod. Genet. 2011, 28, 711–724. [Google Scholar] [CrossRef] [Green Version]
- Larkindale, J.; Knight, M.R. Protection against heat stress-induced oxidative damage in Arabidopsis involves calcium, abscisic acid, ethylene, and salicylic acid. Plant Physiol. 2002, 128, 682–695. [Google Scholar] [CrossRef]
- Bontekoe, S.; Mantikou, E.; van Wely, M.; Seshadri, S.; Repping, S.; Mastenbroek, S. Low oxygen concentrations for embryo culture in assisted reproductive technologies. Cochrane Database Syst. Rev. 2012, 11, CD008950. [Google Scholar] [CrossRef] [PubMed]
- Lampiao, F.; Strijdom, H.; Plessis, S. d. Effects of sperm processing techniques involving centrifugation on nitric oxide, reactive oxygen species generation and sperm function. Open Androl. J. 2010, 2, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Calzi, F.; Papaleo, E.; Rabellotti, E.; Ottolina, J.; Vailati, S.; Viganò, P.; Candiani, M. Exposure of embryos to oxygen at low concentration in a cleavage stage transfer program: Reproductive outcomes in a time-series analysis. Clin. Lab. 2012, 58, 997–1003. [Google Scholar] [CrossRef]
- Cohen, J.; Gilligan, A.; Esposito, W.; Schimmel, T.; Dale, B. Ambient air and its potential effects on conception in vitro. Hum. Reprod. 1997, 12, 1742–1749. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agarwal, A.; Durairajanayagam, D.; du Plessis, S.S. Utility of antioxidants during assisted reproductive techniques: An evidence based review. Reprod. Biol. Endocrinol. 2014, 12, 112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peritore, A.F.; Siracusa, R.; Crupi, R.; Cuzzocrea, S. Therapeutic efficacy of palmitoylethanolamide and its new formulations in synergy with different antioxidant molecules present in diets. Nutrients 2019, 11, 2175. [Google Scholar] [CrossRef] [Green Version]
- Halliwell, B.; Gutteridge, J.M.C. Oxidants, Free Radicals in Biology and Medicine; Oxford University Press: Oxford, UK, 2015; p. 34. [Google Scholar]
- Tan, D.Q.; Suda, T. Reactive Oxygen Species and Mitochondrial Homeostasis as Regulators of Stem Cell Fate and Function. Antioxid. Redox Signal. 2018, 29, 149–168. [Google Scholar] [CrossRef]
- Ekstrand, M.; Trajkovska, M.G.; Perman-Sundelin, J.; Fogelstrand, P.; Adiels, M.; Johansson, M.; Mattsson-Hultén, L.; Borén, J.; Levin, M. Imaging of intracellular and extracellular ROS levels in atherosclerotic mouse aortas ex vivo: Effects of lipid lowering by diet or atorvastatin. PLoS ONE 2015, 10, e0130898. [Google Scholar] [CrossRef]
- Rinaldi, M.; Ceciliani, F.; Lecchi, C.; Moroni, P.; Bannerman, D.D. Differential effects of α1-acid glycoprotein on bovine neutrophil respiratory burst activity and IL-8 production. Vet. Immunol. Immunopathol. 2008, 126, 199–210. [Google Scholar] [CrossRef]
- Khandrika, L.; Kumar, B.; Koul, S.; Maroni, P.; Koul, H.K. Oxidative stress in prostate cancer. Cancer Lett. 2009, 282, 125–136. [Google Scholar] [CrossRef] [Green Version]
- Lenaz, G. The mitochondrial production of reactive oxygen species: Mechanisms and implications in human pathology. IUBMB Life 2001, 52, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Martín-Romero, F.J.; Miguel-Lasobras, E.M.; Domínguez-Arroyo, J.A.; Gonzélez-Carrera, E.; Álvarez, I.S. Contribution of culture media to oxidative stress and its effect on human oocytes. Reprod. Biomed. Online 2008, 17, 652–661. [Google Scholar] [CrossRef]
- Vundru, S.S.; Prasad, N.; Patel, R.; Rani, V.; Yadav, U.C.S. Free Radicals in Human Health and Disease; Springer: Berlin/Heidelberg, Germany, 2015; p. 76. [Google Scholar]
- Guo, Y.; Yu, S.; Zhang, C.; Kong, A.N.T. Epigenetic regulation of Keap1-Nrf2 signaling. Free Radic. Biol. Med. 2015, 88, 337–349. [Google Scholar] [CrossRef] [PubMed]
- Bavineni, M.; Wassenaar, T.M.; Agnihotri, K.; Ussery, D.W.; Lüscher, T.F.; Mehta, J.L. Mechanisms linking preterm birth to onset of cardiovascular disease later in adulthood. Eur. Heart J. 2019, 40, 1107–1112. [Google Scholar] [CrossRef] [Green Version]
- Hoeijmakers, L.; Kempe, H.; Verschure, P.J. Epigenetic imprinting during assisted reproductive technologies: The effect of temporal and cumulative fluctuations in methionine cycling on the DNA methylation state. Mol. Reprod. Dev. 2016, 83, 94–107. [Google Scholar] [CrossRef] [Green Version]
- Mayneris-Perxachs, J.; Lima, A.A.; Guerrant, R.L.; Leite Á, M.; Moura, A.F.; Lima, N.L.; Swann, J.R. Urinary N-methylnicotinamide and β-aminoisobutyric acid predict catch-up growth in undernourished Brazilian children. Sci. Rep. 2016, 6, 19780. [Google Scholar] [CrossRef] [Green Version]
- Kindsfather, A.J.; Czekalski, M.A.; Pressimone, C.A.; Erisman, M.P.; Mann, M.R.W. Perturbations in imprinted methylation from assisted reproductive technologies but not advanced maternal age in mouse preimplantation embryos. Clin. Epigenetics 2019, 11, 162. [Google Scholar] [CrossRef]
- Movahed, E.; Soleimani, M.; Hosseini, S.; Akbari Sene, A.; Salehi, M. Aberrant expression of miR-29a/29b and methylation level of mouse embryos after in vitro fertilization and vitrification at two-cell stage. J. Cell Physiol. 2019, 234, 18942–18950. [Google Scholar] [CrossRef]
- Rexhaj, E.; Bloch, J.; Jayet, P.Y.; Rimoldi, S.F.; Dessen, P.; Mathieu, C.; Tolsa, J.F.; Nicod, P.; Scherrer, U.; Sartori, C. Fetal programming of pulmonary vascular dysfunction in mice: Role of epigenetic mechanisms. Am. J. Physiol. Heart Circ. Physiol. 2011, 301, H247–H252. [Google Scholar] [CrossRef]
- Kietzmann, T.; Petry, A.; Shvetsova, A.; Gerhold, J.M.; Görlach, A. The epigenetic landscape related to reactive oxygen species formation in the cardiovascular system. Br. J. Pharmacol. 2017, 174, 1533–1554. [Google Scholar] [CrossRef]
- Booth, M.J.; Ost, T.W.B.; Beraldi, D.; Bell, N.M.; Branco, M.R.; Reik, W.; Balasubramanian, S. Oxidative bisulfite sequencing of 5-methylcytosine and 5-hydroxymethylcytosine. Nat. Protoc. 2013, 8, 1841–1851. [Google Scholar] [CrossRef] [Green Version]
- Madugundu, G.S.; Cadet, J.; Wagner, J.R. Hydroxyl-radical-induced oxidation of 5-methylcytosine in isolated and cellular DNA. Nucleic Acids Res. 2014, 42, 7450–7460. [Google Scholar] [CrossRef] [PubMed]
- Branco, M.R.; Ficz, G.; Reik, W. Uncovering the role of 5-hydroxymethylcytosine in the epigenome. Nat. Rev. Genet. 2012, 13, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.; Desmarais, T.L.; Tong, Z.; Yao, Y.; Costa, M. Oxidative stress alters global histone modification and DNA methylation. Free Radic. Biol. Med. 2015, 82, 22–28. [Google Scholar] [CrossRef] [Green Version]
- Tsankova, N.; Renthal, W.; Kumar, A.; Nestler, E.J. Epigenetic regulation in psychiatric disorders. Nat. Rev. Neurosci. 2007, 8, 355–367. [Google Scholar] [CrossRef] [PubMed]
- Saenen, N.D.; Martens, D.S.; Neven, K.Y.; Alfano, R.; Bové, H.; Janssen, B.G.; Roels, H.A.; Plusquin, M.; Vrijens, K.; Nawrot, T.S. Air pollution-induced placental alterations: An interplay of oxidative stress, epigenetics, and the aging phenotype? Clin. Epigenetics 2019, 11, 124. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Jiang, B.H. Interplay Between Reactive Oxygen Species and MicroRNAs in Cancer. Curr. Pharmacol. Rep. 2016, 2, 82–90. [Google Scholar] [CrossRef] [Green Version]
- Camici, G.G.; Schiavoni, M.; Francia, P.; Bachschmid, M.; Martin-Padura, I.; Hersberger, M.; Tanner, F.C.; Pelicci, P.G.; Volpe, M.; Anversa, P.; et al. Genetic deletion of p66Shc adaptor protein prevents hyperglycemia-induced endothelial dysfunction and oxidative stress. Proc. Natl. Acad. Sci. USA 2007, 104, 5217–5222. [Google Scholar] [CrossRef] [Green Version]
- Costantino, S.; Paneni, F.; Virdis, A.; Hussain, S.; Mohammed, S.A.; Capretti, G.; Akhmedov, A.; Dalgaard, K.; Chiandotto, S.; Pospisilik, J.A.; et al. Interplay among H3K9-editing enzymes SUV39H1, JMJD2C and SRC-1 drives p66 Shc transcription and vascular oxidative stress in obesity. Eur. Heart J. 2019, 40, 383–391. [Google Scholar] [CrossRef] [Green Version]
- Shimoyama, Y.; Mitsuda, Y.; Hamajima, N.; Niwa, T. Polymorphisms of Nrf2, an antioxidative gene, are associated with blood pressure in Japanese. Nagoya J. Med. Sci. 2014, 76, 113–120. [Google Scholar] [CrossRef]
- Satta, S.; Mahmoud, A.M.; Wilkinson, F.L.; Yvonne Alexander, M.; White, S.J. The Role of Nrf2 in Cardiovascular Function and Disease. Oxid. Med. Cell. Longev. 2017, 2017, 9237263. [Google Scholar] [CrossRef] [PubMed]
- Czogalla, B.; Kahaly, M.; Mayr, D.; Schmoeckel, E.; Niesler, B.; Kolben, T.; Burges, A.; Mahner, S.; Jeschke, U.; Trillsch, F. Interaction of ERα and NRF2 impacts survival in ovarian cancer patients. Int. J. Mol. Sci. 2019, 20, 112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luo, M.; Shang, L.; Brooks, M.D.; Jiagge, E.; Zhu, Y.; Buschhaus, J.M.; Conley, S.; Fath, M.A.; Davis, A.; Gheordunescu, E.; et al. Targeting Breast Cancer Stem Cell State Equilibrium through Modulation of Redox Signaling. Cell Metab. 2018, 28, 69–86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schrier, M.S.; Trivedi, M.S.; Deth, R.C. Redox-related epigenetic mechanisms in glioblastoma: Nuclear factor (erythroid-derived 2)-like 2, cobalamin, and dopamine receptor subtype 4. Front. Oncol. 2017, 7, 46. [Google Scholar] [CrossRef] [Green Version]
- Cuadrado, A.; Rojo, A.I.; Wells, G.; Hayes, J.D.; Cousin, S.P.; Rumsey, W.L.; Attucks, O.C.; Franklin, S.; Levonen, A.L.; Kensler, T.W.; et al. Therapeutic targeting of the NRF2 and KEAP1 partnership in chronic diseases. Nat. Rev. Drug Discov. 2019, 18, 295–317. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Impellizzeri, D.; Siracusa, R.; Cordaro, M.; Crupi, R.; Peritore, A.F.; Gugliandolo, E.; D’Amico, R.; Petrosino, S.; Evangelista, M.; Di Paola, R.; et al. N-Palmitoylethanolamine-oxazoline (PEA-OXA): A new therapeutic strategy to reduce neuroinflammation, oxidative stress associated to vascular dementia in an experimental model of repeated bilateral common carotid arteries occlusion. Neurobiol. Dis. 2019, 125, 77–91. [Google Scholar] [CrossRef]
- Yu, Q.; Li, X.; Cao, X. Linarin could protect myocardial tissue from the injury of Ischemia-reperfusion through activating Nrf-2. Biomed. Pharmacother. 2017, 90, 1–7. [Google Scholar] [CrossRef]
- Bonnefont-Rousselot, D. Resveratrol and cardiovascular diseases. Nutrients 2016, 8, 250. [Google Scholar] [CrossRef]
- Wan, J.; Winn, L.M. In utero-initiated cancer: The role of reactive oxygen species. Birth Defects Res. Part C Embryo Today Rev. 2006, 78, 326–332. [Google Scholar] [CrossRef]
- Giudice, A.; Arra, C.; Turco, M.C. Review of molecular mechanisms involved in the activation of the Nrf2-ARE signaling pathway by chemopreventive agents. Methods Mol. Biol. 2010, 647, 37–74. [Google Scholar] [CrossRef]
- Amin, A. NRF2 Mediated Oxidative Stress Response Activity During Early In Vitro Bovine Embryo Development. Ph.D. Thesis, Universitäts-und Landesbibliothek Bonn, Bonn, Germany, 2015. [Google Scholar]
- Chapple, S.J.; Puszyk, W.M.; Mann, G.E. Keap1-Nrf2 regulated redox signaling in utero: Priming of disease susceptibility in offspring. Free Radic. Biol. Med. 2015, 88, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Mann, G.E.; Niehueser-Saran, J.; Watson, A.; Gao, L.; Ishii, T.; de Winter, P.; Siow, R.C. Nrf2/ARE regulated antioxidant gene expression in endothelial and smooth muscle cells in oxidative stress: Implications for atherosclerosis and preeclampsia. Sheng Li Xue Bao 2007, 59, 117–127. [Google Scholar] [PubMed]
- Kensler, T.W.; Wakabayashi, N.; Biswal, S. Cell Survival Responses to Environmental Stresses Via the Keap1-Nrf2-ARE Pathway. Annu. Rev. Pharmacol. Toxicol. 2007, 47, 89–116. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.D.; Dinkova-Kostova, A.T. The Nrf2 regulatory network provides an interface between redox and intermediary metabolism. Trends Biochem. Sci. 2014, 39, 199–218. [Google Scholar] [CrossRef] [PubMed]
- Kwak, M.K.; Wakabayashi, N.; Itoh, K.; Motohashi, H.; Yamamoto, M.; Kensler, T.W. Modulation of gene expression by cancer chemopreventive dithiolethiones through the Keap1-Nrf2 pathway: Identification of novel gene clusters for cell survival. J. Biol. Chem. 2003, 278, 8135–8145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Macleod, A.K.; Mcmahon, M.; Plummer, S.M.; Higgins, L.G.; Penning, T.M.; Igarashi, K.; Hayes, J.D. Characterization of the cancer chemopreventive NRF2-dependent gene battery in human keratinocytes: Demonstration that the KEAP1-NRF2 pathway, and not the BACH1-NRF2 pathway, controls cytoprotection against electrophiles as well as redox-cycling compounds. Carcinogenesis 2009, 30, 1571–1580. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, J.; Lee, J.M.; Johnson, J.A. Microarray analysis reveals an antioxidant responsive element-driven gene set involved in conferring protection from an oxidative stress-induced apoptosis in IMR-32 cells. J. Biol. Chem. 2002, 277, 388–394. [Google Scholar] [CrossRef] [Green Version]
- Song, D.; Cheng, Y.; Li, X.; Wang, F.; Lu, Z.; Xiao, X.; Wang, Y. Biogenic Nanoselenium Particles Effectively Attenuate Oxidative Stress-Induced Intestinal Epithelial Barrier Injury by Activating the Nrf2 Antioxidant Pathway. ACS Appl. Mater. Interfaces 2017, 9, 14724–14740. [Google Scholar] [CrossRef]
- Singh, A.; Rangasamy, T.; Thimmulappa, R.K.; Lee, H.; Osburn, W.O.; Brigelius-Flohé, R.; Kensler, T.W.; Yamamoto, M.; Biswal, S. Glutathione peroxidase 2, the major cigarette smoke-inducible isoform of GPX in lungs, is regulated by Nrf2. Am. J. Respir. Cell Mol. Biol. 2006, 35, 639–650. [Google Scholar] [CrossRef]
- Cho, H.Y.; Reddy, S.P.; DeBiase, A.; Yamamoto, M.; Kleeberger, S.R. Gene expression profiling of NRF2-mediated protection against oxidative injury. Free Radic. Biol. Med. 2005, 38, 325–343. [Google Scholar] [CrossRef]
- Okawa, H.; Motohashi, H.; Kobayashi, A.; Aburatani, H.; Kensler, T.W.; Yamamoto, M. Hepatocyte-specific deletion of the keap1 gene activates Nrf2 and confers potent resistance against acute drug toxicity. Biochem. Biophys. Res. Commun. 2006, 339, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Banning, A.; Deubel, S.; Kluth, D.; Zhou, Z.; Brigelius-Flohé, R. The GI-GPx Gene Is a Target for Nrf2. Mol. Cell. Biol. 2005, 25, 4914–4923. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Habeos, I.G.; Ziros, P.G.; Chartoumpekis, D.; Psyrogiannis, A.; Kyriazopoulou, V.; Papavassiliou, A.G. Simvastatin activates Keap1/Nrf2 signaling in rat liver. J. Mol. Med. 2008, 86, 1279–1285. [Google Scholar] [CrossRef] [PubMed]
- Reisman, S.A.; Yeager, R.L.; Yamamoto, M.; Klaassen, C.D. Increased Nrf2 activation in livers from keap1-knockdown mice Increases expression of cytoprotective genes that detoxify electrophiles more than those that detoxify reactive oxygen species. Toxicol. Sci. 2009, 108, 35–47. [Google Scholar] [CrossRef]
- Rangasamy, T.; Cho, C.Y.; Thimmulappa, R.K.; Zhen, L.; Srisuma, S.S.; Kensler, T.W.; Yamamoto, M.; Petrache, I.; Tuder, R.M.; Biswal, S. Genetic ablation of Nrf2 enhances susceptibility to cigarette smoke-induced emphysema in mice. J. Clin. Investig. 2004, 114, 1248–1259. [Google Scholar] [CrossRef]
- Thimmulappa, R.K.; Mai, K.H.; Srisuma, S.; Kensler, T.W.; Yamamoto, M.; Biswal, S. Identification of Nrf2-regulated genes induced by the chemopreventive agent sulforaphane by oligonucleotide microarray. Cancer Res. 2002, 62, 5196–5203. [Google Scholar]
- Yates, M.S.; Kwak, M.K.; Egner, P.A.; Groopman, J.D.; Bodreddigari, S.; Sutter, T.R.; Baumgartner, K.J.; Roebuck, B.D.; Liby, K.T.; Yore, M.M.; et al. Potent protection against aflatoxin-induced tumorigenesis through induction of Nrf2-regulated pathways by the triterpenoid 1-[2-cyano-3-,12-dioxooleana-1, 9(11)-dien-28-oyl]imidazole. Cancer Res. 2006, 66, 2488–2494. [Google Scholar] [CrossRef] [Green Version]
- Hu, R.; Xu, C.; Shen, G.; Jain, M.R.; Khor, T.O.; Gopalkrishnan, A.; Lin, W.; Reddy, B.; Chan, J.Y.; Kong, A.N.T. Gene expression profiles induced by cancer chemopreventive isothiocyanate sulforaphane in the liver of C57BL/6J mice and C57BL/6J/Nrf2(−/−) mice. Cancer Lett. 2006, 243, 170–192. [Google Scholar] [CrossRef]
- Hayes, J.D.; Dinkova-Kostova, A.T. Epigenetic Control of NRF2-Directed Cellular Antioxidant Status in Dictating Life-Death Decisions. Mol. Cell 2017, 68, 5–7. [Google Scholar] [CrossRef]
- Chowdhury, I.; Mo, Y.; Gao, L.; Kazi, A.; Fisher, A.B.; Feinstein, S.I. Oxidant stress stimulates expression of the human peroxiredoxin 6 gene by a transcriptional mechanism involving an antioxidant response element. Free Radic. Biol. Med. 2009, 46, 146–153. [Google Scholar] [CrossRef]
- Mahaffey, C.M.; Zhang, H.; Rinna, A.; Holland, W.; Mack, P.C.; Forman, H.J. Multidrug-resistant protein-3 gene regulation by the transcription factor Nrf2 in human bronchial epithelial and non-small-cell lung carcinoma. Free Radic. Biol. Med. 2009, 46, 1650–1657. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Youle, R.J.; Van Der Bliek, A.M. Mitochondrial fission, fusion, and stress. Science 2012, 337, 1062–1065. [Google Scholar] [CrossRef] [Green Version]
- Rodford, J.L.; Torrens, C.; Siow, R.C.M.; Mann, G.E.; Hanson, M.A.; Clough, G.F. Endothelial dysfunction and reduced antioxidant protection in an animal model of the developmental origins of cardiovascular disease. J. Physiol. 2008, 586, 4709–4720. [Google Scholar] [CrossRef] [PubMed]
- Giussani, D.A.; Camm, E.J.; Niu, Y.; Richter, H.G.; Blanco, C.E.; Gottschalk, R.; Blake, E.Z.; Horder, K.A.; Thakor, A.S.; Hansell, J.A.; et al. Developmental programming of cardiovascular dysfunction by prenatal hypoxia and oxidative stress. PLoS ONE 2012, 7, e31017. [Google Scholar] [CrossRef] [Green Version]
- Do Franco, M.C.P.; Dantas, A.P.V.; Akamine, E.H.; Kawamoto, E.M.; Fortes, Z.B.; Scavone, C.; Tostes, R.C.A.; Carvalho, M.H.C.; Nigro, D. Enhanced oxidative stress as a potential mechanism underlying the programming of hypertension in utero. J. Cardiovasc. Pharmacol. 2002, 40, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Da Liao, X.; Wang, L.; Huang, X.; Li, Y.; Dasgupta, C.; Zhang, L. Protective effect of antenatal antioxidant on Nicotine-induced heart ischemia-sensitive phenotype in rat offspring. PLoS ONE 2016, 11, e0150557. [Google Scholar] [CrossRef]
- Cooke, C.L.M.; Shah, A.; Kirschenman, R.D.; Quon, A.L.; Morton, J.S.; Care, A.S.; Davidge, S.T. Increased susceptibility to cardiovascular disease in offspring born from dams of advanced maternal age. J. Physiol. 2018, 596, 5807–5821. [Google Scholar] [CrossRef]
- Shah, A.; Cooke, C.L.M.; Kirschenman, R.D.; Quon, A.L.; Morton, J.S.; Care, A.S.; Davidge, S.T. Sex-specific effects of advanced maternal age on cardiovascular function in aged adult rat offspring. Am. J. Physiol. Heart Circ. Physiol. 2018, 315, H1724–H1734. [Google Scholar] [CrossRef] [Green Version]
- Fox, R.; Kitt, J.; Leeson, P.; Aye, C.Y.L.; Lewandowski, A.J. Preeclampsia: Risk Factors, Diagnosis, Management, and the Cardiovascular Impact on the Offspring. J. Clin. Med. 2019, 8, 1625. [Google Scholar] [CrossRef] [Green Version]
- Tripathi, R.R.; Rifas-Shiman, S.L.; Hawley, N.; Hivert, M.F.; Oken, E. Hypertensive disorders of pregnancy and offspring cardiometabolic health at midchildhood: Project viva findings. J. Am. Heart Assoc. 2018, 7, e007426. [Google Scholar] [CrossRef] [Green Version]
- Yu, Y.; Arah, O.A.; Liew, Z.; Cnattingius, S.; Olsen, J.; Sørensen, H.T.; Qin, G.; Li, J. Maternal diabetes during pregnancy and early onset of cardiovascular disease in offspring: Population based cohort study with 40 years of follow-up. BMJ 2019, 367, l6398. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuo, A.H.; Li, C.; Huber, H.F.; Clarke, G.D.; Nathanielsz, P.W. Intrauterine growth restriction results in persistent vascular mismatch in adulthood. J. Physiol. 2018, 596, 5777–5790. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wen, S.W.; Leader, A.; White, R.R.; Léveillé, M.C.; Wilkie, V.; Zhou, J.; Walker, M.C. A comprehensive assessment of outcomes in pregnancies conceived by in vitro fertilization/intracytoplasmic sperm injection. Eur. J. Obstet. Gynecol. Reprod. Biol. 2010, 150, 160–165. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, Y.; Gu, H.T.; Feng, Q.L.; Liu, J.Y.; Zhou, J.; Yan, F. Association between assisted reproductive technology and cardiac alteration at age 5 years. JAMA Pediatr. 2015, 169, 603–605. [Google Scholar] [CrossRef] [Green Version]
- Meister, T.A.; Rimoldi, S.F.; Soria, R.; von Arx, R.; Messerli, F.H.; Sartori, C.; Scherrer, U.; Rexhaj, E. Association of Assisted Reproductive Technologies with Arterial Hypertension During Adolescence. J. Am. Coll. Cardiol. 2018, 72, 1267–1274. [Google Scholar] [CrossRef] [PubMed]
- Von Arx, R.; Allemann, Y.; Sartori, C.; Rexhaj, E.; Cerny, D.; De Marchi, S.F.; Soria, R.; Germond, M.; Scherrer, U.; Rimoldi, S.F. Right ventricular dysfunction in children and adolescents conceived by assisted reproductive technologies. J. Appl. Physiol. 2015, 118, 1200–1206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scherrer, U.; Rimoldi, S.F.; Rexhaj, E.; Stuber, T.; Duplain, H.; Garcin, S.; De Marchi, S.F.; Nicod, P.; Germond, M.; Allemann, Y.; et al. Systemic and pulmonary vascular dysfunction in children conceived by assisted reproductive technologies. Circulation 2012, 125, 1890–1896. [Google Scholar] [CrossRef] [Green Version]
- Juonala, M.; Lewis, S.; McLachlan, R.; Hammarberg, K.; Kennedy, J.; Saffery, R.; McBain, J.; Welsh, L.; Cheung, M.; Doyle, L.W.; et al. American Heart Association ideal cardiovascular health score and subclinical atherosclerosis in 22-35-year-old adults conceived with and without assisted reproductive technologies. Hum. Reprod. 2020, 35, 232–239. [Google Scholar] [CrossRef]
- Hart, R.; Norman, R.J. The longer-term health outcomes for children born as a result of ivf treatment: Part i-general health outcomes. Hum. Reprod. Update 2013, 19, 232–243. [Google Scholar] [CrossRef] [Green Version]
- Guo, X.Y.; Liu, X.M.; Jin, L.; Wang, T.T.; Ullah, K.; Sheng, J.Z.; Huang, H.F. Cardiovascular and metabolic profiles of offspring conceived by assisted reproductive technologies: A systematic review and meta-analysis. Fertil. Steril. 2017, 107, 622–631. [Google Scholar] [CrossRef] [Green Version]
- Vrooman, L.A.; Bartolomei, M.S. Can assisted reproductive technologies cause adult-onset disease?. Evidence from human and mouse. Reprod. Toxicol. 2017, 68, 72–84. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Steptoe, P.C.; Edwards, R.G. Birth after the reimplantation of a human embryo. Lancet 1978, 2, 366. [Google Scholar] [CrossRef]
- Barandalla, M.; Colleoni, S.; Lazzari, G. Differential response of human embryonic stem and somatic cells to non-cytotoxic hydrogen peroxide exposure: An attempt to model in vitro the effects of oxidative stress on the early embryo. Cell Dev. Biol. 2016, 5, 177. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Gene | Protein Encoded | Synonyms | Species 1 | Refs |
|---|---|---|---|---|
| GSH-based antioxidant genes | ||||
| GCLC | Glutamate-cysteine ligase catalytic subunit | GCS, GLCL, GLCLC | m, h | [131,132,133] |
| GCLM | Glutamate-cysteine ligase modifier subunit | GLCLR | m, h | [131,132,133,134] |
| GGT1 | Gamma-glutamyltransferase 1 | CD224, D22S672, D22S732, GGT | h | [131] |
| GLRX | Glutaredoxin | GRX, GRX1 | h | [131] |
| GLS | Glutaminase | GLS1, KIAA0838 | h | [131] |
| GPX1 | Glutathione peroxidase 1 | - | m | [135,136] |
| GPX2 | Glutathione peroxidase 2 | GSHPX-GI | m, h | [131,137,138,139,140] |
| GPX4 | Glutathione peroxidase 4 | MCSP, PHGPx | m | [131] |
| GSR | Glutathione-disulfide reductase | - | m, h | [133,134] |
| GSTA1 | Glutathione S-transferase alpha 1 | - | m | [131,141,142,143] |
| GSTA2 | Glutathione S-transferase alpha 2 | - | m | [132,142,143] |
| GSTA3 | Glutathione S-transferase alpha 3 | - | m | [141,142,143] |
| GSTA4 | Glutathione S-transferase alpha 4 | - | m | [144] |
| GSTM1 | Glutathione S-transferase mu 1 | GST1, H-B, MU | m | [131,132,143,144] |
| GSTM2 | Glutathione S-transferase mu 2 | GST4 | m | [132,143,144] |
| GSTM3 | Glutathione S-transferase mu 3 | GST5 | m, h | [132,134,143,144] |
| GSTM4 | Glutathione S-transferase mu 4 | - | m | [144] |
| GSTM5 | Glutathione S-transferase mu 5 | - | m | [145] |
| GSTM6 | Glutathione S-transferase mu 6 | - | m | [144] |
| GSTP1 | Glutathione S-transferase pi 1 | FAEES3, GST3, GSTP | m | [131] |
| MGST1 | icrosomal glutathione S-transferase 1 | GST12, MGST-I | m, h | [131] |
| MGST2 | Microsomal glutathione S-transferase 2 | MGST-II | m | [143] |
| MGST3 | Microsomal glutathione S-transferase 3 | GST-III | m | [132,143] |
| SLC6A9 | Solute carrier family 6 member 9 | GLYT1 | m | [131] |
| SLC7A11 | Solute carrier family 7 member 11 | xCT | m, h | [131,146] |
| TXN-based antioxidant genes | ||||
| PRDX1 | Peroxiredoxin 1 | NKEFA, PAGA) | m | [131,138,142] |
| PRDX6 | Peroxiredoxin 6 | 1-Cys, aiPLA2, AOP2, KIAA0106, MGC46173, NSGPx, p29, PRX | h | [131,147] |
| SRXN1 | Sulfiredoxin 1 | C20orf139, dJ850E9.2, Npn3, SRX1, YKL086W | m, h | [131] |
| TXN | Thioredoxin | TRX | m, h | [131,132,135] |
| TXNRD1 | Thioredoxin reductase 1 | GRIM-12, Trxr1, TXNR | m, h | [131,135,144] |
| ATP-binding-based antioxidant genes | ||||
| ABCB6 | ATP binding cassette subfamily B member 6 | EST45597, MTABC3, umat | m, h | [131] |
| ABCC1 | ATP binding cassette subfamily C member 1 | GS-X, MRP, MRP1 | m, h | [131] |
| ABCC2 | ATP binding cassette subfamily C member 2 | CMOAT, cMRP, DJS, MRP2 | m, h | [131,141] |
| ABCC3 | ATP binding cassette subfamily C member 3 | cMOAT2, EST90757, MLP2, MOAT-D, MRP3 | m, h | [131,141,148] |
| ABCC4 | ATP binding cassette subfamily C member 4 | EST170205, MOAT-B, MOATB, MRP4 | m | [131] |
| ABCC5 | ATP binding cassette subfamily C member 5 | EST277145, MOAT-C, MRP5, SMRP | m | [131] |
| Heme/iron metabolism-associated antioxidant genes | ||||
| BLVRA | Biliverdin reductase A | BLVR | h | [131] |
| BLVRB | Biliverdin reductase B | FLR, SDR43U1 | m, h | [131] |
| FTH1 | Ferritin heavy chain 1 | FHC, FTH, FTHL6, PIG15, PLIF | m, h | [131] |
| FTL | Ferritin light chain | MGC71996, NBIA3 | m, h | [131] |
| HMOX1 | Heme oxygenase 1 | bK286B10, HO-1 | m, h | [131,133,134,135,140,145] |
| UDP glucuronosyltransferase-associated antioxidant genes | ||||
| UGT1A1 | UDP glucuronosyltransferase family 1 member A1 | GNT1, UGT1, UGT1A | h | [131] |
| UGT1A6 | UDP glucuronosyltransferase family 1 member A6 | GNT1, HLUGP, UGT1F | m | [138] |
| UGT2B1 | UDP glucuronosyltransferase family 2 member B1 | - | m | [141] |
| UGT2B5 | UDP glucuronosyltransferase family 2 member B5 | - | m | [132,143] |
| UGT2B7 | UDP glucuronosyltransferase family 2 member B7 | UGT2B9 | m, h | [131] |
| Other antioxidant genes | ||||
| ADH7 | Alcohol dehydrogenase 7 (class IV), mu or sigma polypeptide | ADH-4 | m | [131] |
| AKR1A1 | Aldo-keto reductase family 1 member A1 | ALR, DD3 | h | [132,143] |
| AKR1B1 | Aldo-keto reductase family 1 member B1 | ALDR1, AR | m, h | [131] |
| AKR1B8 | Aldo-keto reductase family 1 member B8 | - | m | [142,143] |
| AKR1C1 | Aldo-keto reductase family 1 member C1 | DD1, DDH, DDH1, HAKRC, MBAB | h | [131] |
| ALDH1A1 | ldehyde dehydrogenase 1 family member A1 | ALDH1, PUMB1, RALDH1 | m | [131] |
| ALDH3A1 | Aldehyde dehydrogenase 3 family member A1 | ALDH3 | m, h | [131] |
| ALDH7A1 | Aldehyde dehydrogenase 7 family member A1 | ATQ1, EPD, PDE | m | [131] |
| CAT | Catalase | - | m | [137,141] |
| CBR1 | Carbonyl reductase 1 | CBR, SDR21C1 | h | [131] |
| CYP1B1 | Cytochrome P450 family 1 subfamily B member 1 | CP1B, GLC3A | m | [131] |
| CYP2B9 | Cytochrome P450 family 2 subfamily B member 9 | - | m | [131] |
| G6PD | Glucose-6-phosphate dehydrogenase | G6PD1 | m, h | [131] |
| IDH1 | Isocitrate dehydrogenase (NADP(+)) 1, cytosolic | - | m | [131] |
| ME1 | Malic enzyme 1 | - | m, h | [131] |
| NQO1 | NAD(P)H quinone dehydrogenase 1 | DHQU, DIA4, DTD, NMOR1, QR1 | m, h | [131,132,134,135,142,143] |
| PGD | Phosphogluconate dehydrogenase | - | m, h | [131] |
| PTGR1 | Prostaglandin reductase 1 | LTB4DH, ZADH3 | h | [131] |
| SOD1 | Superoxide dismutase 1 | ALS, ALS1, IPOA | m | [141] |
| SOD2 | Superoxide dismutase 2 | - | m | [141] |
| SOD3 | Superoxide dismutase 3 | EC-SOD | m | [142] |
| TALDO1 | Transaldolase 1 | - | m, h | [131] |
| UGDH | UDP-glucose 6-dehydrogenase | - | h | [131] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, H.; Kuhn, C.; Kolben, T.; Ma, Z.; Lin, P.; Mahner, S.; Jeschke, U.; von Schönfeldt, V. Early Life Oxidative Stress and Long-Lasting Cardiovascular Effects on Offspring Conceived by Assisted Reproductive Technologies: A Review. Int. J. Mol. Sci. 2020, 21, 5175. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21155175
Yang H, Kuhn C, Kolben T, Ma Z, Lin P, Mahner S, Jeschke U, von Schönfeldt V. Early Life Oxidative Stress and Long-Lasting Cardiovascular Effects on Offspring Conceived by Assisted Reproductive Technologies: A Review. International Journal of Molecular Sciences. 2020; 21(15):5175. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21155175
Chicago/Turabian StyleYang, Huixia, Christina Kuhn, Thomas Kolben, Zhi Ma, Peng Lin, Sven Mahner, Udo Jeschke, and Viktoria von Schönfeldt. 2020. "Early Life Oxidative Stress and Long-Lasting Cardiovascular Effects on Offspring Conceived by Assisted Reproductive Technologies: A Review" International Journal of Molecular Sciences 21, no. 15: 5175. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21155175





