Tumor Necrosis Factor α and Interleukin-1β Acutely Inhibit AgRP Neurons in the Arcuate Nucleus of the Hypothalamus
Abstract
:1. Introduction
2. Results
2.1. Characterization of Resting Biophysical Properties of AgRP Neurons
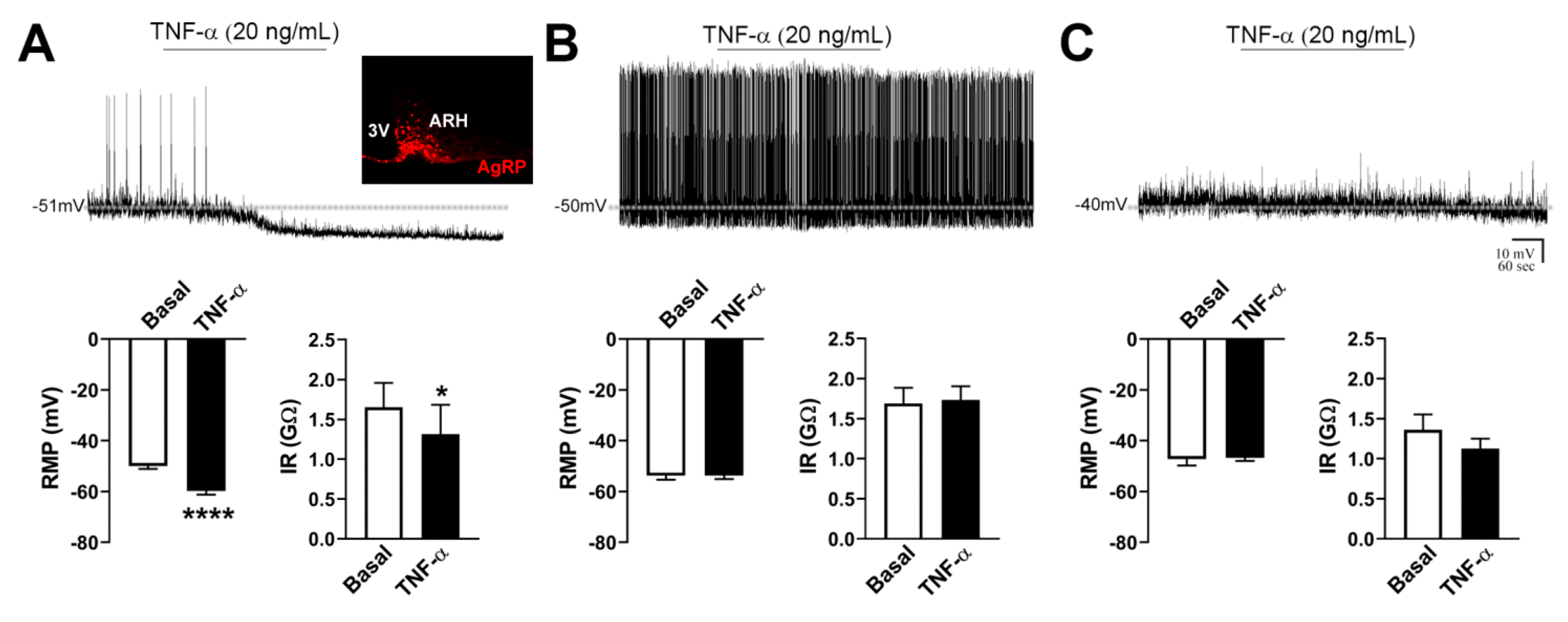
2.2. TNF-α Acutely Inhibits the Activity of AgRP-Producing Neurons
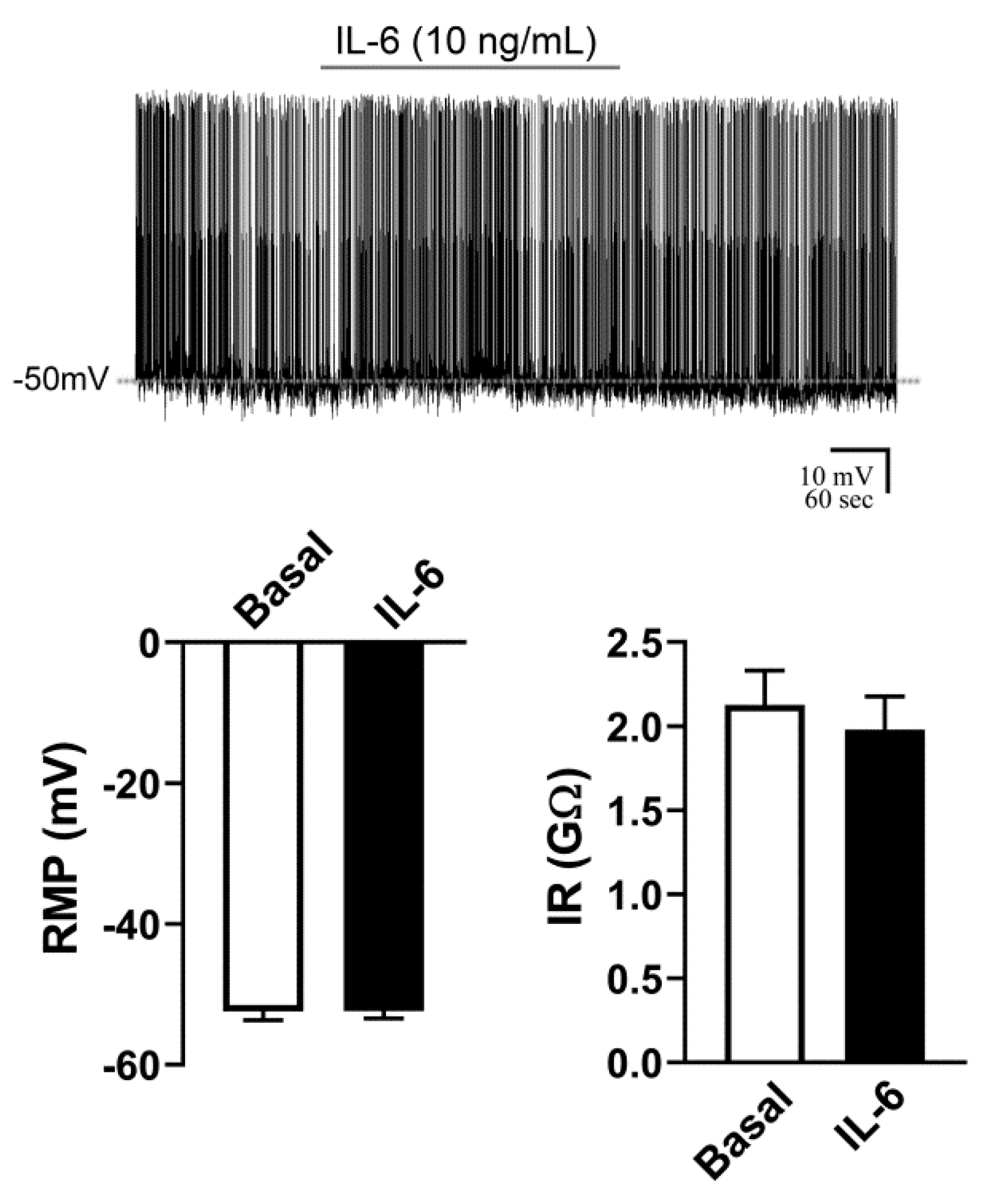
2.3. Activity of AgRP Neurons Is Not Acutely Affected by IL-6
2.4. AgRP Neurons Are Acutely Inhibited by IL-1β
2.5. Characterization of Resting Biophysical Properties of ARH POMC Neurons
2.6. TNF-α Causes Mild Effects on the Activity of POMC Neurons
2.7. IL-6 Causes No Acute Changes in the Activity of ARH POMC Neurons
3. Discussion
4. Materials and Methods
4.1. Mice
4.2. Brain Histology
4.3. Electrophysiology
4.4. Data Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Elmquist, J.K.; Elias, C.F.; Saper, C.B. From lesions to leptin: Hypothalamic control of food intake and body weight. Neuron 1999, 22, 221–232. [Google Scholar] [CrossRef] [Green Version]
- Morgane, P.J. Distinct “feeding” and “hunger motivating” systems in the lateral hypothalamus of the rat. Science 1961, 133, 887–888. [Google Scholar] [CrossRef] [PubMed]
- Brobeck, J.R. Mechanisms of the development of obesity in animals with hypothalamic lesions. Physiol. Rev. 1946, 25, 541–559. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Lobo, A.M.; Donato, J., Jr. The role of leptin in health and disease. Temperature 2017, 4, 258–291. [Google Scholar] [CrossRef] [Green Version]
- Schwartz, M.W.; Woods, S.C.; Porte, D., Jr.; Seeley, R.J.; Baskin, D.G. Central nervous system control of food intake. Nature 2000, 404, 661–671. [Google Scholar] [CrossRef]
- Andermann, M.L.; Lowell, B.B. Toward a wiring diagram understanding of appetite control. Neuron 2017, 95, 757–778. [Google Scholar] [CrossRef]
- Campbell, J.N.; Macosko, E.Z.; Fenselau, H.; Pers, T.H.; Lyubetskaya, A.; Tenen, D.; Goldman, M.; Verstegen, A.M.; Resch, J.M.; McCarroll, S.A.; et al. A molecular census of arcuate hypothalamus and median eminence cell types. Nat. Neurosci. 2017, 20, 484–496. [Google Scholar] [CrossRef] [Green Version]
- Hahn, T.M.; Breininger, J.F.; Baskin, D.G.; Schwartz, M.W. Coexpression of agrp and npy in fasting-activated hypothalamic neurons. Nat. Neurosci. 1998, 1, 271–272. [Google Scholar] [CrossRef]
- Krashes, M.J.; Koda, S.; Ye, C.; Rogan, S.C.; Adams, A.C.; Cusher, D.S.; Maratos-Flier, E.; Roth, B.L.; Lowell, B.B. Rapid, reversible activation of agrp neurons drives feeding behavior in mice. J. Clin. Investig. 2011, 121, 1424–1428. [Google Scholar] [CrossRef] [Green Version]
- Aponte, Y.; Atasoy, D.; Sternson, S.M. Agrp neurons are sufficient to orchestrate feeding behavior rapidly and without training. Nat. Neurosci. 2011, 14, 351–355. [Google Scholar] [CrossRef]
- Cowley, M.A.; Smart, J.L.; Rubinstein, M.; Cerdan, M.G.; Diano, S.; Horvath, T.L.; Cone, R.D.; Low, M.J. Leptin activates anorexigenic pomc neurons through a neural network in the arcuate nucleus. Nature 2001, 411, 480–484. [Google Scholar] [CrossRef] [PubMed]
- Quaresma, P.G.F.; Teixeira, P.D.S.; Furigo, I.C.; Wasinski, F.; Couto, G.C.; Frazao, R.; List, E.O.; Kopchick, J.J.; Donato, J., Jr. Growth hormone/stat5 signaling in proopiomelanocortin neurons regulates glucoprivic hyperphagia. Mol. Cell. Endocrinol. 2019, 498, 110574. [Google Scholar] [CrossRef] [PubMed]
- Krude, H.; Biebermann, H.; Luck, W.; Horn, R.; Brabant, G.; Gruters, A. Severe early-onset obesity, adrenal insufficiency and red hair pigmentation caused by pomc mutations in humans. Nat. Genet. 1998, 19, 155–157. [Google Scholar] [CrossRef] [PubMed]
- Ollmann, M.M.; Wilson, B.D.; Yang, Y.K.; Kerns, J.A.; Chen, Y.; Gantz, I.; Barsh, G.S. Antagonism of central melanocortin receptors in vitro and in vivo by agouti-related protein. Science 1997, 278, 135–138. [Google Scholar] [CrossRef] [PubMed]
- Schaeffer, M.; Langlet, F.; Lafont, C.; Molino, F.; Hodson, D.J.; Roux, T.; Lamarque, L.; Verdie, P.; Bourrier, E.; Dehouck, B.; et al. Rapid sensing of circulating ghrelin by hypothalamic appetite-modifying neurons. Proc. Natl. Acad. Sci. USA 2013, 110, 1512–1517. [Google Scholar] [CrossRef] [Green Version]
- Holzwarth-McBride, M.A.; Hurst, E.M.; Knigge, K.M. Monosodium glutamate induced lesions of the arcuate nucleus. I. Endocrine deficiency and ultrastructure of the median eminence. Anat. Rec. 1976, 186, 185–205. [Google Scholar] [CrossRef]
- Hampl, R.; Bicikova, M.; Sosvorova, L. Hormones and the blood-brain barrier. Horm. Mol. Biol. Clin. Investig. 2015, 21, 159–164. [Google Scholar] [CrossRef]
- Olofsson, L.E.; Unger, E.K.; Cheung, C.C.; Xu, A.W. Modulation of agrp-neuronal function by socs3 as an initiating event in diet-induced hypothalamic leptin resistance. Proc. Natl. Acad. Sci. USA 2013, 110, E697–E706. [Google Scholar] [CrossRef] [Green Version]
- Velloso, L.A.; Folli, F.; Saad, M.J. Tlr4 at the crossroads of nutrients, gut microbiota, and metabolic inflammation. Endocr. Rev. 2015, 36, 245–271. [Google Scholar] [CrossRef] [Green Version]
- Thaler, J.P.; Yi, C.-X.; Schur, E.A.; Guyenet, S.J.; Hwang, B.H.; Dietrich, M.O.; Zhao, X.; Sarruf, D.A.; Izgur, V.; Maravilla, K.R.; et al. Obesity is associated with hypothalamic injury in rodents and humans. J. Clin. Investig. 2012, 122, 153–162. [Google Scholar] [CrossRef] [Green Version]
- Souza, G.F.; Solon, C.; Nascimento, L.F.; De-Lima-Junior, J.C.; Nogueira, G.; Moura, R.; Rocha, G.Z.; Fioravante, M.; Bobbo, V.; Morari, J.; et al. Defective regulation of pomc precedes hypothalamic inflammation in diet-induced obesity. Sci. Rep. 2016, 6, 29290. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Souza, C.T.; Araujo, E.P.; Bordin, S.; Ashimine, R.; Zollner, R.L.; Boschero, A.C.; Saad, M.J.; Velloso, L.A. Consumption of a fat-rich diet activates a proinflammatory response and induces insulin resistance in the hypothalamus. Endocrinology 2005, 146, 4192–4199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Milanski, M.; Degasperi, G.; Coope, A.; Morari, J.; Denis, R.; Cintra, D.E.; Tsukumo, D.M.; Anhe, G.; Amaral, M.E.; Takahashi, H.K.; et al. Saturated fatty acids produce an inflammatory response predominantly through the activation of tlr4 signaling in hypothalamus: Implications for the pathogenesis of obesity. J. Neurosci. 2009, 29, 359–370. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.M.; Olefsky, J.M. The origins and drivers of insulin resistance. Cell 2013, 152, 673–684. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thaler, J.P.; Choi, S.J.; Schwartz, M.W.; Wisse, B.E. Hypothalamic inflammation and energy homeostasis: Resolving the paradox. Front. Neuroendocrinol. 2010, 31, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Borges, B.C.; Garcia-Galiano, D.; Rorato, R.; Elias, L.L.; Elias, C.F. Pi3k p110beta subunit in leptin receptor expressing cells is required for the acute hypophagia induced by endotoxemia. Mol. Metab. 2016, 5, 379–391. [Google Scholar] [CrossRef]
- Liu, Y.; Huang, Y.; Liu, T.; Wu, H.; Cui, H.; Gautron, L. Lipopolysacharide rapidly and completely suppresses agrp neuron-mediated food intake in male mice. Endocrinology 2016, 157, 2380–2392. [Google Scholar] [CrossRef] [Green Version]
- Scarlett, J.M.; Jobst, E.E.; Enriori, P.J.; Bowe, D.D.; Batra, A.K.; Grant, W.F.; Cowley, M.A.; Marks, D.L. Regulation of central melanocortin signaling by interleukin-1 beta. Endocrinology 2007, 148, 4217–4225. [Google Scholar] [CrossRef] [Green Version]
- Teixeira, P.D.S.; Wasinski, F.; Lima, L.B.; Frazao, R.; Bittencourt, J.C.; Donato, J., Jr. Regulation and neurochemical identity of melanin-concentrating hormone neurons in the preoptic area of lactating mice. J. Neuroendocrinol. 2020, 32, e12818. [Google Scholar] [CrossRef]
- Harden, L.M.; du Plessis, I.; Poole, S.; Laburn, H.P. Interleukin (il)-6 and il-1 beta act synergistically within the brain to induce sickness behavior and fever in rats. Brain Behav. Immun. 2008, 22, 838–849. [Google Scholar] [CrossRef]
- de Araujo, T.M.; Razolli, D.S.; Correa-da-Silva, F.; de Lima-Junior, J.C.; Gaspar, R.S.; Sidarta-Oliveira, D.; Victorio, S.C.; Donato, J., Jr.; Kim, Y.B.; Velloso, L.A. The partial inhibition of hypothalamic irx3 exacerbates obesity. EBioMedicine 2019, 39, 448–460. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huo, L.; Grill, H.J.; Bjorbaek, C. Divergent regulation of proopiomelanocortin neurons by leptin in the nucleus of the solitary tract and in the arcuate hypothalamic nucleus. Diabetes 2006, 55, 567–573. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Padilla, S.L.; Carmody, J.S.; Zeltser, L.M. Pomc-expressing progenitors give rise to antagonistic neuronal populations in hypothalamic feeding circuits. Nat. Med. 2010, 16, 403–405. [Google Scholar] [CrossRef] [PubMed]
- Furigo, I.C.; Teixeira, P.D.S.; de Souza, G.O.; Couto, G.C.L.; Romero, G.G.; Perello, M.; Frazao, R.; Elias, L.L.; Metzger, M.; List, E.O.; et al. Growth hormone regulates neuroendocrine responses to weight loss via agrp neurons. Nat. Commun. 2019, 10, 662. [Google Scholar] [CrossRef]
- Teixeira, P.D.S.; Couto, G.C.; Furigo, I.C.; List, E.O.; Kopchick, J.J.; Donato, J., Jr. Central growth hormone action regulates metabolism during pregnancy. Am. J. Physiol. Endocrinol. Metab. 2019, 317, E925–E940. [Google Scholar] [CrossRef]
- Buonfiglio, D.C.; Ramos-Lobo, A.M.; Silveira, M.A.; Furigo, I.C.; Hennighausen, L.; Frazao, R.; Donato, J., Jr. Neuronal stat5 signaling is required for maintaining lactation but not for postpartum maternal behaviors in mice. Horm. Behav. 2015, 71, 60–68. [Google Scholar] [CrossRef]
- Silveira, M.A.; Furigo, I.C.; Zampieri, T.T.; Bohlen, T.M.; de Paula, D.G.; Franci, C.R.; Donato, J., Jr.; Frazao, R. Stat5 signaling in kisspeptin cells regulates the timing of puberty. Mol. Cell. Endocrinol. 2017, 448, 55–65. [Google Scholar] [CrossRef]
- Silveira, M.A.; Zampieri, T.T.; Furigo, I.C.; Abdulkader, F.; Donato, J., Jr.; Frazao, R. Acute effects of somatomammotropin hormones on neuronal components of the hypothalamic-pituitary-gonadal axis. Brain Res. 2019, 1714, 210–217. [Google Scholar] [CrossRef]
- Katafuchi, T.; Motomura, K.; Baba, S.; Ota, K.; Hori, T. Differential effects of tumor necrosis factor-alpha and -beta on rat ventromedial hypothalamic neurons in vitro. Am. J. Physiol. 1997, 272, R1966–R1971. [Google Scholar] [CrossRef]
- Trujillo, M.E.; Sullivan, S.; Harten, I.; Schneider, S.H.; Greenberg, A.S.; Fried, S.K. Interleukin-6 regulates human adipose tissue lipid metabolism and leptin production in vitro. J. Clin. Endocrinol. Metab. 2004, 89, 5577–5582. [Google Scholar] [CrossRef] [Green Version]
- Hao, L.; Sheng, Z.; Potian, J.; Deak, A.; Rohowsky-Kochan, C.; Routh, V.H. Lipopolysaccharide (lps) and tumor necrosis factor alpha (tnfalpha) blunt the response of neuropeptide y/agouti-related peptide (npy/agrp) glucose inhibited (gi) neurons to decreased glucose. Brain Res. 2016, 1648, 181–192. [Google Scholar] [CrossRef] [Green Version]
- Yi, C.X.; Walter, M.; Gao, Y.; Pitra, S.; Legutko, B.; Kalin, S.; Layritz, C.; Garcia-Caceres, C.; Bielohuby, M.; Bidlingmaier, M.; et al. Tnfalpha drives mitochondrial stress in pomc neurons in obesity. Nat. Commun. 2017, 8, 15143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Plata-Salaman, C.R.; Oomura, Y.; Kai, Y. Tumor necrosis factor and interleukin-1 beta: Suppression of food intake by direct action in the central nervous system. Brain Res. 1988, 448, 106–114. [Google Scholar] [CrossRef]
- Bodnar, R.J.; Pasternak, G.W.; Mann, P.E.; Paul, D.; Warren, R.; Donner, D.B. Mediation of anorexia by human recombinant tumor necrosis factor through a peripheral action in the rat. Cancer Res. 1989, 49, 6280–6284. [Google Scholar] [PubMed]
- Kapas, L.; Hong, L.; Cady, A.B.; Opp, M.R.; Postlethwaite, A.E.; Seyer, J.M.; Krueger, J.M. Somnogenic, pyrogenic, and anorectic activities of tumor necrosis factor-alpha and tnf-alpha fragments. Am. J. Physiol. 1992, 263, R708–R715. [Google Scholar] [CrossRef] [PubMed]
- Plata-Salaman, C.R.; Vasselli, J.R.; Sonti, G. Differential responsiveness of obese (fa/fa) and lean (fa/fa) zucker rats to cytokine-induced anorexia. Obes. Res. 1997, 5, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Schele, E.; Benrick, A.; Grahnemo, L.; Egecioglu, E.; Anesten, F.; Palsdottir, V.; Jansson, J.O. Inter-relation between interleukin (il)-1, il-6 and body fat regulating circuits of the hypothalamic arcuate nucleus. J. Neuroendocrinol. 2013, 25, 580–589. [Google Scholar] [CrossRef] [PubMed]
- Wallenius, V.; Wallenius, K.; Ahren, B.; Rudling, M.; Carlsten, H.; Dickson, S.L.; Ohlsson, C.; Jansson, J.O. Interleukin-6-deficient mice develop mature-onset obesity. Nat. Med. 2002, 8, 75–79. [Google Scholar] [CrossRef]
- Tachibana, T.; Kodama, T.; Yamane, S.; Makino, R.; Khan, S.I.; Cline, M.A. Possible role of central interleukins on the anorexigenic effect of lipopolysaccharide in chicks. Br. Poult. Sci. 2017, 58, 305–311. [Google Scholar] [CrossRef]
- Harden, L.M.; du Plessis, I.; Roth, J.; Loram, L.C.; Poole, S.; Laburn, H.P. Differences in the relative involvement of peripherally released interleukin (il)-6, brain il-1beta and prostanoids in mediating lipopolysaccharide-induced fever and sickness behavior. Psychoneuroendocrinology 2011, 36, 608–622. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chaves, F.M.; Mansano, N.S.; Frazão, R.; Donato, J., Jr. Tumor Necrosis Factor α and Interleukin-1β Acutely Inhibit AgRP Neurons in the Arcuate Nucleus of the Hypothalamus. Int. J. Mol. Sci. 2020, 21, 8928. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21238928
Chaves FM, Mansano NS, Frazão R, Donato J Jr. Tumor Necrosis Factor α and Interleukin-1β Acutely Inhibit AgRP Neurons in the Arcuate Nucleus of the Hypothalamus. International Journal of Molecular Sciences. 2020; 21(23):8928. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21238928
Chicago/Turabian StyleChaves, Fernanda M., Naira S. Mansano, Renata Frazão, and Jose Donato, Jr. 2020. "Tumor Necrosis Factor α and Interleukin-1β Acutely Inhibit AgRP Neurons in the Arcuate Nucleus of the Hypothalamus" International Journal of Molecular Sciences 21, no. 23: 8928. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21238928




