Evolutionary History of the Galápagos Rail Revealed by Ancient Mitogenomes and Modern Samples
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Sites and Sampling
2.2. Ancient DNA Extraction and Mitogenome Assembly
2.3. Genetic Diversity
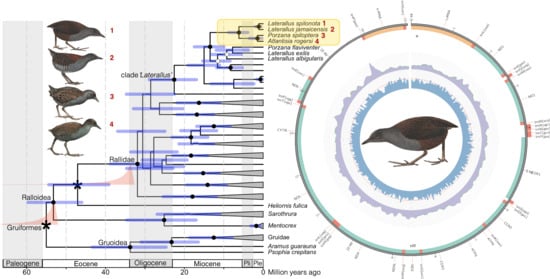
2.4. Phylogenetic Reconstruction and Molecular Dating
2.4.1. Coding Mitogenome Dataset
2.4.2. One Nuclear and Two Mitochondrial Genes
3. Results
3.1. Mitogenome Assembly and Diversity
3.2. Phylogenetic Reconstruction and Molecular Dating
3.3. Intraspecific Genetic Diversity
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Data Accessibility
References
- Darwin, C. Journal of Researches into the Natural History and Geology of the Countries Visited During the Voyage of the H.M.S. Beagle around the World; D. Appleton: New York, NY, USA, 1896. [Google Scholar]
- Gillespie, R. Community assembly through adaptive radiation in Hawaiian spiders. Science 2004, 303, 356–359. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grant, P.R.; Grant, B.R. How and Why Species Multiply: The Radiation of Darwin’s Finches; Princeton University Press: Princeton, NJ, USA, 2011; ISBN 0-691-14999-2. [Google Scholar]
- Losos, J.B.; Ricklefs, R.E. Adaptation and diversification on islands. Nature 2009, 457, 830–836. [Google Scholar] [CrossRef] [PubMed]
- Mayr, E.; Vaurie, C. Evolution in the family Dicruridae (birds). Evolution 1948, 2, 238–265. [Google Scholar] [CrossRef] [PubMed]
- Wallace, A.R. On the physical geography of the Malay Archipelago. J. R. Geogr. Soc. Lond. 1863, 33, 217–234. [Google Scholar] [CrossRef]
- Parent, C.E.; Caccone, A.; Petren, K. Colonization and diversification of Galápagos terrestrial fauna: A phylogenetic and biogeographical synthesis. Philos. Trans. R. Soc. B Biol. Sci. 2008, 363, 3347–3361. [Google Scholar] [CrossRef] [Green Version]
- Shaw, K.L.; Gillespie, R.G. Comparative phylogeography of oceanic archipelagos: Hotspots for inferences of evolutionary process. Proc Natl. Acad. Sci. USA 2016, 113, 7986–7993. [Google Scholar] [CrossRef] [Green Version]
- Poulakakis, N.; Miller, J.M.; Jensen, E.L.; Beheregaray, L.B.; Russello, M.A.; Glaberman, S.; Boore, J.; Caccone, A. Colonization history of Galapagos giant tortoises: Insights from mitogenomes support the progression rule. J. Zool. Syst. Evol. Res. 2020, 58, 1262–1275. [Google Scholar] [CrossRef]
- Lack, D. Darwin’s Finches; Cambridge University Press Archive: Cambridge, UK, 1983; ISBN 0-521-27242-4. [Google Scholar]
- Arbogast, B.S.; Drovetski, S.V.; Curry, R.L.; Boag, P.T.; Seutin, G.; Grant, P.R.; Grant, B.R.; Anderson, D.J. The Origin and Diversification of Galapagos Mockingbirds. Evolution 2006, 60, 370–382. [Google Scholar] [CrossRef]
- Miller, J.M.; Quinzin, M.C.; Edwards, D.L.; Eaton, D.A.; Jensen, E.L.; Russello, M.A.; Gibbs, J.P.; Tapia, W.; Rueda, D.; Caccone, A. Genome-wide assessment of diversity and divergence among extant Galapagos giant tortoise species. J. Hered. 2018, 109, 611–619. [Google Scholar] [CrossRef]
- Caccone, A.; Gibbs, J.P.; Ketmaier, V.; Suatoni, E.; Powell, J.R. Origin and evolutionary relationships of giant Galápagos tortoises. Proc. Natl. Acad. Sci. USA 1999, 96, 13223–13228. [Google Scholar] [CrossRef] [Green Version]
- Sari, E.; Bollmer, J.L. Colonization of Galápagos Birds: Identifying the Closest Relative and Estimating Colonization. In Disease Ecology; Springer: Cham, Switzerland, 2018; pp. 15–43. [Google Scholar]
- Funk, W.C.; McKay, J.K.; Hohenlohe, P.A.; Allendorf, F.W. Harnessing genomics for delineating conservation units. Trends Ecol. Evol. 2012, 27, 489–496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fraser, D.J.; Bernatchez, L. Adaptive evolutionary conservation: Towards a unified concept for defining conservation units. Mol. Ecol. 2001, 10, 2741–2752. [Google Scholar] [CrossRef] [PubMed]
- Cooper, W.E.; Pyron, R.A.; Garland, T., Jr. Island tameness: Living on islands reduces flight initiation distance. Proc. R. Soc. B Biol. Sci. 2014, 281, 20133019. [Google Scholar] [CrossRef] [Green Version]
- Olson, S.L. Evolution of the rails of the South Atlantic islands (Aves: Rallidae). Smithson. Contrib. Zool. 1973, 152, 1–53. [Google Scholar] [CrossRef]
- Wright, N.A.; Steadman, D.W.; Witt, C.C. Predictable evolution toward flightlessness in volant island birds. Proc. Natl. Acad. Sci. USA 2016, 113, 4765–4770. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roff, D.A. The evolution of flightlessness: Is history important? Evol. Ecol. 1994, 8, 639–657. [Google Scholar] [CrossRef]
- Taylor, B.; van Perlo, B. Rails. A Guide to the Rails, Crakes, Gallinules and Coots of the World; Yale University Press: New Haven, CT, USA, 1998; pp. 1–600. [Google Scholar]
- Blackburn, T.M.; Cassey, P.; Duncan, R.P.; Evans, K.L.; Gaston, K.J. Avian extinction and mammalian introductions on oceanic islands. Science 2004, 305, 1955–1958. [Google Scholar] [CrossRef] [Green Version]
- Courchamp, F.; Chapuis, J.-L.; Pascal, M. Mammal invaders on islands: Impact, control and control impact. Biol. Rev. 2003, 78, 347–383. [Google Scholar] [CrossRef] [Green Version]
- Kirchman, J. Speciation of flightless rails on islands: A DNA-based phylogeny of the typical rails of the Pacific. Auk 2012, 129, 56–69. [Google Scholar] [CrossRef]
- Donlan, C.J.; Campbell, K.; Cabrera, W.; Lavoie, C.; Carrion, V.; Cruz, F. Recovery of the Galápagos rail (Laterallus spilonotus) following the removal of invasive mammals. Biol. Conserv. 2007, 138, 520–524. [Google Scholar] [CrossRef]
- Steadman, D.W. Extinction and Biogeography of Tropical Pacific Birds; University of Chicago Press: Chicago, IL, USA, 2006. [Google Scholar]
- Schofield, E.K. Effects of introduced plants and animals on island vegetation: Examples from the Galapagos Archipelago. Conserv. Biol. 1989, 3, 227–238. [Google Scholar] [CrossRef]
- Hamann, O. Vegetation changes over three decades on Santa Fe Island, Galápagos, Ecuador. Nord. J. Bot. 2003, 23, 1–10. [Google Scholar] [CrossRef]
- Phillips, R.; Wiedenfeld, D.; Snell, H. Current status of alien vertebrates in the Galápagos Islands: Invasion history, distribution, and potential impacts. Biol. Invasions 2012, 14, 461–480. [Google Scholar] [CrossRef]
- Harper, G.A.; Bunbury, N. Invasive rats on tropical islands: Their population biology and impacts on native species. Glob. Ecol. Conserv. 2015, 3, 607–627. [Google Scholar] [CrossRef] [Green Version]
- Jiménez-Uzcátegui, G.; Weindenfield, D.; Valle, C.A.; Vargas, H.; Piedrahita, P.; Muñoz-Abril, L.J.; Álava, J.J. Threats and vision for the conservation of Galápagos birds. Open Ornithol. J. 2019, 12, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Vargas, H.; Bensted-Smith, R. Past and present ornithology in Galapagos. Bull. L’Institut R. Sci. Nat. Belg. 2000, 70, 47–52. [Google Scholar]
- Gibbs, J.P.; Shriver, W.G.; Vargas, H. An assessment of a Galapagos Rail population over thirteen years (1986 to 2000). J. Field Ornithol. 2003, 74, 136–140. [Google Scholar] [CrossRef]
- Freile, J.F.; Santander, T.G.; Jiménez-Uzcátegui, G.; Carrasco, L.; Cisneros-Heredia, D.; Guevara, E.A.; Sánchez-Nivicela, M.; Tinoco, B.A. Lista Roja de las Aves del Ecuador continental; Ministerio del Ambiente, Aves y Conservación, Comité Ecuatoriano de Registros Ornitológicos, Universidad del Azuay, Red Aves Ecuador y Universidad San Francisco de Quito: Quito, Ecuador, 2019. [Google Scholar]
- Campbell, K.; Donlan, C.J.; Cruz, F.; Carrion, V. Eradication of feral goats Capra hircus from Pinta Island, Galápagos, Ecuador. Oryx 2004, 38, 328–333. [Google Scholar] [CrossRef] [Green Version]
- Campbell, K.; Donlan, C.J. Feral goat eradications on islands. Conserv. Biol. 2005, 19, 1362–1374. [Google Scholar] [CrossRef]
- Hamann, O. Regeneration of vegetation on Santa Fe and Pinta Islands, Galapagos, after the eradication of goats. Biol. Conserv. 1979, 15, 215–235. [Google Scholar] [CrossRef]
- Shriver, W.G.; Gibbs, J.P.; Woltz, H.W.; Schwarz, N.P.; Pepper, M.A. Galápagos Rail Laterallus spilonotus population change associated with habitat invasion by the Red-barked Quinine Tree Cinchona Pubescens. Bird Conserv. Int. 2011, 21, 221–227. [Google Scholar] [CrossRef] [Green Version]
- Wiedenfeld, D.A.; Jiménez-Uzcátegui, G.A. Critical problems for bird conservation in the Galápagos Islands. Cotinga 2008, 29, 22–27. [Google Scholar]
- Depino, E.A.; Areta, J.I. V-Netting with playback: An active cost-effective method for trapping small rails. Ardeola 2019, 67, 145–156. [Google Scholar] [CrossRef]
- Ewels, P.; Magnusson, M.; Lundin, S.; Käller, M. MultiQC: Summarize analysis results for multiple tools and samples in a single report. Bioinformatics 2016, 32, 3047–3048. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kearse, M.; Moir, R.; Wilson, A.; Stones-Havas, S.; Cheung, M.; Sturrock, S.; Buxton, S.; Cooper, A.; Markowitz, S.; Duran, C. Geneious Basic: An integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 2012, 28, 1647–1649. [Google Scholar] [CrossRef]
- Meng, G.; Li, Y.; Yang, C.; Liu, S. MitoZ: A toolkit for animal mitochondrial genome assembly, annotation and visualization. Nucleic Acids Res. 2019, 47, 63. [Google Scholar] [CrossRef]
- Mindell, D.P.; Sorenson, M.D.; Dimcheff, D.E. An extra nucleotide is not translated in mitochondrial ND3 of some birds and turtles. Mol. Biol. Evol. 1998, 15, 1568–1571. [Google Scholar] [CrossRef] [Green Version]
- Bonaccorso, E.; Peterson, A.T.; Navarro-Sigüenza, A.G.; Fleischer, R.C. Molecular systematics and evolution of the Cyanocorax jays. Mol. Phylogenetics Evol. 2010, 54, 897–909. [Google Scholar] [CrossRef] [Green Version]
- Johansson, U.S.; Parsons, T.J.; Irestedt, M.; Ericson, P.G.P. Clades within the higher land birds’, evaluated by nuclear DNA sequences. J. Zool. Syst. Evol. Res. 2001, 39, 37–52. [Google Scholar] [CrossRef] [Green Version]
- Paradis, E. pegas: An R package for population genetics with an integrated–modular approach. Bioinformatics 2010, 26, 419–420. [Google Scholar] [CrossRef] [Green Version]
- R Core Team R. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Templeton, A.R.; Crandall, K.A.; Sing, C.F. A cladistic analysis of phenotypic associations with haplotypes inferred from restriction endonuclease mapping and DNA sequence data. III. Cladogram Estim. Genet. 1992, 132, 619–633. [Google Scholar]
- Stervander, M.; Ryan, P.G.; Melo, M.; Hansson, B. The origin of the world’s smallest flightless bird, the Inaccessible Island Rail Atlantisia rogersi (Aves: Rallidae). Mol. Phylogenetics Evol. 2019, 130, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Bouckaert, R.; Vaughan, T.G.; Barido-Sottani, J.; Duchêne, S.; Fourment, M.; Gavryushkina, A.; Heled, J.; Jones, G.; Kühnert, D.; De Maio, N. BEAST 2.5: An advanced software platform for Bayesian evolutionary analysis. PLoS Comput. Biol. 2019, 15, 1006650. [Google Scholar]
- Claramunt, S.; Cracraft, J. A new time tree reveals Earth history’s imprint on the evolution of modern birds. Sci. Adv. 2015, 1, 1501005. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Musser, G.; Ksepka, D.T.; Field, D.J. New material of Paleocene-Eocene Pellornis (Aves: Gruiformes) clarifies the pattern and timing of the extant gruiform radiation. Diversity 2019, 11, 102. [Google Scholar] [CrossRef] [Green Version]
- García, J.C.; Lemmon, E.M.; Lemmon, A.R.; French, N. Phylogenomic Reconstruction Sheds Light on New Relationships and Timescale of Rails (Aves: Rallidae) Evolution. Diversity 2020, 12, 70. [Google Scholar] [CrossRef] [Green Version]
- De Pietri, V.L.; Mayr, G. Reappraisal of early Miocene rails (Aves, Rallidae) from central France: Diversity and character evolution. J. Zool. Syst. Evol. Res. 2014, 52, 312–322. [Google Scholar] [CrossRef]
- Rambaut, A.; Drummond, A.J.; Xie, D.; Baele, G.; Suchard, M.A. Posterior summarization in Bayesian phylogenetics using Tracer 1.7. Syst. Biol. 2018, 67, 901–904. [Google Scholar] [CrossRef] [Green Version]
- Rambaut, A.; Drummond, A.J. TreeAnnotator v1.7.0. Available online: http://beast.bio.ed.ac.uk (accessed on 1 August 2018).
- Paradis, E. Analysis of Phylogenetics and Evolution with R.; Springer Science & Business Media: New York, NY, USA, 2011; ISBN 1-4614-1743-0. [Google Scholar]
- Paradis, E.; Claude, J.; Strimmer, K. APE: Analyses of phylogenetics and evolution in R language. Bioinformatics 2004, 20, 289–290. [Google Scholar] [CrossRef] [Green Version]
- Revell, L.J. Phytools: An R package for phylogenetic comparative biology (and other things). Methods Ecol. Evol. 2012, 3, 217–223. [Google Scholar] [CrossRef]
- Lamichhaney, S.; Berglund, J.; Almén, M.S.; Maqbool, K.; Grabherr, M.; Martinez-Barrio, A.; Promerová, M.; Rubin, C.-J.; Wang, C.; Zamani, N.; et al. Evolution of Darwin’s finches and their beaks revealed by genome sequencing. Nature 2015, 518, 371–375. [Google Scholar] [CrossRef]
- Petren, K.; Grant, P.R.; Grant, B.R.; Keller, L.F. Comparative landscape genetics and the adaptive radiation of Darwin’s finches: The role of peripheral isolation. Mol. Ecol. 2005, 14, 2943–2957. [Google Scholar] [CrossRef]
- Carmi, O.; Witt, C.C.; Jaramillo, A.; Dumbacher, J.P. Phylogeography of the Vermilion Flycatcher species complex: Multiple speciation events, shifts in migratory behavior, and an apparent extinction of a Galápagos-endemic bird species. Mol. Phylogenetics Evol. 2016, 102, 152–173. [Google Scholar] [CrossRef] [Green Version]
- Sari, E.; Parker, P.G. Understanding the colonization history of the Galápagos flycatcher (Myiarchus magnirostris). Mol. Phylogenetics Evol. 2012, 63, 244–254. [Google Scholar] [CrossRef] [PubMed]
- Geist, D.J.; Snell, H.; Snell, H.; Goddard, C.; Kurz, M.D. A paleogeographic model of the Galápagos Islands and biogeographical and evolutionary implications. In The Galápagos: A Natural Laboratory for the Earth Sciences; Harpp, K.S., Mittelstaedt, E., d’Ozouville, N., Graham, D.W., Eds.; American Geophysical Union: Washington, DC, USA, 2014; pp. 145–166. [Google Scholar]
- Chaves, J.A.; Parker, P.G.; Smith, T.B. Origin and population history of a recent colonizer, the yellow warbler in Galápagos and Cocos Islands. J. Evol. Biol. 2012, 25, 509–521. [Google Scholar] [CrossRef] [PubMed]
- Leck, C.F. Establishment of New Population Centers with Changes in Migration Patterns. J. Field Ornithol. 1980, 51, 168–173. [Google Scholar]
- Taylor, P.B. Family Rallidae (rails, gallinules and coots). In Handbook of the Birds of the World. Hoatzin to Auks; del Hoyo, J., Elliot, A., Sargata, J., Eds.; Lynx Edicions: Barcelona, Spain, 1996; Volume 3, pp. 108–209. [Google Scholar]
- MacArthur, R.H.; Wilson, E.O. The Theory of Island Biogeography; Princeton University Press: Princeton, NJ, USA, 1967. [Google Scholar]
- Bollmer, J.L.; Kimball, R.T.; Whiteman, N.K.; Sarasola, J.H.; Parker, P.G. Phylogeography of the Galápagos hawk (Buteo galapagoensis): A recent arrival to the Galápagos Islands. Mol. Phylogenet. Evol. 2006, 39, 237–247. [Google Scholar] [CrossRef] [PubMed]
- Sato, A.; O’hUigin, C.; Figueroa, F.; Grant, P.R.; Grant, B.R.; Tichy, H.; Klein, J. Phylogeny of Darwin’s Finches as Revealed by mtDNA Sequences. Proc. Natl. Acad. Sci. USA 1999, 96, 5101–5106. [Google Scholar] [CrossRef] [Green Version]
- Girard, P.; Takekawa, J.Y.; Beissinger, S.R. Uncloaking a cryptic, threatened rail with molecular markers: Origins, connectivity and demography of a recently-discovered population. Conserv. Genet. 2010, 11, 2409–2418. [Google Scholar] [CrossRef] [Green Version]
- Eddleman, W.R.; Flores, R.E.; Legare, M.L. The Birds of North America; Poole, A., Gills, F., Eds.; Acad. Nat. Sci. Philadelphia and Am. Ornithol. Union: Washington, DC, USA, 1994; No. 123; pp. 1–20. [Google Scholar]
- Gill, F.; Donsker, D.; Rasmussen, P. IOC World Bird List (v10.2). Available online: https://0-doi-org.brum.beds.ac.uk/10.14344/IOC.ML.10.2. (accessed on 20 July 2020).
- Dinesen, L.; Chamorro, A.; Fjeldså, J.; Aucca, C. Distribution and habitat description of Junín Rail Laterallus tuerosi, Andean Peru. Bird Conserv. Int. 2017, 27, 388. [Google Scholar] [CrossRef] [Green Version]
- García–R., J.C.; Gibb, G.C.; Trewick, S.A. Eocene diversification of crown group rails (Aves: Gruiformes: Rallidae). PLoS ONE 2014, 9, e109635. [Google Scholar]
- Slikas, B.; Olson, S.L.; Fleischer, R.C. Rapid, independent evolution of flightlessness in four species of Pacific Island rails (Rallidae): An analysis based on mitochondrial sequence data. J. Avian Biol. 2002, 33, 5–14. [Google Scholar] [CrossRef] [Green Version]
- Franklin, A.B.; Clark, D.A.; Clark, D.B. Ecology and Behavior of the Galapagos Rail. Wilson Bull. 1979, 91, 202–221. [Google Scholar]
- Rosenberg, D. The impact of introduced herbivores on the Galapagos Rail (Laterallus spilonotus). Monogr. Syst. Bot. Mo. Bot. Gard. 1990, 32, 169–178. [Google Scholar]
- Kramer, P.; Black, J. Scientific and Conservation Report; Charles Darwin Station: Galapagos, Ecuador, 1970. [Google Scholar]


| Node (MRCA) | Sequence Set | Rallidae Prior | Node Age Estimates (Million Years) | ||
|---|---|---|---|---|---|
| Median | Mean | 95% HPD | |||
| L. spilonota, L. jamaicensis | 2mt1nc | stem | 1.1 | 1.2 | 0.5–1.9 |
| crown | 1.2 | 1.3 | 0.5–2.1 | ||
| L. spilonota, L. rogersi | 2mt1nc | stem | 6.2 | 6.3 | 3.9–8.8 |
| crown | 6.7 | 6.8 | 4.2–9.6 | ||
| mtCDS | stem | 7.6 | 7.7 | 5.1–10.8 | |
| crown | 7.4 | 7.5 | 5.0–10.1 | ||
| Rallidae | 2mt1nc | stem | 31.9 | 32.1 | 25.8–38.8 |
| crown | 33.5 | 34.1 | 32.6–37.1 | ||
| mtCDS | stem | 36.1 | 36.3 | 30.6–41.7 | |
| crown | 34.9 | 35.3 | 32.6–37.1 | ||
| Gruiformes | 2mt1nc | stem | 55.0 | 55.9 | 54.0–60.7 |
| crown | 55.5 | 56.6 | 54.0–62.5 | ||
| mtCDS | stem | 54.9 | 55.8 | 54.0–60.2 | |
| crown | 54.9 | 55.5 | 54.0–59.0 | ||
| Island | Ind. | Haplotypes | Polymorphic Sites | Hb | π |
|---|---|---|---|---|---|
| Santa Cruz | 14 | 3 | 2 | 0.626 ± 0.104 | 0.0007 ± 0.0001 |
| Isabela | 16 | 2 | 1 | 0.125 ± 0.106 | 0.0001 ± 0.0001 |
| Pinta | 9 | 1 | 0 | 0.00 ± 0.00 | 0.00 ± 0.00 |
| Santiago | 21 | 3 | 2 | 0.626 ± 0.082 | 0.0007 ± 0.0001 |
| Total cytb | 56 | 3 | 2 | 0.439 ± 0.069 | 0.0006 ± 0.0001 |
| Santa Cruz | 14 | 4 | 3 | 0.791 ± 0.058 | 0.0010 ± 0.0001 |
| Isabela | 16 | 2 | 1 | 0.458 ± 0.095 | 0.0004 ± 0.0001 |
| Pinta | 3 | 1 | 0 | 0.000 ± 0.000 | 0.0000 ± 0.0000 |
| Santiago | 13 | 3 | 1 | 0.500 ± 0.080 | 0.0004 ± 0.0001 |
| Total ND2 | 46 | 5 | 3 | 0.791 ± 0.058 | 0.0006 ± 0.0001 |
| Santa Cruz | 15 | 1 | 0 | 0.00 ± 0.00 | 0.00 ± 0.00 |
| Isabela | 15 | 1 | 0 | 0.00 ± 0.00 | 0.00 ± 0.00 |
| Pinta | 8 | 1 | 0 | 0.00 ± 0.00 | 0.00 ± 0.00 |
| Santiago | 20 | 2 | 1 | 0.100 ± 0.007 | 0.0003 ± 0.0002 |
| Total RAG-1 | 58 | 2 | 1 | 0.034 ± 0.033 | 0.0001 ± 0.0001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chaves, J.A.; Martinez-Torres, P.J.; Depino, E.A.; Espinoza-Ulloa, S.; García-Loor, J.; Beichman, A.C.; Stervander, M. Evolutionary History of the Galápagos Rail Revealed by Ancient Mitogenomes and Modern Samples. Diversity 2020, 12, 425. https://0-doi-org.brum.beds.ac.uk/10.3390/d12110425
Chaves JA, Martinez-Torres PJ, Depino EA, Espinoza-Ulloa S, García-Loor J, Beichman AC, Stervander M. Evolutionary History of the Galápagos Rail Revealed by Ancient Mitogenomes and Modern Samples. Diversity. 2020; 12(11):425. https://0-doi-org.brum.beds.ac.uk/10.3390/d12110425
Chicago/Turabian StyleChaves, Jaime A., Pedro J. Martinez-Torres, Emiliano A. Depino, Sebastian Espinoza-Ulloa, Jefferson García-Loor, Annabel C. Beichman, and Martin Stervander. 2020. "Evolutionary History of the Galápagos Rail Revealed by Ancient Mitogenomes and Modern Samples" Diversity 12, no. 11: 425. https://0-doi-org.brum.beds.ac.uk/10.3390/d12110425