Identification and Characterization of the Lactating Mouse Mammary Gland Citrullinome
Abstract
:1. Introduction
2. Results
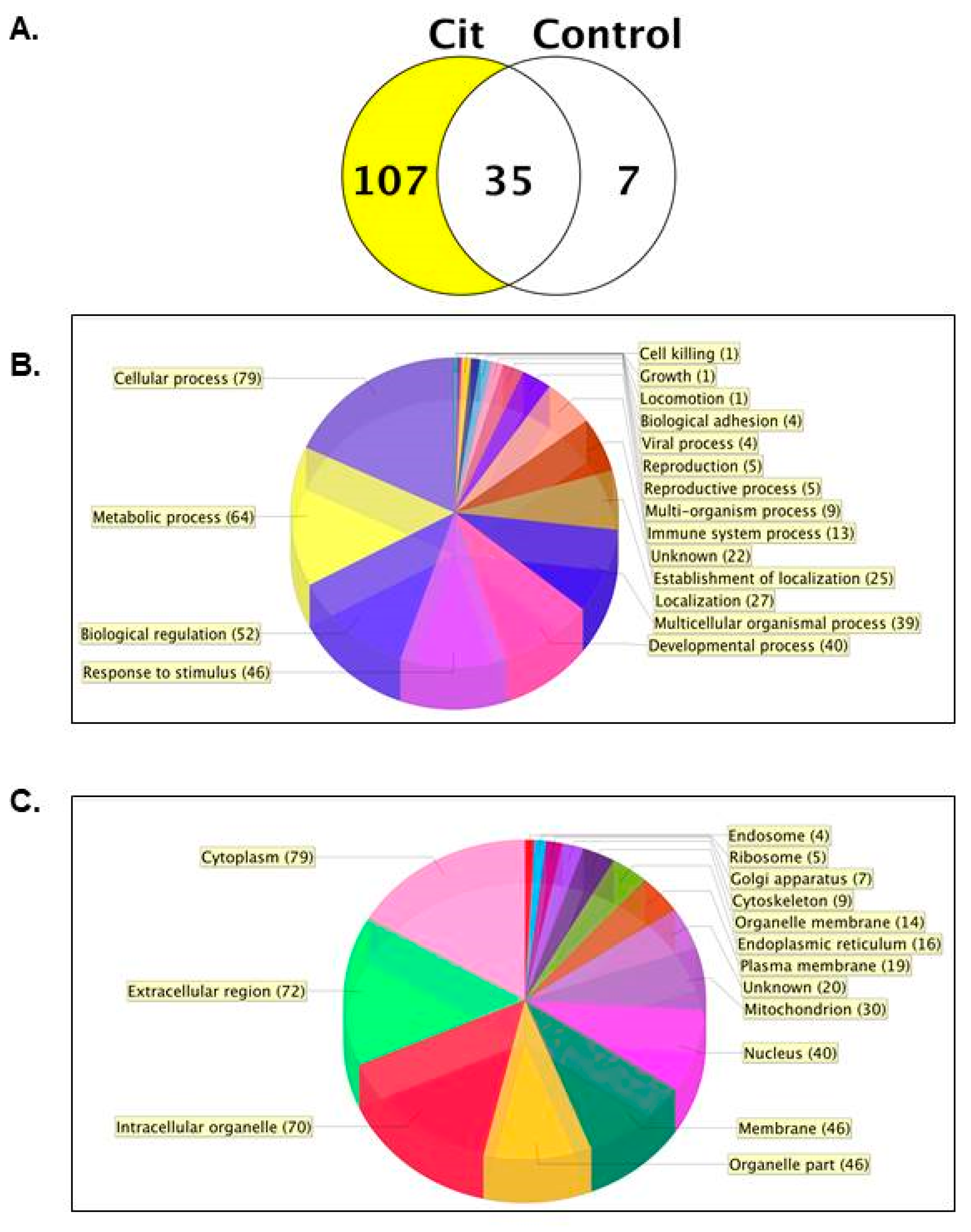
2.1. 107 Citrullinated Proteins Are Present in the Lactation Day 9 (L9) Mouse Mammary Gland
2.2. Prolactin Induces Histone H2A Citrullination in Mouse Mammary Epithelial CID-9 Cells
2.3. Prolactin Stimulates Citrullination to Increase β-Casein and Butyrophilin mRNA Expression in CID-9 Cells
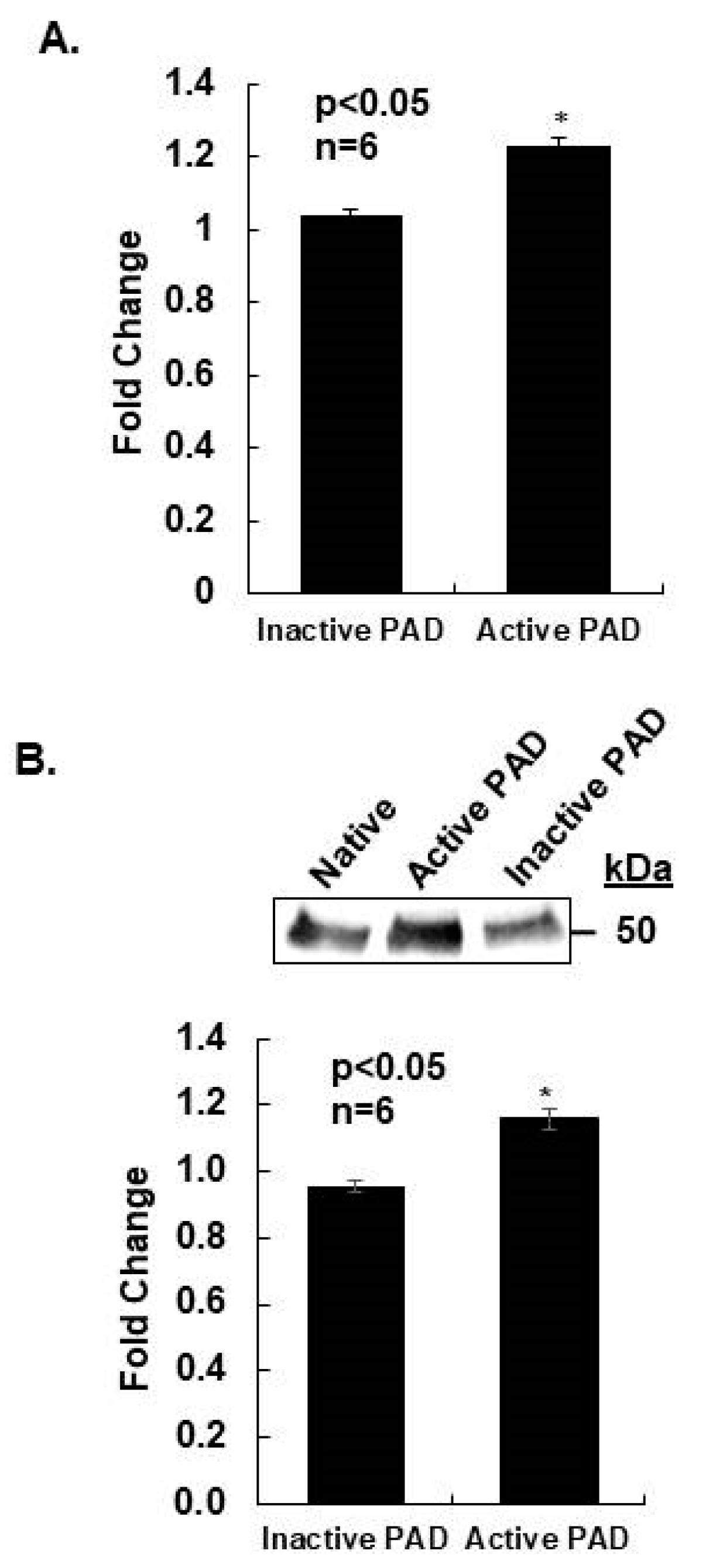
2.4. Prolactin Stimulates Citrullination of α-Tubulin in CID-9 Cells
2.5. Citrullination Alters Tubulin Polymerization Rates
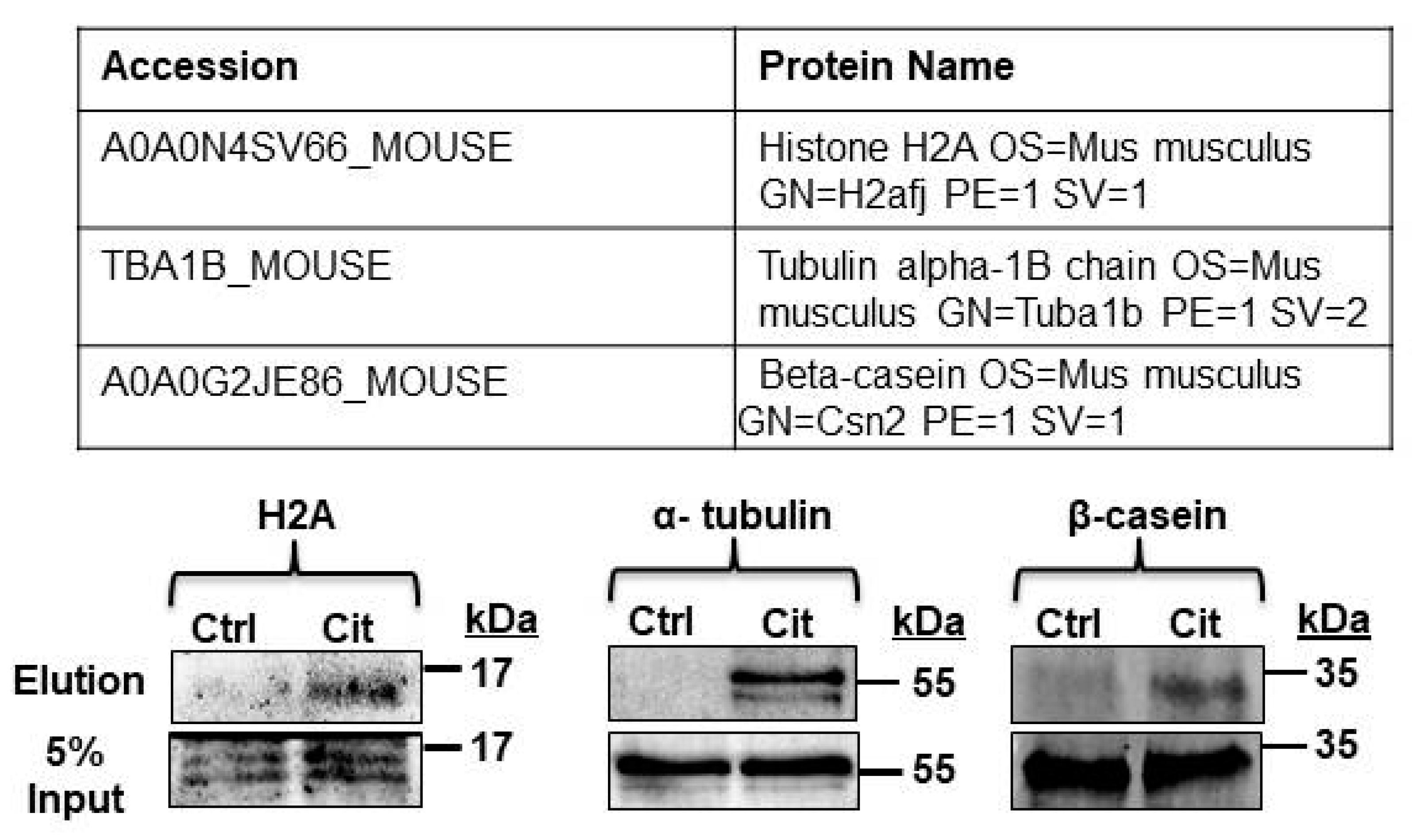
2.6. β-Casein Is Citrullinated in Mouse and Human Milk
3. Discussion
4. Materials and Methods
4.1. Cell Culture and PAD Inhibitor Treatments
4.2. Mouse Mammary Tissue and Human Breast Milk
4.3. Biotin-Phenylglyoxal Enrichment of Citrullinated Proteins
4.4. LC-MS/MS Proteomics
4.4.1. Sample Preparation: In-Gel Trypsin Digestion
4.4.2. Mass Spectrometry Analysis
4.4.3. Data Analysis
4.4.4. Instrument Suitability
4.5. Western Blotting
4.6. qPCR
4.7. In Vitro Tubulin Citrullination and Polymerization
4.7.1. Citrullinated Tubulin Western Blot
4.7.2. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| JAK | Janus kinase |
| STAT | Signal transducer and activator of transcription |
| GAS | Interferon-γ-activated sequence |
| Btn1a1 | Butyrophilin |
| Csn2 | β-casein |
| CLD | Cytoplasmic lipid droplet |
| LC-MS/MS | Liquid chromatography with tandem mass spectrometry |
| Biotin-PG | Biotin-phenylglyoxal |
| PAD/PADI | Peptidylarginine deiminase |
| BB-ClA | Biphenyl-benzimidazole-Cl-amidine |
| PRL | Prolactin |
| RA | Rheumatoid arthritis |
| L9 | Lactation day 9 |
| PTM | Post-translational modification |
References
- Cherrington, B.D.; Morency, E.; Struble, A.M.; Coonrod, S.A.; Wakshlag, J.J. Potential role for peptidylarginine deiminase 2 (pad2) in citrullination of canine mammary epithelial cell histones. PLoS ONE 2010, 5, e11768. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, G.; Hayward, I.N.; Jenkins, B.R.; Rothfuss, H.M.; Young, C.H.; Nevalainen, M.T.; Muth, A.; Thompson, P.R.; Navratil, A.M.; Cherrington, B.D. Peptidylarginine deiminase 3 (pad3) is upregulated by prolactin stimulation of cid-9 cells and expressed in the lactating mouse mammary gland. PLoS ONE 2016, 11, e0147503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cherrington, B.D.; Mohanan, S.; Diep, A.N.; Fleiss, R.; Sudilovsky, D.; Anguish, L.J.; Coonrod, S.A.; Wakshlag, J.J. Comparative analysis of peptidylarginine deiminase-2 expression in canine, feline and human mammary tumours. J. Comp. Pathol. 2012, 147, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Darrah, E.; Rosen, A.; Giles, J.T.; Andrade, F. Peptidylarginine deiminase 2, 3 and 4 have distinct specificities against cellular substrates: Novel insights into autoantigen selection in rheumatoid arthritis. Ann. Rheum. Dis. 2012, 71, 92–98. [Google Scholar] [CrossRef] [Green Version]
- Witalison, E.E.; Thompson, P.R.; Hofseth, L.J. Protein arginine deiminases and associated citrullination: Physiological functions and diseases associated with dysregulation. Curr. Drug Targets 2015, 16, 700–710. [Google Scholar] [CrossRef]
- Badillo-Soto, M.A.; Rodriguez-Rodriguez, M.; Perez-Perez, M.E.; Daza-Benitez, L.; Bollain, Y.G.J.J.; Carrillo-Jimenez, M.A.; Avalos-Diaz, E.; Herrera-Esparza, R. Potential protein targets of the peptidylarginine deiminase 2 and peptidylarginine deiminase 4 enzymes in rheumatoid synovial tissue and its possible meaning. Eur. J. Rheumatol. 2016, 3, 44–49. [Google Scholar] [CrossRef]
- Tilvawala, R.; Nguyen, S.H.; Maurais, A.J.; Nemmara, V.V.; Nagar, M.; Salinger, A.J.; Nagpal, S.; Weerapana, E.; Thompson, P.R. The rheumatoid arthritis-associated citrullinome. Cell Chem. Biol. 2018, 25, 691–704 e696. [Google Scholar] [CrossRef] [Green Version]
- Khan, S.A.; Edwards, B.S.; Muth, A.; Thompson, P.R.; Cherrington, B.D.; Navratil, A.M. Gnrh stimulates peptidylarginine deiminase catalyzed histone citrullination in gonadotrope cells. Mol. Endocrinol. 2016, 30, 1081–1091. [Google Scholar] [CrossRef] [Green Version]
- Young, C.H.; Rothfuss, H.M.; Gard, P.F.; Muth, A.; Thompson, P.R.; Ashley, R.L.; Cherrington, B.D. Citrullination regulates the expression of insulin-like growth factor-binding protein 1 (igfbp1) in ovine uterine luminal epithelial cells. Reproduction 2017, 153, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Jiang, Z.; Cui, Y.; Wang, L.; Zhao, Y.; Yan, S.; Chang, X. Investigating citrullinated proteins in tumour cell lines. World J. Surg Oncol. 2013, 11, 260. [Google Scholar] [CrossRef] [Green Version]
- van Beers, J.J.; Schwarte, C.M.; Stammen-Vogelzangs, J.; Oosterink, E.; Bozic, B.; Pruijn, G.J. The rheumatoid arthritis synovial fluid citrullinome reveals novel citrullinated epitopes in apolipoprotein e, myeloid nuclear differentiation antigen, and beta-actin. Arthritis Rheum. 2013, 65, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Senshu, T.; Kan, S.; Ogawa, H.; Manabe, M.; Asaga, H. Preferential deimination of keratin k1 and filaggrin during the terminal differentiation of human epidermis. Biochem. Biophys. Res. Commun. 1996, 225, 712–719. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Chen, F.F.; Gao, W.B.; Wang, H.Y.; Zhao, N.W.; Xu, M.; Gao, D.Y.; Yu, W.; Yan, X.L.; Zhao, J.N.; et al. Identification of citrullinated peptides in the synovial fluid of patients with rheumatoid arthritis using lc-maldi-tof/tof. Clin. Rheumatol. 2016, 35, 2185–2194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, X.; Robinson, G.W.; Wagner, K.U.; Garrett, L.; Wynshaw-Boris, A.; Hennighausen, L. Stat5a is mandatory for adult mammary gland development and lactogenesis. Genes Dev. 1997, 11, 179–186. [Google Scholar] [CrossRef] [Green Version]
- Radhakrishnan, A.; Raju, R.; Tuladhar, N.; Subbannayya, T.; Thomas, J.K.; Goel, R.; Telikicherla, D.; Palapetta, S.M.; Rahiman, B.A.; Venkatesh, D.D.; et al. A pathway map of prolactin signaling. J. Cell Commun. Signal. 2012, 6, 169–173. [Google Scholar] [CrossRef] [Green Version]
- Ball, R.K.; Friis, R.R.; Schoenenberger, C.A.; Doppler, W.; Groner, B. Prolactin regulation of beta-casein gene expression and of a cytosolic 120-kd protein in a cloned mouse mammary epithelial cell line. EMBO J. 1988, 7, 2089–2095. [Google Scholar] [CrossRef]
- Anderson, S.M.; Rudolph, M.C.; McManaman, J.L.; Neville, M.C. Key stages in mammary gland development. Secretory activation in the mammary gland: It’s not just about milk protein synthesis! Breast Cancer Res. BCR 2007, 9, 204. [Google Scholar] [CrossRef]
- Nickerson, S.C.; Smith, J.J.; Keenan, T.W. Role of microtubules in milk secretion--action of colchicine on microtubules and exocytosis of secretory vesicles in rat mammary epithelial cells. Cell Tissue Res. 1980, 207, 361–376. [Google Scholar] [CrossRef]
- Heid, H.W.; Keenan, T.W. Intracellular origin and secretion of milk fat globules. Eur. J. Cell Biol. 2005, 84, 245–258. [Google Scholar] [CrossRef]
- McManaman, J.L.; Neville, M.C. Mammary physiology and milk secretion. Adv. Drug Deliv. Rev. 2003, 55, 629–641. [Google Scholar] [CrossRef]
- Ogg, S.L.; Weldon, A.K.; Dobbie, L.; Smith, A.J.; Mather, I.H. Expression of butyrophilin (btn1a1) in lactating mammary gland is essential for the regulated secretion of milk-lipid droplets. Proc. Natl. Acad. Sci. USA 2004, 101, 10084–10089. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guerin, M.A.; Loizzi, R.F. Tubulin content and assembly states in guinea pig mammary gland during pregnancy, lactation, and weaning. Proc. Soc. Exp. Biol. Med. Soc. Exp. Biol. Med. 1980, 165, 50–54. [Google Scholar] [CrossRef] [PubMed]
- Nickerson, S.C.; Keenan, T.W. Distribution and orientation of microtubules in milk secreting epithelial cells of rat mammary gland. Cell Tissue Res. 1979, 202, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Rennison, M.E.; Handel, S.E.; Wilde, C.J.; Burgoyne, R.D. Investigation of the role of microtubules in protein secretion from lactating mouse mammary epithelial cells. J. Cell Sci. 1992, 102 Pt 2, 239–247. [Google Scholar]
- Patton, S.; Stemberger, B.H.; Knudson, C.M. The supression of milk fat globule secretion by clochicine: An effect coupled to inhibition of exocytosis. Biochim. Biophys. Acta 1977, 499, 404–410. [Google Scholar] [CrossRef]
- Wloga, D.; Joachimiak, E.; Fabczak, H. Tubulin post-translational modifications and microtubule dynamics. Int. J. Mol. Sci. 2017, 18, 2207. [Google Scholar] [CrossRef] [Green Version]
- Chang, X.; Zhao, Y.; Wang, Y.; Chen, Y.; Yan, X. Screening citrullinated proteins in synovial tissues of rheumatoid arthritis using 2-dimensional western blotting. J. Rheumatol. 2013, 40, 219–227. [Google Scholar] [CrossRef]
- Tsai, A.C.; Pai, H.C.; Wang, C.Y.; Liou, J.P.; Teng, C.M.; Wang, J.C.; Pan, S.L. In vitro and in vivo anti-tumour effects of mpt0b014, a novel derivative aroylquinoline, and in combination with erlotinib in human non-small-cell lung cancer cells. Br. J. Pharmacol. 2014, 171, 122–133. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.H.; Liao, C.H.; Chang, Y.L.; Guh, J.H.; Pan, S.L.; Teng, C.M. Protopine, a novel microtubule-stabilizing agent, causes mitotic arrest and apoptotic cell death in human hormone-refractory prostate cancer cell lines. Cancer Lett. 2012, 315, 1–11. [Google Scholar] [CrossRef]
- Bonne, D.; Heusele, C.; Simon, C.; Pantaloni, D. 4‘,6-diamidino-2-phenylindole, a fluorescent probe for tubulin and microtubules. J. Biol. Chem. 1985, 260, 2819–2825. [Google Scholar]
- Willingham, K.; McNulty, E.; Anderson, K.; Hayes-Klug, J.; Nalls, A.; Mathiason, C. Milk collection methods for mice and reeves‘ muntjac deer. J. Vis. Exp. JoVE 2014, 89, e51007. [Google Scholar] [CrossRef] [PubMed]
- Dieterich, C.M.; Felice, J.P.; O‘Sullivan, E.; Rasmussen, K.M. Breastfeeding and health outcomes for the mother-infant dyad. Pediatr. Clin. N. Am. 2013, 60, 31–48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ip, S.; Chung, M.; Raman, G.; Chew, P.; Magula, N.; DeVine, D.; Trikalinos, T.; Lau, J. Breastfeeding and maternal and infant health outcomes in developed countries. Evid Rep. Technol Assess. (Full Rep.) 2007, 153, 1–186. [Google Scholar]
- Lewallen, D.M.; Bicker, K.L.; Subramanian, V.; Clancy, K.W.; Slade, D.J.; Martell, J.; Dreyton, C.J.; Sokolove, J.; Weerapana, E.; Thompson, P.R. Chemical proteomic platform to identify citrullinated proteins. ACS Chem. Biol. 2015, 10, 2520–2528. [Google Scholar] [CrossRef] [PubMed]
- Horibata, S.; Coonrod, S.A.; Cherrington, B.D. Role for peptidylarginine deiminase enzymes in disease and female reproduction. J. Reprod. Dev. 2012, 58, 274–282. [Google Scholar] [CrossRef] [Green Version]
- Hagiwara, T.; Hidaka, Y.; Yamada, M. Deimination of histone h2a and h4 at arginine 3 in hl-60 granulocytes. Biochemistry 2005, 44, 5827–5834. [Google Scholar] [CrossRef]
- Kabotyanski, E.B.; Huetter, M.; Xian, W.; Rijnkels, M.; Rosen, J.M. Integration of prolactin and glucocorticoid signaling at the beta-casein promoter and enhancer by ordered recruitment of specific transcription factors and chromatin modifiers. Mol. Endocrinol. 2006, 20, 2355–2368. [Google Scholar] [CrossRef]
- Rijnkels, M.; Kabotyanski, E.; Montazer-Torbati, M.B.; Hue Beauvais, C.; Vassetzky, Y.; Rosen, J.M.; Devinoy, E. The epigenetic landscape of mammary gland development and functional differentiation. J. Mammary Gland Biol. Neoplasia 2010, 15, 85–100. [Google Scholar] [CrossRef] [Green Version]
- Lemay, D.G.; Pollard, K.S.; Martin, W.F.; Freeman Zadrowski, C.; Hernandez, J.; Korf, I.; German, J.B.; Rijnkels, M. From genes to milk: Genomic organization and epigenetic regulation of the mammary transcriptome. PLoS ONE 2013, 8, e75030. [Google Scholar] [CrossRef] [Green Version]
- Davey, H.W.; Ogg, S.L.; Husaini, Y.; Snell, R.G.; Korobko, I.V.; Mather, I.H.; Wilkins, R.J. Structure and sequence of the bovine butyrophilin gene. Gene 1997, 199, 57–62. [Google Scholar] [CrossRef]
- Wloga, D.; Gaertig, J. Post-translational modifications of microtubules. J. Cell Sci. 2010, 123, 3447–3455. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holland, J.W. Post-translational modifications of caseins. In Milk Proteins: From Expression to Food; Thompson, A., Ed.; Elsevier B.V.: Amsterdam, The Netherlands, 2009; pp. 107–132. [Google Scholar]
- Schmidhauser, C.; Bissell, M.J.; Myers, C.A.; Casperson, G.F. Extracellular matrix and hormones transcriptionally regulate bovine beta-casein 5’ sequences in stably transfected mouse mammary cells. Proc. Natl. Acad. Sci. USA 1990, 87, 9118–9122. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knight, J.S.; Subramanian, V.; O’Dell, A.A.; Yalavarthi, S.; Zhao, W.; Smith, C.K.; Hodgin, J.B.; Thompson, P.R.; Kaplan, M.J. Peptidylarginine deiminase inhibition disrupts net formation and protects against kidney, skin and vascular disease in lupus-prone mrl/lpr mice. Ann. Rheum. Dis. 2015, 74, 2199–2206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dyballa, N.; Metzger, S. Fast and sensitive colloidal coomassie g-250 staining for proteins in polyacrylamide gels. J. Vis. Exp. JoVE 2009, 30, e1431. [Google Scholar] [CrossRef] [PubMed]
- Saveliev, S.V.; Woodroofe, C.C.; Sabat, G.; Adams, C.M.; Klaubert, D.; Wood, K.; Urh, M. Mass spectrometry compatible surfactant for optimized in-gel protein digestion. Anal. Chem. 2013, 85, 907–914. [Google Scholar] [CrossRef] [PubMed]
- Scopes, R.K. Measurement of protein by spectrophotometry at 205 nm. Anal. Biochem. 1974, 59, 277–282. [Google Scholar] [CrossRef]
- Keller, A.; Nesvizhskii, A.I.; Kolker, E.; Aebersold, R. Empirical statistical model to estimate the accuracy of peptide identifications made by ms/ms and database search. Anal. Chem. 2002, 74, 5383–5392. [Google Scholar] [CrossRef]
- Searle, B.C.; Turner, M.; Nesvizhskii, A.I. Improving sensitivity by probabilistically combining results from multiple ms/ms search methodologies. J. Proteome Res. 2008, 7, 245–253. [Google Scholar] [CrossRef]
- Kall, L.; Storey, J.D.; MacCoss, M.J.; Noble, W.S. Assigning significance to peptides identified by tandem mass spectrometry using decoy databases. J. Proteome Res. 2008, 7, 29–34. [Google Scholar] [CrossRef]
- Nesvizhskii, A.I.; Keller, A.; Kolker, E.; Aebersold, R. A statistical model for identifying proteins by tandem mass spectrometry. Anal. Chem. 2003, 75, 4646–4658. [Google Scholar] [CrossRef]
- Schoenmakers, T.J.; Visser, G.J.; Flik, G.; Theuvenet, A.P. Chelator: An improved method for computing metal ion concentrations in physiological solutions. Biotechniques 1992, 12, 870–874, 876–879. [Google Scholar] [PubMed]
- Musse, A.A.; Polverini, E.; Raijmakers, R.; Harauz, G. Kinetics of human peptidylarginine deiminase 2 (hpad2)--reduction of ca2+ dependence by phospholipids and assessment of proposed inhibition by paclitaxel side chains. Biochem. Cell Biol. 2008, 86, 437–447. [Google Scholar] [CrossRef] [PubMed]




© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, G.; Young, C.H.; Snow, B.; Christensen, A.O.; Demoruelle, M.K.; Nemmara, V.V.; Thompson, P.R.; Rothfuss, H.M.; Cherrington, B.D. Identification and Characterization of the Lactating Mouse Mammary Gland Citrullinome. Int. J. Mol. Sci. 2020, 21, 2634. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21072634
Li G, Young CH, Snow B, Christensen AO, Demoruelle MK, Nemmara VV, Thompson PR, Rothfuss HM, Cherrington BD. Identification and Characterization of the Lactating Mouse Mammary Gland Citrullinome. International Journal of Molecular Sciences. 2020; 21(7):2634. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21072634
Chicago/Turabian StyleLi, Guangyuan, Coleman H. Young, Bryce Snow, Amanda O. Christensen, M. Kristen Demoruelle, Venkatesh V. Nemmara, Paul R. Thompson, Heather M. Rothfuss, and Brian D. Cherrington. 2020. "Identification and Characterization of the Lactating Mouse Mammary Gland Citrullinome" International Journal of Molecular Sciences 21, no. 7: 2634. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21072634




