Therapeutic Potential of Endothelial Colony-Forming Cells in Ischemic Disease: Strategies to Improve their Regenerative Efficacy
Abstract
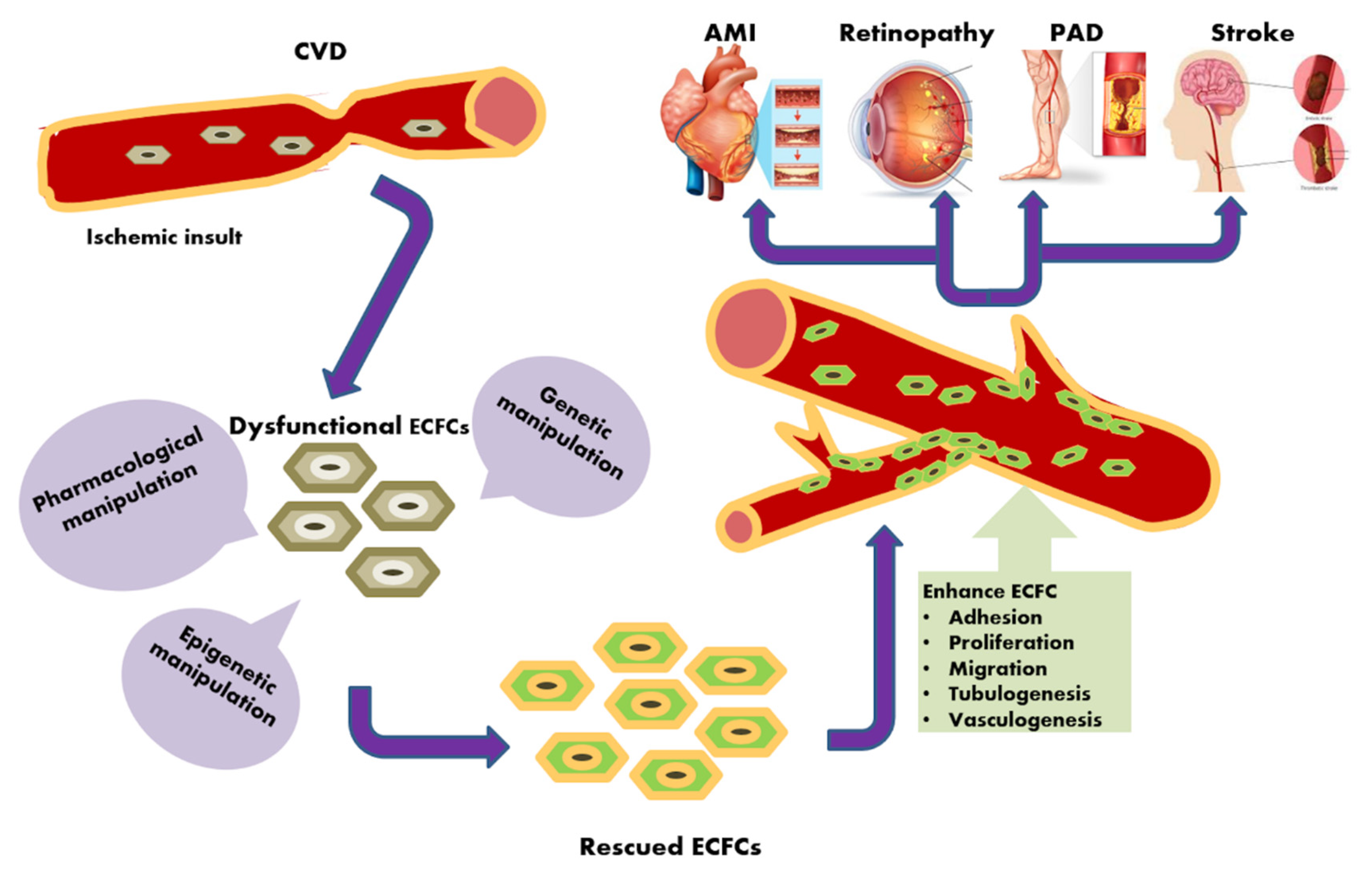
:1. Introduction
2. Endothelial Colony-Forming Cells: Origin, Mechanisms of Actions, and Evidence for Therapeutic Applications
2.1. Endothelial Colony-Forming Cells (ECFCs): Origin, Characterization and Biological Activity
2.2. ECFCs and Ischemic Diseases: An Alternative Strategy to Induce Therapeutic Angiogenesis
2.2.1. Ischemic Brain Disease
2.2.2. Ischemic Retinopathy
2.2.3. Ischemic Cardiovascular Disorders
2.2.4. Peripheral Artery Disease and Critical Limb Ischemia
3. Manipulation of Pro-Angiogenic Signaling Pathways to Improve ECFC Efficiency in Ischemic Diseases
3.1. Boosting ECFC Expansion Ex Vivo with Bioactive Cues
3.2. Priming Dysfunctional ECFCs to Rescue their Angiogenic Activity
3.3. Priming Healthy ECFCs to Enhance their Angiogenic Activity
3.4. Local Injection of Chemoattractants Stimulates ECFC Homing to Sites of Neovessel Formation
3.5. Epigenetic Reprogramming of ECFCs
4. Genetic Manipulation of Pro-Angiogenic Signaling Pathways in ECFCs
5. Priming ECFCs with Mesenchymal Stem Cells
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Benjamin, E.J.; Muntner, P.; Alonso, A.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Das, S.R.; et al. Heart Disease and Stroke Statistics-2019 Update: A Report From the American Heart Association. Circulation 2019, 139, e56–e528. [Google Scholar] [CrossRef] [PubMed]
- Mensah, G.A.; Roth, G.A.; Fuster, V. The Global Burden of Cardiovascular Diseases and Risk Factors. 2020 Beyond 2019, 74, 2529–2532. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.L.; Morrow, D.A. Acute Myocardial Infarction. N. Engl. J. Med. 2017, 376, 2053–2064. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Libby, P.; Theroux, P. Pathophysiology of Coronary Artery Disease. Circulation 2005, 111, 3481–3488. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ylä-Herttuala, S.; Bridges, C.; Katz, M.G.; Korpisalo, P. Angiogenic gene therapy in cardiovascular diseases: Dream or vision? Eur. Heart J. 2017, 38, 1365–1371. [Google Scholar] [CrossRef]
- Hassanshahi, M.; Khabbazi, S.; Peymanfar, Y.; Hassanshahi, A.; Hosseini-Khah, Z.; Su, Y.W.; Xian, C.J. Critical limb ischemia: Current and novel therapeutic strategies. J. Cell. Physiol. 2019, 234, 14445–14459. [Google Scholar] [CrossRef]
- Tompkins, B.A.; Balkan, W.; Winkler, J.; Gyöngyösi, M.; Goliasch, G.; Fernández-Avilés, F.; Hare, J.M. Preclinical Studies of Stem Cell Therapy for Heart Disease. Circ. Res. 2018, 122, 1006–1020. [Google Scholar] [CrossRef]
- Nguyen, P.K.; Rhee, J.W.; Wu, J.C. Adult Stem Cell Therapy and Heart Failure, 2000 to 2016: A Systematic Review. JAMA Cardiol. 2016, 1, 831–841. [Google Scholar] [CrossRef] [Green Version]
- Moccia, F.; Ruffinatti, F.A.; Zuccolo, E. Intracellular Ca2⁺ Signals to Reconstruct A Broken Heart: Still A Theoretical Approach? Curr. Drug Targets 2015, 16, 793–815. [Google Scholar] [CrossRef]
- Lee, P.S.S.; Poh, K.K. Endothelial progenitor cells in cardiovascular diseases. World J. Stem Cells 2014, 6, 355–366. [Google Scholar] [CrossRef]
- Chopra, H.; Hung, M.K.; Kwong, D.L.; Zhang, C.F.; Pow, E.H.N. Insights into Endothelial Progenitor Cells: Origin, Classification, Potentials, and Prospects. Stem Cells Int. 2018, 2018, 9847015. [Google Scholar] [CrossRef] [PubMed]
- Rössig, L.; Urbich, C.; Dimmeler, S. Endothelial Progenitor Cells at Work. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 1977–1979. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murasawa, S.; Asahara, T. Endothelial progenitor cells for vasculogenesis. Physiology 2005, 20, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Asahara, T.; Murohara, T.; Sullivan, A.; Silver, M.; van der Zee, R.; Li, T.; Witzenbichler, B.; Schatteman, G.; Isner, J.M. Isolation of putative progenitor endothelial cells for angiogenesis. Science 1997, 275, 964–967. [Google Scholar] [CrossRef]
- Moccia, F.; Berra-Romani, R.; Rosti, V. Manipulating Intracellular Ca2+ Signals to Stimulate Therapeutic Angiogenesis in Cardiovascular Disorders. Curr. Pharm. Biotechnol. 2018, 19, 686–699. [Google Scholar] [CrossRef]
- Medina, R.J.; Barber, C.L.; Sabatier, F.; Dignat-George, F.; Melero-Martin, J.M.; Khosrotehrani, K.; Ohneda, O.; Randi, A.M.; Chan, J.K.Y.; Yamaguchi, T.; et al. Endothelial Progenitors: A Consensus Statement on Nomenclature. Stem Cells Transl. Med. 2017, 6, 1316–1320. [Google Scholar] [CrossRef]
- Yoder, M.C. Endothelial progenitor cell: A blood cell by many other names may serve similar functions. J. Mol. Med. 2013, 91, 285–295. [Google Scholar] [CrossRef]
- Hirschi, K.K.; Ingram, D.A.; Yoder, M.C. Assessing identity, phenotype, and fate of endothelial progenitor cells. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 1584–1595. [Google Scholar] [CrossRef] [Green Version]
- Critser, P.J.; Yoder, M.C. Endothelial colony-forming cell role in neoangiogenesis and tissue repair. Curr. Opin. Organ Transplant. 2010, 15, 68–72. [Google Scholar] [CrossRef]
- Smadja, D.M.; Melero-Martin, J.M.; Eikenboom, J.; Bowman, M.; Sabatier, F.; Randi, A.M. Standardization of methods to quantify and culture endothelial colony-forming cells derived from peripheral blood: Position paper from the International Society on Thrombosis and Haemostasis SSC. J. Thromb. Haemost. 2019, 17, 1190–1194. [Google Scholar] [CrossRef] [Green Version]
- Reid, E.; Guduric-Fuchs, J.; O’Neill, C.L.; Allen, L.D.; Chambers, S.E.J.; Stitt, A.W.; Medina, R.J. Preclinical Evaluation and Optimization of a Cell Therapy Using Human Cord Blood-Derived Endothelial Colony-Forming Cells for Ischemic Retinopathies. Stem Cells Transl. Med. 2018, 7, 59–67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ingram, D.A.; Mead, L.E.; Tanaka, H.; Meade, V.; Fenoglio, A.; Mortell, K.; Pollok, K.; Ferkowicz, M.J.; Gilley, D.; Yoder, M.C. Identification of a novel hierarchy of endothelial progenitor cells using human peripheral and umbilical cord blood. Blood 2004, 104, 2752–2760. [Google Scholar] [CrossRef] [PubMed]
- Ingram, D.A.; Mead, L.E.; Moore, D.B.; Woodard, W.; Fenoglio, A.; Yoder, M.C. Vessel wall-derived endothelial cells rapidly proliferate because they contain a complete hierarchy of endothelial progenitor cells. Blood 2005, 105, 2783–2786. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Timmermans, F.; Hauwermeiren, F.V.; Smedt, M.D.; Raedt, R.; Plasschaert, F.; Buyzere, M.L.D.; Gillebert, T.C.; Plum, J.; Vandekerckhove, B. Endothelial Outgrowth Cells Are Not Derived From CD133+ Cells or CD45+ Hematopoietic Precursors. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 1572–1579. [Google Scholar] [CrossRef] [Green Version]
- Yoder, M.C.; Mead, L.E.; Prater, D.; Krier, T.R.; Mroueh, K.N.; Li, F.; Krasich, R.; Temm, C.J.; Prchal, J.T.; Ingram, D.A. Redefining endothelial progenitor cells via clonal analysis and hematopoietic stem/progenitor cell principals. Blood 2007, 109, 1801–1809. [Google Scholar] [CrossRef] [Green Version]
- Rossi, E.; Poirault-Chassac, S.; Bieche, I.; Chocron, R.; Schnitzler, A.; Lokajczyk, A.; Bourdoncle, P.; Dizier, B.; Bacha, N.; Gendron, N.; et al. Human Endothelial Colony Forming Cells Express Intracellular CD133 that Modulates their Vasculogenic Properties. Stem Cell Rev. Rep. 2019, 15, s12015–s12019. [Google Scholar] [CrossRef]
- Melero-Martin, J.M.; Khan, Z.A.; Picard, A.; Wu, X.; Paruchuri, S.; Bischoff, J. In vivo vasculogenic potential of human blood-derived endothelial progenitor cells. Blood 2007, 109, 4761–4768. [Google Scholar] [CrossRef] [Green Version]
- Au, P.; Daheron, L.M.; Duda, D.G.; Cohen, K.S.; Tyrrell, J.A.; Lanning, R.M.; Fukumura, D.; Scadden, D.T.; Jain, R.K. Differential in vivo potential of endothelial progenitor cells from human umbilical cord blood and adult peripheral blood to form functional long-lasting vessels. Blood 2008, 111, 1302–1305. [Google Scholar] [CrossRef] [Green Version]
- Bompais, H.; Chagraoui, J.; Canron, X.; Crisan, M.; Liu, X.H.; Anjo, A.; Tolla-Le Port, C.; Leboeuf, M.; Charbord, P.; Bikfalvi, A.; et al. Human endothelial cells derived from circulating progenitors display specific functional properties compared with mature vessel wall endothelial cells. Blood 2004, 103, 2577–2584. [Google Scholar] [CrossRef] [Green Version]
- Putman, D.M.; Liu, K.Y.; Broughton, H.C.; Bell, G.I.; Hess, D.A. Umbilical cord blood-derived aldehyde dehydrogenase-expressing progenitor cells promote recovery from acute ischemic injury. Stem Cells 2012, 30, 2248–2260. [Google Scholar] [CrossRef]
- Drake, K.M.; Federici, C.; Duong, H.T.; Comhair, S.A.; Erzurum, S.C.; Asosingh, K.; Aldred, M.A. Genomic stability of pulmonary artery endothelial colony-forming cells in culture. Pulm. Circ. 2017, 7, 421–427. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reinisch, A.; Hofmann, N.A.; Obenauf, A.C.; Kashofer, K.; Rohde, E.; Schallmoser, K.; Flicker, K.; Lanzer, G.; Linkesch, W.; Speicher, M.R.; et al. Humanized large-scale expanded endothelial colony-forming cells function in vitro and in vivo. Blood 2009, 113, 6716–6725. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Banno, K.; Yoder, M.C. Endothelial Stem and Progenitor Cells for Regenerative Medicine. Curr. Stem Cell Rep. 2019, 5, 101–108. [Google Scholar] [CrossRef]
- Lin, Y.; Weisdorf, D.J.; Solovey, A.; Hebbel, R.P. Origins of circulating endothelial cells and endothelial outgrowth from blood. J. Clin. Investig. 2000, 105, 71–77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fujisawa, T.; Tura-Ceide, O.; Hunter, A.; Mitchell, A.; Vesey, A.; Medine, C.; Gallogly, S.; Hadoke, P.W.F.; Keith, C.; Sproul, A.; et al. Endothelial Progenitor Cells Do Not Originate From the Bone Marrow. Circulation 2019, 140, 1524–1526. [Google Scholar] [CrossRef]
- Tura, O.; Skinner, E.M.; Barclay, G.R.; Samuel, K.; Gallagher, R.C.J.; Brittan, M.; Hadoke, P.W.F.; Newby, D.E.; Turner, M.L.; Mills, N.L. Late Outgrowth Endothelial Cells Resemble Mature Endothelial Cells and Are Not Derived from Bone Marrow. Stem Cells 2013, 31, 338–348. [Google Scholar] [CrossRef]
- Alphonse, R.S.; Vadivel, A.; Zhong, S.; McConaghy, S.; Ohls, R.; Yoder, M.C.; Thebaud, B. The isolation and culture of endothelial colony-forming cells from human and rat lungs. Nat. Protoc. 2015, 10, 1697–1708. [Google Scholar] [CrossRef] [Green Version]
- Duong, H.T.; Comhair, S.A.; Aldred, M.A.; Mavrakis, L.; Savasky, B.M.; Erzurum, S.C.; Asosingh, K. Pulmonary artery endothelium resident endothelial colony-forming cells in pulmonary arterial hypertension. Pulm. Circ. 2011, 1, 475–486. [Google Scholar] [CrossRef] [Green Version]
- Green, L.; Ofstein, R.H.; Rapp, B.; Saadatzadeh, M.R.; Bhavsar, J.R.; Fajardo, A.; Dalsing, M.C.; Ingram, D.A.; Murphy, M.P. Adult venous endothelium is a niche for highly proliferative and vasculogenic endothelial colony-forming cells. J. Vasc. Surg. 2017, 66, 1854–1863. [Google Scholar] [CrossRef] [Green Version]
- Solomon, I.; O’Reilly, M.; Ionescu, L.; Alphonse, R.S.; Rajabali, S.; Zhong, S.; Vadivel, A.; Shelley, W.C.; Yoder, M.C.; Thebaud, B. Functional Differences Between Placental Micro- and Macrovascular Endothelial Colony-Forming Cells. Stem Cells Transl. Med. 2016, 5, 291–300. [Google Scholar] [CrossRef] [Green Version]
- Lin, R.Z.; Moreno-Luna, R.; Munoz-Hernandez, R.; Li, D.; Jaminet, S.C.; Greene, A.K.; Melero-Martin, J.M. Human white adipose tissue vasculature contains endothelial colony-forming cells with robust in vivo vasculogenic potential. Angiogenesis 2013, 16, 735–744. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prasain, N.; Lee, M.R.; Vemula, S.; Meador, J.L.; Yoshimoto, M.; Ferkowicz, M.J.; Fett, A.; Gupta, M.; Rapp, B.M.; Saadatzadeh, M.R.; et al. Differentiation of human pluripotent stem cells to cells similar to cord-blood endothelial colony-forming cells. Nat. Biotechnol. 2014, 32, 1151–1157. [Google Scholar] [CrossRef] [PubMed]
- Kutikhin, A.G.; Tupikin, A.E.; Matveeva, V.G.; Shishkova, D.K.; Antonova, L.V.; Kabilov, M.R.; Velikanova, E.A. Human Peripheral Blood-Derived Endothelial Colony-Forming Cells Are Highly Similar to Mature Vascular Endothelial Cells yet Demonstrate a Transitional Transcriptomic Signature. Cells 2020, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Toshner, M.; Dunmore, B.J.; McKinney, E.F.; Southwood, M.; Caruso, P.; Upton, P.D.; Waters, J.P.; Ormiston, M.L.; Skepper, J.N.; Nash, G.; et al. Transcript Analysis Reveals a Specific HOX Signature Associated with Positional Identity of Human Endothelial Cells. PLoS ONE 2014, 9, e91334. [Google Scholar] [CrossRef] [Green Version]
- McDonald, A.I.; Shirali, A.S.; Aragón, R.; Ma, F.; Hernandez, G.; Vaughn, D.A.; Mack, J.J.; Lim, T.Y.; Sunshine, H.; Zhao, P.; et al. Endothelial Regeneration of Large Vessels Is a Biphasic Process Driven by Local Cells with Distinct Proliferative Capacities. Cell Stem Cell 2018, 23, 210–225. [Google Scholar] [CrossRef] [Green Version]
- Malinverno, M.; Corada, M.; Ferrarini, L.; Formicola, L.; Marazzi, G.; Sassoon, D.; Dejana, E. Peg3/PW1 Is a Marker of a Subset of Vessel Associated Endothelial Progenitors. Stem Cells 2017, 35, 1328–1340. [Google Scholar] [CrossRef] [Green Version]
- Yu, Q.C.; Song, W.; Wang, D.; Zeng, Y.A. Identification of blood vascular endothelial stem cells by the expression of protein C receptor. Cell Res. 2016, 26, 1079–1098. [Google Scholar] [CrossRef] [Green Version]
- Naito, H.; Kidoya, H.; Sakimoto, S.; Wakabayashi, T.; Takakura, N. Identification and characterization of a resident vascular stem/progenitor cell population in preexisting blood vessels. EMBO J. 2012, 31, 842–855. [Google Scholar] [CrossRef] [Green Version]
- Wolmarans, E.; Nel, S.; Durandt, C.; Mellet, J.; Pepper, M.S. Side Population: Its Use in the Study of Cellular Heterogeneity and as a Potential Enrichment Tool for Rare Cell Populations. Stem Cells Int. 2018, 2018, 2472137. [Google Scholar] [CrossRef] [Green Version]
- Paschalaki, K.E.; Randi, A.M. Recent Advances in Endothelial Colony Forming Cells Toward Their Use in Clinical Translation. Front. Med. 2018, 5. [Google Scholar] [CrossRef] [Green Version]
- Smadja, D.M.; Bièche, I.; Susen, S.; Mauge, L.; Laurendeau, I.; d’Audigier, C.; Grelac, F.; Emmerich, J.; Aiach, M.; Gaussem, P. Interleukin 8 is differently expressed and modulated by PAR-1 activation in early and late endothelial progenitor cells. J. Cell. Mol. Med. 2009, 13, 2534–2546. [Google Scholar] [CrossRef] [PubMed]
- Blandinières, A.; Gendron, N.; Bacha, N.; Bièche, I.; Chocron, R.; Nunes, H.; Nevo, N.; Rossi, E.; Crestani, B.; Lecourt, S.; et al. Interleukin-8 release by endothelial colony-forming cells isolated from idiopathic pulmonary fibrosis patients might contribute to their pathogenicity. Angiogenesis 2019, 22, 325–339. [Google Scholar] [CrossRef] [PubMed]
- d’Audigier, C.; Cochain, C.; Rossi, E.; Guérin, C.L.; Bièche, I.; Blandinières, A.; Marsac, B.; Silvestre, J.S.; Gaussem, P.; Smadja, D.M. Thrombin receptor PAR-1 activation on endothelial progenitor cells enhances chemotaxis-associated genes expression and leukocyte recruitment by a COX-2-dependent mechanism. Angiogenesis 2015, 18, 347–359. [Google Scholar] [CrossRef] [PubMed]
- Lin, R.Z.; Moreno-Luna, R.; Li, D.; Jaminet, S.C.; Greene, A.K.; Melero-Martin, J.M. Human endothelial colony-forming cells serve as trophic mediators for mesenchymal stem cell engraftment via paracrine signaling. Proc. Natl. Acad. Sci. USA 2014, 111, 10137–10142. [Google Scholar] [CrossRef] [Green Version]
- Lin, R.Z.; Moreno-Luna, R.; Zhou, B.; Pu, W.T.; Melero-Martin, J.M. Equal modulation of endothelial cell function by four distinct tissue-specific mesenchymal stem cells. Angiogenesis 2012, 15, 443–455. [Google Scholar] [CrossRef] [Green Version]
- Boscolo, E.; Stewart, C.L.; Greenberger, S.; Wu, J.K.; Durham, J.T.; Herman, I.M.; Mulliken, J.B.; Kitajewski, J.; Bischoff, J. JAGGED1 signaling regulates hemangioma stem cell-to-pericyte/vascular smooth muscle cell differentiation. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 2181–2192. [Google Scholar] [CrossRef] [Green Version]
- Kang, K.T.; Lin, R.Z.; Kuppermann, D.; Melero-Martin, J.M.; Bischoff, J. Endothelial colony forming cells and mesenchymal progenitor cells form blood vessels and increase blood flow in ischemic muscle. Sci. Rep. 2017, 7, 770. [Google Scholar] [CrossRef]
- Traktuev, D.O.; Prater, D.N.; Merfeld-Clauss, S.; Sanjeevaiah, A.R.; Saadatzadeh, M.R.; Murphy, M.; Johnstone, B.H.; Ingram, D.A.; March, K.L. Robust functional vascular network formation in vivo by cooperation of adipose progenitor and endothelial cells. Circ. Res. 2009, 104, 1410–1420. [Google Scholar] [CrossRef]
- Banno, K.; Yoder, M.C. Tissue regeneration using endothelial colony-forming cells: Promising cells for vascular repair. Pediatric Res. 2018, 83, 283–290. [Google Scholar] [CrossRef] [Green Version]
- Basile, D.P.; Yoder, M.C. Circulating and Tissue Resident Endothelial Progenitor Cells. J. Cell. Physiol. 2014, 229, 10–16. [Google Scholar] [CrossRef] [Green Version]
- Ding, J.; Zhao, Z.; Wang, C.; Wang, C.X.; Li, P.C.; Qian, C.; Teng, G.J. Bioluminescence imaging of transplanted human endothelial colony-forming cells in an ischemic mouse model. Brain Res. 2016, 1642, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Bayraktutan, U. Endothelial progenitor cells: Potential novel therapeutics for ischaemic stroke. Pharmacol. Res. 2019, 144, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Kang, K.T.; Coggins, M.; Xiao, C.; Rosenzweig, A.; Bischoff, J. Human vasculogenic cells form functional blood vessels and mitigate adverse remodeling after ischemia reperfusion injury in rats. Angiogenesis 2013, 16, 773–784. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Minami, Y.; Nakajima, T.; Ikutomi, M.; Morita, T.; Komuro, I.; Sata, M.; Sahara, M. Angiogenic potential of early and late outgrowth endothelial progenitor cells is dependent on the time of emergence. Int. J. Cardiol. 2015, 186, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Alphonse, R.S.; Vadivel, A.; Fung, M.; Shelley, W.C.; Critser, P.J.; Ionescu, L.; O’Reilly, M.; Ohls, R.K.; McConaghy, S.; Eaton, F.; et al. Existence, functional impairment, and lung repair potential of endothelial colony-forming cells in oxygen-induced arrested alveolar growth. Circulation 2014, 129, 2144–2157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Viñas, J.L.; Burger, D.; Zimpelmann, J.; Haneef, R.; Knoll, W.; Campbell, P.; Gutsol, A.; Carter, A.; Allan, D.S.; Burns, K.D. Transfer of microRNA-486-5p from human endothelial colony forming cell-derived exosomes reduces ischemic kidney injury. Kidney Int. 2016, 90, 1238–1250. [Google Scholar] [CrossRef] [Green Version]
- Burger, D.; Vinas, J.L.; Akbari, S.; Dehak, H.; Knoll, W.; Gutsol, A.; Carter, A.; Touyz, R.M.; Allan, D.S.; Burns, K.D. Human endothelial colony-forming cells protect against acute kidney injury: Role of exosomes. Am. J. Pathol. 2015, 185, 2309–2323. [Google Scholar] [CrossRef]
- Guerra, G.; Lucariello, A.; Perna, A.; Botta, L.; De Luca, A.; Moccia, F. The Role of Endothelial Ca(2+) Signaling in Neurovascular Coupling: A View from the Lumen. Int. J. Mol. Sci. 2018, 19. [Google Scholar] [CrossRef] [Green Version]
- Wardlaw, J.M.; Sandercock, P.A.G.; Murray, V. Should more patients with acute ischaemic stroke receive thrombolytic treatment? BMJ 2009, 339, b4584. [Google Scholar] [CrossRef]
- O’Neill, C.L.; McLoughlin, K.J.; Chambers, S.E.J.; Guduric-Fuchs, J.; Stitt, A.W.; Medina, R.J. The Vasoreparative Potential of Endothelial Colony Forming Cells: A Journey Through Pre-clinical Studies. Front. Med. 2018, 5. [Google Scholar] [CrossRef] [Green Version]
- Moubarik, C.; Guillet, B.; Youssef, B.; Codaccioni, J.L.; Piercecchi, M.D.; Sabatier, F.; Lionel, P.; Dou, L.; Foucault-Bertaud, A.; Velly, L.; et al. Transplanted late outgrowth endothelial progenitor cells as cell therapy product for stroke. Stem Cell Rev. Rep. 2011, 7, 208–220. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, Y.; Wang, S.; Han, Z.; Huang, X.; Li, S.; Chen, F.; Niu, R.; Dong, J.F.; Jiang, R.; et al. Transplantation of expanded endothelial colony-forming cells improved outcomes of traumatic brain injury in a mouse model. J. Surg. Res. 2013, 185, 441–449. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.T.; Zhang, Y.Q.; Li, S.J.; Li, S.H.; Tang, Q.; Wang, Z.T.; Dong, J.F.; Zhang, J.N. Intracerebroventricular transplantation of ex vivo expanded endothelial colony-forming cells restores blood-brain barrier integrity and promotes angiogenesis of mice with traumatic brain injury. J. Neurotrauma 2013, 30, 2080–2088. [Google Scholar] [CrossRef] [Green Version]
- Bertelli, P.M.; Pedrini, E.; Guduric-Fuchs, J.; Peixoto, E.; Pathak, V.; Stitt, A.W.; Medina, R.J. Vascular Regeneration for Ischemic Retinopathies: Hope from Cell Therapies. Curr. Eye Res. 2020, 45, 372–384. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, R.; Caspi, R.R. Ocular immune privilege. F1000 Biol. Rep. 2010, 2. [Google Scholar] [CrossRef] [PubMed]
- Medina, R.J.; O’Neill, C.L.; Humphreys, M.W.; Gardiner, T.A.; Stitt, A.W. Outgrowth endothelial cells: Characterization and their potential for reversing ischemic retinopathy. Investig. Ophthalmol. Vis. Sci. 2010, 51, 5906–5913. [Google Scholar] [CrossRef] [Green Version]
- Cahoon, J.M.; Rai, R.R.; Carroll, L.S.; Uehara, H.; Zhang, X.; O’Neil, C.L.; Medina, R.J.; Das, S.K.; Muddana, S.K.; Olson, P.R.; et al. Intravitreal AAV2.COMP-Ang1 Prevents Neurovascular Degeneration in a Murine Model of Diabetic Retinopathy. Diabetes 2015, 64, 4247–4259. [Google Scholar] [CrossRef] [Green Version]
- O’Leary, O.E.; Canning, P.; Reid, E.; Bertelli, P.M.; McKeown, S.; Brines, M.; Cerami, A.; Du, X.; Xu, H.; Chen, M.; et al. The vasoreparative potential of endothelial colony-forming cells in the ischemic retina is enhanced by cibinetide, a non-hematopoietic erythropoietin mimetic. Exp. Eye Res. 2019, 182, 144–155. [Google Scholar] [CrossRef]
- Sakimoto, S.; Marchetti, V.; Aguilar, E.; Lee, K.; Usui, Y.; Murinello, S.; Bucher, F.; Trombley, J.K.; Fallon, R.; Wagey, R.; et al. CD44 expression in endothelial colony-forming cells regulates neurovascular trophic effect. JCI Insight 2017, 2, e89906. [Google Scholar] [CrossRef] [Green Version]
- Li Calzi, S.; Shaw, L.C.; Moldovan, L.; Shelley, W.C.; Qi, X.; Racette, L.; Quigley, J.L.; Fortmann, S.D.; Boulton, M.E.; Yoder, M.C.; et al. Progenitor cell combination normalizes retinal vascular development in the oxygen-induced retinopathy (OIR) model. JCI Insight 2019, 4. [Google Scholar] [CrossRef] [Green Version]
- Dellett, M.; Brown, E.D.; Guduric-Fuchs, J.; O’Connor, A.; Stitt, A.W.; Medina, R.J.; Simpson, D.A. MicroRNA-containing extracellular vesicles released from endothelial colony-forming cells modulate angiogenesis during ischaemic retinopathy. J. Cell. Mol. Med. 2017, 21, 3405–3419. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitsos, S.; Katsanos, K.; Koletsis, E.; Kagadis, G.C.; Anastasiou, N.; Diamantopoulos, A.; Karnabatidis, D.; Dougenis, D. Therapeutic angiogenesis for myocardial ischemia revisited: Basic biological concepts and focus on latest clinical trials. Angiogenesis 2012, 15, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Prasad, M.; Corban, M.T.; Henry, T.D.; Dietz, A.B.; Lerman, L.O.; Lerman, A. Promise of autologous CD34+ stem/progenitor cell therapy for treatment of cardiovascular disease. Cardiovasc. Res. 2020, 116, 1424–1433. [Google Scholar] [CrossRef] [PubMed]
- Kawamoto, A.; Gwon, H.C.; Iwaguro, H.; Yamaguchi, J.I.; Uchida, S.; Masuda, H.; Silver, M.; Ma, H.; Kearney, M.; Isner, J.M.; et al. Therapeutic potential of ex vivo expanded endothelial progenitor cells for myocardial ischemia. Circulation 2001, 103, 634–637. [Google Scholar] [CrossRef] [PubMed]
- Kocher, A.A.; Schuster, M.D.; Szabolcs, M.J.; Takuma, S.; Burkhoff, D.; Wang, J.; Homma, S.; Edwards, N.M.; Itescu, S. Neovascularization of ischemic myocardium by human bone-marrow-derived angioblasts prevents cardiomyocyte apoptosis, reduces remodeling and improves cardiac function. Nat. Med. 2001, 7, 430–436. [Google Scholar] [CrossRef]
- Sung, P.H.; Lee, F.Y.; Tong, M.S.; Chiang, J.Y.; Pei, S.N.; Ma, M.C.; Li, Y.C.; Chen, Y.L.; Wu, C.J.; Sheu, J.J.; et al. The Five-Year Clinical and Angiographic Follow-Up Outcomes of Intracoronary Transfusion of Circulation-Derived CD34+ Cells for Patients With End-Stage Diffuse Coronary Artery Disease Unsuitable for Coronary Intervention-Phase I Clinical Trial. Crit. Care Med. 2018, 46, e411–e418. [Google Scholar] [CrossRef]
- Lee, F.Y.; Chen, Y.L.; Sung, P.H.; Ma, M.C.; Pei, S.N.; Wu, C.J.; Yang, C.H.; Fu, M.; Ko, S.F.; Leu, S.; et al. Intracoronary Transfusion of Circulation-Derived CD34+ Cells Improves Left Ventricular Function in Patients With End-Stage Diffuse Coronary Artery Disease Unsuitable for Coronary Intervention. Crit. Care Med. 2015, 43, 2117–2132. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, S.; Rabinovich, B.; Bidaut, L.; Soghomonyan, S.; Alauddin, M.M.; Bankson, J.A.; Shpall, E.; Willerson, J.T.; Gelovani, J.G.; et al. Human CD34+ cells in experimental myocardial infarction: Long-term survival, sustained functional improvement, and mechanism of action. Circ. Res. 2010, 106, 1904–1911. [Google Scholar] [CrossRef] [Green Version]
- Losordo, D.W.; Henry, T.D.; Davidson, C.; Sup Lee, J.; Costa, M.A.; Bass, T.; Mendelsohn, F.; Fortuin, F.D.; Pepine, C.J.; Traverse, J.H.; et al. Intramyocardial, autologous CD34+ cell therapy for refractory angina. Circ. Res. 2011, 109, 428–436. [Google Scholar] [CrossRef] [Green Version]
- Henry, T.D.; Schaer, G.L.; Traverse, J.H.; Povsic, T.J.; Davidson, C.; Lee, J.S.; Costa, M.A.; Bass, T.; Mendelsohn, F.; Fortuin, F.D.; et al. Autologous CD34(+) Cell Therapy for Refractory Angina: 2-Year Outcomes From the ACT34-CMI Study. Cell Transplant. 2016, 25, 1701–1711. [Google Scholar] [CrossRef] [Green Version]
- Psaltis, P.J.; Schwarz, N.; Toledo-Flores, D.; Nicholls, S.J. Cellular Therapy for Heart Failure. Curr. Cardiol. Rev. 2016, 12, 195–215. [Google Scholar] [CrossRef]
- Bervar, M.; Kozelj, M.; Poglajen, G.; Sever, M.; Zemljic, G.; Frljak, S.; Cukjati, M.; Cernelc, P.; Haddad, F.; Vrtovec, B. Effects of Transendocardial CD34(+) Cell Transplantation on Diastolic Parameters in Patients with Nonischemic Dilated Cardiomyopathy. Stem Cells Transl. Med. 2017, 6, 1515–1521. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, T.M.; Leicht, S.F.; Radic, T.; Rodriguez-Arabaolaza, I.; Hermann, P.C.; Berger, F.; Saif, J.; Böcker, W.; Ellwart, J.W.; Aicher, A.; et al. Vascular incorporation of endothelial colony-forming cells is essential for functional recovery of murine ischemic tissue following cell therapy. Arterioscler. Thromb. Vasc. Biol. 2012, 32, e13–e21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dubois, C.; Liu, X.; Claus, P.; Marsboom, G.; Pokreisz, P.; Vandenwijngaert, S.; Depelteau, H.; Streb, W.; Chaothawee, L.; Maes, F.; et al. Differential effects of progenitor cell populations on left ventricular remodeling and myocardial neovascularization after myocardial infarction. J. Am. Coll. Cardiol. 2010, 55, 2232–2243. [Google Scholar] [CrossRef] [PubMed]
- Deutsch, M.A.; Brunner, S.; Grabmaier, U.; David, R.; Ott, I.; Huber, B.C. Cardioprotective Potential of Human Endothelial-Colony Forming Cells from Diabetic and Nondiabetic Donors. Cells 2020, 9. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.W.; Jin, H.L.; Kang, S.M.; Kim, S.; Yoo, K.J.; Jang, Y.; Kim, H.O.; Yoon, Y.S. Therapeutic effects of late outgrowth endothelial progenitor cells or mesenchymal stem cells derived from human umbilical cord blood on infarct repair. Int. J. Cardiol. 2016, 203, 498–507. [Google Scholar] [CrossRef] [Green Version]
- Massa, M.; Campanelli, R.; Bonetti, E.; Ferrario, M.; Marinoni, B.; Rosti, V. Rapid and large increase of the frequency of circulating endothelial colony-forming cells (ECFCs) generating late outgrowth endothelial cells in patients with acute myocardial infarction. Exp. Hematol. 2009, 37, 8–9. [Google Scholar] [CrossRef]
- Meneveau, N.; Deschaseaux, F.; Seronde, M.F.; Chopard, R.; Schiele, F.; Jehl, J.; Tiberghien, P.; Bassand, J.P.; Kantelip, J.P.; Davani, S. Presence of endothelial colony-forming cells is associated with reduced microvascular obstruction limiting infarct size and left ventricular remodelling in patients with acute myocardial infarction. Basic Res. Cardiol. 2011, 106, 1397–1410. [Google Scholar] [CrossRef]
- Shahrivari, M.; Wise, E.; Resende, M.; Shuster, J.J.; Zhang, J.; Bolli, R.; Cooke, J.P.; Hare, J.M.; Henry, T.D.; Khan, A.; et al. Peripheral Blood Cytokine Levels After Acute Myocardial Infarction: IL-1β- and IL-6-Related Impairment of Bone Marrow Function. Circ. Res. 2017, 120, 1947–1957. [Google Scholar] [CrossRef]
- Qadura, M.; Terenzi, D.C.; Verma, S.; Al-Omran, M.; Hess, D.A. Concise Review: Cell Therapy for Critical Limb Ischemia: An Integrated Review of Preclinical and Clinical Studies. Stem Cells 2018, 36, 161–171. [Google Scholar] [CrossRef] [Green Version]
- Inampudi, C.; Akintoye, E.; Ando, T.; Briasoulis, A. Angiogenesis in peripheral arterial disease. Curr. Opin. Pharmacol. 2018, 39, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Morley, R.L.; Sharma, A.; Horsch, A.D.; Hinchliffe, R.J. Peripheral artery disease. BMJ 2018, 360, j5842. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Odent Grigorescu, G.; Preda, M.B.; Radu, E.; Rosca, A.M.; Tutuianu, R.; Mitroi, D.N.; Simionescu, M.; Burlacu, A. Combinatorial approach for improving the outcome of angiogenic therapy in ischemic tissues. Biomaterials 2015, 60, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Rossi, E.; Smadja, D.M.; Boscolo, E.; Langa, C.; Arevalo, M.A.; Pericacho, M.; Gamella-Pozuelo, L.; Kauskot, A.; Botella, L.M.; Gaussem, P.; et al. Endoglin regulates mural cell adhesion in the circulatory system. Cell. Mol. Life Sci. 2016, 73, 1715–1739. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rossi, E.; Smadja, D.; Goyard, C.; Cras, A.; Dizier, B.; Bacha, N.; Lokajczyk, A.; Guerin, C.L.; Gendron, N.; Planquette, B.; et al. Co-injection of mesenchymal stem cells with endothelial progenitor cells accelerates muscle recovery in hind limb ischemia through an endoglin-dependent mechanism. Thromb. Haemost. 2017, 117, 1908–1918. [Google Scholar] [CrossRef]
- Garrigue, P.; Hache, G.; Bennis, Y.; Brige, P.; Stalin, J.; Pellegrini, L.; Velly, L.; Orlandi, F.; Castaldi, E.; Dignat-George, F.; et al. Erythropoietin Pretreatment of Transplanted Endothelial Colony-Forming Cells Enhances Recovery in a Cerebral Ischemia Model by Increasing Their Homing Ability: A SPECT/CT Study. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2016, 57, 1798–1804. [Google Scholar] [CrossRef] [Green Version]
- Abdulkadir, R.; Alwjwaj, M.; Othman, O.; Rakkar, K.; Bayraktutan, U. Outgrowth endothelial cells form a functional cerebral barrier and restore its integrity after damage. Neural Regen. Res. 2020, 15, 1071–1078. [Google Scholar] [CrossRef]
- Gao, W.; Li, F.; Liu, L.; Xu, X.; Zhang, B.; Wu, Y.; Yin, D.; Zhou, S.; Sun, D.; Huang, Y.; et al. Endothelial colony-forming cell-derived exosomes restore blood-brain barrier continuity in mice subjected to traumatic brain injury. Exp. Neurol. 2018, 307, 99–108. [Google Scholar] [CrossRef]
- Mena, H.A.; Zubiry, P.R.; Dizier, B.; Mignon, V.; Parborell, F.; Schattner, M.; Boisson-Vidal, C.; Negrotto, S. Ceramide 1-Phosphate Protects Endothelial Colony-Forming Cells from Apoptosis and Increases Vasculogenesis In Vitro and In Vivo. Arterioscler. Thromb. Vasc. Biol. 2019, 39, e219–e232. [Google Scholar] [CrossRef]
- Tasev, D.; Koolwijk, P.; van Hinsbergh, V.W. Therapeutic Potential of Human-Derived Endothelial Colony-Forming Cells in Animal Models. Tissue Eng. Part B Rev. 2016, 22, 371–382. [Google Scholar] [CrossRef]
- Keighron, C.; Lyons, C.J.; Creane, M.; O’Brien, T.; Liew, A. Recent Advances in Endothelial Progenitor Cells Toward Their Use in Clinical Translation. Front. Med. 2018, 5, 354. [Google Scholar] [CrossRef] [Green Version]
- Moccia, F.; Fotia, V.; Tancredi, R.; Della Porta, M.G.; Rosti, V.; Bonetti, E.; Poletto, V.; Marchini, S.; Beltrame, L.; Gallizzi, G.; et al. Breast and renal cancer-Derived endothelial colony forming cells share a common gene signature. Eur. J. Cancer 2017, 77, 155–164. [Google Scholar] [CrossRef]
- Campanelli, R.; Codazzi, A.C.; Poletto, V.; Abba, C.; Catarsi, P.; Fois, G.; Avanzini, M.A.; Brazzelli, V.; Tzialla, C.; De Silvestri, A.; et al. Kinetic and Angiogenic Activity of Circulating Endothelial Colony Forming Cells in Patients with Infantile Haemangioma Receiving Propranolol. Thromb. Haemost. 2019, 119, 274–284. [Google Scholar] [CrossRef]
- Moccia, F.; Lodola, F.; Dragoni, S.; Bonetti, E.; Bottino, C.; Guerra, G.; Laforenza, U.; Rosti, V.; Tanzi, F. Ca2+ signalling in endothelial progenitor cells: A novel means to improve cell-based therapy and impair tumour vascularisation. Curr. Vasc. Pharmacol. 2014, 12, 87–105. [Google Scholar] [CrossRef]
- Smadja, D.M. Vasculogenic Stem and Progenitor Cells in Human: Future Cell Therapy Product or Liquid Biopsy for Vascular Disease. Adv. Exp. Med. Biol. 2019, 1201, 215–237. [Google Scholar] [CrossRef]
- Guven, H.; Shepherd, R.M.; Bach, R.G.; Capoccia, B.J.; Link, D.C. The number of endothelial progenitor cell colonies in the blood is increased in patients with angiographically significant coronary artery disease. J. Am. Coll. Cardiol. 2006, 48, 1579–1587. [Google Scholar] [CrossRef] [Green Version]
- Smadja, D.M.; Duong-van-Huyen, J.P.; Dal Cortivo, L.; Blanchard, A.; Bruneval, P.; Emmerich, J.; Gaussem, P. Early endothelial progenitor cells in bone marrow are a biomarker of cell therapy success in patients with critical limb ischemia. Cytotherapy 2012, 14, 232–239. [Google Scholar] [CrossRef]
- Mauge, L.; Sabatier, F.; Boutouyrie, P.; D’Audigier, C.; Peyrard, S.; Bozec, E.; Blanchard, A.; Azizi, M.; Dizier, B.; Dignat-George, F.; et al. Forearm ischemia decreases endothelial colony-forming cell angiogenic potential. Cytotherapy 2014, 16, 213–224. [Google Scholar] [CrossRef] [Green Version]
- Toshner, M.; Voswinckel, R.; Southwood, M.; Al-Lamki, R.; Howard, L.S.; Marchesan, D.; Yang, J.; Suntharalingam, J.; Soon, E.; Exley, A.; et al. Evidence of dysfunction of endothelial progenitors in pulmonary arterial hypertension. Am. J. Respir. Crit. Care Med. 2009, 180, 780–787. [Google Scholar] [CrossRef] [Green Version]
- Su, S.H.; Wu, C.H.; Chiu, Y.L.; Chang, S.J.; Lo, H.H.; Liao, K.H.; Tsai, C.F.; Tsai, T.N.; Lin, C.H.; Cheng, S.M.; et al. Dysregulation of Vascular Endothelial Growth Factor Receptor-2 by Multiple miRNAs in Endothelial Colony-Forming Cells of Coronary Artery Disease. J. Vasc. Res. 2017, 54, 22–32. [Google Scholar] [CrossRef]
- Shelley, W.C.; Leapley, A.C.; Huang, L.; Critser, P.J.; Zeng, P.; Prater, D.; Ingram, D.A.; Tarantal, A.F.; Yoder, M.C. Changes in the frequency and in vivo vessel-forming ability of rhesus monkey circulating endothelial colony-forming cells across the lifespan (birth to aged). Pediatr. Res. 2012, 71, 156–161. [Google Scholar] [CrossRef] [Green Version]
- Jarajapu, Y.P.; Hazra, S.; Segal, M.; Li Calzi, S.; Jadhao, C.; Qian, K.; Mitter, S.K.; Raizada, M.K.; Boulton, M.E.; Grant, M.B. Vasoreparative dysfunction of CD34+ cells in diabetic individuals involves hypoxic desensitization and impaired autocrine/paracrine mechanisms. PLoS ONE 2014, 9, e93965. [Google Scholar] [CrossRef]
- Sung, S.H.; Wu, T.C.; Chen, J.S.; Chen, Y.H.; Huang, P.H.; Lin, S.J.; Shih, C.C.; Chen, J.W. Reduced number and impaired function of circulating endothelial progenitor cells in patients with abdominal aortic aneurysm. Int. J. Cardiol. 2013, 168, 1070–1077. [Google Scholar] [CrossRef]
- Hernandez-Lopez, R.; Chavez-Gonzalez, A.; Torres-Barrera, P.; Moreno-Lorenzana, D.; Lopez-DiazGuerrero, N.; Santiago-German, D.; Isordia-Salas, I.; Smadja, D.; Yoder, C.M.; Majluf-Cruz, A.; et al. Reduced proliferation of endothelial colony-forming cells in unprovoked venous thromboembolic disease as a consequence of endothelial dysfunction. PLoS ONE 2017, 12, e0183827. [Google Scholar] [CrossRef]
- Fujinaga, H.; Fujinaga, H.; Watanabe, N.; Kato, T.; Tamano, M.; Terao, M.; Takada, S.; Ito, Y.; Umezawa, A.; Kuroda, M. Cord blood-derived endothelial colony-forming cell function is disrupted in congenital diaphragmatic hernia. Am. J. Physiol. Lung Cell Mol. Physiol. 2016, 310, L1143–L1154. [Google Scholar] [CrossRef]
- Komici, K.; Faris, P.; Negri, S.; Rosti, V.; Garcia-Carrasco, M.; Mendoza-Pinto, C.; Berra-Romani, R.; Cervera, R.; Guerra, G.; Moccia, F. Systemic lupus erythematosus, endothelial progenitor cells and intracellular Ca(2+) signaling: A novel approach for an old disease. J. Autoimmun. 2020. [Google Scholar] [CrossRef]
- Choi, S.A.; Chong, S.; Kwak, P.A.; Moon, Y.J.; Jangra, A.; Phi, J.H.; Lee, J.Y.; Park, S.H.; Kim, S.K. Impaired functional recovery of endothelial colony-forming cells from moyamoya disease in a chronic cerebral hypoperfusion rat model. J. Neurosurg. Pediatr. 2018, 23, 204–213. [Google Scholar] [CrossRef] [Green Version]
- Gupta, S.K.; Liu, Z.; Sims, E.C.; Repass, M.J.; Haneline, L.S.; Yoder, M.C. Endothelial Colony-Forming Cell Function Is Reduced During HIV Infection. J. Infect. Dis. 2019, 219, 1076–1083. [Google Scholar] [CrossRef]
- Mena, H.A.; Zubiry, P.R.; Dizier, B.; Schattner, M.; Boisson-Vidal, C.; Negrotto, S. Acidic preconditioning of endothelial colony-forming cells (ECFC) promote vasculogenesis under proinflammatory and high glucose conditions in vitro and in vivo. Stem Cell Res. Ther. 2018, 9, 120. [Google Scholar] [CrossRef]
- Gremmels, H.; de Jong, O.G.; Hazenbrink, D.H.; Fledderus, J.O.; Verhaar, M.C. The Transcription Factor Nrf2 Protects Angiogenic Capacity of Endothelial Colony-Forming Cells in High-Oxygen Radical Stress Conditions. Stem Cells Int. 2017, 2017, 4680612. [Google Scholar] [CrossRef] [Green Version]
- He, M.; Ma, S.; Cai, Q.; Wu, Y.; Shao, C.; Kong, H.; Wang, H.; Zeng, X.; Xie, W. Hypoxia induces the dysfunction of human endothelial colony-forming cells via HIF-1alpha signaling. Respir. Physiol. Neurobiol. 2018, 247, 87–95. [Google Scholar] [CrossRef]
- Tasev, D.; Dekker-Vroling, L.; van Wijhe, M.; Broxterman, H.J.; Koolwijk, P.; van Hinsbergh, V.W.M. Hypoxia Impairs Initial Outgrowth of Endothelial Colony Forming Cells and Reduces Their Proliferative and Sprouting Potential. Front. Med. 2018, 5, 356. [Google Scholar] [CrossRef] [Green Version]
- Mena, H.A.; Carestia, A.; Scotti, L.; Parborell, F.; Schattner, M.; Negrotto, S. Extracellular histones reduce survival and angiogenic responses of late outgrowth progenitor and mature endothelial cells. J. Thromb. Haemost. 2016, 14, 397–410. [Google Scholar] [CrossRef]
- Moccia, F.; Lucariello, A.; Guerra, G. TRPC3-mediated Ca(2+) signals as a promising strategy to boost therapeutic angiogenesis in failing hearts: The role of autologous endothelial colony forming cells. J. Cell. Physiol. 2018, 233, 3901–3917. [Google Scholar] [CrossRef]
- Suarez, Y.; Shepherd, B.R.; Rao, D.A.; Pober, J.S. Alloimmunity to human endothelial cells derived from cord blood progenitors. J. Immunol. 2007, 179, 7488–7496. [Google Scholar] [CrossRef] [Green Version]
- Merola, J.; Reschke, M.; Pierce, R.W.; Qin, L.; Spindler, S.; Baltazar, T.; Manes, T.D.; Lopez-Giraldez, F.; Li, G.; Bracaglia, L.G.; et al. Progenitor-derived human endothelial cells evade alloimmunity by CRISPR/Cas9-mediated complete ablation of MHC expression. JCI Insight 2019, 4. [Google Scholar] [CrossRef] [Green Version]
- Tasev, D.; van Wijhe, M.H.; Weijers, E.M.; van Hinsbergh, V.W.; Koolwijk, P. Long-Term Expansion in Platelet Lysate Increases Growth of Peripheral Blood-Derived Endothelial-Colony Forming Cells and Their Growth Factor-Induced Sprouting Capacity. PLoS ONE 2015, 10, e0129935. [Google Scholar] [CrossRef] [Green Version]
- Siegel, G.; Fleck, E.; Elser, S.; Hermanutz-Klein, U.; Waidmann, M.; Northoff, H.; Seifried, E.; Schafer, R. Manufacture of endothelial colony-forming progenitor cells from steady-state peripheral blood leukapheresis using pooled human platelet lysate. Transfusion 2018, 58, 1132–1142. [Google Scholar] [CrossRef]
- Kim, H.; Prasain, N.; Vemula, S.; Ferkowicz, M.J.; Yoshimoto, M.; Voytik-Harbin, S.L.; Yoder, M.C. Human platelet lysate improves human cord blood derived ECFC survival and vasculogenesis in three dimensional (3D) collagen matrices. Microvasc. Res. 2015, 101, 72–81. [Google Scholar] [CrossRef] [Green Version]
- Ogunye, A.A.; McNiece, I.K.; Daniliuc, S.; Genser-Nir, M.; Miropolski, Y.Y.; Fiorentini, D.; Kellner, J.N. Ex vivo expansion of cord blood-derived endothelial cells using a novel xeno-free culture media. Future Sci. OA 2019, 5, FSO376. [Google Scholar] [CrossRef] [Green Version]
- Fortunato, T.; Beltrami, C.; Emanueli, C.; De Bank, P.A.; Pula, G. Platelet lysate gel and endothelial progenitors stimulate microvascular network formation in vitro: Tissue engineering implications. Sci. Rep. 2016, 6, 25326. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zemani, F.; Benisvy, D.; Galy-Fauroux, I.; Lokajczyk, A.; Colliec-Jouault, S.; Uzan, G.; Fischer, A.M.; Boisson-Vidal, C. Low-molecular-weight fucoidan enhances the proangiogenic phenotype of endothelial progenitor cells. Biochem. Pharmacol. 2005, 70, 1167–1175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, J.H.; Lee, S.H.; Choi, S.H.; Asahara, T.; Kwon, S.M. The sulfated polysaccharide fucoidan rescues senescence of endothelial colony-forming cells for ischemic repair. Stem Cells 2015, 33, 1939–1951. [Google Scholar] [CrossRef] [PubMed]
- Sarlon, G.; Zemani, F.; David, L.; Duong Van Huyen, J.P.; Dizier, B.; Grelac, F.; Colliec-Jouault, S.; Galy-Fauroux, I.; Bruneval, P.; Fischer, A.M.; et al. Therapeutic effect of fucoidan-stimulated endothelial colony-forming cells in peripheral ischemia. J. Thromb. Haemost. 2012, 10, 38–48. [Google Scholar] [CrossRef] [Green Version]
- Zemani, F.; Silvestre, J.S.; Fauvel-Lafeve, F.; Bruel, A.; Vilar, J.; Bieche, I.; Laurendeau, I.; Galy-Fauroux, I.; Fischer, A.M.; Boisson-Vidal, C. Ex vivo priming of endothelial progenitor cells with SDF-1 before transplantation could increase their proangiogenic potential. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 644–650. [Google Scholar] [CrossRef]
- Sun, J.; Li, Y.; Graziani, G.M.; Filion, L.; Allan, D.S. E-selectin mediated adhesion and migration of endothelial colony forming cells is enhanced by SDF-1α/CXCR4. PLoS ONE 2013, 8, e60890. [Google Scholar] [CrossRef] [Green Version]
- Smadja, D.M.; Bieche, I.; Uzan, G.; Bompais, H.; Muller, L.; Boisson-Vidal, C.; Vidaud, M.; Aiach, M.; Gaussem, P. PAR-1 activation on human late endothelial progenitor cells enhances angiogenesis in vitro with upregulation of the SDF-1/CXCR4 system. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 2321–2327. [Google Scholar] [CrossRef] [Green Version]
- Smadja, D.M.; Laurendeau, I.; Avignon, C.; Vidaud, M.; Aiach, M.; Gaussem, P. The angiopoietin pathway is modulated by PAR-1 activation on human endothelial progenitor cells. J. Thromb. Haemost. 2006, 4, 2051–2058. [Google Scholar] [CrossRef]
- Bennis, Y.; Sarlon-Bartoli, G.; Guillet, B.; Lucas, L.; Pellegrini, L.; Velly, L.; Blot-Chabaud, M.; Dignat-Georges, F.; Sabatier, F.; Pisano, P. Priming of late endothelial progenitor cells with erythropoietin before transplantation requires the CD131 receptor subunit and enhances their angiogenic potential. J. Thromb. Haemost. 2012, 10, 1914–1928. [Google Scholar] [CrossRef]
- Wang, D.; Song, Y.; Zhang, J.; Pang, W.; Wang, X.; Zhu, Y.; Li, X. AMPK-KLF2 signaling pathway mediates the proangiogenic effect of erythropoietin in endothelial colony-forming cells. Am. J. Physiol. Cell Physiol. 2017, 313, C674–C685. [Google Scholar] [CrossRef]
- Pellegrini, L.; Bennis, Y.; Guillet, B.; Velly, L.; Garrigue, P.; Sabatier, F.; Dignat-George, F.; Bruder, N.; Pisano, P. Therapeutic benefit of a combined strategy using erythropoietin and endothelial progenitor cells after transient focal cerebral ischemia in rats. Neurol. Res. 2013, 35, 937–947. [Google Scholar] [CrossRef]
- Hache, G.; Garrigue, P.; Bennis, Y.; Stalin, J.; Moyon, A.; Cerami, A.; Brines, M.; Blot-Chabaud, M.; Sabatier, F.; Dignat-George, F.; et al. ARA290, a Specific Agonist of Erythropoietin/CD131 Heteroreceptor, Improves Circulating Endothelial Progenitors’ Angiogenic Potential and Homing Ability. Shock 2016, 46, 390–397. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Lee, J.H.; Asahara, T.; Kim, Y.S.; Jeong, H.C.; Ahn, Y.; Jung, J.S.; Kwon, S.M. Genistein promotes endothelial colony-forming cell (ECFC) bioactivities and cardiac regeneration in myocardial infarction. PLoS ONE 2014, 9, e96155. [Google Scholar] [CrossRef]
- Liu, B.; Xu, L.; Yu, X.; Jiao, X.; Yan, J.; Li, W.; Guo, M. Genistein Inhibited Estradiol-Induced Vascular Endothelial Cell Injury by Downregulating the FAK/Focal Adhesion Pathway. Cell. Physiol. Biochem. 2018, 49, 2277–2292. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Liu, Q.; Sun, J.; Yi, K.; Wu, L.; Tan, X. Nicotine improves the functional activity of late endothelial progenitor cells via nicotinic acetylcholine receptors. Biochem. Cell Biol. 2011, 89, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Mena, H.A.; Lokajczyk, A.; Dizier, B.; Strier, S.E.; Voto, L.S.; Boisson-Vidal, C.; Schattner, M.; Negrotto, S. Acidic preconditioning improves the proangiogenic responses of endothelial colony forming cells. Angiogenesis 2014, 17, 867–879. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, S.H.; Lee, J.H.; Han, Y.S.; Ryu, J.M.; Yoon, Y.M.; Han, H.J. Hypoxia accelerates vascular repair of endothelial colony-forming cells on ischemic injury via STAT3-BCL3 axis. Stem Cell Res. Ther. 2015, 6, 139. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.H.; Lee, J.H.; Yoo, S.Y.; Hur, J.; Kim, H.S.; Kwon, S.M. Hypoxia inhibits cellular senescence to restore the therapeutic potential of old human endothelial progenitor cells via the hypoxia-inducible factor-1alpha-TWIST-p21 axis. Arterioscler. Thromb. Vasc. Biol. 2013, 33, 2407–2414. [Google Scholar] [CrossRef] [Green Version]
- Moccia, F.; Guerra, G. Ca(2+) Signalling in Endothelial Progenitor Cells: Friend or Foe? J. Cell. Physiol. 2016, 231, 314–327. [Google Scholar] [CrossRef]
- Dragoni, S.; Reforgiato, M.; Zuccolo, E.; Poletto, V.; Lodola, F.; Ruffinatti, F.A.; Bonetti, E.; Guerra, G.; Barosi, G.; Rosti, V.; et al. Dysregulation of VEGF-induced proangiogenic Ca2+ oscillations in primary myelofibrosis-derived endothelial colony-forming cells. Exp. Hematol. 2015, 43, 1019.e1013–1030.e1013. [Google Scholar] [CrossRef]
- Lodola, F.; Laforenza, U.; Cattaneo, F.; Ruffinatti, F.A.; Poletto, V.; Massa, M.; Tancredi, R.; Zuccolo, E.; Khdar, A.D.; Riccardi, A.; et al. VEGF-induced intracellular Ca2+ oscillations are down-regulated and do not stimulate angiogenesis in breast cancer-derived endothelial colony forming cells. Oncotarget 2017, 8, 95223–95246. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moccia, F.; Bonetti, E.; Dragoni, S.; Fontana, J.; Lodola, F.; Romani, R.B.; Laforenza, U.; Rosti, V.; Tanzi, F. Hematopoietic progenitor and stem cells circulate by surfing on intracellular Ca2+ waves: A novel target for cell-based therapy and anti-cancer treatment? Curr. Signal Transd. Ther. 2012, 7, 161–176. [Google Scholar] [CrossRef]
- Negri, S.; Faris, P.; Pellavio, G.; Botta, L.; Orgiu, M.; Forcaia, G.; Sancini, G.; Laforenza, U.; Moccia, F. Group 1 metabotropic glutamate receptors trigger glutamate-induced intracellular Ca(2+) signals and nitric oxide release in human brain microvascular endothelial cells. Cell. Mol. Life Sci. 2019, 77, 2235–2253. [Google Scholar] [CrossRef]
- Faris, P.; Pellavio, G.; Ferulli, F.; Di Nezza, F.; Shekha, M.; Lim, D.; Maestri, M.; Guerra, G.; Ambrosone, L.; Pedrazzoli, P.; et al. Nicotinic Acid Adenine Dinucleotide Phosphate (NAADP) Induces Intracellular Ca(2+) Release through the Two-Pore Channel TPC1 in Metastatic Colorectal Cancer Cells. Cancers 2019, 11, E542. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berra-Romani, R.; Faris, P.; Negri, S.; Botta, L.; Genova, T.; Moccia, F. Arachidonic Acid Evokes an Increase in Intracellular Ca(2+) Concentration and Nitric Oxide Production in Endothelial Cells from Human Brain Microcirculation. Cells 2019, 8, 689. [Google Scholar] [CrossRef] [Green Version]
- Berra-Romani, R.; Faris, P.; Pellavio, G.; Orgiu, M.; Negri, S.; Forcaia, G.; Var-Gaz-Guadarrama, V.; Garcia-Carrasco, M.; Botta, L.; Sancini, G.; et al. Histamine induces intracellular Ca(2+) oscillations and nitric oxide release in endothelial cells from brain microvascular circulation. J. Cell. Physiol. 2020, 235, 1515–1530. [Google Scholar] [CrossRef] [PubMed]
- Dragoni, S.; Guerra, G.; Fiorio Pla, A.; Bertoni, G.; Rappa, A.; Poletto, V.; Bottino, C.; Aronica, A.; Lodola, F.; Cinelli, M.P.; et al. A functional transient receptor potential vanilloid 4 (TRPV4) channel is expressed in human endothelial progenitor cells. J. Cell. Physiol. 2015, 230, 95–104. [Google Scholar] [CrossRef]
- Zuccolo, E.; Dragoni, S.; Poletto, V.; Catarsi, P.; Guido, D.; Rappa, A.; Reforgiato, M.; Lodola, F.; Lim, D.; Rosti, V.; et al. Arachidonic acid-evoked Ca2+ signals promote nitric oxide release and proliferation in human endothelial colony forming cells. Vascul. Pharmacol. 2016, 87, 159–171. [Google Scholar] [CrossRef] [PubMed]
- Bouvard, C.; Galy-Fauroux, I.; Grelac, F.; Carpentier, W.; Lokajczyk, A.; Gandrille, S.; Colliec-Jouault, S.; Fischer, A.M.; Helley, D. Low-Molecular-Weight Fucoidan Induces Endothelial Cell Migration via the PI3K/AKT Pathway and Modulates the Transcription of Genes Involved in Angiogenesis. Mar. Drugs 2015, 13, 7446–7462. [Google Scholar] [CrossRef] [Green Version]
- Di Nezza, F.; Zuccolo, E.; Poletto, V.; Rosti, V.; De Luca, A.; Moccia, F.; Guerra, G.; Ambrosone, L. Liposomes as a Putative Tool to Investigate NAADP Signaling in Vasculogenesis. J. Cell. Biochem. 2017, 118, 3722–3729. [Google Scholar] [CrossRef]
- Huang, Z.; Miao, X.; Patarroyo, M.; Nilsson, G.P.; Pernow, J.; Li, N. Tetraspanin CD151 and integrin α6β1 mediate platelet-enhanced endothelial colony forming cell angiogenesis. J. Thromb. Haemost. 2016, 14, 606–618. [Google Scholar] [CrossRef]
- Smadja, D.M.; Bièche, I.; Emmerich, J.; Aiach, M.; Gaussem, P. PAR-1 activation has different effects on the angiogenic activity of endothelial progenitor cells derived from human adult and cord blood. J. Thromb. Haemost. 2006, 4, 2729–2731. [Google Scholar] [CrossRef] [PubMed]
- Langford-Smith, A.W.W.; Hasan, A.; Weston, R.; Edwards, N.; Jones, A.M.; Boulton, A.J.M.; Bowling, F.L.; Rashid, S.T.; Wilkinson, F.L.; Alexander, M.Y. Diabetic endothelial colony forming cells have the potential for restoration with glycomimetics. Sci. Rep. 2019, 9, 2309. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Williamson, K.A.; Hamilton, A.; Reynolds, J.A.; Sipos, P.; Crocker, I.; Stringer, S.E.; Alexander, Y.M. Age-related impairment of endothelial progenitor cell migration correlates with structural alterations of heparan sulfate proteoglycans. Aging Cell 2013, 12, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, A.M.; Wilkinson, F.L.; Jones, A.M.; Wilkinson, J.A.; Romero, M.; Duarte, J.; Alexander, M.Y. A novel role for small molecule glycomimetics in the protection against lipid-induced endothelial dysfunction: Involvement of Akt/eNOS and Nrf2/ARE signaling. Biochim. Biophys. Acta Gen. Subj. 2017, 1861, 3311–3322. [Google Scholar] [CrossRef] [PubMed]
- Leicht, S.F.; Schwarz, T.M.; Hermann, P.C.; Seissler, J.; Aicher, A.; Heeschen, C. Adiponectin pretreatment counteracts the detrimental effect of a diabetic environment on endothelial progenitors. Diabetes 2011, 60, 652–661. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.W.; Su, S.H.; Wang, Y.L.; Chang, S.T.; Liao, K.H.; Lo, H.H.; Chiu, Y.L.; Hsieh, T.H.; Huang, T.S.; Lin, C.S.; et al. MicroRNA-134 Contributes to Glucose-Induced Endothelial Cell Dysfunction and This Effect Can Be Reversed by Far-Infrared Irradiation. PLoS ONE 2016, 11, e0147067. [Google Scholar] [CrossRef]
- Li, D.W.; Li, J.H.; Wang, Y.D.; Li, G.R. Atorvastatin protects endothelial colony--forming cells against H2O2--induced oxidative damage by regulating the expression of annexin A2. Mol. Med. Rep. 2015, 12, 7941–7948. [Google Scholar] [CrossRef]
- He, M.; Cui, T.; Cai, Q.; Wang, H.; Kong, H.; Xie, W. Iptakalim ameliorates hypoxia-impaired human endothelial colony-forming cells proliferation, migration, and angiogenesis via Akt/eNOS pathways. Pulm. Circ. 2019, 9, 2045894019875417. [Google Scholar] [CrossRef]
- Wang, H.; Tang, Y.; Zhang, Y.L. Hypoxic pulmonary hypertension (HPH) and iptakalim, a novel ATP-sensitive potassium channel opener targeting smaller arteries in hypertension. Cardiovasc. Drug Rev. 2005, 23, 293–316. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; He, M.Y.; Ye, J.K.; Ma, S.Y.; Huang, W.; Wei, Y.Y.; Kong, H.; Wang, H.; Zeng, X.N.; Xie, W.P. Activation of ATP-sensitive potassium channels facilitates the function of human endothelial colony-forming cells via Ca(2+)/Akt/eNOS pathway. J. Cell. Mol. Med. 2017, 21, 609–620. [Google Scholar] [CrossRef] [PubMed]
- Moccia, F.; Berra-Romani, R.; Tanzi, F. Update on vascular endothelial Ca2+ signalling: A tale of ion channels, pumps and transporters. World J. Biol. Chem. 2012, 3, 127–158. [Google Scholar] [CrossRef] [PubMed]
- Zuccolo, E.; Bottino, C.; Diofano, F.; Poletto, V.; Codazzi, A.C.; Mannarino, S.; Campanelli, R.; Fois, G.; Marseglia, G.L.; Guerra, G.; et al. Constitutive Store-Operated Ca(2+) Entry Leads to Enhanced Nitric Oxide Production and Proliferation in Infantile Hemangioma-Derived Endothelial Colony-Forming Cells. Stem Cells Dev. 2016, 25, 301–319. [Google Scholar] [CrossRef]
- Dragoni, S.; Laforenza, U.; Bonetti, E.; Lodola, F.; Bottino, C.; Berra-Romani, R.; Carlo Bongio, G.; Cinelli, M.P.; Guerra, G.; Pedrazzoli, P.; et al. Vascular endothelial growth factor stimulates endothelial colony forming cells proliferation and tubulogenesis by inducing oscillations in intracellular Ca2+ concentration. Stem Cells 2011, 29, 1898–1907. [Google Scholar] [CrossRef] [PubMed]
- Tsai, W.C.; Chiang, W.H.; Wu, C.H.; Li, Y.C.; Campbell, M.; Huang, P.H.; Lin, M.W.; Lin, C.H.; Cheng, S.M.; Chang, P.C.; et al. miR-548aq-3p is a novel target of Far infrared radiation which predicts coronary artery disease endothelial colony forming cell responsiveness. Sci. Rep. 2020, 10, 6805. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zuccolo, E.; Di Buduo, C.; Lodola, F.; Orecchioni, S.; Scarpellino, G.; Kheder, D.A.; Poletto, V.; Guerra, G.; Bertolini, F.; Balduini, A.; et al. Stromal Cell-Derived Factor-1alpha Promotes Endothelial Colony-Forming Cell Migration Through the Ca(2+)-Dependent Activation of the Extracellular Signal-Regulated Kinase 1/2 and Phosphoinositide 3-Kinase/AKT Pathways. Stem Cells Dev. 2018, 27, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Li, X.; Wang, D.; Fu, C.; Zhu, Z.; Zou, M.H.; Zhu, Y. Transcription factor Krüppel-like factor 2 plays a vital role in endothelial colony forming cells differentiation. Cardiovasc. Res. 2013, 99, 514–524. [Google Scholar] [CrossRef] [Green Version]
- Negri, S.; Faris, P.; Rosti, V.; Antognazza, M.R.; Lodola, F.; Moccia, F. Endothelial TRPV1 as an Emerging Molecular Target to Promote Therapeutic Angiogenesis. Cells 2020, 9. [Google Scholar] [CrossRef]
- Smadja, D.M.; Bieche, I.; Silvestre, J.S.; Germain, S.; Cornet, A.; Laurendeau, I.; Duong-Van-Huyen, J.P.; Emmerich, J.; Vidaud, M.; Aiach, M.; et al. Bone morphogenetic proteins 2 and 4 are selectively expressed by late outgrowth endothelial progenitor cells and promote neoangiogenesis. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 2137–2143. [Google Scholar] [CrossRef]
- Balbi, C.; Lodder, K.; Costa, A.; Moimas, S.; Moccia, F.; van Herwaarden, T.; Rosti, V.; Campagnoli, F.; Palmeri, A.; De Biasio, P.; et al. Reactivating endogenous mechanisms of cardiac regeneration via paracrine boosting using the human amniotic fluid stem cell secretome. Int. J. Cardiol. 2019, 287, 87–95. [Google Scholar] [CrossRef]
- Balbi, C.; Lodder, K.; Costa, A.; Moimas, S.; Moccia, F.; van Herwaarden, T.; Rosti, V.; Campagnoli, F.; Palmeri, A.; De Biasio, P.; et al. Supporting data on in vitro cardioprotective and proliferative paracrine effects by the human amniotic fluid stem cell secretome. Data Brief 2019, 25, 104324. [Google Scholar] [CrossRef] [PubMed]
- Smadja, D.M.; d’Audigier, C.; Bieche, I.; Evrard, S.; Mauge, L.; Dias, J.V.; Labreuche, J.; Laurendeau, I.; Marsac, B.; Dizier, B.; et al. Thrombospondin-1 is a plasmatic marker of peripheral arterial disease that modulates endothelial progenitor cell angiogenic properties. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 551–559. [Google Scholar] [CrossRef] [Green Version]
- Dias, J.V.; Benslimane-Ahmim, Z.; Egot, M.; Lokajczyk, A.; Grelac, F.; Galy-Fauroux, I.; Juliano, L.; Le-Bonniec, B.; Takiya, C.M.; Fischer, A.M.; et al. A motif within the N-terminal domain of TSP-1 specifically promotes the proangiogenic activity of endothelial colony-forming cells. Biochem. Pharmacol. 2012, 84, 1014–1023. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Benslimane-Ahmim, Z.; Heymann, D.; Dizier, B.; Lokajczyk, A.; Brion, R.; Laurendeau, I.; Bieche, I.; Smadja, D.M.; Galy-Fauroux, I.; Colliec-Jouault, S.; et al. Osteoprotegerin, a new actor in vasculogenesis, stimulates endothelial colony-forming cells properties. J. Thromb. Haemost. 2011, 9, 834–843. [Google Scholar] [CrossRef] [PubMed]
- Benslimane-Ahmim, Z.; Poirier, F.; Delomenie, C.; Lokajczyk, A.; Grelac, F.; Galy-Fauroux, I.; Mohamedi, A.; Fischer, A.M.; Heymann, D.; Lutomski, D.; et al. Mechanistic study of the proangiogenic effect of osteoprotegerin. Angiogenesis 2013, 16, 575–593. [Google Scholar] [CrossRef]
- Stalin, J.; Harhouri, K.; Hubert, L.; Garrigue, P.; Nollet, M.; Essaadi, A.; Muller, A.; Foucault-Bertaud, A.; Bachelier, R.; Sabatier, F.; et al. Soluble CD146 boosts therapeutic effect of endothelial progenitors through proteolytic processing of short CD146 isoform. Cardiovasc. Res. 2016, 111, 240–251. [Google Scholar] [CrossRef] [Green Version]
- Caliceti, C.; Zambonin, L.; Rizzo, B.; Fiorentini, D.; Vieceli Dalla Sega, F.; Hrelia, S.; Prata, C. Role of plasma membrane caveolae/lipid rafts in VEGF-induced redox signaling in human leukemia cells. BioMed Res. Int. 2014, 2014, 857504. [Google Scholar] [CrossRef] [Green Version]
- Essaadi, A.; Nollet, M.; Moyon, A.; Stalin, J.; Simoncini, S.; Balasse, L.; Bertaud, A.; Bachelier, R.; Leroyer, A.S.; Sarlon, G.; et al. Stem cell properties of peripheral blood endothelial progenitors are stimulated by soluble CD146 via miR-21: Potential use in autologous cell therapy. Sci. Rep. 2018, 8, 9387. [Google Scholar] [CrossRef] [Green Version]
- Heo, S.C.; Kwon, Y.W.; Jang, I.H.; Jeong, G.O.; Yoon, J.W.; Kim, C.D.; Kwon, S.M.; Bae, Y.S.; Kim, J.H. WKYMVm-induced activation of formyl peptide receptor 2 stimulates ischemic neovasculogenesis by promoting homing of endothelial colony-forming cells. Stem Cells 2014, 32, 779–790. [Google Scholar] [CrossRef]
- Kim, B.R.; Jang, I.H.; Shin, S.H.; Kwon, Y.W.; Heo, S.C.; Choi, E.J.; Lee, J.S.; Kim, J.H. Therapeutic angiogenesis in a murine model of limb ischemia by recombinant periostin and its fasciclin I domain. Biochim. Biophys. Acta 2014, 1842, 1324–1332. [Google Scholar] [CrossRef] [Green Version]
- Kim, B.R.; Kwon, Y.W.; Park, G.T.; Choi, E.J.; Seo, J.K.; Jang, I.H.; Kim, S.C.; Ko, H.C.; Lee, S.C.; Kim, J.H. Identification of a novel angiogenic peptide from periostin. PLoS ONE 2017, 12, e0187464. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harhouri, K.; Kebir, A.; Guillet, B.; Foucault-Bertaud, A.; Voytenko, S.; Piercecchi-Marti, M.D.; Berenguer, C.; Lamy, E.; Vely, F.; Pisano, P.; et al. Soluble CD146 displays angiogenic properties and promotes neovascularization in experimental hind-limb ischemia. Blood 2010, 115, 3843–3851. [Google Scholar] [CrossRef] [PubMed]
- Holliday, R. The inheritance of epigenetic defects. Science 1987, 238, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Schübeler, D. Function and information content of DNA methylation. Nature 2015, 517, 321–326. [Google Scholar] [CrossRef]
- Ho, E.; Clarke, J.D.; Dashwood, R.H. Dietary Sulforaphane, a Histone Deacetylase Inhibitor for Cancer Prevention. J. Nutr. 2009, 139, 2393–2396. [Google Scholar] [CrossRef]
- He, L.; Hannon, G.J. MicroRNAs: Small RNAs with a big role in gene regulation. Nat. Rev. Genet. 2004, 5, 522–531. [Google Scholar] [CrossRef]
- Fraineau, S.; Palii, C.G.; McNeill, B.; Ritso, M.; Shelley, W.C.; Prasain, N.; Chu, A.; Vion, E.; Rieck, K.; Nilufar, S.; et al. Epigenetic Activation of Pro-angiogenic Signaling Pathways in Human Endothelial Progenitors Increases Vasculogenesis. Stem Cell Rep. 2017, 9, 1573–1587. [Google Scholar] [CrossRef] [Green Version]
- Fraineau, S.; Palii, C.G.; Allan, D.S.; Brand, M. Epigenetic regulation of endothelial-cell-mediated vascular repair. FEBS J. 2015, 282, 1605–1629. [Google Scholar] [CrossRef]
- Prasain, N.; Meador, J.L.; Yoder, M.C. Phenotypic and functional characterization of endothelial colony forming cells derived from human umbilical cord blood. J. Vis. Exp. 2012, 13, 3872. [Google Scholar] [CrossRef] [Green Version]
- Palii, C.G.; Vulesevic, B.; Fraineau, S.; Pranckeviciene, E.; Griffith, A.J.; Chu, A.; Faralli, H.; Li, Y.; McNeill, B.; Sun, J.; et al. Trichostatin A enhances vascular repair by injected human endothelial progenitors through increasing the expression of TAL1-dependent genes. Cell Stem Cell 2014, 14, 644–657. [Google Scholar] [CrossRef] [Green Version]
- Jangra, A.; Choi, S.A.; Koh, E.J.; Moon, Y.J.; Wang, K.C.; Phi, J.H.; Lee, J.Y.; Kim, S.K. Panobinostat, a histone deacetylase inhibitor, rescues the angiogenic potential of endothelial colony-forming cells in moyamoya disease. Child’s Nerv. Syst. 2019, 35, 823–831. [Google Scholar] [CrossRef]
- Lee, J.Y.; Moon, Y.J.; Lee, H.O.; Park, A.K.; Choi, S.A.; Wang, K.C.; Han, J.W.; Joung, J.G.; Kang, H.S.; Kim, J.E.; et al. Deregulation of Retinaldehyde Dehydrogenase 2 Leads to Defective Angiogenic Function of Endothelial Colony-Forming Cells in Pediatric Moyamoya Disease. Arterioscler. Thromb. Vasc. Biol. 2015, 35, 1670–1677. [Google Scholar] [CrossRef] [Green Version]
- Chang, T.Y.; Tsai, W.C.; Huang, T.S.; Su, S.H.; Chang, C.Y.; Ma, H.Y.; Wu, C.H.; Yang, C.Y.; Lin, C.H.; Huang, P.H.; et al. Dysregulation of endothelial colony-forming cell function by a negative feedback loop of circulating miR-146a and -146b in cardiovascular disease patients. PLoS ONE 2017, 12, e0181562. [Google Scholar] [CrossRef] [PubMed]
- Laurenzana, A.; Margheri, F.; Chillà, A.; Biagioni, A.; Margheri, G.; Calorini, L.; Fibbi, G.; Del Rosso, M. Endothelial Progenitor Cells as Shuttle of Anticancer Agents. Hum. Gene Ther. 2016, 27, 784–791. [Google Scholar] [CrossRef] [PubMed]
- Iwaguro, H.; Yamaguchi, J.; Kalka, C.; Murasawa, S.; Masuda, H.; Hayashi, S.; Silver, M.; Li, T.; Isner, J.M.; Asahara, T. Endothelial progenitor cell vascular endothelial growth factor gene transfer for vascular regeneration. Circulation 2002, 105, 732–738. [Google Scholar] [CrossRef]
- Lin, R.Z.; Dreyzin, A.; Aamodt, K.; Li, D.; Jaminet, S.C.; Dudley, A.C.; Melero-Martin, J.M. Induction of erythropoiesis using human vascular networks genetically engineered for controlled erythropoietin release. Blood 2011, 118, 5420–5428. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, Y.; Chang, L.; Solovey, A.; Healey, J.F.; Lollar, P.; Hebbel, R.P. Use of blood outgrowth endothelial cells for gene therapy for hemophilia A. Blood 2002, 99, 457–462. [Google Scholar] [CrossRef] [Green Version]
- Gao, K.; Kumar, P.; Cortez-Toledo, E.; Hao, D.; Reynaga, L.; Rose, M.; Wang, C.; Farmer, D.; Nolta, J.; Zhou, J.; et al. Potential long-term treatment of hemophilia A by neonatal co-transplantation of cord blood-derived endothelial colony-forming cells and placental mesenchymal stromal cells. Stem Cell Res. Ther. 2019, 10, 34. [Google Scholar] [CrossRef] [Green Version]
- De Meyer, S.F.; Vanhoorelbeke, K.; Chuah, M.K.; Pareyn, I.; Gillijns, V.; Hebbel, R.P.; Collen, D.; Deckmyn, H.; VandenDriessche, T. Phenotypic correction of von Willebrand disease type 3 blood-derived endothelial cells with lentiviral vectors expressing von Willebrand factor. Blood 2006, 107, 4728–4736. [Google Scholar] [CrossRef] [Green Version]
- de Boer, S.; Eikenboom, J. Von Willebrand Disease: From In Vivo to In Vitro Disease Models. HemaSphere 2019, 3, e297. [Google Scholar] [CrossRef]
- Bodempudi, V.; Ohlfest, J.R.; Terai, K.; Zamora, E.A.; Vogel, R.I.; Gupta, K.; Hebbel, R.P.; Dudek, A.Z. Blood outgrowth endothelial cell-based systemic delivery of antiangiogenic gene therapy for solid tumors. Cancer Gene Ther. 2010, 17, 855–863. [Google Scholar] [CrossRef] [PubMed]
- Dudek, A.Z.; Bodempudi, V.; Welsh, B.W.; Jasinski, P.; Griffin, R.J.; Milbauer, L.; Hebbel, R.P. Systemic inhibition of tumour angiogenesis by endothelial cell-based gene therapy. Br. J. Cancer 2007, 97, 513–522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramamoorth, M.; Narvekar, A. Non viral vectors in gene therapy- an overview. J. Clin. Diagn. Res. 2015, 9, GE01-06. [Google Scholar] [CrossRef] [PubMed]
- Hardee, C.L.; Arévalo-Soliz, L.M.; Hornstein, B.D.; Zechiedrich, L. Advances in Non-Viral DNA Vectors for Gene Therapy. Genes 2017, 8, 65. [Google Scholar] [CrossRef]
- Lim, D.; Bertoli, A.; Sorgato, M.C.; Moccia, F. Generation and usage of aequorin lentiviral vectors for Ca(2+) measurement in sub-cellular compartments of hard-to-transfect cells. Cell Calcium 2016, 59, 228–239. [Google Scholar] [CrossRef]
- Nowakowski, A.; Andrzejewska, A.; Janowski, M.; Walczak, P.; Lukomska, B. Genetic engineering of stem cells for enhanced therapy. Acta Neurobiol. Exp. 2013, 73, 1–18. [Google Scholar]
- Matsui, H.; Shibata, M.; Brown, B.; Labelle, A.; Hegadorn, C.; Andrews, C.; Hebbel, R.P.; Galipeau, J.; Hough, C.; Lillicrap, D. Ex vivo gene therapy for hemophilia A that enhances safe delivery and sustained in vivo factor VIII expression from lentivirally engineered endothelial progenitors. Stem Cells 2007, 25, 2660–2669. [Google Scholar] [CrossRef]
- Ozelo, M.C.; Vidal, B.; Brown, C.; Notley, C.; Hegadorn, C.; Webster, S.; Harpell, L.; Ahlin, J.; Winterborn, A.; Handforth, J.; et al. Omental implantation of BOECs in hemophilia dogs results in circulating FVIII antigen and a complex immune response. Blood 2014, 123, 4045–4053. [Google Scholar] [CrossRef]
- Cubbon, R.M.; Yuldasheva, N.Y.; Viswambharan, H.; Mercer, B.N.; Baliga, V.; Stephen, S.L.; Askham, J.; Sukumar, P.; Skromna, A.; Mughal, R.S.; et al. Restoring Akt1 activity in outgrowth endothelial cells from South Asian men rescues vascular reparative potential. Stem Cells 2014, 32, 2714–2723. [Google Scholar] [CrossRef] [Green Version]
- Goto, K.; Takemura, G.; Takahashi, T.; Okada, H.; Kanamori, H.; Kawamura, I.; Watanabe, T.; Morishita, K.; Tsujimoto, A.; Miyazaki, N.; et al. Intravenous Administration of Endothelial Colony-Forming Cells Overexpressing Integrin β1 Augments Angiogenesis in Ischemic Legs. Stem Cells Transl. Med. 2016, 5, 218–226. [Google Scholar] [CrossRef]
- Stupack, D.G.; Cheresh, D.A. Get a ligand, get a life: Integrins, signaling and cell survival. J. Cell Sci. 2002, 115, 3729–3738. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bloch, W.; Forsberg, E.; Lentini, S.; Brakebusch, C.; Martin, K.; Krell, H.W.; Weidle, U.H.; Addicks, K.; Fassler, R. Beta 1 integrin is essential for teratoma growth and angiogenesis. J. Cell Biol. 1997, 139, 265–278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Neill, K.M.; Campbell, D.C.; Edgar, K.S.; Gill, E.K.; Moez, A.; McLoughlin, K.J.; O’Neill, C.L.; Dellett, M.; Hargey, C.J.; Abudalo, R.A.; et al. NOX4 is a major regulator of cord blood-derived endothelial colony-forming cells which promotes post-ischaemic revascularization. Cardiovasc. Res. 2020, 116, 393–405. [Google Scholar] [CrossRef]
- Hakami, N.Y.; Ranjan, A.K.; Hardikar, A.A.; Dusting, G.J.; Peshavariya, H.M. Role of NADPH Oxidase-4 in Human Endothelial Progenitor Cells. Front. Physiol. 2017, 8, 150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sturtzel, C.; Lipnik, K.; Hofer-Warbinek, R.; Testori, J.; Ebner, B.; Seigner, J.; Qiu, P.; Bilban, M.; Jandrositz, A.; Preisegger, K.H.; et al. FOXF1 Mediates Endothelial Progenitor Functions and Regulates Vascular Sprouting. Front. Bioeng. Biotechnol. 2018, 6. [Google Scholar] [CrossRef] [Green Version]
- Dragoni, S.; Laforenza, U.; Bonetti, E.; Lodola, F.; Bottino, C.; Guerra, G.; Borghesi, A.; Stronati, M.; Rosti, V.; Tanzi, F.; et al. Canonical transient receptor potential 3 channel triggers vascular endothelial growth factor-induced intracellular Ca2+ oscillations in endothelial progenitor cells isolated from umbilical cord blood. Stem Cells Dev. 2013, 22, 2561–2580. [Google Scholar] [CrossRef]
- Moccia, F.; Dragoni, S.; Cinelli, M.; Montagnani, S.; Amato, B.; Rosti, V.; Guerra, G.; Tanzi, F. How to utilize Ca2+ signals to rejuvenate the repairative phenotype of senescent endothelial progenitor cells in elderly patients affected by cardiovascular diseases: A useful therapeutic support of surgical approach? BMC Surg. 2013, 13, S46. [Google Scholar] [CrossRef] [Green Version]
- d’Audigier, C.; Susen, S.; Blandinieres, A.; Mattot, V.; Saubamea, B.; Rossi, E.; Nevo, N.; Lecourt, S.; Guerin, C.L.; Dizier, B.; et al. Egfl7 Represses the Vasculogenic Potential of Human Endothelial Progenitor Cells. Stem Cell Rev. Rep. 2018, 14, 82–91. [Google Scholar] [CrossRef]
- Fortunato, T.M.; Vara, D.S.; Wheeler-Jones, C.P.; Pula, G. Expression of protease-activated receptor 1 and 2 and anti-tubulogenic activity of protease-activated receptor 1 in human endothelial colony-forming cells. PLoS ONE 2014, 9, e109375. [Google Scholar] [CrossRef] [Green Version]
- Souidi, N.; Stolk, M.; Rudeck, J.; Strunk, D.; Schallmoser, K.; Volk, H.D.; Seifert, M. Stromal Cells Act as Guardians for Endothelial Progenitors by Reducing Their Immunogenicity After Co-Transplantation. Stem Cells 2017, 35, 1233–1245. [Google Scholar] [CrossRef] [Green Version]
- Joo, H.J.; Seo, H.R.; Jeong, H.E.; Choi, S.C.; Park, J.H.; Yu, C.W.; Hong, S.J.; Chung, S.; Lim, D.S. Smooth muscle progenitor cells from peripheral blood promote the neovascularization of endothelial colony-forming cells. Biochem. Biophys. Res. Commun. 2014, 449, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Shafiee, A.; Patel, J.; Wong, H.Y.; Donovan, P.; Hutmacher, D.W.; Fisk, N.M.; Khosrotehrani, K. Priming of endothelial colony-forming cells in a mesenchymal niche improves engraftment and vasculogenic potential by initiating mesenchymal transition orchestrated by NOTCH signaling. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2017, 31, 610–624. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Poitevin, S.; Cussac, D.; Leroyer, A.S.; Albinet, V.; Sarlon-Bartoli, G.; Guillet, B.; Hubert, L.; Andrieu-Abadie, N.; Couderc, B.; Parini, A.; et al. Sphingosine kinase 1 expressed by endothelial colony-forming cells has a critical role in their revascularization activity. Cardiovasc. Res. 2014, 103, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Keshavarz, S.; Nassiri, S.M.; Siavashi, V.; Alimi, N.S. Regulation of plasticity and biological features of endothelial progenitor cells by MSC-derived SDF-1. Biochim. Biophys. Acta Mol. Cell Res. 2019, 1866, 296–304. [Google Scholar] [CrossRef] [PubMed]
- Peters, E.B.; Christoforou, N.; Moore, E.; West, J.L.; Truskey, G.A. CD45+ Cells Present Within Mesenchymal Stem Cell Populations Affect Network Formation of Blood-Derived Endothelial Outgrowth Cells. BioRes. Open Access 2015, 4, 75–88. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Wang, F.; Mei, H.; Wang, S.; Cheng, L. Human Adipose Mesenchymal Stem Cells Show More Efficient Angiogenesis Promotion on Endothelial Colony-Forming Cells than Umbilical Cord and Endometrium. Stem Cells Int. 2018, 2018, 7537589. [Google Scholar] [CrossRef] [Green Version]
- Shafiee, A.; Patel, J.; Lee, J.S.; Hutmacher, D.W.; Fisk, N.M.; Khosrotehrani, K. Mesenchymal stem/stromal cells enhance engraftment, vasculogenic and pro-angiogenic activities of endothelial colony forming cells in immunocompetent hosts. Sci. Rep. 2017, 7, 13558. [Google Scholar] [CrossRef]
- Lodola, F.; Rosti, V.; Tullii, G.; Desii, A.; Tapella, L.; Catarsi, P.; Lim, D.; Moccia, F.; Antognazza, M.R. Conjugated polymers optically regulate the fate of endothelial colony-forming cells. Sci. Adv. 2019, 5, eaav4620. [Google Scholar] [CrossRef] [Green Version]
| Disease | ECFC Source | Effect | Reference |
|---|---|---|---|
| Ischemic stroke | UCB | Neurological functional recovery, improved angiogenesis and neurogenesis, and decreased apoptosis | [61] |
| Ischemic stroke | UCB | Rescue of BBB integrity, reduced cerebral apoptosis, increased CBF | [106] |
| Ischemic stroke | PB | Rescue of BBB structure and function | [107] |
| MCAO | UCB | Neurological functional recovery, improved angiogenesis and neurogenesis, reduced astrogliosis | [71] |
| TBI | UCB | Neurological functional recovery and improved angiogenesis | [72] |
| TBI | UCB | Rescue of BBB integrity and angiogenesis | [73,108] |
| CA | UCB | Protection against degeneration of the aneurysmal wall | [54] |
| OIR | PB | Vascular repair, decreased avascular areas, and increased normovascular areas | [76] |
| OIR | hiPSCs-derived ECFCs | Increased neovessel formation and decreased neovascular tufts | [42] |
| OIR | UCB | Improved angiogenesis and reduced avascular area | [81] |
| OIR | UCB | Vascular repair | [21,79] |
| OIR | UCB | Improved angiogenesis and vascular repair | [80] |
| Diabetic retinopathy | UCB | Stabilized vision and improved angiogenesis | [77] |
| AMI | UCB | Improved cardiac function, enhanced neovascularization, and decreased myocardial fibrosis | [10,93] |
| AMI | PB | Improved cardiac function and enhanced neovascularization | [95] |
| AMI | UCB | Improved cardiac function and enhanced neovascularization and reduced fibrosis | [96] |
| AMI | UCB | Improved cardiac function and mitigation of adverse remodeling | [63] |
| CLI | UCB | Blood flow restoration | [57] |
| CLI | UCB | Rescued of blood perfusion and enhanced neovascularization | [70,93] |
| CLI | hiPSCs-derived ECFCs | Promote neovessel formation and rescue blood perfusion | [42] |
| CLI | UCB | Improved muscle perfusion and hindlimb regeneration | [103] |
| CLI | UCB | Rescued of blood perfusion and enhanced neovascularization | [105] |
| CLI | UCB | Enhanced vascularization, leg reperfusion and muscle repair | [109] |
| Strategy to Improve Regenerative Activity | ECFC Source | Disease or Pathological Condition | Effecton ECFCs | Mechanism of Action | Reference |
|---|---|---|---|---|---|
| Acidosis | UCB | Hindlimb ischemia | In vitro proliferation and tubulogenesis, in vivo revascularization | Activates Akt and ERK1/2, inhibits p38 | [156] |
| Acidosis | UCB | T2DM and Hindlimb ischemia | In vitro adhesion and anticytotoxic effect, in vivo revascularization | bFGF, TGFβ1, IL-8, IL-4, VEGF, PDGF, and IL-10 | [129] |
| AA | PB | In vitro proliferation | Activates TRPV4 channel and Ca2+-dependent NO release | [167,168] | |
| EPO | UCB | In vitro migration and tube formation | AMPK-Krüppel-like factor 2 (KLF2) and eNOS | [150] | |
| EPO | UCB | Cerebral ischemia | In vivo homing, reduction of BBB disruption, and cerebral apoptosis | CD146 expression | [106] |
| EPO | UCB | Cerebral ischemia | In vivo angiogenesis and neurogenesis, reduction of infarct volume and neurological deficit | Activation HSP27, STAT-5, Bcl-2, down-regulation of Bax and DP5.Akt-1, BDNF, and VEGF expression | [151] |
| EPO | UCB | Hindlimb ischemia | In vitro proliferation, migration, tube formation, resistance to H2O2-induced apoptosis, in vivo revascularization and rescue of blood flow | CD131 and PI3K/Akt | [149] |
| Fucoidan | UCB | Hindlimb ischemia | In vivo improvement of residual muscle blood flow and increased collateral vessel formation | SDF-1α | [144] |
| Fucoidan | UCB | Hindlimb ischemia | In vitro rescue from cellular senescence and tube formation, in vivo proliferation, survival, incorporation, and differentiation within neovesselsand recovery of blood flow | FAK, ERK, Akt | [143] |
| Fucoidan | UCB | In vitro migration | PI3K/Akt | [169] | |
| Genistein | UCB | AMI | In vitro proliferation and migration, in vivo revascularization, improvement of cardiac function and reduction of fibrosis | ILK, α-parvin, F-actin, and ERK1/2 | [153] |
| PL | UCB | In vitro survival, vasculogenesis, and augments blood vessel formation by inhibiting apoptosis | Akt, Bad, and Bcl-xL | [139] | |
| HPL-gel | PB | In vitro proliferation in 2D culture and formation of a complete microvascular network in 3D cultures | VEGF | [141] | |
| Hypoxia | PB | Hindlimb ischemia | In vitro inhibition of cellular senescence, enhances proliferation survival, and angiogenic. In vivo accelerates vascular repair capacity, increases blood flow ratio and capillary density | Hypoxia-inducible factor-1α-TWIST-p21 axis | [158] |
| Hypoxia | PB | Hindlimb ischemia | In vitro proliferation and survival. In vivo enhanced blood flow ratio, capillary density, and angiogenic cytokine secretion | STAT3–BCL3 axis | [157] |
| NAADP | PB | In vitro proliferation | Ca2+-dependent manner | [170] | |
| Nicotine | UCB | In vitro enhanced viability, adhesion, migration, and tube formation | a7-nAChR | [155] | |
| PRs | PB | In vitro tube formation | Tetraspanin CD151, α6β1 integrin, and Src–PI3K signaling pathway | [171] | |
| SDF-1α | UCB | In vitro enhanced adhesion and migration | SDF-1α and CXCR4 | [146] | |
| SDF-1α | UCB | Hindlimb ischemia | In vitro adhesion and tube formation. In vivo accumulation of transplanted ECFCs at sites of ischemia and enhanced neovascularization | MMP-2, FGF-2 CXCR4 | [145] |
| SFLLRN peptide | PB and UCB | Hindlimb ischemia | In vivo increased chemotactic gene expression and leukocyte recruitment at ischemic sites | COX-2 and PAR-1 | [53] |
| SFLLRN peptide | PB and UCB | Hindlimb ischemia | In vitro proliferation | Angiopoietin 2 and PAR-1 | [172] |
| Strategy to Improve Regenerative Activity | ECFC Source | Disease or Pathological Condition | Effect on ECFCs | Mechanism of Action | Reference |
|---|---|---|---|---|---|
| Adiponectin | PB | T2DM, hindlimb ischemia (normo- and hyperglycemic conditions) | In vitro proliferation and migration and in vivo neovascularization | COX-2 | [176] |
| Acidosis | UCB | T2DM, hindlimb ischemia | In vitro adhesion and release of pro-angiogenic molecules. In vivo increase in capillary density and rescued local blood flow | bFGF, TGFβ1, IL-8, IL-4, VEGF, PDGF, and IL release | [129] |
| Atorvastatin | H2O2-induced oxidative damage | In vitro resistance to cell death | Annexin A2 up-regulation | [178] | |
| C1P | UCB | Hindlimb ischemia | In vitro adhesion, proliferation, migration, and tube formation, in vivo vascularization of gel plugs and rescue of blood flow | ERK1/2 and Akt | [109] |
| FIR | PB of DM and healthy donors | High glucose-induced endothelial dysfunction and hindlimb ischemia | In vitro rescue of migration and tube formation, in vivo revascularization | Down-regulation of miR-134 | [177] |
| FIR | PB of CAD patients and healthy donor | CAD | In vitro rescue of migration and tube formation | Down-regulation of miR-486-5p | [185] |
| Fucoidan | UCB | Senescence andhindlimb ischemia | In vitro rescue from cellular senescence and tube formation, proliferation, and survival. In vivo incorporation and differentiation within neovesselsand recovery of blood flow | FAK, ERK, and Akt | [143] |
| Glycomimetic C3 | PB | DM with NI or NP foot ulcers, hindlimb ischemia | In vitro rescue of proliferation and tube formation (mainly in NI) | Akt/eNOS and Nrf2/ARE | [173] |
| Iptakalim | PB | In vitro proliferation and bidimensional tube formation | Akt and eNOS | [179] |
| Strategy to Improve Regenerative Activity | ECFC Source | Disease or Pathological Condition | Effect on ECFCs | Mechanism of Action | Reference |
|---|---|---|---|---|---|
| Acidosis | UCB | Hindlimb ischemia | In vitro proliferation and tubulogenesis, in vivo revascularization | Activates Akt and ERK1/2, inhibits p38 | [156] |
| BMP4 | UCB and PB | Hindlimb ischemia | In vitro proliferation and tube formation, in vivo revascularization | [189] | |
| EPO | UCB | In vitro migration and tube formation | AMPK-Krüppel-like factor 2 (KLF2) and eNOS | [150] | |
| EPO | UCB | Cerebral ischemia | In vivo homing, reduction of BBB disruption and cerebral apoptosis, rescue of CBF | CD146 | [106] |
| EPO | UCB | Cerebral ischemia | In vivo angiogenesis and neurogenesis, and reduction of infarct volume and neurological deficit | Activation of HSP27, STAT-5, Bcl-2, and down-regulation of Bax and DP5.Akt-1, BDNF, and VEGF expression | [151] |
| EPO | UCB | Hindlimb ischemia | In vitro proliferation, migration, tube formation, and resistance to H2O2-induced apoptosis, in vivo neovascularization and rescue of blood flow | CD131 and PI3K/Akt | [149] |
| Fucoidan | UCB | Hindlimb ischemia | In vivo improvement of residual muscle blood flow and increased collateral vessel formation | SDF-1α | [144] |
| Fucoidan | UCB | Senescence and hindlimb ischemia | In vitro rescue from cellular senescence and tube formation, proliferation, and survival. In vivo incorporation, differentiation within neovessels, and recovery of blood flow | FAK, ERK, and Akt | [143] |
| Fucoidan | UCB | In vitro migration | PI3K/Akt | [169] | |
| Genistein | UCB | AMI | In vitro proliferation and migration, in vivo revascularization and improvement of cardiac function, reduction of fibrosis | ILK, α-parvin, F-actin, and ERK1/2 | [153] |
| hAFS-CM | PB | AMI | In vitro bidimensional tube formation and promotion, in vivo revascularization | Through an oscillatory increase in [Ca2+]i | [190] |
| OPG | UCB | In vitro migration, chemotaxis, and vascular cord formation. In vivo microvessel formation | SDF-1α, ERK, PI3K/Akt/mTOR | [194] | |
| SDF-1α | UCB | In vitro enhanced adhesion and migration | CXCR4 | [146] | |
| SDF-1α | UCB | Hindlimb ischemia | In vitro adhesion and tube formation, in vivo accumulation of transplanted ECFCs at sites of ischemia and enhanced neovascularization | MMP-2, FGF-2 CXCR4 | [145] |
| sCD146 | Hindlimb ischemia | In vitro survival and enhanced angiogenesis in vivo | FADD, Bcl-xl, and eNOS | [196] | |
| sCD146 | PB from PADpatients | Hindlimb ischemia | In vitro increased clonogenic activity and inhibition of cellular senescence, in vivo neovascularization and recovery of blood flow | miR-21 and embryonic transcription factors | [198] |
| TSP1 | UCB | In vitro migration and tube formation in Matrigel plug, adhesion to an endothelial monolayer | Syndecan 4 and α6-integrin | [193] |
| Strategy to Improve Regenerative Activity | ECFC Source | Disease or Pathological Condition | Effect on ECFCs | Mechanism of Action | Reference |
|---|---|---|---|---|---|
| C1P | UCB | Hindlimb ischemia | In vitro adhesion, proliferation, migration, and tube formation, in vivo vascularization and rescue blood flow | ERK1/2 and Akt | [109] |
| Periostin | UCB | Hindlimb ischemia | In vitro proliferation and adhesion, in vivo homing, blood flow perfusion, and leg salvage | FAS-1, β3, and β5 integrins | [200] |
| sCD146 | UCB | Hindlimb ischemia | In vitro survival and in vivo angiogenesis | FADD, Bcl-xl, and eNOS | [196] |
| sCD146 | PB | Hindlimb ischemia | In vitro migration and tube formation | eNOS, VEGFR2, MMP-2, and uPA | [202] |
| WKYMVm | UCB | Hindlimb ischemia | In vitro chemotaxis, proliferation, and tube formation. In vivo attenuated tissue necrosis, neovascularization, and recovery of blood flow | FPR2 | [199] |
| Strategy to Improve Regenerative Activity | ECFC Source | Disease or Pathological Condition | Effect on ECFCs | Mechanism of Action | Reference |
|---|---|---|---|---|---|
| Egfl7 silencing | PB | Hindlimb ischemia | In vitro proliferation, differentiation, and migration, in vivo revascularization | [238] | |
| Epigenetic drugs (GSK-343 and Panobinostat, (either alone or in combination) | UCB | Hindlimb ischemia | In vitro migration and tube formation, and resistance to serum starvation-induced apoptosis, in vivo revascularization and rescue of blood flow | VEGFR2, CXCR4, WNT, Notch, and SHH | [207] |
| FIR | From PB of healthy donor and CAD patients | CAD | In vitro rescue of migration and tube formation | Down-regulation of miR-486-5p | [185] |
| FIR | From PB of DM and healthy donors | High glucose-induced endothelial dysfunction and hindlimb ischemia | In vitro rescue of migration and tube formation, in vivo revascularization | Down-regulation of miR-134 | [177] |
| FOXF1 overexpression | UCB | Sprouting angiogenesis in vitro and in a zebrafish model | Notch2 and VEGFR2 expression | [235] | |
| NOX4 overexpression | UCB | Hindlimb ischemia | In vitro proliferation, migration, and tubulogenesis. In vivo vasoreparative function and enhanced blood flow | H2O2 production, PMA-induced superoxide in a NOX4-dependent manner | [233] |
| Integrin β1 overexpression | PB | Hindlimb ischemia | ECFC homing, in vivo angiogenesis and recovery of blood flow | [230] | |
| Panobinostat | PB | MMD | Restoring capillary-like network formation | Derepressing RALDH | [211] |
| Akt1 overexpression | PB of South Asian men (at risk for cardiovascular events) | Hindlimb ischemia | In vitro angiogenesis, in vivo re-endothelialization and perfusion recovery | Akt and NO | [229] |
| Strategy to Improve Regenerative Activity | ECFC Source | Disease or Pathological Condition | Effect on ECFCs | Mechanism of Action | Reference |
|---|---|---|---|---|---|
| ECFCs + MPCs | UCB | Hindlimb ischemia | Enhanced blood flow | [57] | |
| ECFCs + MSCs | UCB | Lower risk of ECFC rejection, improved in vivo vascularization | Endothelial HLA-DR expression | [240] | |
| ECFCs + MSCs | UCB | Hindlimb ischemia | In vitro proliferation, migration and tubulogenesis, in vivo enhanced neovessel density | VEGF, HGF and MMP-9 | [55] |
| ECFCs + MSCs | Fetal term placental | In vitro matrigel plugs cell engraftment, and neovascularization | Via direct contact and Notch signaling | [247] | |
| ECFCs + MSCs | UCB | Ischemic gastrocnemius muscle | In vitro proliferation and capillary-like formation, in vivo increase in capillary density | Up-regulating SphK1 | [243] |
| ECFCs + MSCs | Mouse BM | In vitro proliferation, colony formation, migration, and angiogenesis | SDF1-α | [244] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Faris, P.; Negri, S.; Perna, A.; Rosti, V.; Guerra, G.; Moccia, F. Therapeutic Potential of Endothelial Colony-Forming Cells in Ischemic Disease: Strategies to Improve their Regenerative Efficacy. Int. J. Mol. Sci. 2020, 21, 7406. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21197406
Faris P, Negri S, Perna A, Rosti V, Guerra G, Moccia F. Therapeutic Potential of Endothelial Colony-Forming Cells in Ischemic Disease: Strategies to Improve their Regenerative Efficacy. International Journal of Molecular Sciences. 2020; 21(19):7406. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21197406
Chicago/Turabian StyleFaris, Pawan, Sharon Negri, Angelica Perna, Vittorio Rosti, Germano Guerra, and Francesco Moccia. 2020. "Therapeutic Potential of Endothelial Colony-Forming Cells in Ischemic Disease: Strategies to Improve their Regenerative Efficacy" International Journal of Molecular Sciences 21, no. 19: 7406. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21197406





