Development of Genetic Modification Tools for Hanseniasporauvarum
Abstract
:1. Introduction
2. Results
2.1. Plasmid Design and Construction
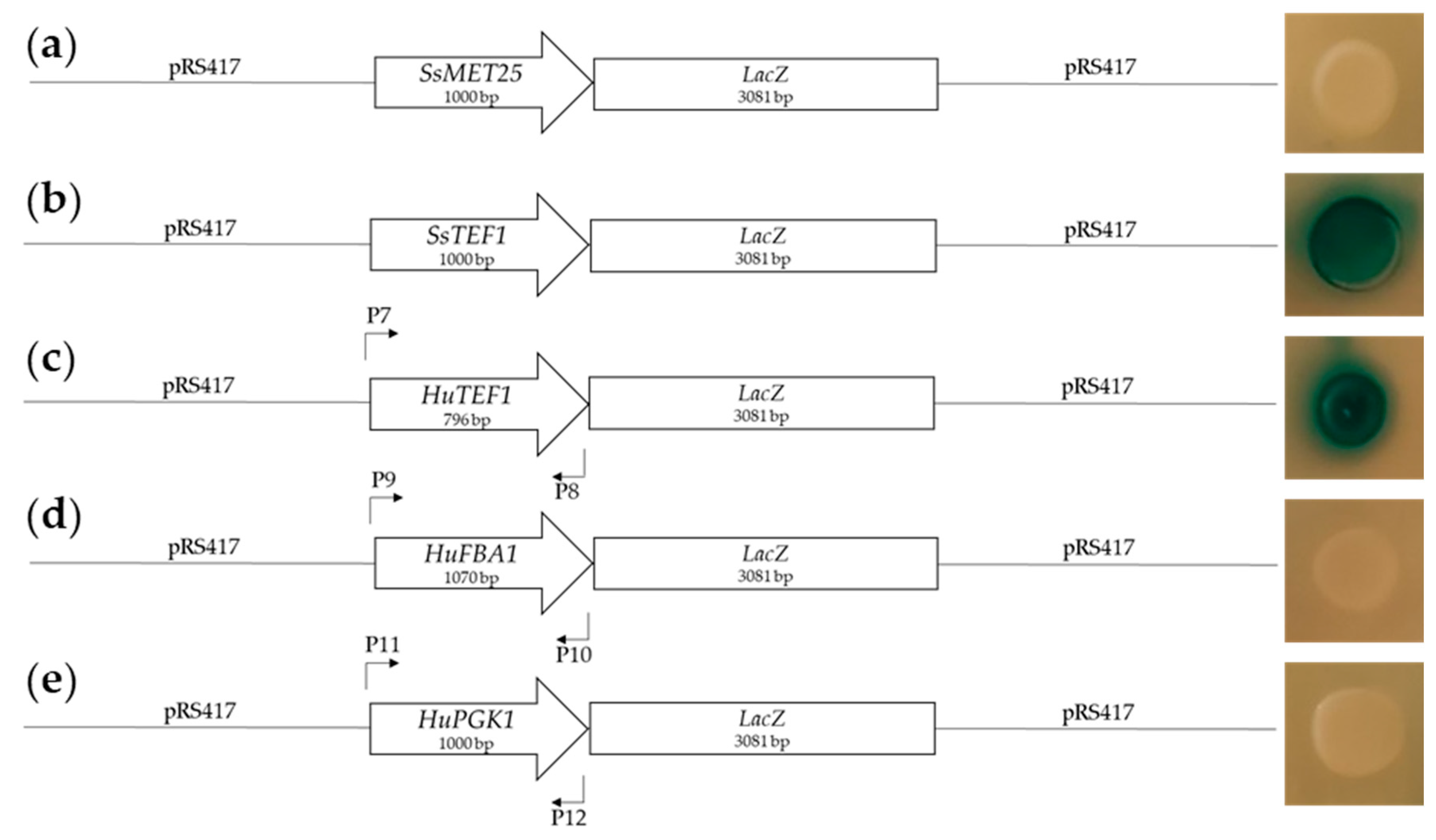
2.1.1. Design and Testing of Promoters
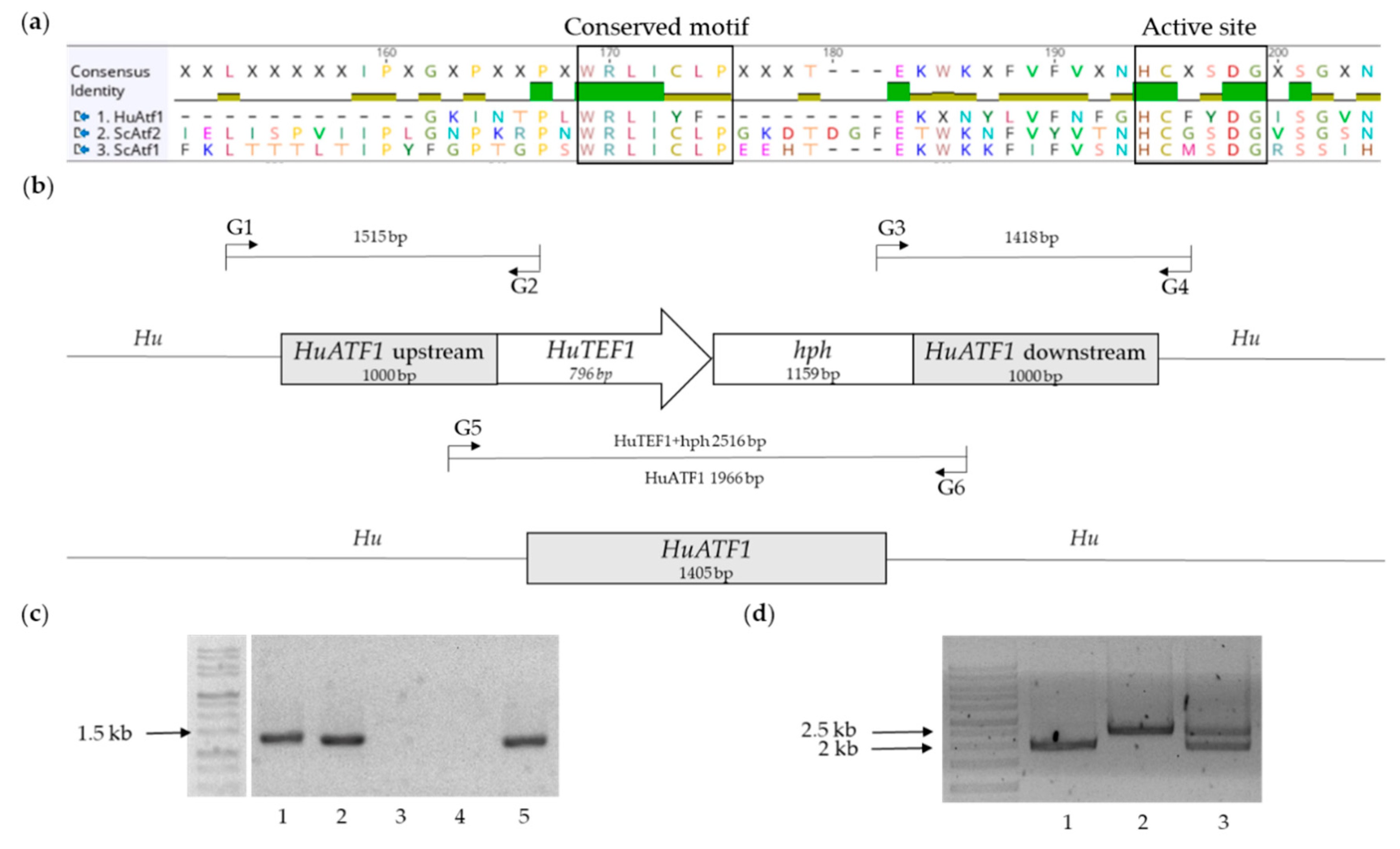
2.1.2. Choice of Candidate Gene and Design of the Knock-Out Cassette
2.2. Generation of Strains and Verification of Transformants
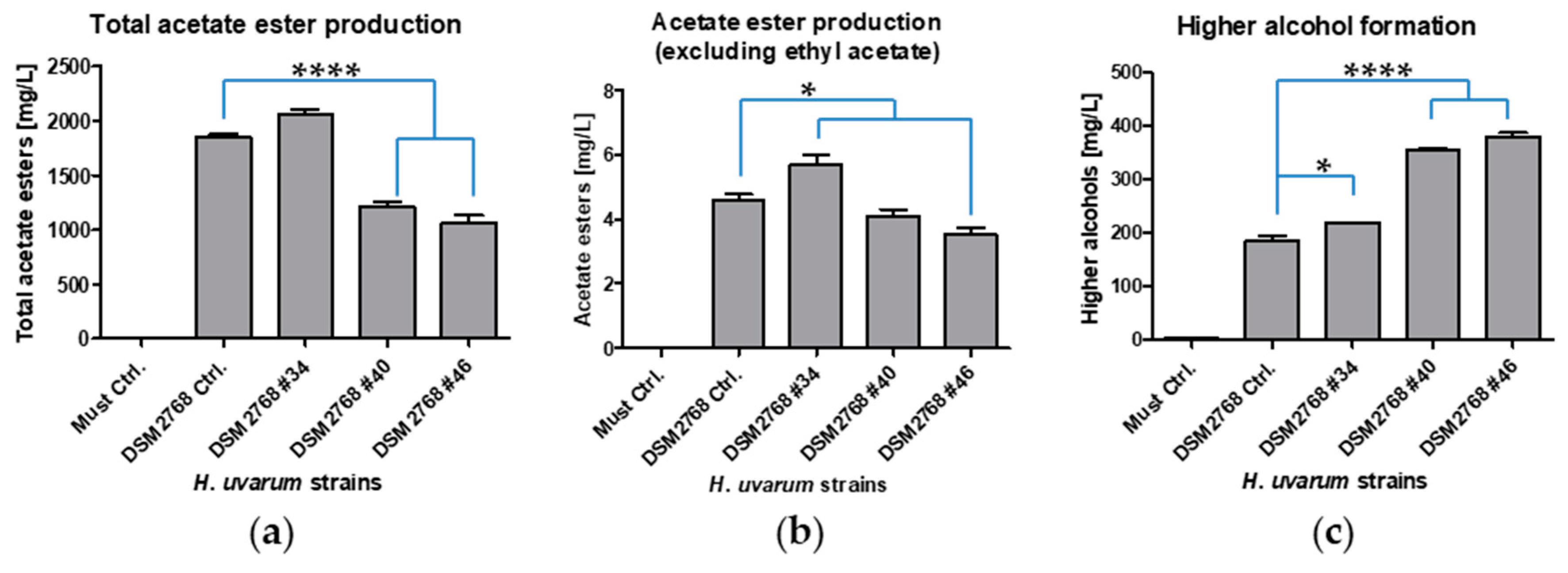
2.3. Fermentations and Phenotypical Characterisation
3. Discussion
4. Materials and Methods
4.1. Strains and Culture Conditions
4.2. Plasmid Design and Construction
4.3. Yeast Transformation
4.4. Verification of Yeast Transformants
4.5. Fermentations
4.6. High Performance Liquid Chromatography (HPLC) Analysis
4.7. Volatile Compound Analysis
4.8. Data Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MDPI | Multidisciplinary Digital Publishing Institute |
| ATF1 | Alcohol acetyltransferase I |
| TEF1 | Translational elongation factor EF-1α |
| bp | Base pair |
| DNA | Deoxyribonucleic acid |
| SO2 | Sulphur dioxide |
| kanMX | Geneticin resistance marker |
| hygMX | Hygromycin resistance marker |
| ORF | Open reading frame |
| PCR | Polymerase chain reaction |
| PGK1 | 3-Phosphoglycerate kinase 1 |
| FBA1 | Fructose-1,6-bisphosphate aldolase 1 |
| ATF2 | Alcohol acetyltransferase II |
| Hph | Hygromycin b phosphotransferase |
| neoR | Neomycin-geneticin resistance gene |
| natI | Nourseothricin N-acetyltransferase |
| Sh Ble | Bleomycin resistance protein |
| acetyl-CoA | Acetyl coenzyme A |
| YPD | Yeast extract peptone dextrose media |
| ANOVA | Analysis of variance |
| TE | Tris-ethylenediaminetetraacetic acid |
| HCl | Hydrogen chloride |
| EDTA | Ethylenediaminetetraacetic acid |
| HPLC | High performance liquid chromatography |
| VCA | Volatile compound analysis |
| MWD | Multi-wavelength detector |
| RID | Refractive index detector |
| HS-SPME-GC-MS | Headspace-solid phase microextraction gas chromatography mass Spectrometry |
| GC | Gas chromatography |
| MS | Mass spectrometry |
| MPS | Multipurpose sampler |
| CIS | Cooled injection system |
| NaCl | Sodium chloride |
References
- Martin, V.; Valera, M.; Medina, K.; Boido, E.; Carrau, F. Oenological impact of the Hanseniaspora/Kloeckera yeast genus on wines—A review. Fermentation 2018, 4, 76. [Google Scholar] [CrossRef] [Green Version]
- Heard, G.M.; Fleet, G.H. Evaluation of selective media for enumeration of yeasts during wine fermentation. J. Appl. Bacteriol. 1986, 60, 477–481. [Google Scholar] [CrossRef]
- Fleet, G. Yeast interactions and wine flavour. Int. J. Food Microbiol. 2003, 86, 11–22. [Google Scholar] [CrossRef]
- Heard, G.M.; Fleet, G.H. Growth of natural yeast flora during the fermentation of inoculated wines. Appl. Environ. Microbiol. 1985, 50, 727–728. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holloway, P.; Subden, R.E.; Lachance, M.-A. The yeasts in a Riesling must from the Niagara grape-growing region of Ontario. Can. Inst. Food Sci. Technol. J. 1990, 23, 212–216. [Google Scholar] [CrossRef]
- Comi, G.; Romano, P.; Cocolin, L.; Fiore, C. Characterization of Kloeckera apiculata strains from the Friuli region in Northern Italy. World J. Microbiol. Biotechnol. 2001, 17, 391–394. [Google Scholar] [CrossRef]
- Jolly, N.P.; Augustyn, O.; Pretorius, I.S. The occurrence of non-Saccharomyces cerevisiae yeast species over three vintages in four vineyards and grape musts from four production regions of the Western Cape, South Africa. S. Afr. J. Enol. Vitic. 2003, 24. [Google Scholar] [CrossRef] [Green Version]
- Li, S.-S.; Cheng, C.; Li, Z.; Chen, J.-Y.; Yan, B.; Han, B.-Z.; Reeves, M. Yeast species associated with wine grapes in China. Int. J. Food Microbiol. 2010, 138, 85–90. [Google Scholar] [CrossRef]
- Belda, I.; Ruiz, J.; Alastruey-Izquierdo, A.; Navascués, E.; Marquina, D.; Santos, A. Unraveling the enzymatic basis of wine “flavorome”: A phylo-functional study of wine related yeast species. Front. Microbiol. 2016, 7, 12. [Google Scholar] [CrossRef] [Green Version]
- Tofalo, R.; Patrignani, F.; Lanciotti, R.; Perpetuini, G.; Schirone, M.; Di Gianvito, P.; Pizzoni, D.; Arfelli, G.; Suzzi, G. Aroma profile of Montepulciano d’Abruzzo wine fermented by single and co-culture starters of autochthonous Saccharomyces and non-Saccharomyces yeasts. Front. Microbiol. 2016, 7, 610. [Google Scholar] [CrossRef]
- Jolly, N.P.; Varela, C.; Pretorius, I.S. Not your ordinary yeast: Non-Saccharomyces yeasts in wine production uncovered. FEMS Yeast Res. 2014, 14, 215–237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, C.; Mas, A.; Esteve-Zarzoso, B. Interaction between Hanseniaspora uvarum and Saccharomyces cerevisiae during alcoholic fermentation. Int. J. Food Microbiol. 2015, 206, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Pina, C.; Santos, C.; Couto, J.A.; Hogg, T. Ethanol tolerance of five non-Saccharomyces wine yeasts in comparison with a strain of Saccharomyces cerevisiae—Influence of different culture conditions. Food Microbiol. 2004, 21, 439–447. [Google Scholar] [CrossRef]
- Akan, M.; Michling, F.; Matti, K.; Krause, S.; Muno-Bender, J.; Wendland, J. Snails as taxis for a large yeast biodiversity. Fermentation 2020, 6, 90. [Google Scholar] [CrossRef]
- Comitini, F.; Ciani, M. The zymocidial activity of Tetrapisispora phaffii in the control of Hanseniaspora uvarum during the early stages of winemaking. Lett. Appl. Microbiol. 2010, 50, 50–56. [Google Scholar] [CrossRef]
- Tristezza, M.; Tufariello, M.; Capozzi, V.; Spano, G.; Mita, G.; Grieco, F. The oenological potential of Hanseniaspora uvarum in simultaneous and sequential co-fermentation with Saccharomyces cerevisiae for industrial wine production. Front. Microbiol. 2016, 7, 670. [Google Scholar] [CrossRef] [Green Version]
- Petruzzi, L.; Capozzi, V.; Berbegal, C.; Corbo, M.R.; Bevilacqua, A.; Spano, G.; Sinigaglia, M. Microbial resources and enological significance: Opportunities and benefits. Front. Microbiol. 2017, 8, 995. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roudil, L.; Russo, P.; Berbegal, C.; Albertin, W.; Spano, G.; Capozzi, V. Non-Saccharomyces commercial starter cultures: Scientific trends, recent patents and innovation in the wine sector. Recent Pat. Food Nutr. Agric. 2020, 11, 27–39. [Google Scholar] [CrossRef]
- Van Wyk, N.; von Wallbrunn, C.; Swiegers, J.H.; Pretorius, I.S. Biotechnology of wine yeasts. In Encyclopedia of Mycology; Elsevier: Amsterdam, The Netherlands, 2020; ISBN 9780128096338. [Google Scholar]
- Valles, B.S.; Bedriñana, R.P.; Tascón, N.F.; Simón, A.Q.; Madrera, R.R. Yeast species associated with the spontaneous fermentation of cider. Food Microbiol. 2007, 24, 25–31. [Google Scholar] [CrossRef]
- Lachance, M.A. Yeast communities in a natural tequila fermentation. Antonie van Leeuwenhoek 1995, 68, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Masoud, W.; Cesar, L.B.; Jespersen, L.; Jakobsen, M. Yeast involved in fermentation of Coffea arabica in East Africa determined by genotyping and by direct denaturating gradient gel electrophoresis. Yeast 2004, 21, 549–556. [Google Scholar] [CrossRef]
- Batista, N.N.; Ramos, C.L.; Ribeiro, D.D.; Pinheiro, A.C.M.; Schwan, R.F. Dynamic behavior of Saccharomyces cerevisiae, Pichia kluyveri and Hanseniaspora uvarum during spontaneous and inoculated cocoa fermentations and their effect on sensory characteristics of chocolate. LWT—Food Sci. Technol. 2015, 63, 221–227. [Google Scholar] [CrossRef] [Green Version]
- Arias, C.R.; Burns, J.K.; Friedrich, L.M.; Goodrich, R.M.; Parish, M.E. Yeast species associated with orange juice: Evaluation of different identification methods. Appl. Environ. Microbiol. 2002, 68, 1955–1961. [Google Scholar] [CrossRef] [Green Version]
- Vadkertiová, R.; Dudášová, H.; Stratilová, E.; Balaščáková, M. Diversity of yeasts in the soil adjacent to fruit trees of the Rosaceae family. Yeast 2019, 36, 617–631. [Google Scholar] [CrossRef] [PubMed]
- Sláviková, E.; Vadkertiová, R.; Vránová, D. Yeasts colonizing the leaves of fruit trees. Ann. Microbiol. 2009, 59, 419–424. [Google Scholar] [CrossRef]
- Nguyen, N.H.; Suh, S.-O.; Blackwell, M. Five novel Candida species in insect-associated yeast clades isolated from Neuroptera and other insects. Mycologia 2007, 99, 842–858. [Google Scholar] [CrossRef] [PubMed]
- Kocan, R.M.; Hasenclever, H.F. Normal yeast flora of the upper digestive tract of some wild columbids. J. Wildl. Dis. 1972, 8, 365–368. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Araujo, F.V.; Soares, C.A.; Hagler, A.N.; Mendonça-Hagler, L.C. Ascomycetous yeast communities of marine invertebrates in a southeast Brazilian mangrove ecosystem. Antonie van Leeuwenhoek 1995, 68, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Pagnocca, F.G.; Mendonca-Hagler, L.C.; Hagler, A.N. Yeasts associated with the white shrimp Penaeus schmitti, sediment, and water of Sepetiba Bay, Rio de Janeiro, Brasil. Yeast 1989, 5, 479–483. [Google Scholar]
- Long, C.; Wu, Z.; Deng, B.-X. Biological control of Penicillium italicum of citrus and Botrytis cinerea of grape by strain 34–9 of Kloeckera apiculata. Eur. Food Res. Technol. 2005, 221, 197–201. [Google Scholar] [CrossRef]
- Basha, H.; Ramanujam, B. Growth promotion effect of Pichia guilliermondii in chilli and biocontrol potential of Hanseniaspora uvarum against Colletotrichum capsici causing fruit rot. Biocontrol Sci. Technol. 2015, 25, 185–206. [Google Scholar] [CrossRef]
- Liu, H.M.; Guo, J.H.; Cheng, Y.J.; Luo, L.; Liu, P.; Wang, B.Q.; Deng, B.X.; Long, C.A. Control of gray mold of grape by Hanseniaspora uvarum and its effects on postharvest quality parameters. Ann. Microbiol. 2010, 60, 31–35. [Google Scholar] [CrossRef]
- Sternes, P.R.; Lee, D.; Kutyna, D.R.; Borneman, A.R. Genome sequences of three species of Hanseniaspora isolated from spontaneous wine fermentations. Genome Announc. 2016, 4. [Google Scholar] [CrossRef] [Green Version]
- Seixas, I.; Barbosa, C.; Mendes-Faia, A.; Güldener, U.; Tenreiro, R.; Mendes-Ferreira, A.; Mira, N.P. Genome sequence of the non-conventional wine yeast Hanseniaspora guilliermondii UTAD222 unveils relevant traits of this species and of the Hanseniaspora genus in the context of wine fermentation. DNA Res. 2019, 26, 67–83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guaragnella, N.; Chiara, M.; Capece, A.; Romano, P.; Pietrafesa, R.; Siesto, G.; Manzari, C.; Pesole, G. Genome sequencing and comparative analysis of three Hanseniaspora uvarum indigenous wine strains reveal remarkable biotechnological potential. Front. Microbiol. 2019, 10, 3133. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, K.; Yang, X.; Zheng, F.; Long, C. Genome sequencing and analysis of Kloeckera apiculata strain 34-9, a biocontrol agent against postharvest pathogens in citrus. Genes Genom. 2017, 39, 87–99. [Google Scholar] [CrossRef]
- Langenberg, A.-K.; Bink, F.J.; Wolff, L.; Walter, S.; von Wallbrunn, C.; Grossmann, M.; Heinisch, J.J.; Schmitz, H.-P. Glycolytic functions are conserved in the genome of the wine yeast Hanseniaspora uvarum, and pyruvate kinase limits its capacity for alcoholic fermentation. Appl. Environ. Microbiol. 2017, 83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Steenwyk, J.L.; Opulente, D.A.; Kominek, J.; Shen, X.-X.; Zhou, X.; Labella, A.L.; Bradley, N.P.; Eichman, B.F.; Čadež, N.; Libkind, D.; et al. Extensive loss of cell-cycle and DNA repair genes in an ancient lineage of bipolar budding yeasts. PLoS Biol. 2019, 17, e3000255. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saubin, M.; Devillers, H.; Proust, L.; Brier, C.; Grondin, C.; Pradal, M.; Legras, J.-L.; Neuvéglise, C. Investigation of genetic relationships between Hanseniaspora species found in grape musts revealed interspecific hybrids with dynamic genome structures. Front. Microbiol. 2019, 10, 2960. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kayacan, Y.; Griffiths, A.; Wendland, J. A script for initiating molecular biology studies with non-conventional yeasts based on Saccharomycopsis schoenii. Microbiol. Res. 2019, 229, 126342. [Google Scholar] [CrossRef] [PubMed]
- Nancolas, B.; Bull, I.D.; Stenner, R.; Dufour, V.; Curnow, P. Saccharomyces cerevisiae Atf1p is an alcohol acetyltransferase and a thioesterase in vitro. Yeast 2017, 34, 239–251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bernardi, B.; Kayacan, Y.; Akan, M.; Wendland, J. Overexpression of RAD51 enables PCR-based gene targeting in lager yeast. Microorganisms 2019, 7, 192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sakai, K.; Yamamoto, M. Transformation of the yeast, Saccharomyces carlsbergensis, using an antibiotic resistance marker. Agric. Biol. Chem. 1986, 50, 1177–1182. [Google Scholar] [CrossRef]
- Nakazawa, N.; Hashimoto, H.; Harashima, S.; Oshima, Y. Use of the PDR4 gene as a dominant selective marker in combination with cerulenin for prototrophic strains in Saccharomyces cerevisiae. J. Ferment. Bioeng. 1993, 76, 60–63. [Google Scholar] [CrossRef]
- Kondo, K.; Saito, T.; Kajiwara, S.; Takagi, M.; Misawa, N. A transformation system for the yeast Candida utilis: Use of a modified endogenous ribosomal protein gene as a drug-resistant marker and ribosomal DNA as an integration target for vector DNA. J. Bacteriol. 1995, 177, 7171–7177. [Google Scholar] [CrossRef] [Green Version]
- Hashida-Okado, T.; Ogawa, A.; Kato, I.; Takesako, K. Transformation system for prototrophic industrial yeasts using the AUR1 gene as a dominant selection marker. FEBS Lett. 1998, 425, 117–122. [Google Scholar] [CrossRef] [Green Version]
- Lilly, M.; Bauer, F.F.; Lambrechts, M.G.; Swiegers, J.H.; Cozzolino, D.; Pretorius, I.S. The effect of increased yeast alcohol acetyltransferase and esterase activity on the flavour profiles of wine and distillates. Yeast 2006, 23, 641–659. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verstrepen, K.J.; van Laere, S.D.M.; Vanderhaegen, B.M.P.; Derdelinckx, G.; Dufour, J.-P.; Pretorius, I.S.; Winderickx, J.; Thevelein, J.M.; Delvaux, F.R. Expression levels of the yeast alcohol acetyltransferase genes ATF1, Lg-ATF1, and ATF2 control the formation of a broad range of volatile esters. Appl. Environ. Microbiol. 2003, 69, 5228–5237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Christiaens, J.F.; Franco, L.M.; Cools, T.L.; de Meester, L.; Michiels, J.; Wenseleers, T.; Hassan, B.A.; Yaksi, E.; Verstrepen, K.J. The fungal aroma gene ATF1 promotes dispersal of yeast cells through insect vectors. Cell Rep. 2014, 9, 425–432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Wyk, N.; Kroukamp, H.; Espinosa, M.I.; von Wallbrunn, C.; Wendland, J.; Pretorius, I.S. Blending wine yeast phenotypes with the aid of CRISPR DNA editing technologies. Int. J. Food Microbiol. 2020, 324, 108615. [Google Scholar] [CrossRef] [PubMed]
- Sikorski, R.S.; Hieter, P. A System of shuttle vectors and yeast host strains designed for efficient manipulation of DNA in Saccharomyces cerevisiae. Genetics 1989, 122, 19–27. [Google Scholar] [CrossRef]
- Chee, M.K.; Haase, S.B. New and redesigned pRS plasmid shuttle vectors for genetic manipulation of Saccharomyces cerevisiae. G3 Genes|Genomes|Genetics 2012, 2, 515–526. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wach, A.; Brachat, A.; Pöhlmann, R.; Philippsen, P. New heterologous modules for classical or PCR-based gene disruptions in Saccharomyces cerevisiae. Yeast 1994, 10, 1793–1808. [Google Scholar] [CrossRef] [PubMed]
- Schaub, Y.; Dünkler, A.; Walther, A.; Wendland, J. New pFA-cassettes for PCR-based gene manipulation in Candida albicans. J. Basic Microbiol. 2006, 46, 416–429. [Google Scholar] [CrossRef] [PubMed]
- Gatignol, A.; Dassain, M.; Tiraby, G. Cloning of Saccharomyces cerevisiae promoters using a probe vector based on phleomycin resistance. Gene 1990, 91, 35–41. [Google Scholar] [CrossRef]
- Gietz, R.D.; Schiestl, R.H. High-efficiency yeast transformation using the LiAc/SS carrier DNA/PEG method. Nat. Protoc. 2007, 2, 31–34. [Google Scholar] [CrossRef]
- Thompson, J.R.; Register, E.; Curotto, J.; Kurtz, M.; Kelly, R. An improved protocol for the preparation of yeast cells for transformation by electroporation. Yeast 1998, 14, 565–571. [Google Scholar] [CrossRef]
- Schneider, A.; Gerbi, V.; Redoglia, M. A rapid HPLC method for separation and determination of major organic acids in grape musts and wines. Am. J. Enol. Vitic. 1987, 38, 151. [Google Scholar]
- Mecca, D.; Benito, S.; Beisert, B.; Brezina, S.; Fritsch, S.; Semmler, H.; Rauhut, D. Influence of nutrient supplementation on Torulaspora Delbrueckii wine fermentation aroma. Fermentation 2020, 6, 35. [Google Scholar] [CrossRef] [Green Version]
- Câmara, J.S.; Alves, M.A.; Marques, J.C. Development of headspace solid-phase microextraction-gas chromatography–mass spectrometry methodology for analysis of terpenoids in Madeira wines. Anal. Chim. Acta 2006, 555, 191–200. [Google Scholar] [CrossRef]
- Dutraive, O.; Benito, S.; Fritsch, S.; Beisert, B.; Patz, C.-D.; Rauhut, D. Effect of sequential inoculation with non-Saccharomyces and Saccharomyces yeasts on Riesling wine chemical composition. Fermentation 2019, 5, 79. [Google Scholar] [CrossRef] [Green Version]

| Strain | Feature/Genotype | Source |
|---|---|---|
| BY4741 | Saccharomyces cerevisiae MATa his3Δ1, leu2Δ0, met15Δ0, ura3Δ0 | Euroscarf, Oberursel, Germany |
| DSM2768 | Hanseniaspora uvarum ATF1/ATF1 | [38] |
| DSM2768 (WT/Δ) | Hanseniaspora uvarum ATF1/atf1:HuTEF1-hph | This study |
| DSM2768 (Δ/Δ) | Hanseniaspora uvarum atf1:HuTEF1-hph/atf1:HuTEF1-hph | This study |
| Strain | Feature/Genotype | Source |
|---|---|---|
| pRS415 | bla, LEU2 | [52] |
| pGEM | bla | Promega, Madison, WI, USA |
| pJET | bla | Thermo Fisher Scientific, Waltham, MA, USA |
| pRS417-SsTEF-lacZ | bla, GEN3, lacZ driven by S. schoenii TEF1 promoter | [41] |
| pRS417-SsMET25-lacZ | bla, GEN3, lacZ driven by S. schoenii MET25 promoter | [41] |
| pRS417-HuTEF-lacZ | bla, GEN3, lacZ driven by H. uvarum TEF1 promoter | This study |
| pRS417-HuFBA-lacZ | bla, GEN3, lacZ driven by H. uvarum FBA1 promoter | This study |
| pRS417-HuPGK-lacZ | bla, GEN3, lacZ driven by H. uvarum PGK1 promoter | This study |
| pRS40H | HygMX | [53] |
| pTEF | pJET-HuTEF1 | This study |
| pJB1-HuTEF-hph | pRS415-HuTEF1-hph | This study |
| pJB2-HuTEF-hph-ATF1up/down | pRS415-HuTEF1-hph-1000 bp HuATF1 upstream/downstream | This study |
| pFA-KanMX6 | KanMX6 | [54] |
| pJB3-HuTEF-neoR | pRS415-HuTEF1-neoR | This study |
| pFA-NatMX3 | NatMX3 | [55] |
| pJB4-HuTEF-natI | pRS415-HuTEF1-natI | This study |
| pZeoR | BleMX6 | [56] |
| pJB5-HuTEF-Sh Ble | pRS415-HuTEF1-Sh Ble | This study |
| pJB6-HuTEF-Sh Ble-ATF1up/down | pRS415-HuTEF1-Sh Ble-1000 bp HuATF1 upstream/downstream | This study |
| Primer Number | Primer Name | Sequence 5′ → 3′ |
|---|---|---|
| Hu-Promoter Test | ||
| 1 | TEF_L | TTGATGGATAACTTGAAGGC |
| 2 | TEF_R | TCTATATACTGTATACCTTAG |
| 3 | FBA_L | GAAGATATACTAAATTTGTCCC |
| 4 | FBA_R | AATGTATGTATTTGTATAATTGATATTATTATGG |
| 5 | PGK_L | TGATTATGACTCTGATAGCAAC |
| 6 | PGK_R | TTTTAAGATTTGTAGTAATTTAATTGTTTATATG |
| 7 | TEF forw. LacZ | CGCCAGGGTTTTCCCAGTCACGACGTTGTAAAACG ACGGCCAGTGTTGATGGATAACTTGAAGGC |
| 8 | TEF rev. LacZ | CAATCTTTGGATCGTTTAAATAAGTTTGAATTTTT TCAGTCATGTTTCTATATACTGTATACCTTAG |
| 9 | FBA forw. LacZ | CGCCAGGGTTTTCCCAGTCACGACGTTGTAAAACG ACGGCCAGTGGAAGATATACTAAATTTGTCCC |
| 10 | FBA rev. LacZ | CAATCTTTGGATCGTTTAAATAAGTTTGAATTTTTTCAGTCA TGTTAATGTATGTATTTGTATAATTGATAT TATTATGG |
| 11 | PGK forw. LacZ | CGCCAGGGTTTTCCCAGTCACGACGTTGTAAAAC GACGGCCAGTGTGATTATGACTCTGATAGCAAC |
| 12 | PGK rev. LacZ | CAATCTTTGGATCGTTTAAATAAGTTTGAATTTTTTCAGTC ATGTTTTTTAAGATTTGTAGTAATTTAATTGTTTATATG |
| Construction Knock-Out Cassettehph | ||
| 13 | pRS415 (ov) + TEF_L | ATTGGGTACCGGGCCCCCCCTCGAGGTCGACGGTA TCGATTTGATGGATAACTTGAAGGC |
| 14 | TEF_R+hyg (ov) | TTTTTCAACAGAAGTAGCAGTCAATTCTGGTTTTT TCATTCTATATACTGTATACCTTAG |
| 15 | TEF (ov) + hyg_L | AGCGGTATAACCATAGAAACTAAGGTATACAGTAT ATAGAATGAAAAAACCAGAATTGAC |
| 16 | hyg_R + pRS415 (ov) | GCTGGAGCTCCACCGCGGTGGCGGCCGCTCTAGAA CTAGTAGGACCACCTTTGATTGTAA |
| 17 | pRS415 (ov) + ATFup_L | ATTGGGTACCGGGCCCCCCCTCGAGGTCGACGGTA TCGATGTACATCTGTTTTTAATGCT |
| 18 | ATFup_R + TEF (ov) | AGTATGGCCATTGTTATGATGCCTTCAAGTTATCC ATCAACTCCGAGGTAATGTTTTTGA |
| 19 | Hyg (ov) + ATFdown_L NEW | AGGGTGGTAATTATTACTATTTACAATCAAAGGTG GTCCTTCAAAATATTGTATTTTCTT |
| 20 | ATFdown_R + pRS415 (ov) NEW | CTGGAGCTCCACCGCGGTGGCGGCCGCTCTAGAAC TAGTGTATCCCAACAAAAGATAGAA |
| 21 | ATFup (ov) + TEF_L | TTAACCCCAGCAGGAATACATCAAAAACATTACCT CGGAGTTGATGGATAACTTGAAGGC |
| 22 | Hyg_R + ATFdown (ov) NEW | TAAAAAGAATAAAACTTTGAAAGAAAATACAATAT TTTGAAGGACCACCTTTGATTGTAA |
| Construction Knock-Out CassetteneoR, natI, Sh Ble | ||
| 23 | TEF_R + kanMX (ov) | CGTATAAATCAGCATCCATTCTATATACTGTATACC TTAGTTTCTATGGTTATACCGCTA |
| 24 | TEF (ov) + kanMX_L | AGCGGTATAACCATAGAAACTAAGGTATACAGTAT ATAGAATGGATGCTGATTTATACGG |
| 25 | kanMX_R + pRS415 (ov) | GCTGGAGCTCCACCGCGGTGGCGGCCGCTCTAGAA CTAGTTAATAAATTATTTTTATTGT |
| 26 | kanMX_R + ATFdown (ov) | GGATCGTAAAGTCTATTAAAACTTTTAAAGTAATT GAACTTAATAAATTATTTTTATTGT |
| 27 | kanMX (ov) + ATFdown_L | TTCTTGCTTTATAAATAACAACAATAAAAATAATTT ATTAAGTTCAATTACTTTAAAAGT |
| 28 | TEF_R + clo (ov) | CCAATGTTTCAGCAACTTGTTCAGGAATAACAGAAA TTTTTCTATATACTGTATACCTTA |
| 29 | clo_L + TEF (ov) | AGCGGTATAACCATAGAAACTAAGGTATACAGTAT ATAGAAAAATTTCTGTTATTCCTGA |
| 30 | TEF_R + Zeo (ov) | GAGCAGTCAGGACTGGAACAGCAGAGGTGAGTTTA GCCATTCTATATACTGTATACCTTA |
| 31 | Zeo_L + TEF (ov) | AGCGGTATAACCATAGAAACTAAGGTATACAGTAT ATAGAATGGCTAAACTCACCTCTGC |
| 32 | ATFdown_L + Zeo (ov) | GTTTTATTATCTATTTATGCCCTTATATTCTGTAACTA TCTCAAAATATTGTATTTTCTT |
| 33 | Zeo_R + ATFdown (ov) | TAAAAAGAATAAAACTTTGAAAGAAAATACAATAT TTTGAGATAGTTACAGAATATAAGG |
| 34 | Zeo_R + pRS415 (ov) | CTGGAGCTCCACCGCGGTGGCGGCCGCTCTAGAACT AGTGGATAGTTACAGAATATAAGG |
| Verification of Gene Deletion inH. uvarum(see Figure 2) | ||
| 35 | G1 ATF1 F | ATTCCTGCGCAGTCTTAAGCTT |
| 36 | G2 R | CAAATCGCTGAAATGGGTGCT |
| 37 | G3 F | CAGGTGCTGGTACTGTTGGT |
| 38 | G4 ATF1 R NEW | AGAATCTTTTGACCGAGCATGA |
| 39 | ATF1_L | TTAATTAAATGCTTACGCTTTCGGATGTTC |
| 40 | HuATF1 R | AAAGGCGCGCCTACAATATTTTGACTAAATGTTAT |
| 41 | G5 ATFup-TEF_L | CAAAAGGCAACCATTCCCCC |
| 42 | G6 ATFdown-Hyg_R | CTGCCATGGCCAATATTCCA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Badura, J.; van Wyk, N.; Brezina, S.; Pretorius, I.S.; Rauhut, D.; Wendland, J.; von Wallbrunn, C. Development of Genetic Modification Tools for Hanseniasporauvarum. Int. J. Mol. Sci. 2021, 22, 1943. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22041943
Badura J, van Wyk N, Brezina S, Pretorius IS, Rauhut D, Wendland J, von Wallbrunn C. Development of Genetic Modification Tools for Hanseniasporauvarum. International Journal of Molecular Sciences. 2021; 22(4):1943. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22041943
Chicago/Turabian StyleBadura, Jennifer, Niël van Wyk, Silvia Brezina, Isak S. Pretorius, Doris Rauhut, Jürgen Wendland, and Christian von Wallbrunn. 2021. "Development of Genetic Modification Tools for Hanseniasporauvarum" International Journal of Molecular Sciences 22, no. 4: 1943. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22041943






