Chemically and Green Synthesized ZnO Nanoparticles Alter Key Immunological Molecules in Common Carp (Cyprinus carpio) Skin Mucus
Abstract
:1. Introduction
2. Results
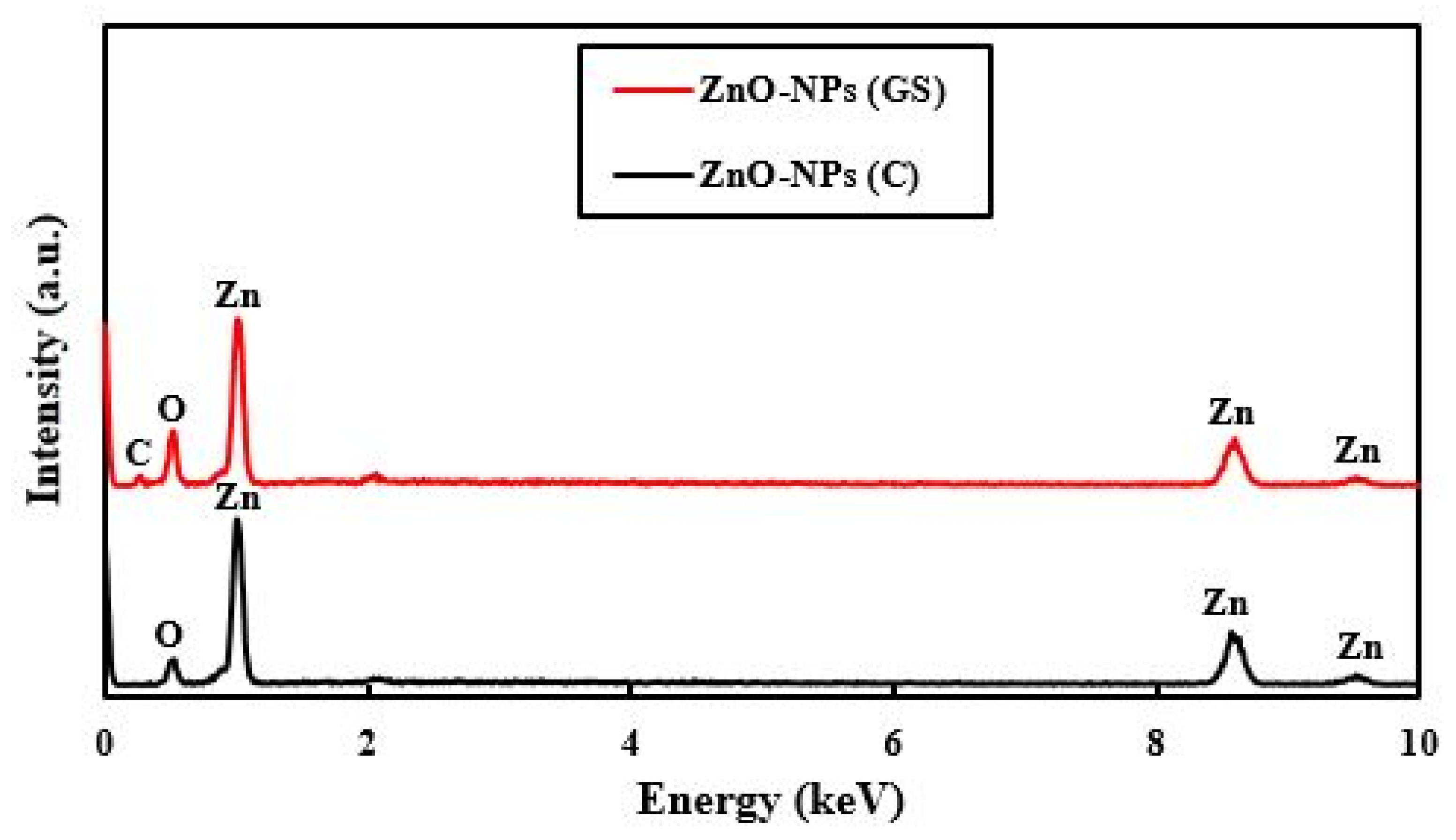
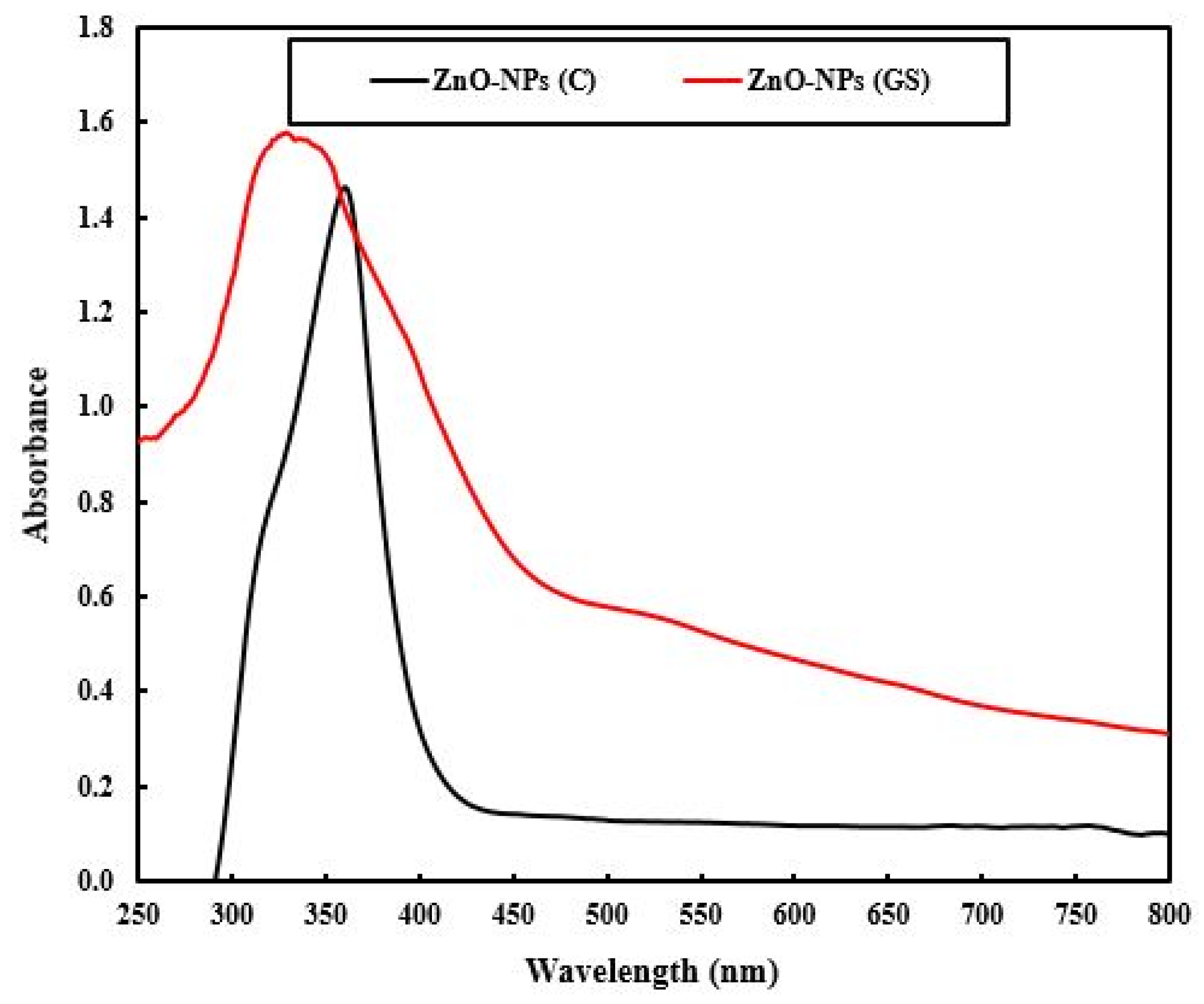
2.1. Characterization of the Fabricated ZnO-NPs
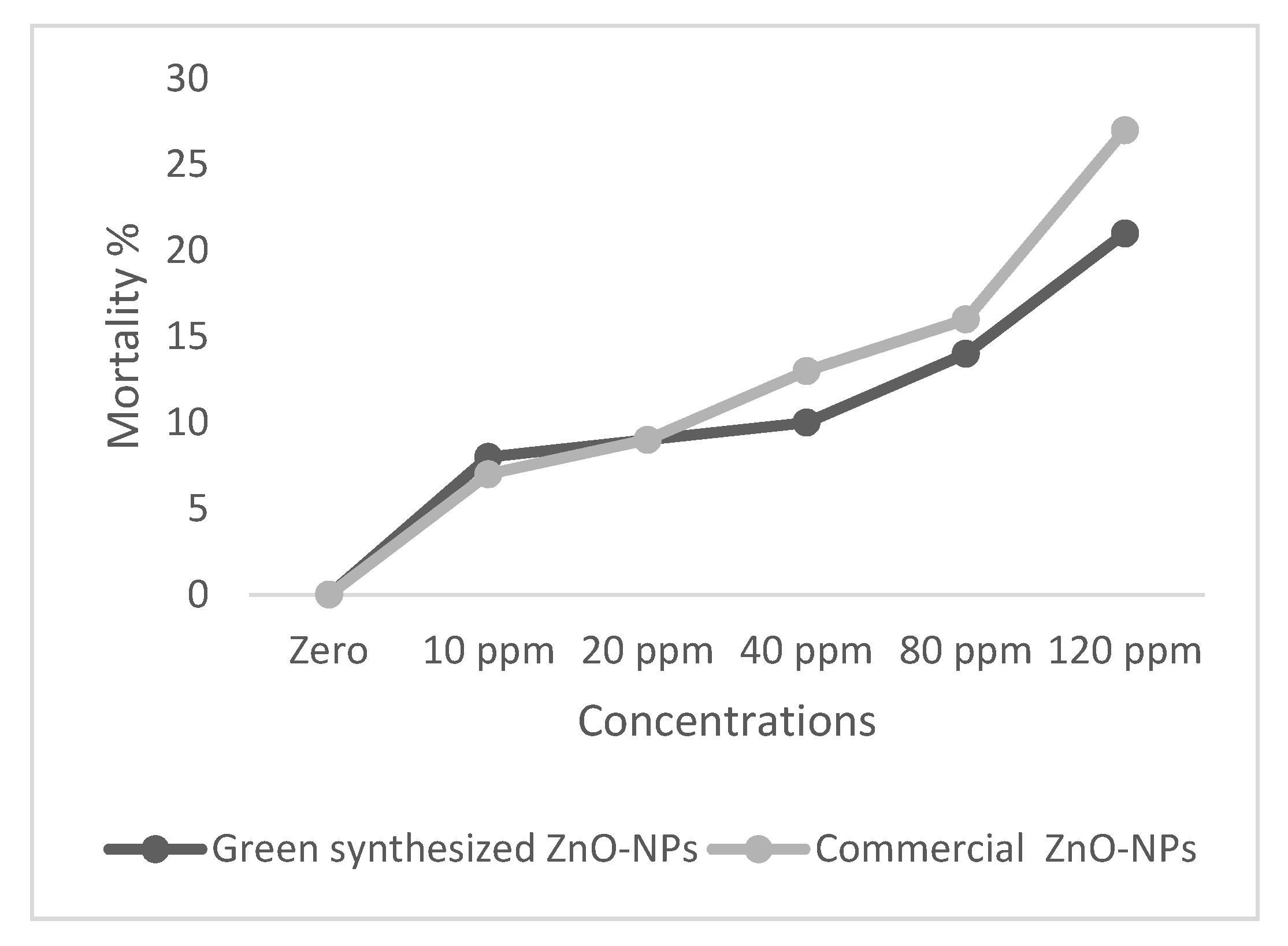
2.2. Toxicity Assessment of ZnO-NPs
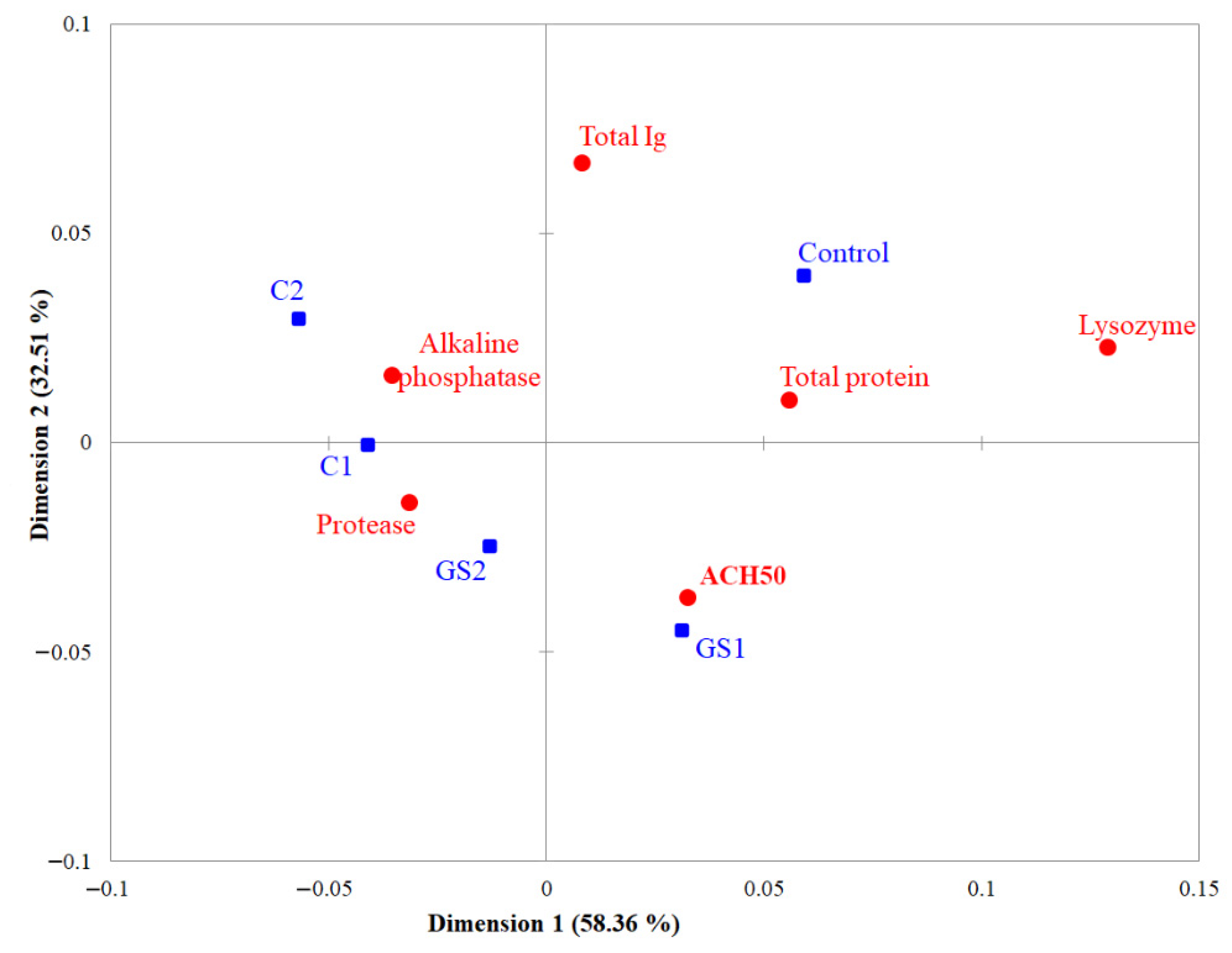
2.3. Skin Mucus Immunological Parameters
3. Discussion
4. Materials and Methods
4.1. Preparation of the Extract and Nanoparticles
4.2. Characterization of Fabricated NPs
4.3. Assessment of LC50 of ZnO-NPs
4.4. Fish Husbandry
4.5. Exposure Experiment
4.6. Sampling Process
4.7. Skin Mucus Immunological Parameters
4.7.1. Lysozyme Activity
4.7.2. Total Immunoglobulin (total Ig)
4.7.3. Alternative Complement Activity (ACH50)
4.7.4. Protease Activity
4.7.5. Alkaline Phosphatase Activity
4.7.6. Total Protein
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rossetti, R.; Brus, L.E. Picosecond resonance Raman scattering study of methylviologen reduction on the surface of photoexcited colloidal CdS crystallites. J. Phys. Chem. 1986, 90, 558–560. [Google Scholar] [CrossRef]
- Forouhar Vajargah, M.; Mohamadi Yalsuyi, A.; Hedayati, A.; Faggio, C. Histopathological lesions and toxicity in common carp (Cyprinus carpio L. 1758) induced by copper nanoparticles. Microsc. Res. Tech. 2018, 81, 724–729. [Google Scholar] [CrossRef]
- Forouhar Vajargah, M.; Mohamadi Yalsuyi, A.; Sattari, M.; Prokić, M.D.; Faggio, C. Effects of copper oxide nanoparticles (cuo-nps) on parturition time, survival rate and reproductive success of guppy fish, Poecilia reticulata. J. Clust. Sci. 2020, 31, 499–506. [Google Scholar] [CrossRef]
- Forouhar Vajargah, M.; Imanpoor, M.R.; Shabani, A.; Hedayati, A.; Faggio, C. Effect of long-term exposure of silver nanoparticles on growth indices, hematological and biochemical parameters and gonad histology of male goldfish (Carassius auratus gibelio). Microsc. Res. Tech. 2019, 82, 1224–1230. [Google Scholar] [CrossRef] [PubMed]
- Mohsenpour, R.; Mousavi-Sabet, H.; Hedayati, A.; Rezaei, A.; Yalsuyi, A.M.; Faggio, C. In vitro effects of silver nanoparticles on gills morphology of female Guppy (Poecilia reticulate) after a short-term exposure. Microsc. Res. Tech. 2020, 83, 1552–1557. [Google Scholar] [CrossRef]
- Vijayakumar, S.; Vaseeharan, B.; Sudhakaran, R.; Jeyakandan, J.; Ramasamy, P.; Sonawane, A.; Padhi, A.; Velusamy, P.; Anbu, P.; Faggio, C. Bioinspired zinc oxide nanoparticles using Lycopersicon esculentum for antimicrobial and anticancer applications. J. Clust. Sci. 2019, 30, 1465–1479. [Google Scholar] [CrossRef]
- Lowry, G.V.; Gregory, K.B.; Apte, S.C.; Lead, J.R. Transformations of nanomaterials in the environment. Environ. Sci. Technol. 2012, 7, 55–87. [Google Scholar] [CrossRef]
- Alarifi, S.; Ali, D.; Alkahtani, S.; Verma, A.; Ahamed, M.; Ahmed, M.; Alhadlaq, H.A. Induction of oxidative stress, DNA damage, and apoptosis in a malignant human skin melanoma cell line after exposure to zinc oxide nanoparticles. Int. J. Nanomed. 2013, 8, 983. [Google Scholar] [CrossRef] [Green Version]
- Ma, H.; Williams, P.L.; Diamond, S.A. Ecotoxicity of manufactured ZnO nanoparticles—A review. Environ. Pollut. 2013, 172, 76–85. [Google Scholar] [CrossRef]
- Malhotra, N.; Ger, T.R.; Uapipatanakul, B.; Huang, J.C.; Chen, K.H.C.; Hsiao, C. Der Review of copper and copper nanoparticle toxicity in fish. Nanomaterials 2020, 10, 1126. [Google Scholar] [CrossRef]
- Manke, A.; Wang, L.; Rojanasakul, Y. Mechanisms of nanoparticle-induced oxidative stress and toxicity. Biomed Res. Int. 2013, 2013, 942916. [Google Scholar] [CrossRef] [Green Version]
- Ilinskaya, A.N.; Dobrovolskaia, M.A. Immunosuppressive and anti-inflammatory properties of engineered nanomaterials. Br. J. Pharmacol. 2014, 171, 3988–4000. [Google Scholar] [CrossRef] [PubMed]
- Jovanović, B.; Palić, D.Š. Immunotoxicology of non-functionalized engineered nanoparticles in aquatic organisms with special emphasis on fish-Review of current knowledge, gap identification, and call for further research. Aquat. Toxicol. 2012, 118, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Mahboub, H.; Shahin, K.; Zaglool, A.; Roushdy, E.; Ahmed, S. Efficacy of nano zinc oxide dietary supplements on growth performance, immunomodulation and disease resistance of African catfish Clarias gariepinus. Dis. Aquat. Organ. 2020, 142, 147–160. [Google Scholar] [CrossRef] [PubMed]
- Li, X.F.; Wang, P.F.; Feng, C.C.L.; Liu, D.Q.; Chen, J.K.; Wu, F.C. Acute toxicity and hazardous concentrations of zinc to native freshwater organisms under different pH values in China. Bull. Environ. Contam. Toxicol. 2019, 103, 120–126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pagano, M.; Porcino, C.; Briglia, M.; Fiorino, E.; Vazzana, M.; Silvestro, S.; Faggio, C. The influence of exposure of cadmium chloride and zinc chloride on haemolymph and digestive gland cells from Mytilus galloprovincialis. Int. J. Environ. Res. 2017, 11, 207–216. [Google Scholar] [CrossRef]
- Yu, L.P.; Fang, T.; Xiong, D.W.; Zhu, W.T.; Sima, X.F. Comparative toxicity of nano-ZnO and bulk ZnO suspensions to zebrafish and the effects of sedimentation, OH production and particle dissolution in distilled water. J. Environ. Monit. 2011, 13, 1975–1982. [Google Scholar] [CrossRef]
- Blinova, I.; Ivask, A.; Heinlaan, M.; Mortimer, M.; Kahru, A. Ecotoxicity of nanoparticles of CuO and ZnO in natural water. Environ. Pollut. 2010, 158, 41–47. [Google Scholar] [CrossRef]
- Balta, S.; Sotto, A.; Luis, P.; Benea, L.; Van der Bruggen, B.; Kim, J. A new outlook on membrane enhancement with nanoparticles: The alternative of ZnO. J. Memb. Sci. 2012, 389, 155–161. [Google Scholar] [CrossRef]
- Wong, S.W.Y.; Leung, P.T.Y.; Djurišić, A.B.; Leung, K.M.Y. Toxicities of nano zinc oxide to five marine organisms: Influences of aggregate size and ion solubility. Anal. Bioanal. Chem. 2010, 396, 609–618. [Google Scholar] [CrossRef]
- Mortimer, M.; Kasemets, K.; Kahru, A. Toxicity of ZnO and CuO nanoparticles to ciliated protozoa Tetrahymena thermophila. Toxicology 2010, 269, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Brayner, R.; Dahoumane, S.A.; Yéprémian, C.; Djediat, C.; Meyer, M.; Couté, A.; Fiévet, F. ZnO nanoparticles: Synthesis, characterization, and ecotoxicological studies. Langmuir 2010, 26, 6522–6528. [Google Scholar] [CrossRef]
- Bystrzejewska-Piotrowska, G.; Golimowski, J.; Urban, P.L. Nanoparticles: Their potential toxicity, waste and environmental management. Waste Manag. 2009, 29, 2587–2595. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Jiang, Y.; Ding, Y.; Povey, M.; York, D. Investigation into the antibacterial behaviour of suspensions of ZnO nanoparticles (ZnO nanofluids). J. Nanoparticle Res. 2007, 9, 479–489. [Google Scholar] [CrossRef]
- Zhao, H.Z.; Lu, G.H.; Xia, J.; Jin, S.G. Toxicity of nanoscale CuO and ZnO to Daphnia magna. Chem. Res. Chinese Univ. 2012, 28, 209–213. [Google Scholar]
- Zeumer, R.; Galhano, V.; Monteiro, M.S.; Kuehr, S.; Knopf, B.; Meisterjahn, B.; Soares, A.M.V.M.; Loureiro, S.; Lopes, I.; Schlechtriem, C. Chronic effects of wastewater-borne silver and titanium dioxide nanoparticles on the rainbow trout (Oncorhynchus mykiss). Sci. Total Environ. 2020, 723, 137974. [Google Scholar] [CrossRef] [PubMed]
- Haghighat, F.; Kim, Y.; Sourinejad, I.; Yu, I.J.; Johari, S.A. Titanium dioxide nanoparticles affect the toxicity of silver nanoparticles in common carp (Cyprinus carpio). Chemosphere 2021, 262, 127805. [Google Scholar] [CrossRef] [PubMed]
- Abdelkhalek, A.; Al-Askar, A.A. Green synthesized ZnO nanoparticles mediated by Mentha spicata extract induce plant systemic resistance against Tobacco mosaic virus. Appl. Sci. 2020, 10, 5054. [Google Scholar] [CrossRef]
- Salem, S.S.; Fouda, A. Green synthesis of metallic nanoparticles and their prospective biotechnological applications: An Overview. Biol. Trace Elem. Res. 2020, 2020, 1–27. [Google Scholar] [CrossRef]
- Mahdavi, M.; Namvar, F.; Ahmad, M.B.; Mohamad, R. Green biosynthesis and characterization of magnetic iron oxide (Fe 3O4) nanoparticles using seaweed (Sargassum muticum) aqueous extract. Molecules 2013, 18, 5954–5964. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, H.; Venkat Kumar, S.; Rajeshkumar, S. A review on green synthesis of zinc oxide nanoparticles – An eco-friendly approach. Resour. Technol. 2017, 3, 406–413. [Google Scholar] [CrossRef]
- Mashjoor, S.; Alishahi, M.; Dezfuly, Z.T. Comparative toxicity assessment of chemical nanosilver and biosynthetic silver nanoparticles produced by marine macroalgae from the Persian Gulf in biomarker: Artemia nauplii. J. Vet. Res. 2019, 74, 73–82. [Google Scholar] [CrossRef]
- Aldalbahi, A.; Alterary, S.; Ali Abdullrahman Almoghim, R.; Awad, M.A.; Aldosari, N.S.; Fahad Alghannam, S.; Nasser Alabdan, A.; Alharbi, S.; Ali Mohammed Alateeq, B.; Abdulrahman Al Mohsen, A.; et al. Greener synthesis of zinc oxide nanoparticles: Characterization and multifaceted applications. Molecules 2020, 25, 4198. [Google Scholar] [CrossRef] [PubMed]
- Hao, L.; Chen, L. Oxidative stress responses in different organs of carp (Cyprinus carpio) with exposure to ZnO nanoparticles. Ecotoxicol. Environ. Saf. 2012, 80, 103–110. [Google Scholar] [CrossRef]
- Connolly, M.; Fernández, M.; Conde, E.; Torrent, F.; Navas, J.M.; Fernández-Cruz, M.L. Tissue distribution of zinc and subtle oxidative stress effects after dietary administration of ZnO nanoparticles to rainbow trout. Sci. Total Environ. 2016, 551, 334–343. [Google Scholar] [CrossRef] [PubMed]
- Chupani, L.; Niksirat, H.; Velíšek, J.; Stará, A.; Hradilová, Š.; Kolařík, J.; Panáček, A.; Zusková, E. Chronic dietary toxicity of zinc oxide nanoparticles in common carp (Cyprinus carpio L.): Tissue accumulation and physiological responses. Ecotoxicol. Environ. Saf. 2018, 147, 110–116. [Google Scholar] [CrossRef]
- Abdelazim, A.M.; Saadeldin, I.M.; Swelum, A.A.A.; Afifi, M.M.; Alkaladi, A. Oxidative stress in the muscles of the fish Nile tilapia caused by zinc oxide nanoparticles and its modulation by vitamins C and E. Oxid. Med. Cell. Longev. 2018, 2018, 6926712. [Google Scholar] [CrossRef] [PubMed]
- Marisa, I.; Matozzo, V.; Munari, M.; Binelli, A.; Parolini, M.; Martucci, A.; Franceschinis, E.; Brianese, N.; Marin, M.G. In vivo exposure of the marine clam Ruditapes philippinarum to zinc oxide nanoparticles: Responses in gills, digestive gland and haemolymph. Environ. Sci. Pollut. Res. 2016, 23, 15275–15293. [Google Scholar] [CrossRef]
- Vale, G.; Mehennaoui, K.; Cambier, S.; Libralato, G.; Jomini, S.; Domingos, R.F. Manufactured nanoparticles in the aquatic environment-biochemical responses on freshwater organisms: A critical overview. Aquat. Toxicol. 2016, 170, 162–174. [Google Scholar] [CrossRef]
- FAO. The State of World Fisheries and Aquaculture 2020: Sustainability in Action; FAO: Rome, Italy, 2020; ISBN 9789251326923. [Google Scholar]
- Bayrami, A.; Parvinroo, S.; Habibi-Yangjeh, A.; Rahim Pouran, S. Bio-extract-mediated ZnO nanoparticles: Microwave-assisted synthesis, characterization and antidiabetic activity evaluation. Artif. Cells, Nanomed. Biotechnol. 2018, 46, 730–739. [Google Scholar] [CrossRef]
- Mohammadi-Aloucheh, R.; Habibi-Yangjeh, A.; Bayrami, A.; Latifi-Navid, S.; Asadi, A. Green synthesis of ZnO and ZnO/CuO nanocomposites in Mentha longifolia leaf extract: Characterization and their application as anti-bacterial agents. J. Mater. Sci. Mater. Electron. 2018, 29, 13596–13605. [Google Scholar] [CrossRef]
- Pirhashemi, M.; Habibi-Yangjeh, A. Ultrasonic-assisted preparation of novel ternary ZnO/Ag3VO4/Ag2CrO4 nanocomposites and their enhanced visible-light activities in degradation of different pollutants. Solid State Sci. 2016, 55, 58–68. [Google Scholar] [CrossRef]
- Moore, M.N. Do nanoparticles present ecotoxicological risks for the health of the aquatic environment? Environ. Int. 2006, 32, 967–976. [Google Scholar] [CrossRef] [PubMed]
- Chupani, L.; Zusková, E.; Niksirat, H.; Panáček, A.; Lünsmann, V.; Haange, S.B.; von Bergen, M.; Jehmlich, N. Effects of chronic dietary exposure of zinc oxide nanoparticles on the serum protein profile of juvenile common carp (Cyprinus carpio L.). Sci. Total Environ. 2017, 579, 1504–1511. [Google Scholar] [CrossRef] [PubMed]
- Sula, E.; Aliko, V.; Barceló, D.; Faggio, C. Combined effects of moderate hypoxia, pesticides and PCBs upon crucian carp fish, Carassius carassius, from a freshwater lake- in situ ecophysiological approach. Aquat. Toxicol. 2020, 228, 105644. [Google Scholar] [CrossRef]
- Liew, H.J.; Pelle, A.; Chiarella, D.; Faggio, C.; Tang, C.H.; Blust, R.; De Boeck, G. Common carp, Cyprinus carpio, prefer branchial ionoregulation at high feeding rates and kidney ionoregulation when food supply is limited: Additional effects of cortisol and exercise. Fish Physiol. Biochem. 2020, 46, 451–469. [Google Scholar] [CrossRef] [PubMed]
- Fiorino, E.; Sehonova, P.; Plhalova, L.; Blahova, J.; Svobodova, Z.; Faggio, C. Effects of glyphosate on early life stages: Comparison between Cyprinus carpio and Danio rerio. Environ. Sci. Pollut. Res. 2018, 25, 8542–8549. [Google Scholar] [CrossRef] [PubMed]
- Pietrzak, E.; Mazurkiewicz, J.; Slawinska, A. Innate immune responses of skin mucosa in common carp (Cyprinus carpio) fed a diet supplemented with galactooligosaccharides. Animals 2020, 10, 438. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krotman, Y.; Yergaliyev, T.M.; Alexander Shani, R.; Avrahami, Y.; Szitenberg, A. Dissecting the factors shaping fish skin microbiomes in a heterogeneous inland water system. Microbiome 2020, 8, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Cabillon, N.A.R.; Lazado, C.C. Mucosal barrier functions of fish under changing environmental conditions. Fishes 2019, 4, 2. [Google Scholar] [CrossRef] [Green Version]
- Swain, P.; Dash, S.; Sahoo, P.K.; Routray, P.; Sahoo, S.K.; Gupta, S.D.; Meher, P.K.; Sarangi, N. Non-specific immune parameters of brood Indian major carp Labeo rohita and their seasonal variations. Fish Shellfish Immunol. 2007, 22, 38–43. [Google Scholar] [CrossRef]
- Arasu, A.; Kumaresan, V.; Sathyamoorthi, A.; Palanisamy, R.; Prabha, N.; Bhatt, P.; Roy, A.; Thirumalai, M.K.; Gnanam, A.J.; Pasupuleti, M.; et al. Fish lily type lectin-1 contains β-prism architecture: Immunological characterization. Mol. Immunol. 2013, 56, 497–506. [Google Scholar] [CrossRef]
- Patel, M.; Ashraf, M.S.; Siddiqui, A.J.; Ashraf, S.A.; Sachidanandan, M.; Snoussi, M.; Adnan, M.; Hadi, S. Profiling and role of bioactive molecules from puntius sophore (Freshwater/brackish fish) skin mucus with its potent antibacterial, antiadhesion, and antibiofilm activities. Biomolecules 2020, 10, 920. [Google Scholar] [CrossRef] [PubMed]
- Adam, N.; Vergauwen, L.; Blust, R.; Knapen, D. Gene transcription patterns and energy reserves in Daphnia magna show no nanoparticle specific toxicity when exposed to ZnO and CuO nanoparticles. Environ. Res. 2015, 138, 82–92. [Google Scholar] [CrossRef]
- Falfushynska, H.; Gnatyshyna, L.; Yurchak, I.; Sokolova, I.; Stoliar, O. The effects of zinc nanooxide on cellular stress responses of the freshwater mussels Unio tumidus are modulated by elevated temperature and organic pollutants. Aquat. Toxicol. 2015, 162, 82–93. [Google Scholar] [CrossRef] [PubMed]
- Mansouri, B.; Johari, S.A.; Azadi, N.A.; Sarkheil, M. Effects of waterborne ZnO nanoparticles and Zn2+ions on the gills of rainbow trout (Oncorhynchus mykiss): Bioaccumulation, histopathological and ultrastructural changes. Turk. J. Fish. Aquat. Sci. 2018, 18, 739–746. [Google Scholar] [CrossRef]
- Torrealba, D.; More-Bayona, J.A.; Wakaruk, J.; Barreda, D.R. Innate immunity provides biomarkers of health for teleosts exposed to nanoparticles. Front. Immunol. 2019, 10, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Mir, A.H.; Qamar, A.; Qadir, I.; Naqvi, A.H.; Begum, R. Accumulation and trafficking of zinc oxide nanoparticles in an invertebrate model, Bombyx mori, with insights on their effects on immuno-competent cells. Sci. Rep. 2020, 10, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.; Tvarijonaviciute, A.; Trindade, T.; Soares, A.M.V.M.; Tort, L.; Teles, M. Can non-invasive methods be used to assess effects of nanoparticles in fish? Ecol. Indic. 2018, 95, 1118–1127. [Google Scholar] [CrossRef]
- Hawkins, A.D.; Thornton, C.; Steevens, J.A.; Willett, K.L. Alteration in Pimephales promelas mucus production after exposure to nanosilver or silver nitrate. Environ. Toxicol. Chem. 2014, 33, 2869–2872. [Google Scholar] [CrossRef]
- Cherian, T.; Ali, K.; Fatima, S.; Saquib, Q.; Ansari, S.M.; Alwathnani, H.A.; Al-Khedhairy, A.A.; Al-Shaeri, M.; Musarrat, J. Myristica fragrans bio-active ester functionalized ZnO nanoparticles exhibit antibacterial and antibiofilm activities in clinical isolates. J. Microbiol. Methods 2019, 166, 105716. [Google Scholar] [CrossRef] [PubMed]
- Guardiola, F.A.; Cuesta, A.; Abellán, E.; Meseguer, J.; Esteban, M.A. Comparative analysis of the humoral immunity of skin mucus from several marine teleost fish. Fish Shellfish Immunol. 2014, 40, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Rashidian, G.; Kajbaf, K.; Prokić, M.D.; Faggio, C. Extract of common mallow (Malvae sylvestris) enhances growth, immunity, and resistance of rainbow trout (Oncorhynchus mykiss) fingerlings against Yersinia ruckeri infection. Fish Shellfish Immunol. 2020, 96, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Hoseinifar, S.H.; Shakouri, M.; Yousefi, S.; Van Doan, H.; Shafiei, S.; Yousefi, M.; Mazandarani, M.; Torfi Mozanzadeh, M.; Tulino, M.G.; Faggio, C. Humoral and skin mucosal immune parameters, intestinal immune related genes expression and antioxidant defense in rainbow trout (Oncorhynchus mykiss) fed olive (Olea europea L.) waste. Fish Shellfish Immunol. 2020, 100, 171–178. [Google Scholar] [CrossRef]
- Abdelfatah, E.N.; Mahboub, H.H.H. Studies on the effect of Lactococcus garvieae of dairy origin on both cheese and Nile tilapia (O. niloticus). Int. J. Vet. Sci. Med. 2018, 6, 201–207. [Google Scholar] [CrossRef] [Green Version]
- Fernández-Alacid, L.; Sanahuja, I.; Ordóñez-Grande, B.; Sánchez-Nuño, S.; Viscor, G.; Gisbert, E.; Herrera, M.; Ibarz, A. Skin mucus metabolites in response to physiological challenges: A valuable non-invasive method to study teleost marine species. Sci. Total Environ. 2018, 644, 1323–1335. [Google Scholar] [CrossRef]
- Wang, T.; Long, X.; Liu, Z.; Cheng, Y.; Yan, S. Effect of copper nanoparticles and copper sulphate on oxidation stress, cell apoptosis and immune responses in the intestines of juvenile Epinephelus coioides. Fish Shellfish Immunol. 2015, 44, 674–682. [Google Scholar] [CrossRef]
- Ates, M.; Demir, V.; Arslan, Z.; Kaya, H.; Yılmaz, S.; Camas, M. Chronic exposure of tilapia (Oreochromis niloticus) to iron oxide nanoparticles: Effects of particle morphology on accumulation, elimination, hematology and immune responses. Aquat. Toxicol. 2016, 177, 22–32. [Google Scholar] [CrossRef] [Green Version]
- Kaya, H.; Aydin, F.; Gürkan, M.; Yilmaz, S.; Ates, M.; Demir, V.; Arslan, Z. A comparative toxicity study between small and large size zinc oxide nanoparticles in tilapia (Oreochromis niloticus): Organ pathologies, osmoregulatory responses and immunological parameters. Chemosphere 2016, 144, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Wang, Q.; Huang, Z.; Ding, L.; Xu, Z. Immunoglobulins, mucosal immunity and vaccination in teleost fish. Front. Immunol. 2020, 11, 2597. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Sharma, N.; Maitra, S.S. In vitro and in vivo toxicity assessment of nanoparticles. Int. Nano Lett. 2017, 7, 243–256. [Google Scholar] [CrossRef] [Green Version]
- Jiang, J.; Pi, J.; Cai, J. The advancing of zinc oxide nanoparticles for biomedical applications. Bioinorg. Chem. Appl. 2018, 2018, 1062562. [Google Scholar] [CrossRef]
- Upadhyay, P.K.; Jain, V.K.; Sharma, S.; Shrivastav, A.K.; Sharma, R. Green and chemically synthesized ZnO nanoparticles: A comparative study. In Proceedings of the IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2020. [Google Scholar]
- Lee, J.; Kim, J.; Shin, Y.; Ryu, J.; Eom, I.; Lee, J.S.; Kim, Y.; Kim, P.; Choi, K.; Lee, B. cheun Serum and ultrastructure responses of common carp (Cyprinus carpio L.) during long-term exposure to zinc oxide nanoparticles. Ecotoxicol. Environ. Saf. 2014, 104, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Muralisankar, T.; Bhavan, P.S.; Radhakrishnan, S.; Seenivasan, C.; Manickam, N.; Srinivasan, V. Dietary supplementation of zinc nanoparticles and its influence on biology, physiology and immune responses of the freshwater prawn, Macrobrachium rosenbergii. Biol. Trace Elem. Res. 2014, 160, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Sanahuja, I.; Ibarz, A. Skin mucus proteome of gilthead sea bream: A non-invasive method to screen for welfare indicators. Fish Shellfish Immunol. 2015, 46, 426–435. [Google Scholar] [CrossRef] [PubMed]
- Sanahuja, I.; Fernández-Alacid, L.; Ordóñez-Grande, B.; Sánchez-Nuño, S.; Ramos, A.; Araujo, R.M.; Ibarz, A. Comparison of several non-specific skin mucus immune defences in three piscine species of aquaculture interest. Fish Shellfish Immunol. 2019, 89, 428–436. [Google Scholar] [CrossRef]
- Safari, R.; Hoseinifar, S.H.; Van Doan, H.; Dadar, M. The effects of dietary Myrtle (Myrtus communis) on skin mucus immune parameters and mRNA levels of growth, antioxidant and immune related genes in zebrafish (Danio rerio). Fish Shellfish Immunol. 2017, 66, 264–269. [Google Scholar] [CrossRef]
- Ellis, A.E. Lysozyme assays. Tech. Fish Immunol. 1990, 1, 101–103. [Google Scholar]
- Siwicki AK, A.D. Nonspecific defense mechanisms assay in fish: II. lmmunoglobulin levels in fish sera measured by polyethylene glycol and spectrophotometric methods in microtiter plates serum. Vet. Immunol. Immunopathol. 1994, 1994, 105–111. [Google Scholar]
- Yano, T. The Fish Immune System: Organism, Pathogen, and Environment; The Nonspecific Immune System: Humoral Defense; Academic Press: San Diego, CA, USA, 1996. [Google Scholar]
- Ross, N.W.; Firth, K.J.; Wang, A.; Burka, J.F.; Johnson, S.C. Changes in hydrolytic enzyme activities of naive Atlantic salmon Salmo salar skin mucus due to infection with the salmon louse Lepeophtheirus salmonis and cortisol implantation. Dis. Aquat. Organ. 2000, 41, 43–51. [Google Scholar] [CrossRef]




| Concentration (ppm) | ||||
|---|---|---|---|---|
| Point | 24 h | 48 h | 72 h | 96 h |
| LC30 | 230.36 | 125.95 | 73.08 | 32.1 |
| LC50 | 312.51 | 184.51 | 117.62 | 59.95 |
| LC70 | 394.66 | 243.08 | 162.16 | 87.49 |
| LC90 | 513.27 | 327.64 | 226.47 | 127.26 |
| LC99 | 676.94 | 444.33 | 315.22 | 182.14 |
| Concentration (ppm) | ||||
|---|---|---|---|---|
| Point | 24 h | 48 h | 72 h | 96 h |
| LC30 | 145.76 | 92.46 | 43.9 | 39.31 |
| LC50 | 179.51 | 161.43 | 87.84 | 78.9 |
| LC70 | 213.26 | 230.4 | 131.87 | 118.49 |
| LC90 | 261.99 | 329.98 | 195.22 | 175 |
| LC99 | 329.23 | 467.39 | 282.77 | 254.52 |
| Treatment | Control | GS1 | GS2 | C1 | C2 |
|---|---|---|---|---|---|
| Lysozyme (U mL−1) | 9.89 ± 0.37 a | 8.07 ± 0.21 b | 6.71 ± 0.38 c | 5.83 ± 0.21 cd | 5.4 ± 0.18 d |
| ACH50 (U mL−1) | 32.88 ± 1.31 a | 33.64 ± 1.32 a | 27.93 ± 0.87 b | 25.69 ± 0.33 bc | 24.11 ± 0.70 c |
| Alkaline phosphatase (U L−1) | 27.61 ± 0.70 a | 24.96 ± 0.61 b | 24.85 ± 0.57 b,c | 22.89 ± 0.40 c | 23.60 ± 0.85 b,c |
| Protease (U mL−1) | 42.58 ± 1.58 a | 42.84 ± 1.41 a | 38.04 ± 0.77 b | 38.38 ± 1.00 b | 36.21 ± 1.04 b |
| Total Ig (mg mL−1) | 19.33 ± 0.37 a | 15.67 ± 0.37 b | 13.47 ± 0.32 c | 14.47 ± 0.40 c,d | 14.70 ± 0.29 b,d |
| Total protein (mg mL−1) | 3.04 ± 0.08 a | 2.64 ± 0.07 b | 2.35 ± 0.07 c | 2.13 ± 0.04 d | 2.01 ± 0.06 d |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rashidian, G.; Lazado, C.C.; Mahboub, H.H.; Mohammadi-Aloucheh, R.; Prokić, M.D.; Nada, H.S.; Faggio, C. Chemically and Green Synthesized ZnO Nanoparticles Alter Key Immunological Molecules in Common Carp (Cyprinus carpio) Skin Mucus. Int. J. Mol. Sci. 2021, 22, 3270. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22063270
Rashidian G, Lazado CC, Mahboub HH, Mohammadi-Aloucheh R, Prokić MD, Nada HS, Faggio C. Chemically and Green Synthesized ZnO Nanoparticles Alter Key Immunological Molecules in Common Carp (Cyprinus carpio) Skin Mucus. International Journal of Molecular Sciences. 2021; 22(6):3270. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22063270
Chicago/Turabian StyleRashidian, Ghasem, Carlo C. Lazado, Heba H. Mahboub, Ramin Mohammadi-Aloucheh, Marko D. Prokić, Hend S. Nada, and Caterina Faggio. 2021. "Chemically and Green Synthesized ZnO Nanoparticles Alter Key Immunological Molecules in Common Carp (Cyprinus carpio) Skin Mucus" International Journal of Molecular Sciences 22, no. 6: 3270. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms22063270








