A Neuroprotective Bovine Colostrum Attenuates Apoptosis in Dexamethasone-Treated MC3T3-E1 Osteoblastic Cells
Abstract
:1. Introduction
2. Results
2.1. Cell Model of Neurotoxicity by SH-SY5Y Cell Exposure to Dexamethasone (DEX)
2.2. Neuroprotective Effect of Colostrum on DEX-Induced SH-SY5Y Cell Damage
2.3. ROS Scavenger Activity of Bovine Colostrum in SH-SY5Y Cells
2.4. DEX-Induced Caspase-3 Activation SH-5Y5Y Is Attenuated by Colostrum Treatment
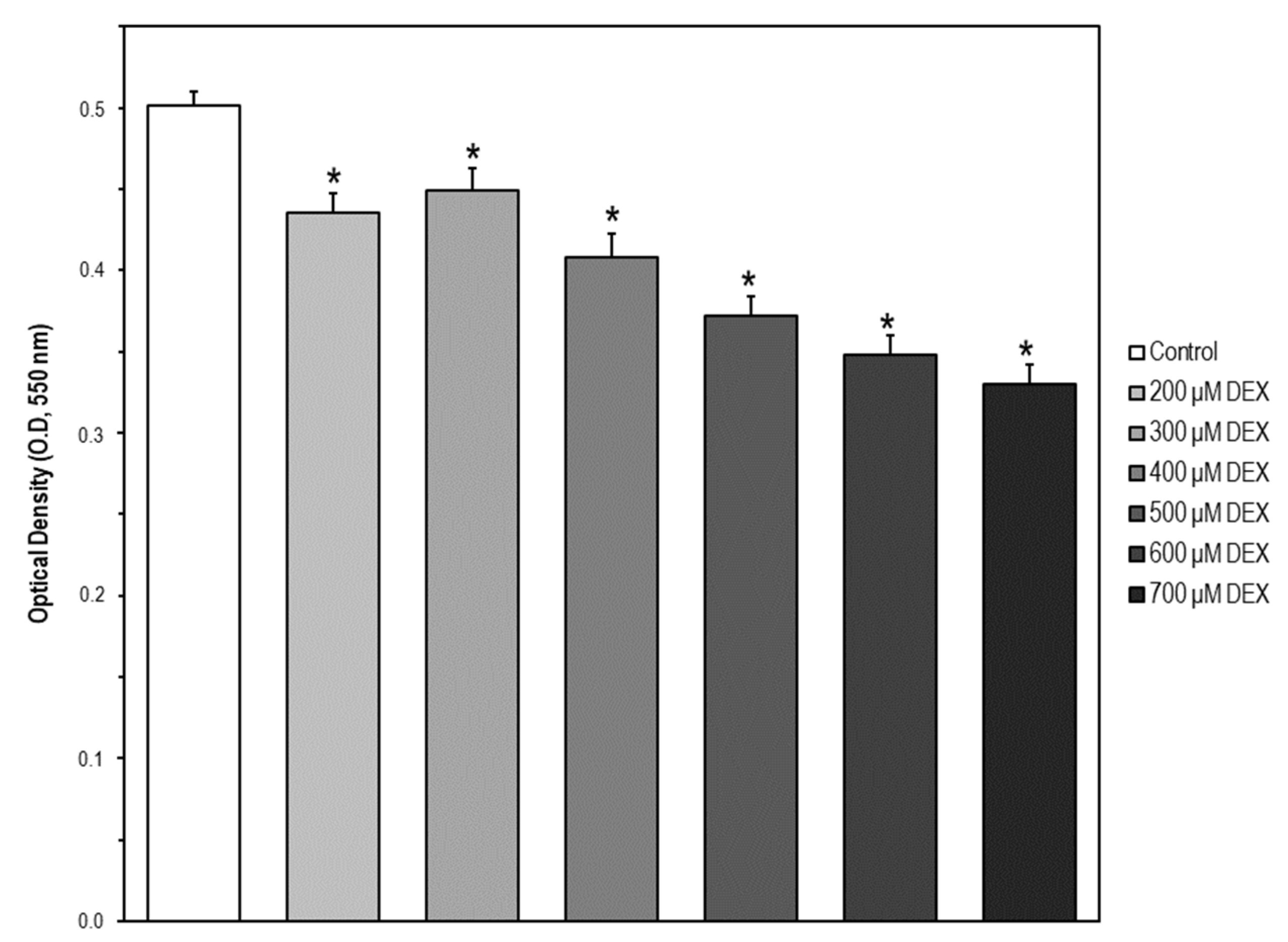
2.5. Cell Model of Apoptosis by MC3T3-E1 Cell Exposure to Dexamethasone (DEX)
2.6. Suitable Concentration Range of Colostrum Which Is Non-Cytotoxic for MC3T3-E1 Cells
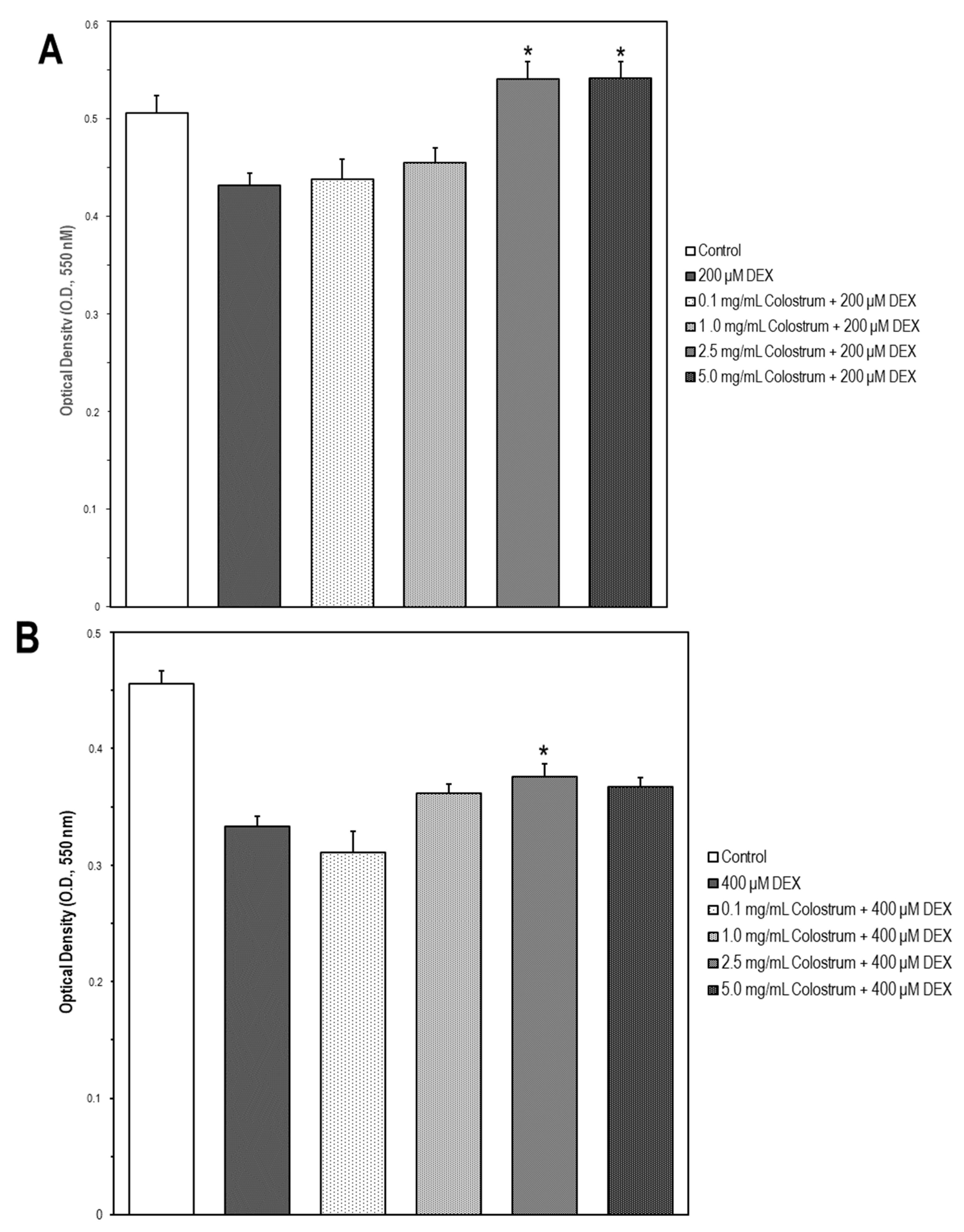
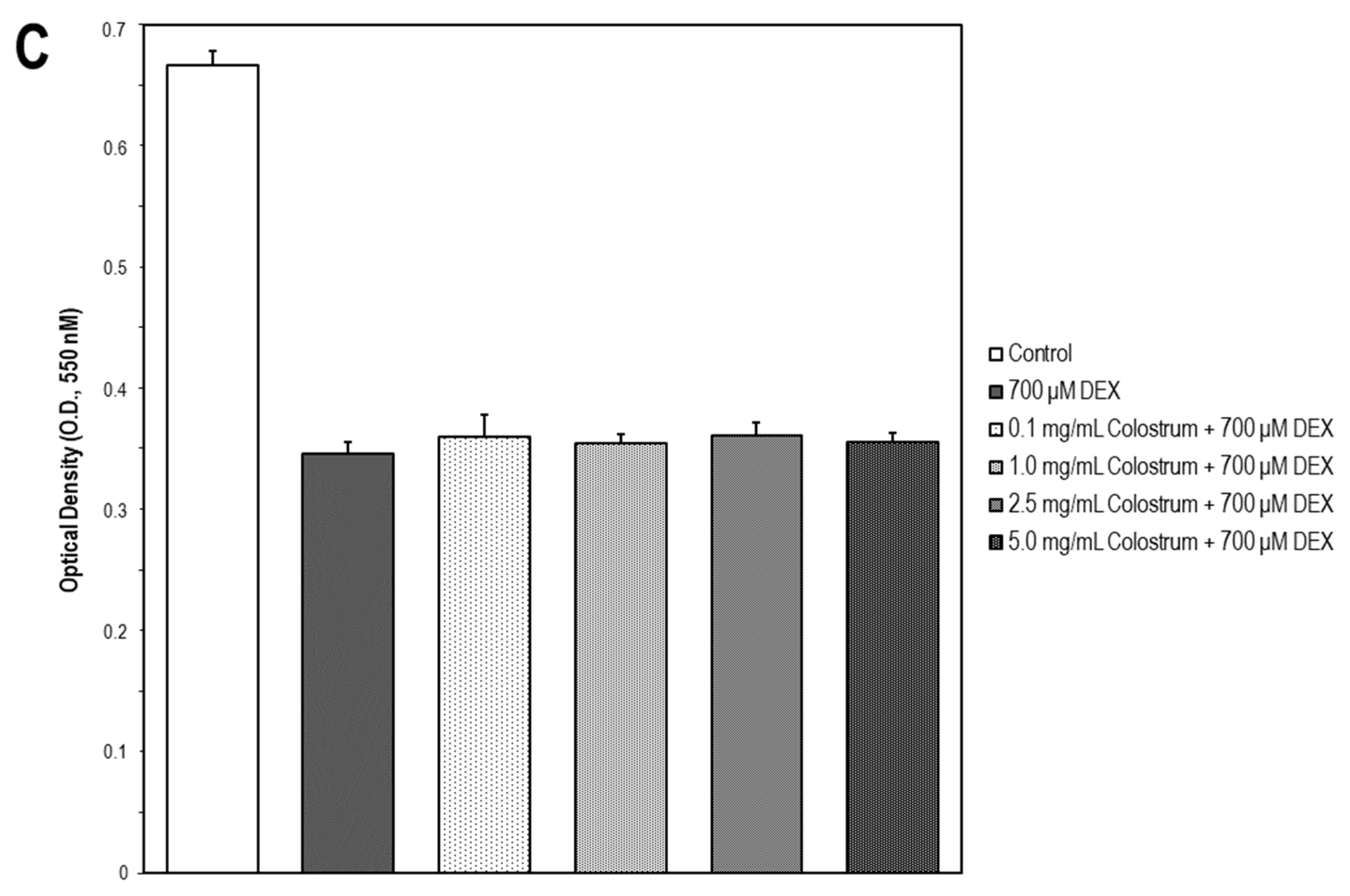
2.7. Colostrum Rescues MC3T3-E1 Cells from DEX Cytotoxicity
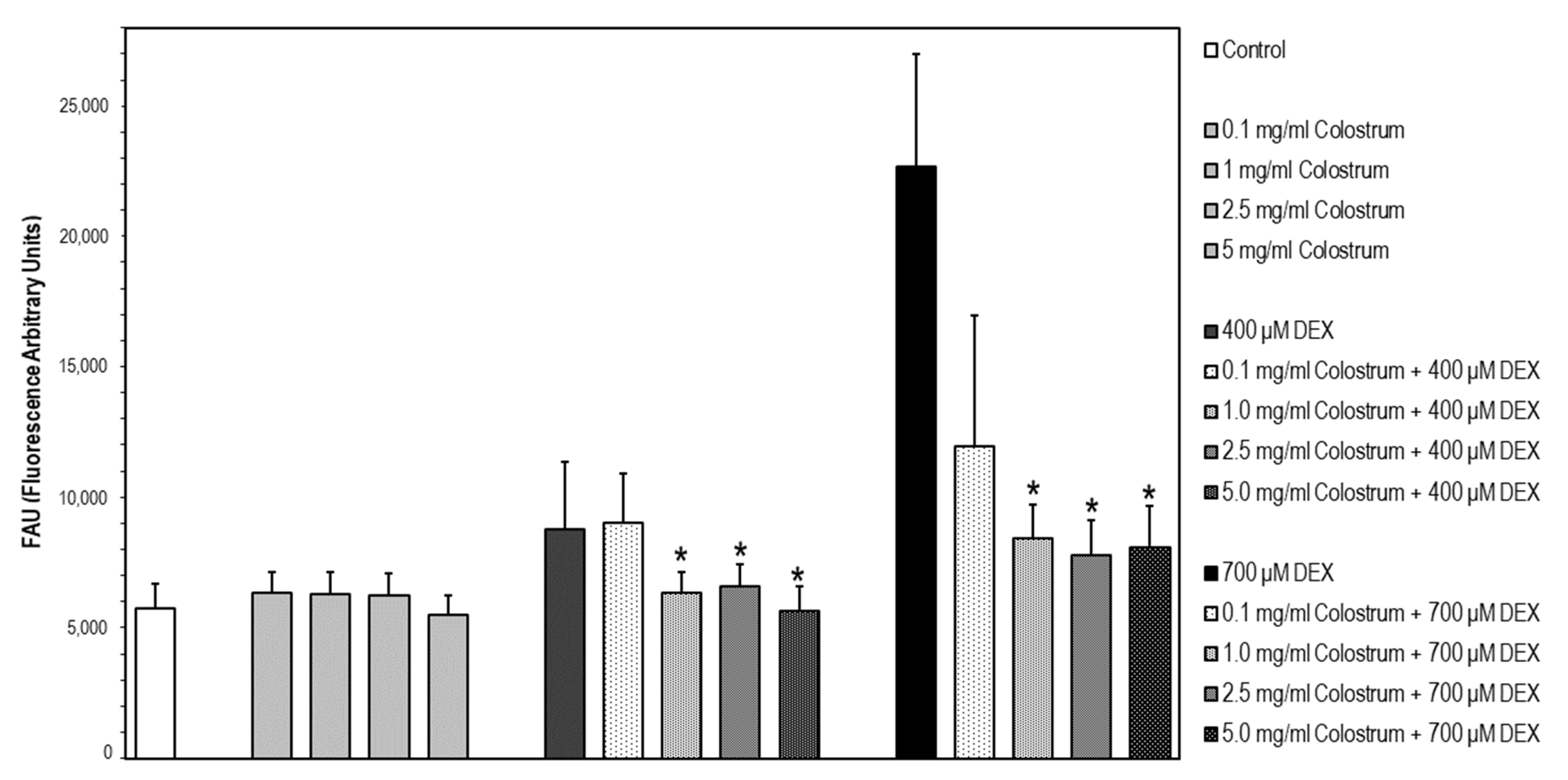
2.8. DEX-Induced Caspase-3 Activation Is Attenuated by Colostrum Treatment
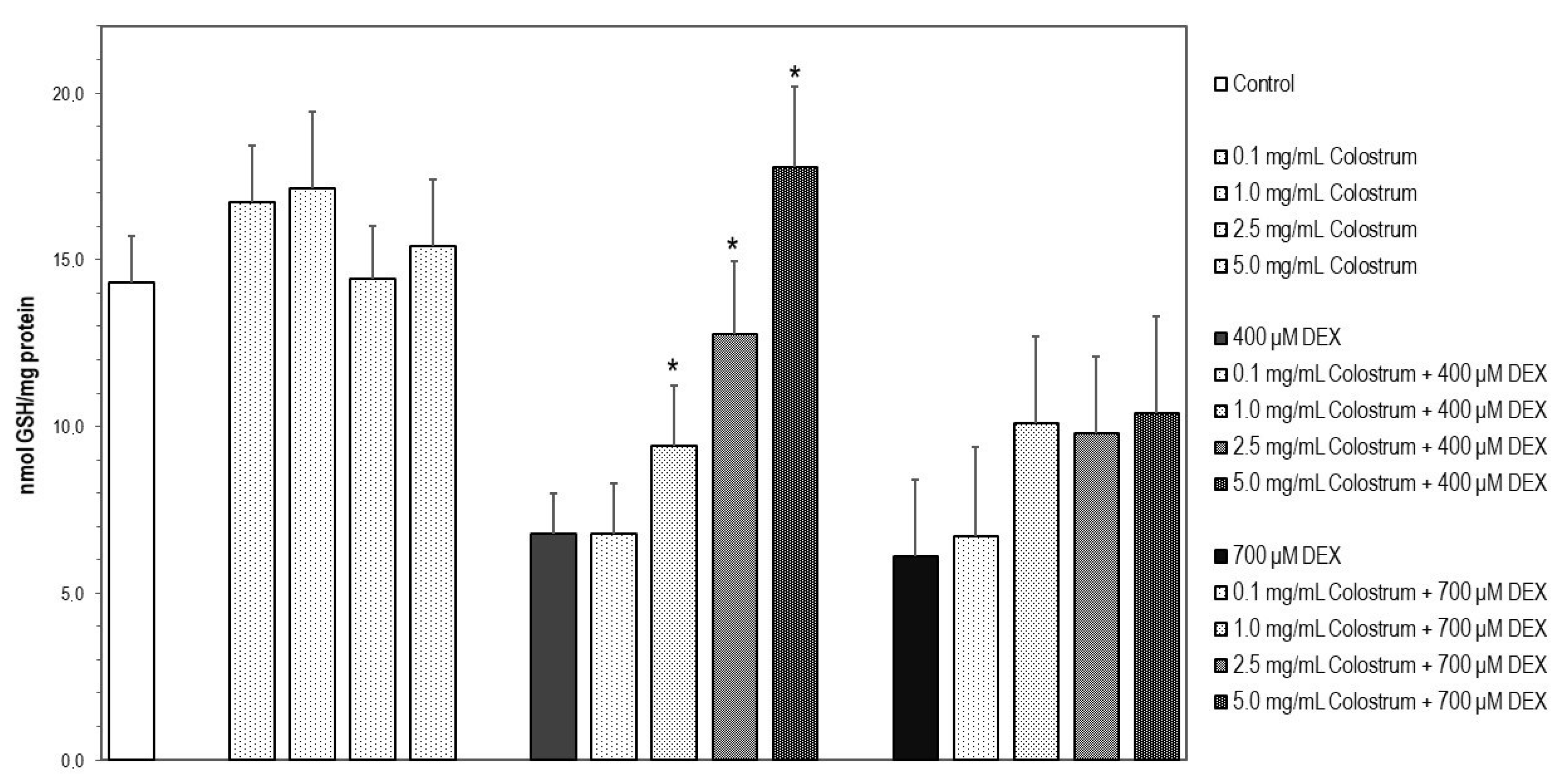
2.9. Colostrum Treatment Restores GSH Levels in DEX-Treated MC3T3-E1 Cells
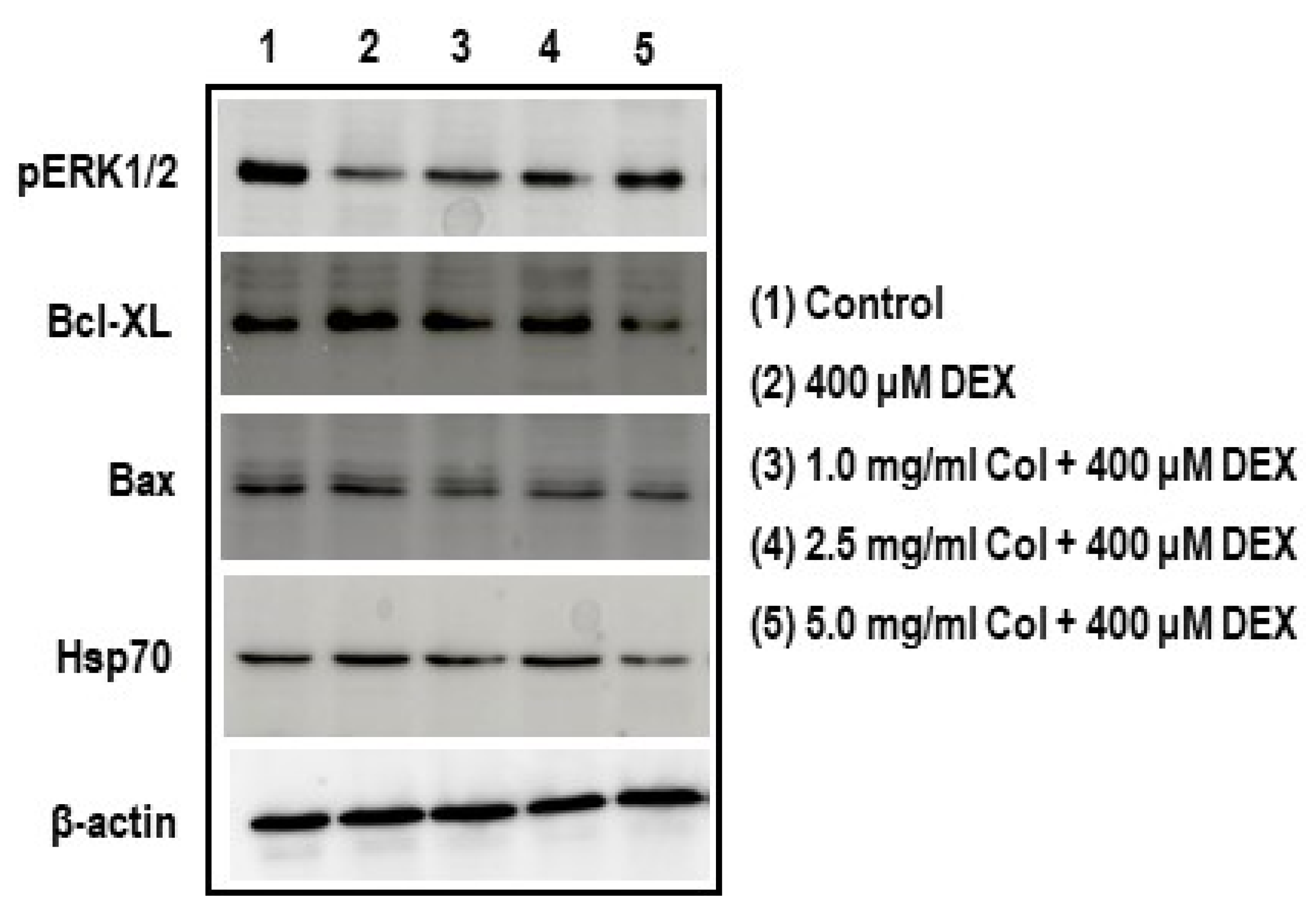
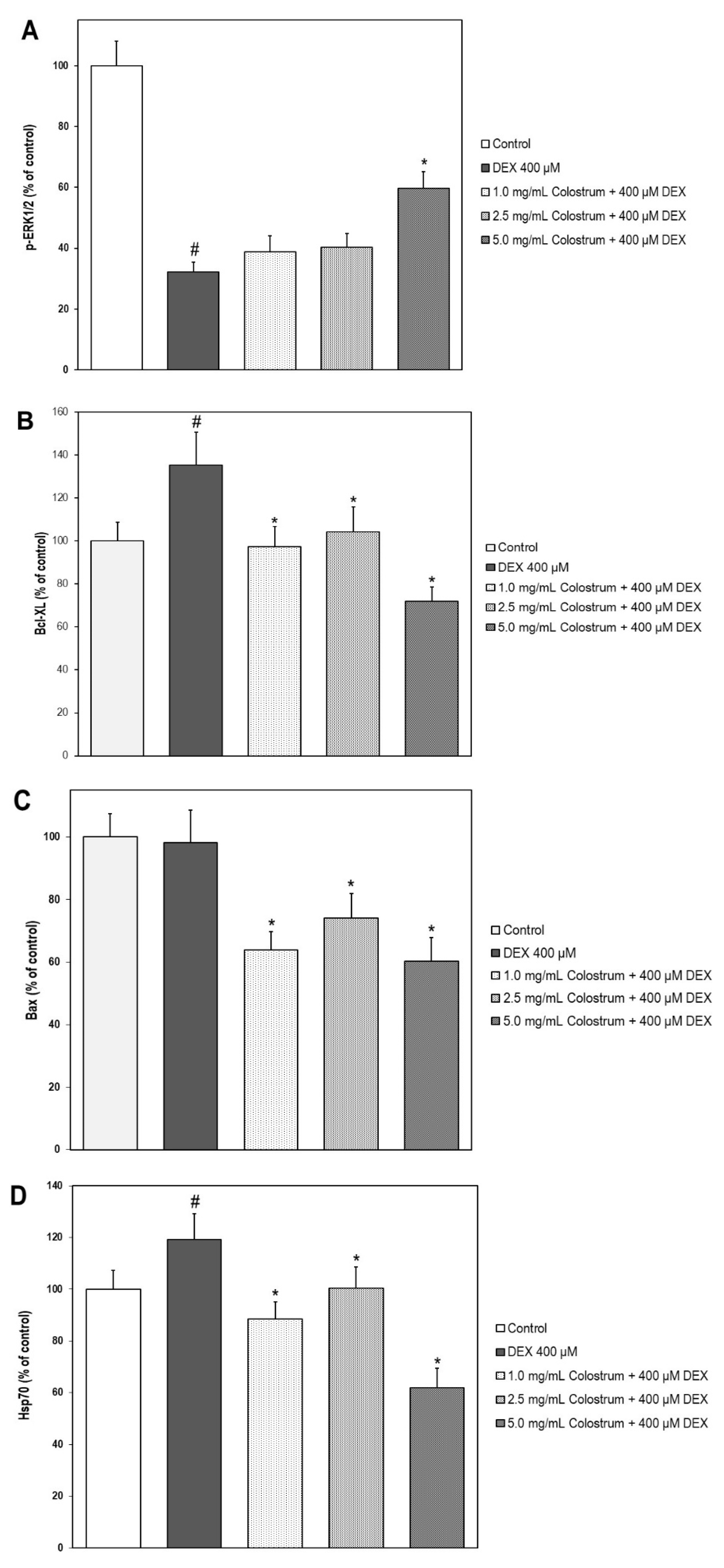
2.10. Colostrum Treatment Increases p-ERK1/2 Levels in DEX-Treated MC3T3-E1 Cells
2.11. Colostrum Treatment Diminishes Bcl-XL Levels in DEX-Treated MC3T3-E1 Cells
2.12. Colostrum Treatment Diminishes Bax Levels in DEX-Treated MC3T3-E1 Cells
2.13. Colostrum Treatment Attenuates Hsp70 Levels in DEX-Treated MC3T3-E1 Cells
3. Discussion
4. Materials and Methods
4.1. Bovine Colostrum
4.2. Culture of the Human Neuroblastoma SH-SY5Y Cells
4.3. Culture of the Mouse MC3T3-E1 Osteoblast Cell Line
4.4. Cell Treatment
4.5. Cell Viability Assay
4.6. Reactive Oxygen Species (ROS) Measurement in SH-SY5Y Cells
4.7. Determination of Caspase-3 Activity
4.8. Glutathione Content
4.9. Analysis of pERK1/2, Bcl-XL, Bax and Hsp70 by Western Blot
4.10. Statistical Analyses
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| DEX | Dexamethasone |
| FAU | Fluorescence arbitrary units |
| FBS | Fetal bovine serum |
| GIO | Glucocorticoid-induced osteoporosis |
| GSH | Glutathione reduced |
| MTT | 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide |
| OD | Optical density |
| ROS | Reactive oxygen species |
References
- Fonseca, H.; Moreira-Gonçalves, D.; Coriolano, H.J.; Duarte, J.A. Bone quality: The determinants of bone strength and fragility. Sports Med. 2014, 44, 37–53. [Google Scholar] [CrossRef] [PubMed]
- Shepstone, L.; Lenaghan, E.; Cooper, C.; Clarke, S.; Fong-Soe-Khioe, R.; Fordham, R.; Gittoes, N.; Harvey, I.; Harvey, N.; Heawood, A.; et al. Screening in the community to reduce fractures in older women (SCOOP): A randomised controlled trial. Lancet 2018, 391, 741–747. [Google Scholar] [CrossRef] [Green Version]
- Amorim, T.; Wyon, M.; Maia, J.; Machado, J.C.; Franklim, M.; Metsios, G.S.; Flouris, A.D.; Koutedakis, Y. Prevalence of Low Bone Mineral Density in Female Dancers. Sports Med. 2015, 45, 257–268. [Google Scholar] [CrossRef] [PubMed]
- Amorim, T.; Koutedakis, Y.; Nevill, A.; Wyon, M.; Maia, J.; Machado, J.C.; Marques, F.; Metsios, G.S.; Flouris, A.D.; Adubeiro, N.; et al. Bone mineral density in vocational and professional ballet dancers. Osteoporos. Int. 2017, 28, 2903–2912. [Google Scholar] [CrossRef]
- Binks, S.; Dobson, R. Risk Factors, Epidemiology and Treatment Strategies for Metabolic Bone Disease in Patients with Neurological Disease. Curr. Osteoporos. Rep. 2016, 14, 199–210. [Google Scholar] [CrossRef]
- Lee, T.H.; Formolo, D.A.; Kong, T.; Lau, S.W.; Ho, C.S.; Leung, R.Y.H.; Hung, F.H.; Yau, S. Potential exerkines for physical exercise-elicited pro-cognitive effects: Insight from clinical and animal research. Int. Rev. Neurobiol. 2019, 147, 361–395. [Google Scholar]
- Tan, Z.S.; Seshadri, S.; Beiser, A.; Zhang, Y.; Felson, D.; Marian, T.H.; Au, R.; Philip, A.W.; Douglas, P.K. Bone Mineral Density and the Risk of Alzheimer Disease. Arch. Neurol. 2005, 62, 107–111. [Google Scholar] [CrossRef]
- Chena, Y.; Raymond, Y.L. Alzheimer’s disease and osteoporosis. Tzu-Chi Med. J. 2017, 29, 138–142. [Google Scholar]
- Henneicke, H.; Gasparini, S.J.; Brennan-Speranza, T.C.; Zhou, H.; Seibel, M.J. Glucocorticoids and bone: Local effects and systemic implications. Trends Endocrinol. Metab. 2014, 25, 197–211. [Google Scholar] [CrossRef]
- Compston, J. Glucocorticoid-induced osteoporosis: An update. Endocrine 2018, 61, 7–16. [Google Scholar] [CrossRef] [Green Version]
- Suwanjang, W.; Wu, K.L.H.; Prachayasittikul, S.; Chetsawang, B.; Charngkaew, K. Mitochondrial Dynamics Impairment in Dexamethasone-Treated Neuronal Cells. Neurochem. Res. 2019, 44, 1567–1581. [Google Scholar] [CrossRef] [PubMed]
- Uenishi, K. Prevention of osteoporosis by foods and dietary supplements. Prevention of osteoporosis by milk and dairy products. Clin. Calcium. 2006, 16, 1606–1614. [Google Scholar] [PubMed]
- Mila, H.; Feugier, A.; Grellet, A.; Anne, J.; Gonnier, M.; Martin, M.; Rossig, L.; Chastant-Maillard, S. Immunoglobulin G concentration in canine colostrum: Evaluation and variability. J. Reprod. Immunol. 2015, 112, 24–28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Batty, B.S.; Bionaz, M. Graduate Student Literature Review: The milk behind the mustache: A review of milk and bone biology. J. Dairy Sci. 2019, 102, 7608–7617. [Google Scholar] [CrossRef]
- Stańczykiewicz, B.; Jakubik-Witkowska, M.; Polanowski, A.; Trziszka, T.; Rymaszewska, J. An animal model of the procognitive properties of cysteine protease inhibitor and immunomodulatory peptides based on colostrum. Adv. Clin. Exp. Med. 2017, 26, 563–569. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Przybylska, J.; Albera, E.; Kankofer, M. Antioxidants in Bovine Colostrum. Reprod. Domest. Anim. 2007, 42, 402–409. [Google Scholar] [CrossRef]
- Moretti, D.B.; Santos, C.B.; Alencar, S.M.; Machado-Neto, R. Colostrum from primiparous Holstein cows shows higher antioxidant activity than colostrum of multiparous ones. J. Dairy Res. 2020, 87, 356–359. [Google Scholar] [CrossRef]
- Petzold, C.; Monjo, M.; Rubert, M.; Reinholt, F.P.; Gomez-Florit, M.; Ramis, J.M.; Ellingsen, J.E.; Lyngstadaas, S.P. Effect of proline-rich synthetic peptide-coated titanium implants on bone healing in a rabbit model. Int. J. Oral. Maxillofac. Implants. 2013, 28, e547–e555. [Google Scholar] [CrossRef]
- Yenkoyan, K.; Fereshetyan, K.; Matinyan, S.; Chavushyan, V.; Aghajanov, M. The role of monoamines in the development of Alzheimer’s disease and neuroprotective effect of a proline rich polypeptide. Prog. Neuropsychopharmacol. Biol. Psychiatry 2018, 86, 76–82. [Google Scholar] [CrossRef]
- Schlimme, E.; Martin, D.; Meisel, H. Nucleosides and nucleotides: Natural bioactive substances in milk and colostrum. Br. J. Nutr. 2000, 84 (Suppl. 1), S59–S68. [Google Scholar] [CrossRef] [Green Version]
- Douraghi-Zadeh, D.; Matharu, B.; Razvi, A.; Austen, B. The protective effects of the nutraceutical, colostrinin, against Alzheimer’s disease, is mediated via prevention of apoptosis in human neurones induced by aggregated beta-amyloid. J. Nutr. Health Aging 2009, 13, 522–527. [Google Scholar] [CrossRef]
- Park, Y.G.; Jeong, J.K.; Moon, M.H.; Lee, J.H.; Lee, Y.J.; Seol, J.W.; Kim, S.J.; Kang, S.J.; Park, S.Y. Insulin-like growth factor-1 protects against prion peptide-induced cell death in neuronal cells via inhibition of Bax translocation. Int. J. Mol. Med. 2012, 30, 1069–1074. [Google Scholar] [CrossRef]
- Kim, S.-K.; Kwon, J.-Y.; Nam, T.-J. Involvement of ligand occupancy in Insulin-like growth factor-I (IGF-I) induced cell growth in osteoblast like MC3T3-E1 cells. Biofactors 2007, 29, 187–202. [Google Scholar] [CrossRef] [PubMed]
- Bagwe-Parab, S.; Yadav, P.; Kaur, G.; Tuli, H.S.; Buttar, H.S. Therapeutic Applications of Human and Bovine Colostrum in the Treatment of Gastrointestinal Diseases and Distinctive Cancer Types: The Current Evidence. Front. Pharmacol. 2020, 11, 01100. [Google Scholar] [CrossRef] [PubMed]
- Aranda, P.; Sanchez, L.; Perez, M.D.; Ena, J.M.; Calvo, M. Insulin in bovine colostrum and milk: Evolution throughout lactation and binding to caseins. J. Dairy Sci. 1991, 74, 4320–4325. [Google Scholar] [CrossRef]
- Pinotti, L.; Rosi, F. Leptin in bovine colostrum and milk. Horm. Metab. Res. 2006, 38, 89–93. [Google Scholar] [CrossRef]
- Kim, Y.; Kelly, O.J.; Ilich, J.Z. Synergism of α-Linolenic Acid, Conjugated Linoleic Acid and Calcium in Decreasing Adipocyte and Increasing Osteoblast Cell Growth. Lipids 2013, 48, 787–802. [Google Scholar] [CrossRef]
- Liu, M.; Fan, F.; Shi, P.; Tu, M.; Yu, C.; Yu, C.; Du, M. Lactoferrin promotes MC3T3-E1 osteoblast cells proliferation via MAPK signaling pathways. Int. J. Biol Macromol. 2018, 107 Pt A, 137–143. [Google Scholar] [CrossRef]
- Canalis, E.; Mazziotti, G.; Giustina, A.; Bilezikian, J.P. Glucocorticoid-induced osteoporosis: Pathophysiology and therapy. Osteoporos. Int. 2007, 18, 1319–1328. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.-L.; Tang, X.-L. Effect of glucocorticoid-induced oxidative stress on the expression of Cbfa1. Chem. Biol. Interact. 2014, 207, 26–31. [Google Scholar] [CrossRef]
- Domazetovic, V.; Marcucci, G.; Iantomasi, T.; Brandi, M.L.; Vincenzini, M.T. Oxidative stress in bone remodeling: Role of antioxidants. Clin. Cases Miner Bone Metab. 2017, 14, 209–216. [Google Scholar] [CrossRef]
- Wong, M.M.; Rao, L.G.; Ly, H.; Hamilton, L.; Tong, J.; Sturtridge, W.; McBroom, R.; Aubin, J.E.; Murray, T.M. Long-term effects of physiologic concentrations of dexamethasone on human bone-derived cells. J. Bone Miner. Res. 1990, 5, 803–813. [Google Scholar] [CrossRef] [PubMed]
- Gopi, I.K.; Rattan, S.I.S. Biphasic Dose Response and Hormetic Effects of Stress Hormone Hydrocortisone on Telomerase-Immortalized Human Bone Marrow Stem Cells In Vitro. Dose Response. 2019, 17, 1559325819889819. [Google Scholar] [CrossRef]
- Mussano, F.; Bartorelli, A.; Brossa, A.; Carossa, S.; Bussolati, G.; Bussolati, B. Presence of osteoinductive factors in bovine colostrum. Biosci. Biotechnol. Biochem. 2014, 78, 662–671. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Liu, X.; He, C. Glucocorticoid-induced autophagy and apoptosis in bone. Apoptosis 2020, 25, 157–168. [Google Scholar] [CrossRef] [PubMed]
- Senft, A.P.; Dalton, T.P.; Shertzer, H.G. Determining glutathione and glutathione disulfide using the fluorescence probe o-phthalaldehyde. Anal. Biochem. 2000, 280, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Nuutinen, U.; Postila, V.; Mättö, M.; Eeva, J.; Ropponen, A.; Eray, M.; Riikonen, P.; Pelkonen, J. Inhibition of PI3-kinase-Akt pathway enhances dexamethasone-induced apoptosis in a human follicular lymphoma cell line. Exp. Cell Res. 2006, 312, 322–330. [Google Scholar] [CrossRef] [PubMed]
- Leskiewicz, M.; Jantas, D.; Regulska, M.; Kaczanowska, J.; Basta-Kaim, A.; Budziszewska, B.; Kubera, M.; Lason, W. Antidepressants attenuate the dexamethasone induced decrease in viability and proliferation of human neuroblastoma SH-SY5Y cells: A involvement of extracellular regulated kinase (ERK1/2). Neurochem. Int. 2013, 63, 354–362. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, C.; Kiely, A.P.; Coakley, M.F.; Manning, S.; Long-Smith, C.M. Insulin and IGF-1 signalling: Longevity, protein homoeostasis and Alzheimer’s disease. Biochem. Soc. Trans. 2012, 40, 721–727. [Google Scholar] [CrossRef]
- Kruzel, M.L.; Janusz, M.; Lisowski, J.; Fischleigh, R.V.; Georgiades, J.A. Towards an understanding of biological role of Colostrinin peptides. Mol. Neurosci. 2001, 17, 379–389. [Google Scholar] [CrossRef]
- Boldogh, I.; Kruzel, M.L. ColostrininTM: An Oxidative Stress Modulator for Prevention and Treatment of Age-related Disorders. J. Alzheimers Dis. 2008, 13, 303–321. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suwanjang, W.; Abramov, A.Y.; Charngkaew, K.; Govitrapong, P.; Chetsawang, B. Melatonin prevents cytosolic calcium overload, mitochondrial damage and cell death due to toxically high doses of dexamethasone-induced oxidative stress in human neuroblastoma SH-SY5Y cells. Neurochem. Int. 2016, 97, 34–41. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, X.W.; Li, Y.H.; Zhang, M.J.; Chen, Z.; Ke, D.S.; Xue, Y.; Hou, J.M. Lactoferrin ameliorates aging-suppressed osteogenesis via IGF1 signaling. J. Mol. Endocrinol. 2019, 63, 63–75. [Google Scholar] [CrossRef] [PubMed]
- Vitali, A. Proline-rich peptides: Multifunctional bioactive molecules as new potential therapeutic drugs. Curr. Protein. Pept. Sci. 2015, 16, 147–162. [Google Scholar] [CrossRef] [PubMed]
- Clem, R.J.; Cheng, E.H.; Karp, C.L.; Kirsch, D.G.; Ueno, K.; Takahashi, A.; Kastan, M.B.; Griffin, D.E.; Earnshaw, W.C.; Veliuona, M.A.; et al. Modulation of cell death by Bcl-XL through caspase interaction. Proc. Natl. Acad. Sci. USA 1998, 95, 554–559. [Google Scholar] [CrossRef] [Green Version]
- Zalavras, C.; Shah, S.; Birnbaum, M.J.; Frenkel, B. Role of apoptosis in glucocorticoid-induced osteoporosis and osteonecrosis. Crit. Rev. Eukaryot. Gene Expr. 2003, 13, 2–4. [Google Scholar] [CrossRef]
- Guo, B.; Yang, M.; Liang, D.; Yang, L.; Cao, J.; Zhang, L. Cell apoptosis induced by zinc deficiency in osteoblastic MC3T3-E1 cells via a mitochondrial-mediated pathway. Mol. Cell. Biochem. 2012, 361, 209–216. [Google Scholar] [CrossRef]
- Chen, Y.-H.; Peng, S.-Y.; Cheng, M.-T.; Hsu, Y.-P.; Huang, Z.-X.; Cheng, W.T.-K.; Wu, S.-C. Different susceptibilities of osteoclasts and osteoblasts to glucocorticoid-induced oxidative stress and mitochondrial alterations. Chin. J. Physiol. 2019, 62, 70–79. [Google Scholar]
- Miraoui, H.; Oudina, K.; Petite, H.; Tanimoto, Y.; Moriyama, K.; Marie, P.J. Fibroblast growth factor receptor 2 promotes osteogenic differentiation in mesenchymal cells via ERK1/2 and protein kinase C signaling. J. Biol. Chem. 2009, 284, 4897–4904. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Porta, A.; Peng, X.; Gengaro, K.; Cunningham, E.B.; Li, H. Prevention of glucocorticoid-induced apoptosis in osteocytes and osteoblasts by calbindin-D28k. J. Bone Miner Res. 2004, 19, 479–490. [Google Scholar] [CrossRef]
- Merriman, H.L.; La Tour, D.; Linkhart, T.A.; Mohan, S.; Baylink, D.J.; Strong, D.D. Insulin-like growth factor-I and insulin-like growth factor-II induce c-fos in mouse osteoblastic cells. Calcif. Tissue Int. 1990, 46, 258–262. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, Y.; Yin, Q.; Xia, H.; Wang, J. Platelet-derived growth factor promotes osteoblast proliferation by activating G-protein-coupled receptor kinase interactor-1. Mol. Med. Rep. 2014, 10, 1349–1354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chaudhary, L.R.; Hruska, K.A. The cell survival signal Akt is differentially activated by PDGF-BB, EGF, and FGF-2 in osteoblastic cells. J. Cell Biochem. 2001, 81, 304–311. [Google Scholar] [CrossRef]
- Ramalingam, M.; Kwon, Y.D.; Kim, S.J. Insulin as a Potent Stimulator of Akt, ERK and Inhibin-βE signaling in Osteoblast-Like UMR-106 Cells. Biomol. Ther. 2016, 24, 589–594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kozawa, O.; Niwa, M.; Hatakeyama, D.; Tokuda, H.; Oiso, Y.; Matsuno, H.; Kato, K.; Uematsu, T. Specific induction of heat shock protein 27 by glucocorticoid in osteoblasts. J. Cell Biochem. 2002, 86, 357–364. [Google Scholar] [CrossRef]
- Arya, R.; Mallik, M.; Lakhotia, S.C. Heat shock genes—integrating cell survival and death. J. Biosci. 2007, 32, 595–610. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, M.; D’Annessa, I.; Moroni, E.; Morra, G.; Paladino, A.; Rinaldi, S.; Compostella, F.; Colombo, G. Allosteric Modulators of HSP90 and HSP70: Dynamics Meets Function through Structure-Based Drug Design. Rev. J. Med. Chem. 2019, 62, 60–87. [Google Scholar] [CrossRef]
- Marchbank, T.; Davison, G.; Oakes, J.R.; Ghatei, M.A.; Patterson, M.; Moyer, M.P.; Playford, R.J. The nutraceutical bovine colostrum truncates the increase in gut permeability caused by heavy exercise in athletes. Am. J. Physiol. Gastrointest. Liver Physiol. 2011, 300, G477–G484. [Google Scholar] [CrossRef] [Green Version]
- Kim, T.; Na, H.; Lee, W.; Jeoung, M.; Lee, S. Heat shock protein 70-1A is a novel angiogenic regulator. Biochem. Biophys. Res. Commun. 2016, 469, 222. [Google Scholar] [CrossRef]
- Schoedel, K.; Miller, V.; Osei-Hwedieh, D.; Watters, R.; Duensing, A.; John, I.; Chandran, U.; Chang, A.; Soman, V.; Weiss, K. Differential expression of angiogenesis markers HSP70, HSP90, VEGF and pERK1/2 in both components of dedifferentiated chondrosarcomas. J. Bone Oncol. 2021, 29, 100370. [Google Scholar] [CrossRef]
- Deng, S.; Dai, G.; Chen, S.; Nie, Z.; Zhou, J.; Fang, H.; Peng, H. Dexamethasone induces osteoblast apoptosis through ROS-PI3K/AKT/GSK3β signaling pathway. Biomed. Pharmacother. 2019, 110, 602–608. [Google Scholar] [CrossRef] [PubMed]
- Han, D.; Gao, J.; Gu, X.; Hengstler, J.G.; Zhang, L.; Shahid, M.; Ali, T.; Han, B. P21(Waf1/Cip1) depletion promotes dexamethasone-induced apoptosis in osteoblastic MC3T3-E1 cells by inhibiting the Nrf2/HO-1 pathway. Arch. Toxicol. 2018, 92, 679–692. [Google Scholar] [CrossRef] [PubMed]
- Chau, J.F.; Leong, W.F.; Li, B. Signaling pathways governing osteoblast proliferation, differentiation and function. Histol. Histopathol. 2009, 24, 1593–1606. [Google Scholar]
- Lavenius, E.; Parrow, V.; Nånberg, E.; Påhlman, S. Basic FGF and IGF-I promote differentiation of human SH-SY5Y neuroblastoma cells in culture. Growth Factors. 1994, 10, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Rimando, M.; Wu, H.H.; Liu, Y.A.; Lee, C.W.; Kuo, S.W.; Lo, Y.P.; Tseng, K.F.; Liu, Y.S.; Kuang-Sheng, O. Glucocorticoid receptor and Histone deacetylase 6 mediate the differential effect of dexamethasone during osteogenesis of mesenchymal stromal cells (MSCs). Sci. Rep. 2016, 6, 37371. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Wang, B.; Liu, X.; Pan, Z.; Liu, C.; Ma, H. The dosage effects of dexamethasone on osteogenic activity and biocompatibility of poly(lactic-co-glycolic acid)/hydroxyapatite nanofibers. Artif. Cells Nanomed. Biotechnol. 2019, 47, 1823–1832. [Google Scholar] [CrossRef] [Green Version]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martin-Aragon, S.; Bermejo-Bescós, P.; Benedí, J.; Raposo, C.; Marques, F.; Kydonaki, E.K.; Gkiata, P.; Koutedakis, Y.; Ntina, G.; Carrillo, A.E.; et al. A Neuroprotective Bovine Colostrum Attenuates Apoptosis in Dexamethasone-Treated MC3T3-E1 Osteoblastic Cells. Int. J. Mol. Sci. 2021, 22, 10195. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms221910195
Martin-Aragon S, Bermejo-Bescós P, Benedí J, Raposo C, Marques F, Kydonaki EK, Gkiata P, Koutedakis Y, Ntina G, Carrillo AE, et al. A Neuroprotective Bovine Colostrum Attenuates Apoptosis in Dexamethasone-Treated MC3T3-E1 Osteoblastic Cells. International Journal of Molecular Sciences. 2021; 22(19):10195. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms221910195
Chicago/Turabian StyleMartin-Aragon, Sagrario, Paloma Bermejo-Bescós, Juana Benedí, Carlos Raposo, Franklim Marques, Eirini K. Kydonaki, Paraskevi Gkiata, Yiannis Koutedakis, Georgia Ntina, Andres E. Carrillo, and et al. 2021. "A Neuroprotective Bovine Colostrum Attenuates Apoptosis in Dexamethasone-Treated MC3T3-E1 Osteoblastic Cells" International Journal of Molecular Sciences 22, no. 19: 10195. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms221910195






