Effect of Dissolved Humic Substances on the Photochemical Degradation Rate of 1-Aminopyrene and Atrazine
Abstract
:Introduction
Experimental
Materials
1-Aminopyrene and atrazine photo-degradation
Results and Discussion
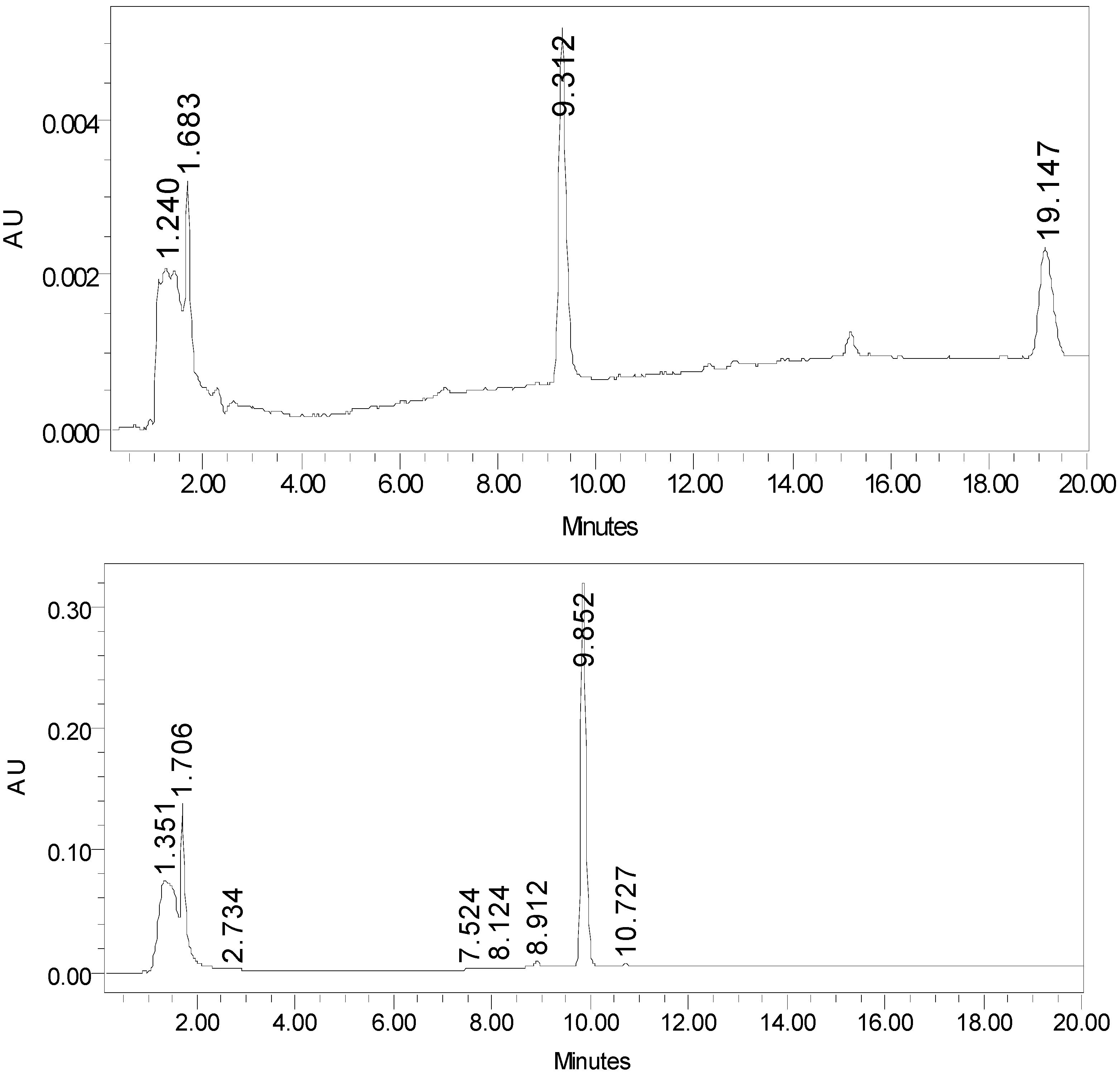
Separation of 1-AP, atrazine and their photoproducts
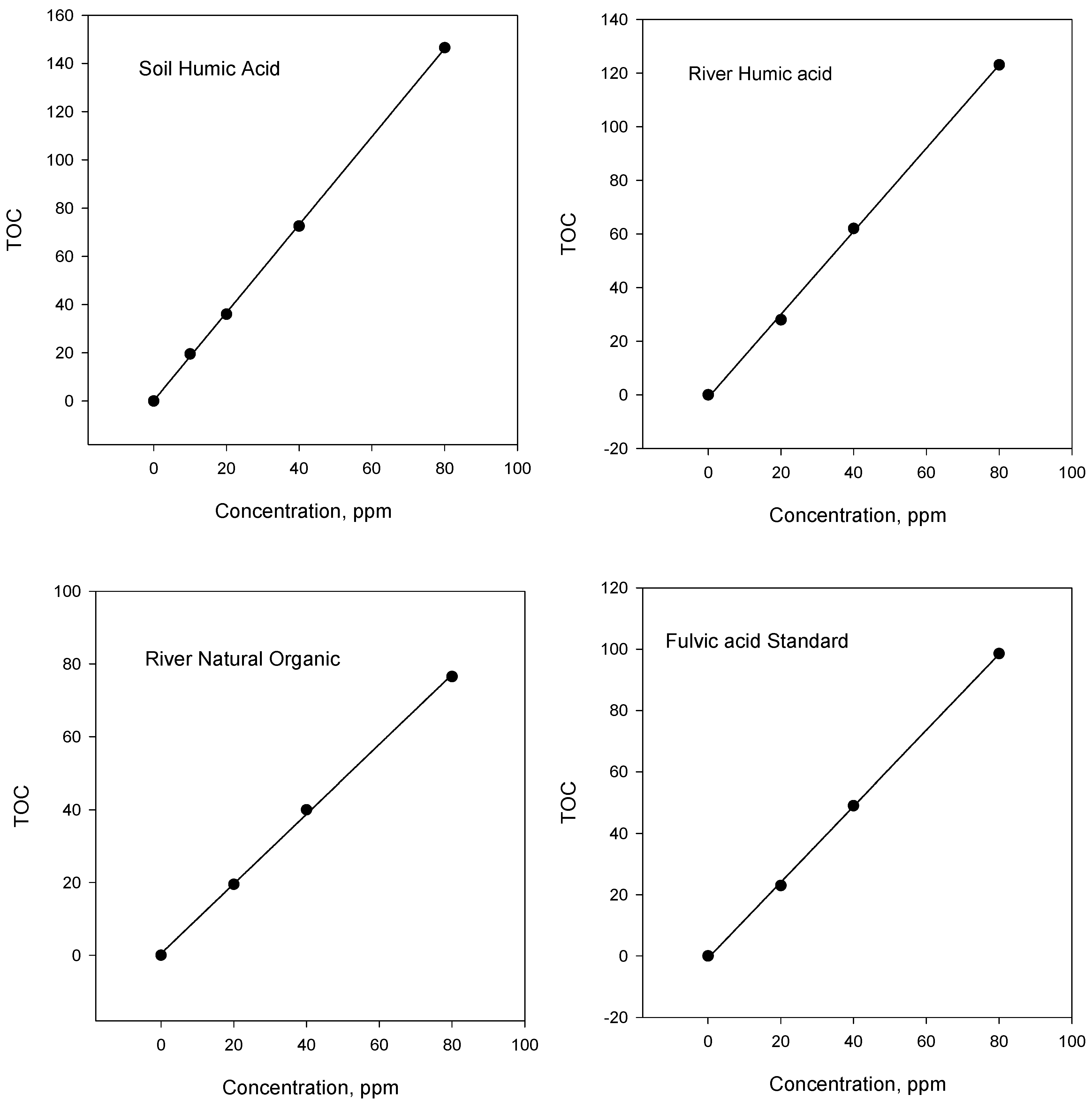
Total organic carbon content of HS
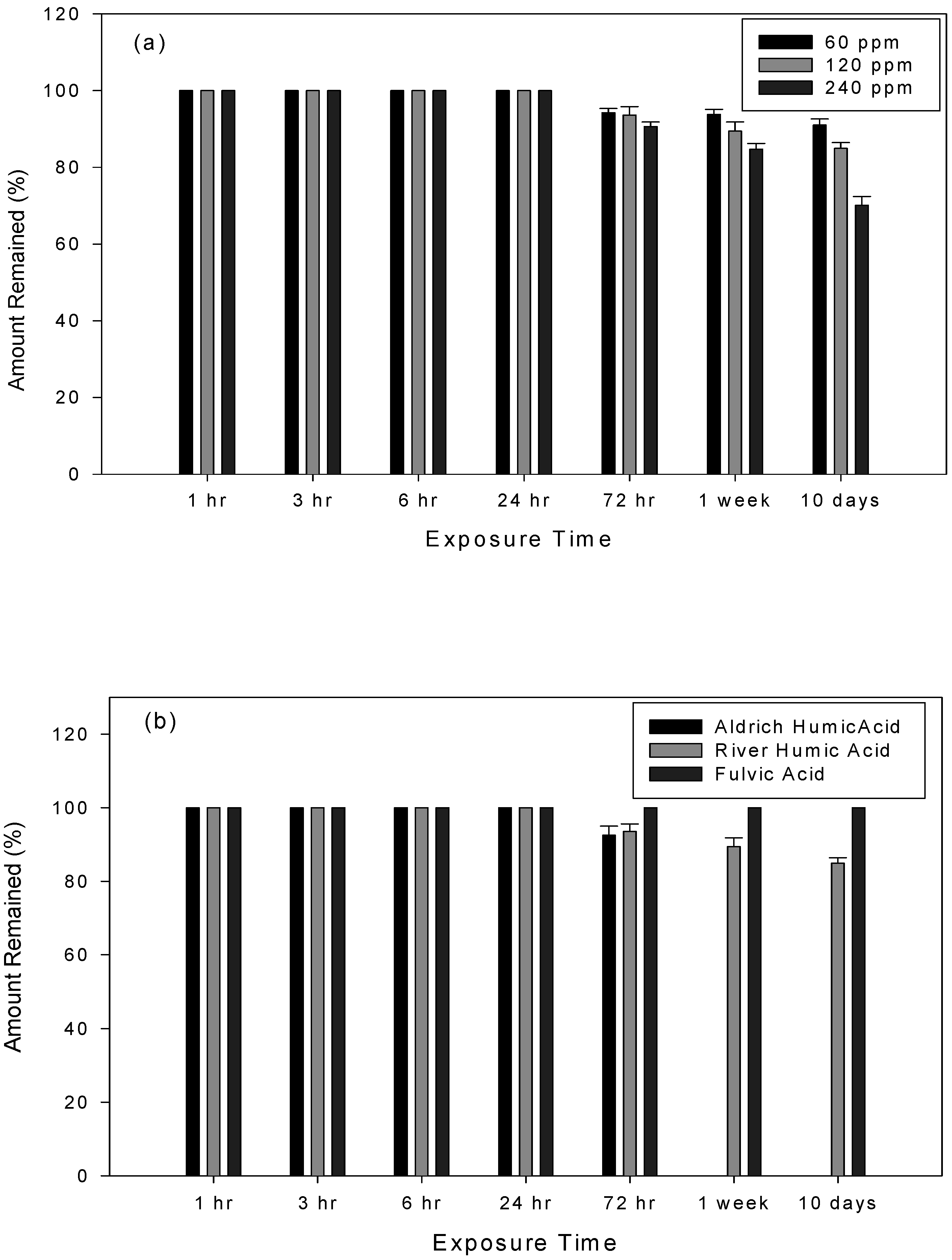
The effect of river water on the photolysis rate of 1-AP
The effect of commercial HS types and concentration on the photolysis rate of 1-AP

| HS concentration, ppm | ||||
| HS Type | 20 | 40 | 80 | 0 |
| 1 mM PBS (pH 7.0) | 17.4±1.1 | |||
| Aldrich Humic Acid | 3.5±0.5 | 3.3±0.5 | 8.1±0.7 | |
| River Humic Acid | 6.7±0.6 | 6.5±0.6 | 6.9±0.7 | |
| River Fulvic Acid | 16.0±1.3 | 14.9±1.2 | 11.6±1.2 | |
| River Natural Organic | 10.7±1.1 | 9.7±1.2 | 8.2±1.1 | |
| Fulvic Acid | 10.6±1.4 | 10.4±1.2 | 10.2±1.2 | |
| Soil Humic Acid | 7.7±0.5 | 26.0±1.6 | 63.0±4.3 | |
The effect of HS types and concentration on the photolysis rate of atrazine
Acknowledgements.
References
- Matthews, J. C.; Ekuta, J. E.; Drake, K. D.; Watson, C. F.; Benson, W. H. Nature and extent of the interactions of humic acids with water treatment algicide and a fungicide. Chemosphere 1995, 30, 1565–1572. [Google Scholar] [CrossRef]
- Dawson, H. J.; Hrutfiord, B. F.; Zasoski, R. J.; and Ugolini, F. C. The molecular weight and origin of yellow organic acids. Soil Sci. 1981, 132, 191–199. [Google Scholar] [CrossRef]
- Thurman, E. M. Organic Geochemistry of Natural WatersMartinus Nijhoff/Dr. Junk, W. Publishers, 1985, 1st ed. [Google Scholar]
- Cooper, W. J.; Zika, R. G.; Petasne, R. G; Fischer, A. M. Sunlight-induced photochemistry pf humic substances in natural waters: major reactive species. In Aquatic Humic Substances: Influence on Fate and Treatment of Pollutants; Suffet, I. H., MacCarthy, P., Eds.; American Chemical Society: Washington, DC, 1989; pp. 333–362. [Google Scholar]
- Frimmel, F. R.; Hessler, D. P. Photochemical degradation of triazine and anilide pesticides in natural waters. In Aquatic and Surface Photochemistry; Relz, G. R., Zepp, R. G., Crosby, D. G., Eds.; Lewis Publishers: Boca Raton, FL, 1994; pp. 137–147. [Google Scholar]
- Zepp, R. G. Environmental photoprocesses involving natural organic matter. In Humic Substances and Their Role in the Environment; Frimmel, F.H., Christman, R. F., Eds.; Wiley: New York, 1988; pp. 193–214. [Google Scholar]
- Bushaw, K. L.; Zepp, R. G.; Tarr, M. A.; Schulz-Jander, D.; Bourbonniere, R. A.; Hodson, R. E.; Miller, W. L.; Bronk, D. A.; Moran, M.A. Photochemical release of biologically available nitrogen from aquatic dissolved organic matter. Nature 1996, 381, 404–407. [Google Scholar] [CrossRef]
- IARC (International Agency for Research on Cancer). Polynuclear aromatic compounds. Part I: chemical, environmental and experimental data. Lyon, 1983. [Google Scholar]
- U.S. Department of Health and Human Services. P.H.S., ATSDR Toxicological Profile for Polycyclic Aromatic Hydrocarbons (PAHs). Atlanta, GA, 1995. [Google Scholar]
- Keith, L. H.; Telliard, W. A. Priority pollutants I – A perspective view. Environ. Sci. Technol. 1979, 13, 15. [Google Scholar]
- Landrum, P. F.; Giesy, J. P.; Oris, J. T.; Allred, P. M. Oil in freshwater: chemistry, biology Countermeasure Technology; Vandermeulen, J.H., Hrudy, H., Eds.; Pergamon: Elmsford, USA, 1987; p. 304. [Google Scholar]
- McConkey, B. J.; Duxbury, C. L.; Dixon, D. G.; Greenberg, B. M. Toxicity of a PAH photooxidation product to the bacteria photobacterium phosphoreum and the Duckeweed lemna gibba: effects of phenanthrene and its primary photoproduct, phenanthrenequinenone. Environ. Toxicol. Chem. 1997, 16, 892–899. [Google Scholar] [CrossRef]
- Hatch, A. C.; Burton, G. A., Jr. Effects of photoinduced toxicity of fluoranthene on amphibian embryos and larvae. Environ. Toxicol. Chem. 1998, 17, 1777–1785. [Google Scholar] [CrossRef]
- Fujikawa, K.; Fort, F.L.; Samejima, K.; Sakamoto, Y. Genotoxic potency in Drosophila melanogaster of selected aromatic amines and polycyclic aromatic hydrocarbons as assayed in the DNA repair test. Mutat. Res. 1993, 290(2), 175–182. [Google Scholar] [CrossRef] [PubMed]
- Lafi, A; Parry, J. M. Chromosome aberrations induced by nitro-, nitroso- and aminopyrenes in cultured Chinese hamster cells. Mutagenesis 1987, 2(1), 23–26. [Google Scholar] [CrossRef] [PubMed]
- Dong, S.; Hwang, H.-M.; Harrison, C.; Holloway, L.; Shi, X.; Yu, H. UVA light-induced DNA cleavage by selected polycyclic aromatic hydrocarbons. Bull. Environ. Contam. Toxicol. 2000, 64, 467–476. [Google Scholar] [CrossRef] [PubMed]
- Hapeman-Somich, C.J. Pesticide Transformation Products; Somasundaran, Ed.; ACS Symp. Series 459; Washington DC, 1991; Chap. 10. [Google Scholar]
- Fallon, R. D. Evidence of hydrolytic route for anaerobic cyanide degradation. Appl. Environ. Microbiol. 1992, 58, 3163–3164. [Google Scholar] [PubMed]
© 2002 by MDPI (http://www.mdpi.org).
Share and Cite
Zeng, K.; Hwang, H.-m.; Yuzuri, H. Effect of Dissolved Humic Substances on the Photochemical Degradation Rate of 1-Aminopyrene and Atrazine. Int. J. Mol. Sci. 2002, 3, 1048-1057. https://0-doi-org.brum.beds.ac.uk/10.3390/i3101048
Zeng K, Hwang H-m, Yuzuri H. Effect of Dissolved Humic Substances on the Photochemical Degradation Rate of 1-Aminopyrene and Atrazine. International Journal of Molecular Sciences. 2002; 3(10):1048-1057. https://0-doi-org.brum.beds.ac.uk/10.3390/i3101048
Chicago/Turabian StyleZeng, Kui, Huey-min Hwang, and Hongtao Yuzuri. 2002. "Effect of Dissolved Humic Substances on the Photochemical Degradation Rate of 1-Aminopyrene and Atrazine" International Journal of Molecular Sciences 3, no. 10: 1048-1057. https://0-doi-org.brum.beds.ac.uk/10.3390/i3101048




