Interaction of Plant Epicuticular Waxes and Extracellular Esterases of Curvularia eragrostidis during Infection of Digitaria sanguinalis and Festuca arundinacea by the Fungus
Abstract
:1. Introduction
2. Results and Discussion
2.1 Results
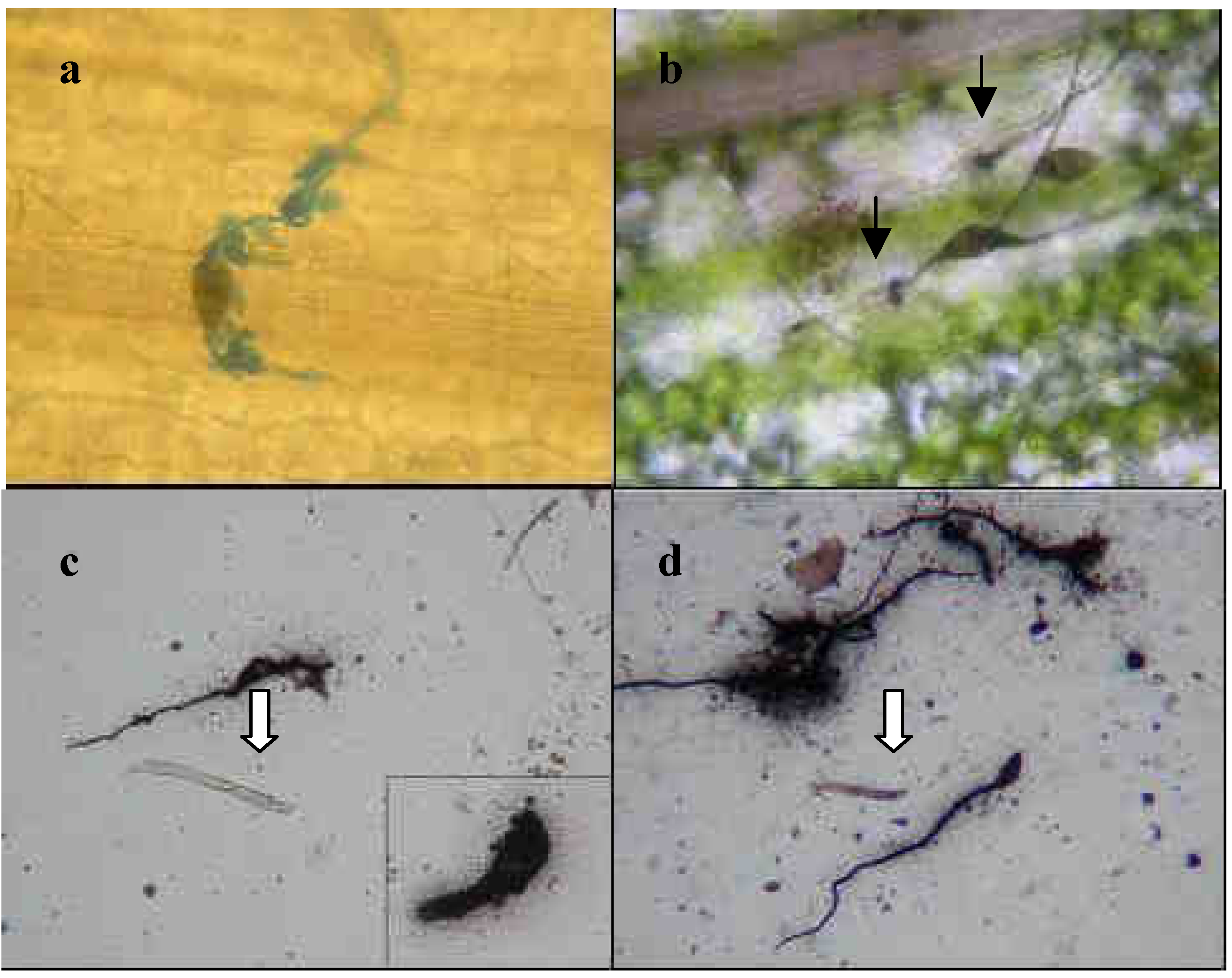
2.1.1 Localization of esterase secreted by conidia
2.1.2 Conidia adhesion
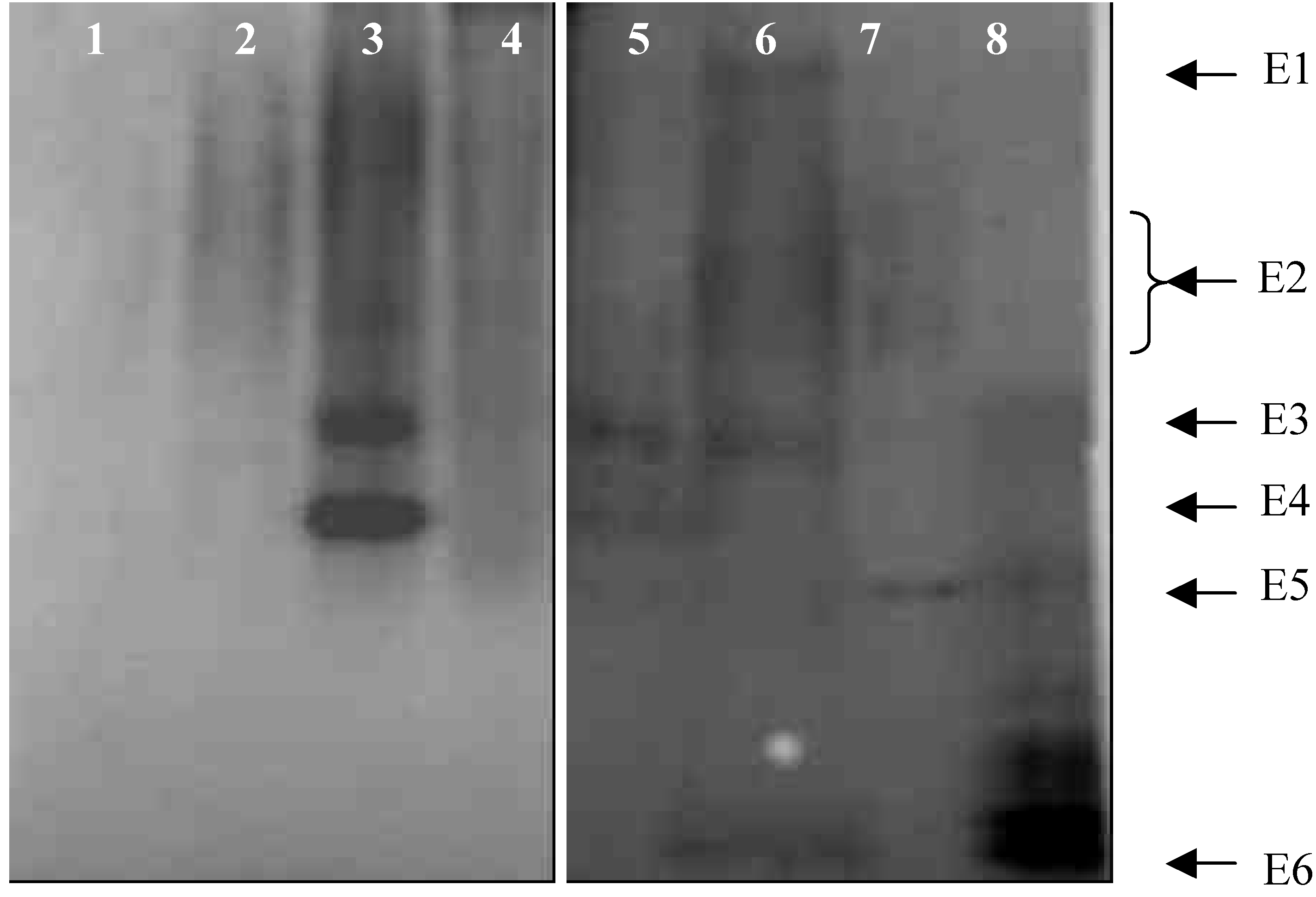
2.1.3 Extracellular esterase isoenzymes from germinating spore
- Extracellular esterase was extracted with water as extracting solution.
- 0.1 M phosphates buffer (pH 7.0) as extracting solution.
- Extracting solution was 0.1% Tween-20 in phosphate buffer.
- 0.1% SDS in phosphate buffer as extracting solution.
- Conidia germinated for 6 h on glass surface and extracellular esterases extracted.
- Conidia germinated for 6 h on leaf surface of D. sanguinalis and extracellular esterase of conidia were collected.
- Intracellular esterase isoenzymes of conidia germinated for 6 h, with extracellular material removed.
- Esterase isoenzymes from leaves of D. sanguinalis.
- Extracellular esterase was extracted with water as extracting solution.
- 0.1 M phosphates buffer (pH 7.0) as extracting solution.
- Extracting solution was 0.1% Tween-20 in phosphate buffer.
- 0.1% SDS in phosphate buffer as extracting solution.
- Conidia germinated for 6 h on glass surface and extracellular esterases extracted.
- Conidia germinated for 6 h on leaf surface of D. sanguinalis and extracellular esterase of conidia were collected.
- Intracellular esterase isoenzymes of conidia germinated for 6 h, with extracellular material removed.
- Esterase isoenzymes from leaves of D. sanguinalis.
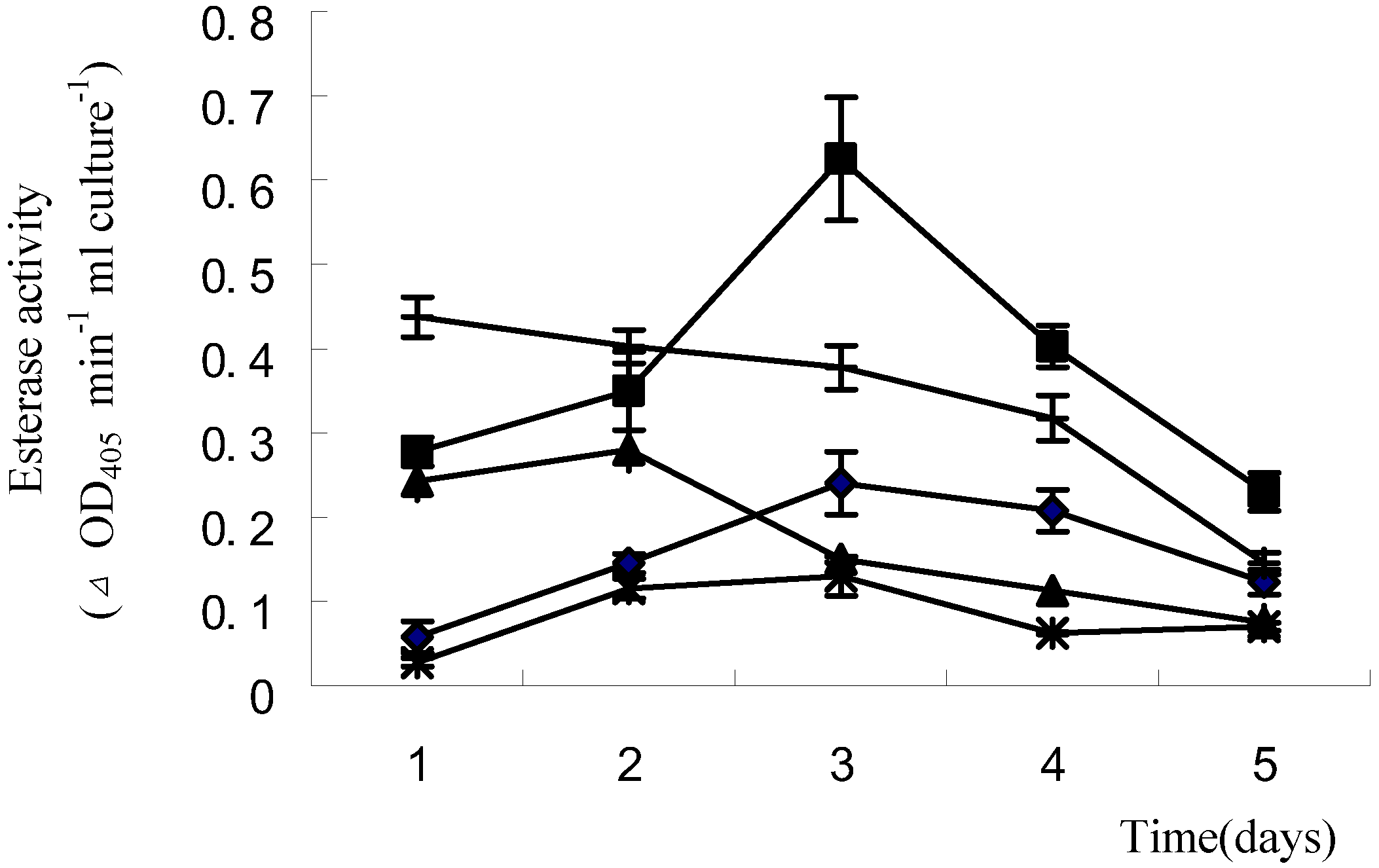
2.1.4 Induction of esterase activity by waxes
2.1.5 Degradation of two plants waxes by extracellular esterase of C. eragrostidis conidia
2.2 Discussion
2.3 Conclusion
3.Experimental Section
3.1 Plants and Fungal Material
3.2 Assay of surface esterase activity
3.3 Isolation of epicuticular waxes
3.4 Adhesion assay
3.5 Extraction of extracellular esterases of conidia
3.6 Esterases from germinating conidia induced by waxes and organic acids
3.7 Esterase assay
3.8 Degradation of epicuticular waxes by the extracellular esterase of conidia
Acknowledgements
References
- Zhu, Y.Z.; Qiang, S. Isolation, pathogenicity and safety of Curvularia eragrostidis isolate QZ-2000 as a bioherbicide agent for large crabgrass (Digitaria sanguinalis). Biocontrol Sci. Techn. 2004, 14(8), 769–782. [Google Scholar]
- Zhu, Y.Z.; Qiang, S. The biological characteristics of fungus strain QZ-200 of Curvularia sp. for controlling Digitaria sanguinalis. Acta Phytopathologica Sinica 2003, 33(6), 524–529. [Google Scholar]
- Nicholson, R.L.; Yoshioka, H.; Yamaoka, N.; Kunoh, H. Preparation of the infection court by Erysiphe graminis. II. Release of esterase enzyme activity from conidia in response to a contact stimulus. Exp. Mycol. 1988, 12, 336–349. [Google Scholar] [CrossRef]
- Deising, H.; Nicholson, R.L.; Haug, M.; Howard, R.J.; Mendgen, K. Adhesion pad formation and the involvement of surface localized cutinase and esterases in the attachment of Uredospores to the host cuticle. Plant Cell 1992, 4, 1011–1111. [Google Scholar]
- DeJong, J.C.; McCormack, B.J.; Smirnoff, N.; Talbot, N.J. Glycerol generates turgor in rice blast. Nature 1997, 389, 244–245. [Google Scholar] [CrossRef]
- Stahl, D.J.; Schafer, W. Cutinase is not required for fungal pathogenicity on pea. Plant Cell 1992, 4, 621–629. [Google Scholar]
- Yao, C.; Koëller, W. Diversity of cutinases from plant pathogenic fungi: Different cutinases are expressed during saprophytic and pathogenic stages of Alternaria brassicicola. Mol. Plant-Microbe Interact. 1995, 8, 122–130. [Google Scholar]
- Davies, K.A.; Delorono, I.; Foster, S.J.; LI, D.; Johnstone, K.; Ashby, A.M. Evidence for a role of cutinase in pathogenicity of Pyrenopeziza brassicae on brassicas. Physiol. Mol. Plant Pathol. 2000, 57, 63–75. [Google Scholar] [CrossRef]
- Walton, T.J. Waxes, cutin and suberin. Methods in Plant Bioch. 1990, 4, 105–158. [Google Scholar]
- Schonherr, J. Resistance of plant surfaces to water loss. In Physiological Plant Ecology IIÐWater Relations and Carbon Assimilation I; Lange, OL, Nobel, PS, Osmond, CB, Ziegler, H, Eds.; Springer-Verlag: Berlin, 1982; pp. 173–179. [Google Scholar]
- Carver, T.L.W; Thomas, B.J.; Ingersonmorris, S.M.; Roderick, H.W. The role of the abaxial leaf surface waxes of Lolium spp. in resistance to Erysiphe graminis. Plant Pathol 1990, 39, 573–583. [Google Scholar] [CrossRef]
- Podila, G.K.; Rogers, L.M.; Kolattukudy, P.E. Chemical signals from avocado surface wax trigger germination and appressorium formation in Colletotrichum gloeosporioides. Plant Physiol 1993, 103, 267–272. [Google Scholar]
- Flaishman, M.A.; Hwang, C-S.; Kolattukudy, P.E. Involvement of protein phosphorylation in the induction of appressorium formation in Colletotrichum gloeosporioides by its host surface wax and ethylene. Physiol Mol Plant Pathol. 1995, 47, 103–117. [Google Scholar] [CrossRef]
- Maier, C.G.; Post-Beittenmiller, D. Epicuticular wax on leek in vitro developmental stages and seedlings under varied growth conditions. Plant Science 1998, 134, 53–67. [Google Scholar] [CrossRef]
- Jetter, R.; Scha¨ffer, S. Chemical composition of the Prunus laurocerasus leaf surface. Dynamic changes of the epicuticular wax film during leaf development. Plant Physiol. 2001, 126, 1725–1737. [Google Scholar] [CrossRef]
- Koch, K.; Neinhuis, C.; Ensikat, H.J.; Barthlott, W. Self assembly of epicuticular waxes on living plant surfaces imaged by atomic force microscopy (AFM). J. Exp. Bot. 2004, 55, 711–718. [Google Scholar] [CrossRef]
- Tomaszewski, D. The wax layer and its morphological variability in four European Salix species. Flora. Jena. 2004, 199(4), 320–326. [Google Scholar] [CrossRef]
- Knight, T.G.; Wallwork, M.; Sedgley, M. Leaf epicuticular wax and cuticle ultrastructure of four Eucalyptus species and their hybrids. Int. J.Plant Sci. 2004, 165(1), 27–36. [Google Scholar] [CrossRef]
- Tsuba, M.; Katagiri, C.; Takeuchi, Y.; Takada, Y.;Yamaoka, N. Chemical factors of the leaf surface involved in the morphogenesis of Blumeria graminis. Physiol Mol Plant Pathol. 2002, 60, 51–57. [Google Scholar]
- Shepherd, T.; Robertson, G.W.; Griffiths, D.W.; Birch, A.N.E. Epicuticular wax ester and triacylglycerol composition in relation to aphid infestation and resistance in red raspberry (Rubus idaeus L.). Phytochemistry 1999, 52, 1255–1267. [Google Scholar] [CrossRef]
- Griffiths, D.W.; Robertson, G.W.; Shepherd, T.; Ramsay, G. Epicuticular waxes and volatiles from faba bean (Vicia faba ) flowers. Phytochemistry 1999, 52, 607–612. [Google Scholar] [CrossRef]
- Jenks, M.A.; Joly, R.J.; Peters, P.J.; Rich, P.J.; Axtell, J.D.; Ashworth, E.N. Chemically induced cuticle mutation affecting epidermal conductance to water vapor and disease susceptibility in sorghum bicolor (L.) moench. Plant Physiol. 1994, 105, 1239–1245. [Google Scholar]
- Alcerito, T.; Brabo, F.E.; Negri, G.; Santos, D.Y.A.C.; Meda, C.I.; Young, M.C.M.; Cha′vez, D.; Blatt, C.T.T. Folicular epicuticular wax of Arrabidea brachypoda: flavonoids and antifungal activity. Biochem. Syst. Ecol. 2002, 30, 677–683. [Google Scholar] [CrossRef]
- Kerstiens, G. Signalling across the divide: a wider perspective of cuticular structure–function relationships. Trends Plant Sci 1996, 1, 125–129. [Google Scholar] [CrossRef]
- Pascholati, S.F.; Deising, H.; Leite, B.; Anderson, D.; Nicholson, R.L. Cutinase and non-specific esterase activities in the conidial mucilage of Colletotrichum graminicola. Physiol Mol Plant Pathol. 1993, 42, 37–51. [Google Scholar] [CrossRef]
- Sugui, J.A.; Pascholati, S.F.; Kunoh, H.; Howard, R.; Nicholson, R.L. Cutinase and non-specific esterase produced by Pestalotia malicola. Physiol. Mol. Plant Pathol. 1998, 52, 213–221. [Google Scholar] [CrossRef]
- Jansson, H.B.; Åkesson, H. Extracellular matrix, esterase and the phytotoxin prehelminthosporol infection of barley leaves by Bipolaris sorokiniana. European Journal of Plant Pathology 2003, 109, 599–605. [Google Scholar] [CrossRef]
- Osman, S.F.; Gerard, H.C.; Moreau, R.A.; Fett, W.F. Model substrates for cutinases. Chemistry and Physics of Lipids 1993, 66(3), 215–218. [Google Scholar] [CrossRef]
- Griffiths, D.W.; Robertson, G.W.; Shepherd, T.; Birch, A.N.E.; Gordon, S.C.; Woodford, J.A.T. A comparison of the composition of epicuticular wax from red raspberry (Rubus idaeus L.) and hawthorn (Crataegus monogyna Jacq.) flowers. Phytochemistry 2000, 55, 111–116. [Google Scholar] [CrossRef]
- Szafranek, B.M.; Synak, E.E. Cuticular waxes from potato (Solanum tuberosum) leaves. Phytochemistry 2006, 67, 80–90. [Google Scholar] [CrossRef]
- Tesch, C.; Nikolet, K.; Gnau, V.; Gotz, F.; Bormann, C. Biochemical and molecular characterization of the extracellular esterase from Streptomyces diastatochromogenes. J Bacteriol 1996, 179, 1858–1865. [Google Scholar]
- Berto, P.; Commenil, P.; Belingheri, L.; Dehorter, B. Occurrence of a lipase in spores of Alternaria brassicicola with a crucial role in the infection of cauliflower leaves. FEMS Microbiology Letter 1999, 180, 183–89. [Google Scholar] [CrossRef]
- Huang, T.L.; Székács, A.; Uematsu, T.; Kuwano, E.; Parkinson, A.; Hammock, B.D. Hydrolysis of carbonates, thiocarbonates, carbamates, and carboxylic esters of α-napthol and β-napthol, and p-nitophenol by Human, rat, and mouse liver carboxylesterases. Pharm. Res. 1992, 10(5), 639–648. [Google Scholar]
- Rumbolz, J.; Kassemeyer, H-H.; Steinmetz, V.; Deising, H.B.; Mendgen, K.; Mathys, D.; Wirtz, S.; Guggenheim, R. Differentiation of infection structures of the powdery mildew fungus Uncinula necator and adhesion to the host cuticle. Canadian Journal of Botany 2000, 78(3), 409–421. [Google Scholar]
- Davies, K. A.; Deloronoil, I.; Foster, S. J.; Lli, D.; Johnstone, K.; Ashby, A M. Evidence for a role of cutinase in pathogenicity of Pyrenopeziza brassicae on brassicas. Physiological and Molecular Plant Pathology 2000, 57, 63–75. [Google Scholar] [CrossRef]
- Fett, W.;F.; Gerard, H.C.; Moreau, R.A.; Osman, S.F.; Jones, L.E. Cutinase production by Streptomyces spp. Curr. Microbiol. 1992, 25, 165–171. [Google Scholar] [CrossRef]
- Kolattukudy, P.E.; Purdy, R.E.; Maiti, I.B. Cutinases from fungi and pollen. Methods Enzymol. 1981, 7l, 652–664. [Google Scholar] [CrossRef]
- Powell, G.; Maniar, S.P.; Pickett, J.A.; Hardie, J. Aphid responses to non-host epicuticular lipids. Entomol. Exp. Appl. 1999, 91, 115–123. [Google Scholar]
- Wang, C.H.L.; Chen, C.H.K.; Gianfangna, T. Relationship between cutin monomers and tomato resistance to powdery mildew infection. Physiol Mol Plant Pathol. 2000, 57, 55–61. [Google Scholar] [CrossRef]
© 2006 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Wang, F.; Zhang, P.; Qiang, S.; Xu, L.-L. Interaction of Plant Epicuticular Waxes and Extracellular Esterases of Curvularia eragrostidis during Infection of Digitaria sanguinalis and Festuca arundinacea by the Fungus. Int. J. Mol. Sci. 2006, 7, 346-357. https://0-doi-org.brum.beds.ac.uk/10.3390/i7090346
Wang F, Zhang P, Qiang S, Xu L-L. Interaction of Plant Epicuticular Waxes and Extracellular Esterases of Curvularia eragrostidis during Infection of Digitaria sanguinalis and Festuca arundinacea by the Fungus. International Journal of Molecular Sciences. 2006; 7(9):346-357. https://0-doi-org.brum.beds.ac.uk/10.3390/i7090346
Chicago/Turabian StyleWang, Fei, Peng Zhang, Sheng Qiang, and Lang-Lai Xu. 2006. "Interaction of Plant Epicuticular Waxes and Extracellular Esterases of Curvularia eragrostidis during Infection of Digitaria sanguinalis and Festuca arundinacea by the Fungus" International Journal of Molecular Sciences 7, no. 9: 346-357. https://0-doi-org.brum.beds.ac.uk/10.3390/i7090346






