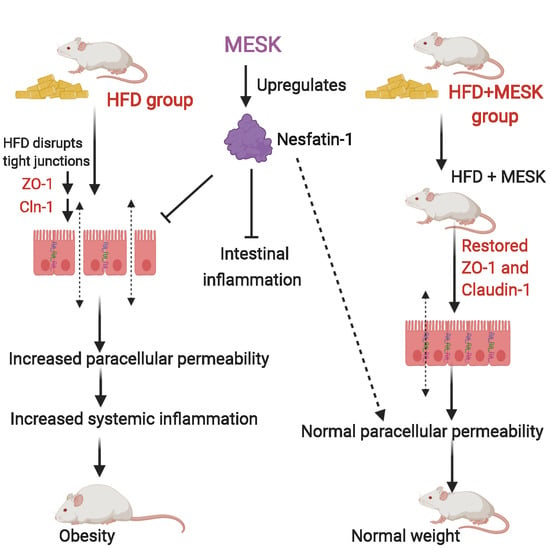
Reversal of Increase in Intestinal Permeability by Mangifera indica Seed Kernel Extract in High-Fat Diet-Induced Obese Mice
Abstract
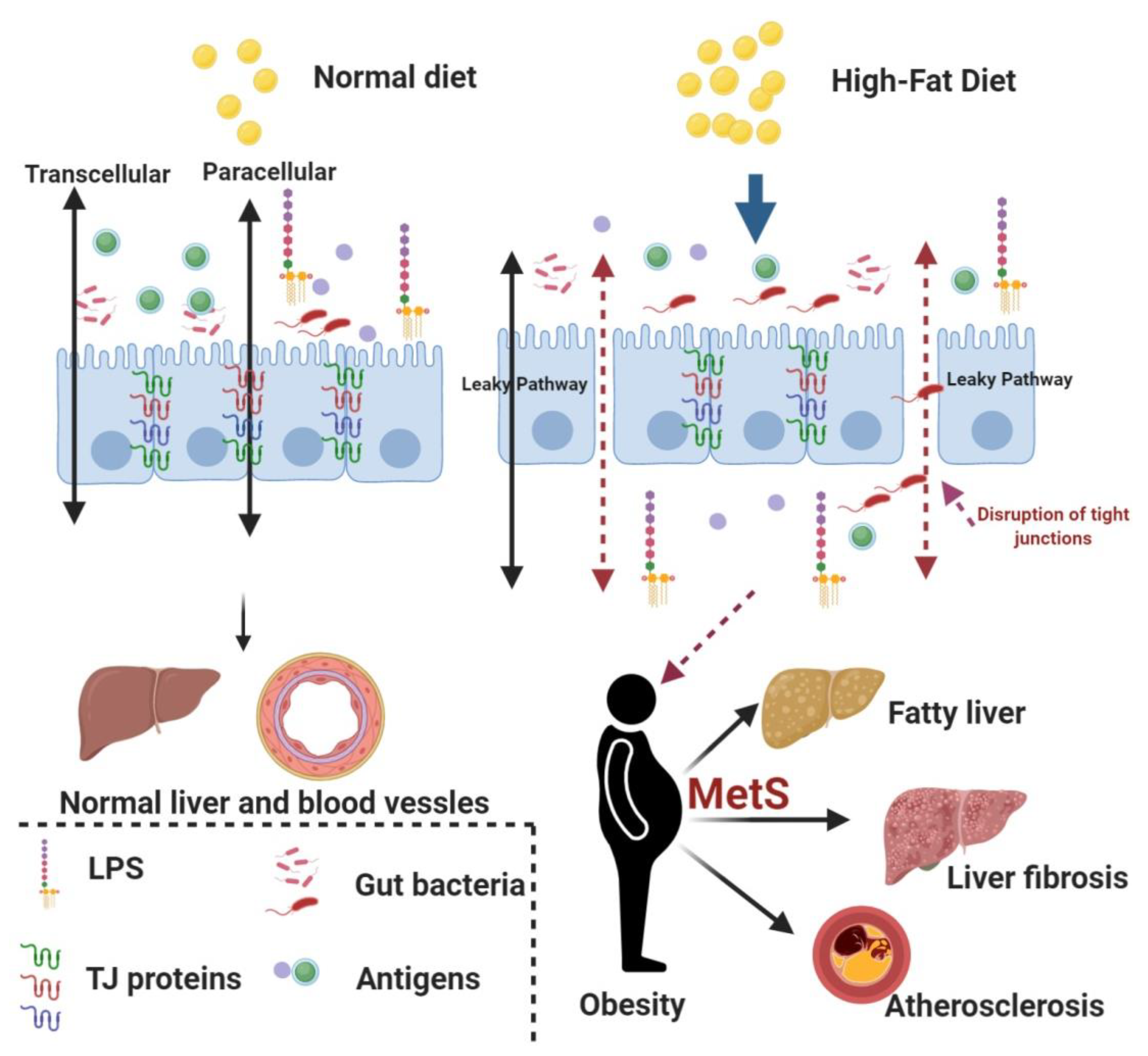
:1. Introduction
2. Materials and Methods
2.1. Collection and Preparation of Herbal Extract
2.2. Phyto-Constituent Analysis of MESK by High Performance Liquid Chromatography (HPLC)
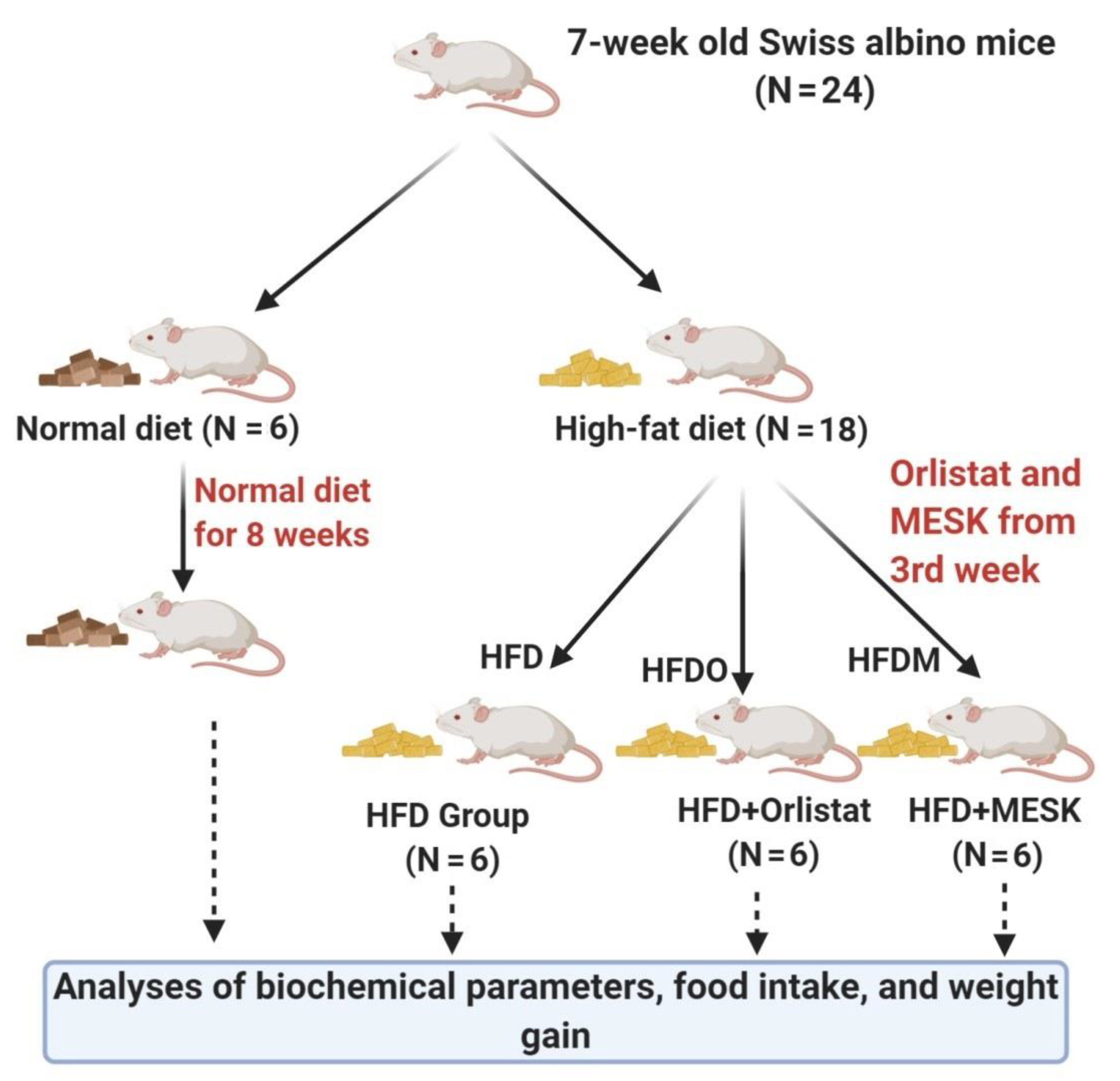
2.3. Experimental Animals
2.4. Food Consumption, Energy Intake, and Body Weight Measurement
2.5. Determination of Biochemical Parameters
2.6. Epididymal Adipose Tissue (EAT) and Liver Weight Analyses
2.7. Histopathology
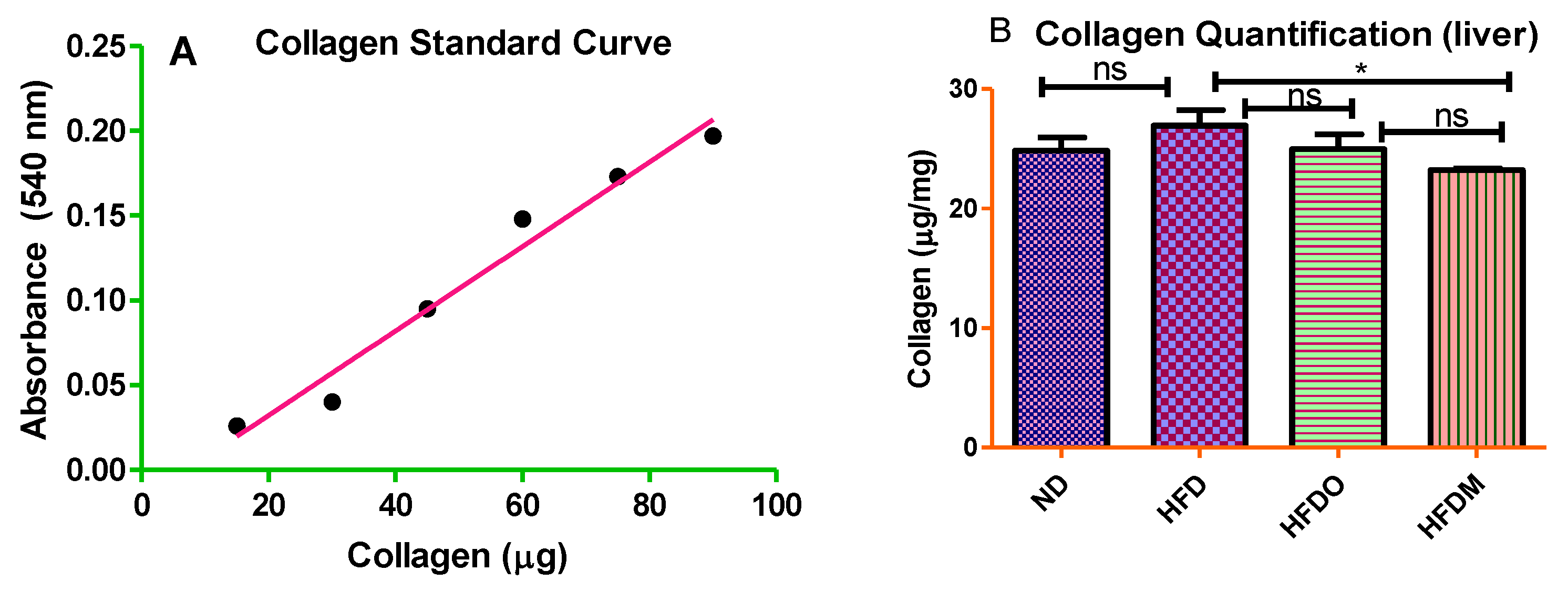
2.8. Sirius Red Staining
2.9. In Vivo Intestinal Permeability Measurement Using the FITC-Dextran Method
2.10. Protein Expression Analysis of ZO-1 and Nesfatin-1/NUCB-2 Using Immunofluorescence
2.11. Immunohistochemical Analysis of Claudin-1
2.12. Statistical Analysis
3. Results
3.1. Phytoconstituents in MESK
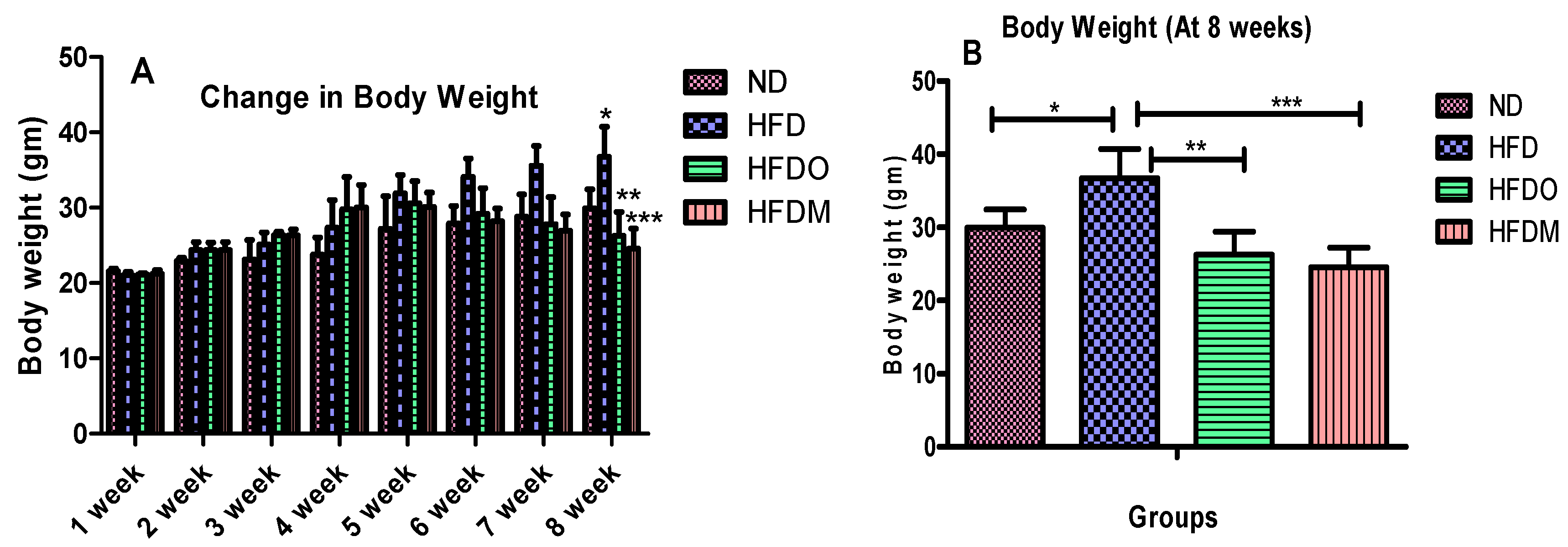
3.2. Effects of Mangifera Indica on Body Weight, Food, and Energy Intake
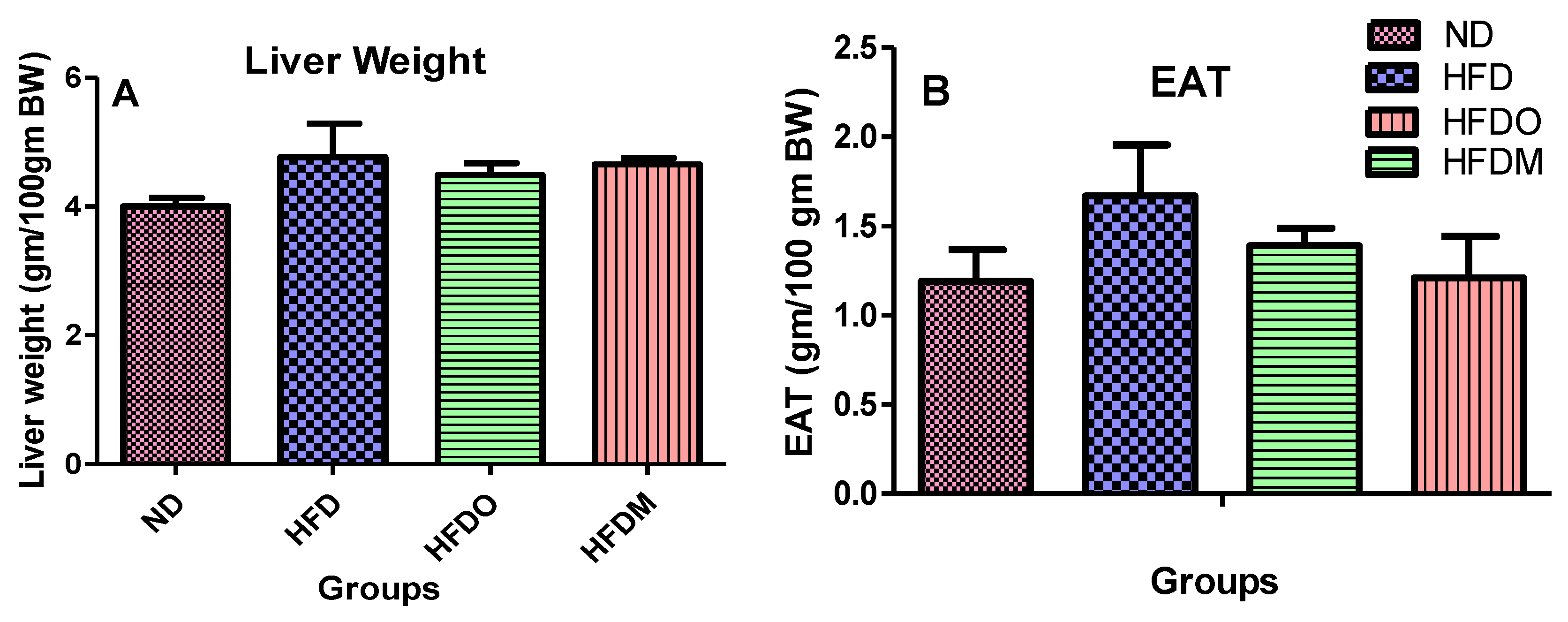
3.3. Effect of MESK on the Weight of Epididymal Adipose Tissue (EAT) and Liver
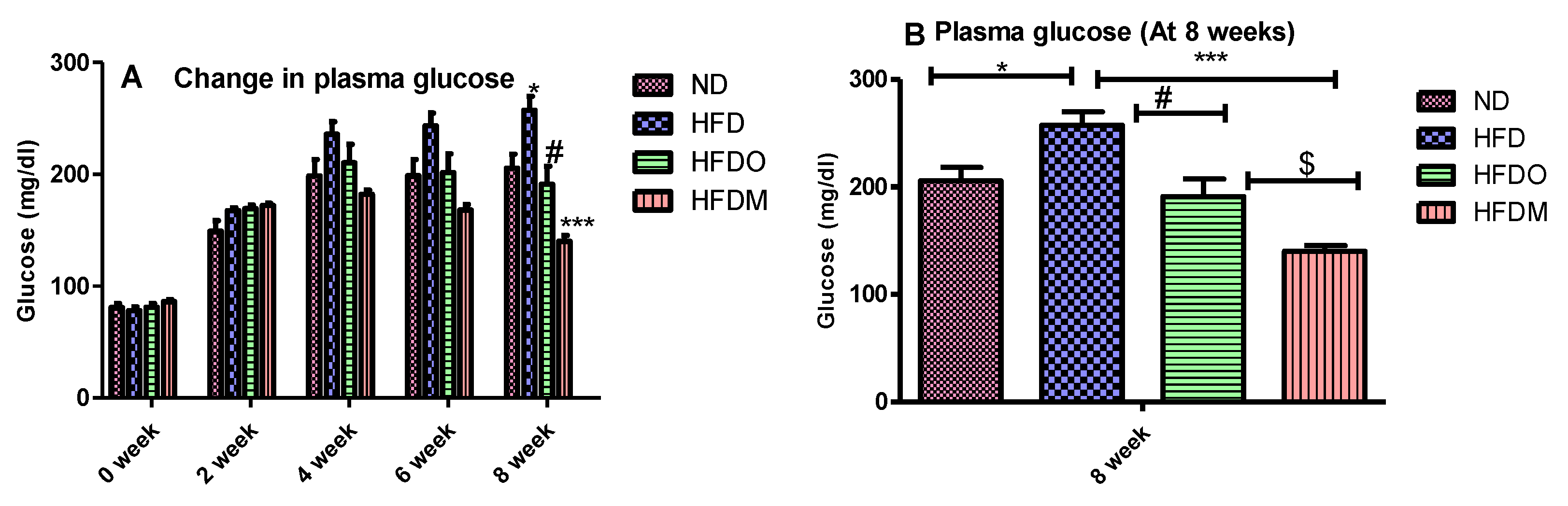
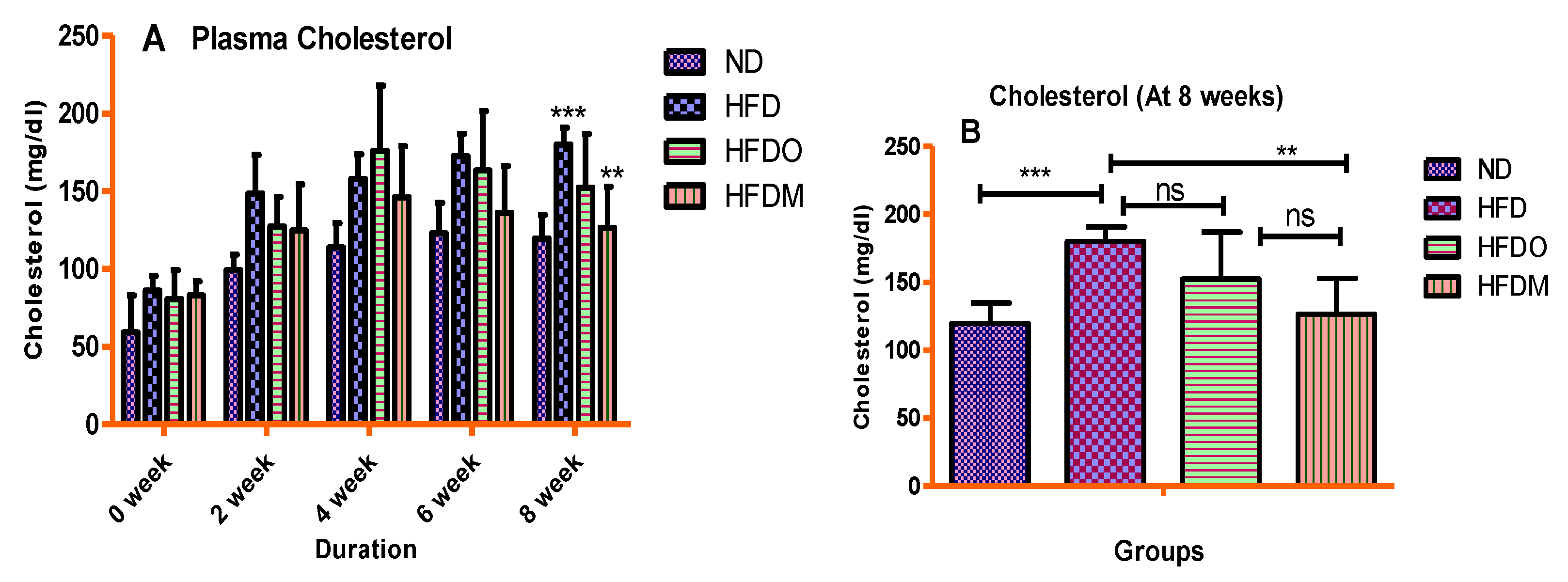
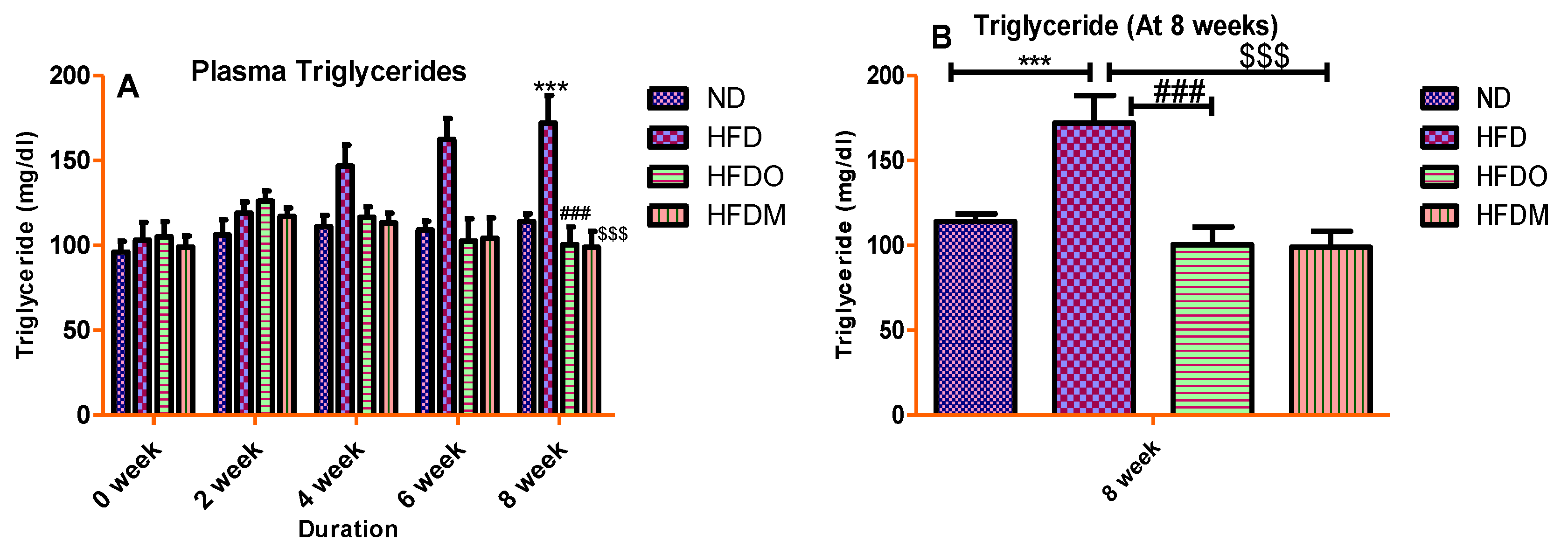
3.4. Biochemical Parameters
3.5. Effect of MESK on Histological Changes in Liver
3.6. Mangifera indica Extract Prevents Hepatic Fibrosis
3.7. Assessment of Intestinal Permeability
3.8. Expression of Zonula Occludens-1 in the Small Intestine
3.9. Expression of Claudin-1 in the Small Intestine
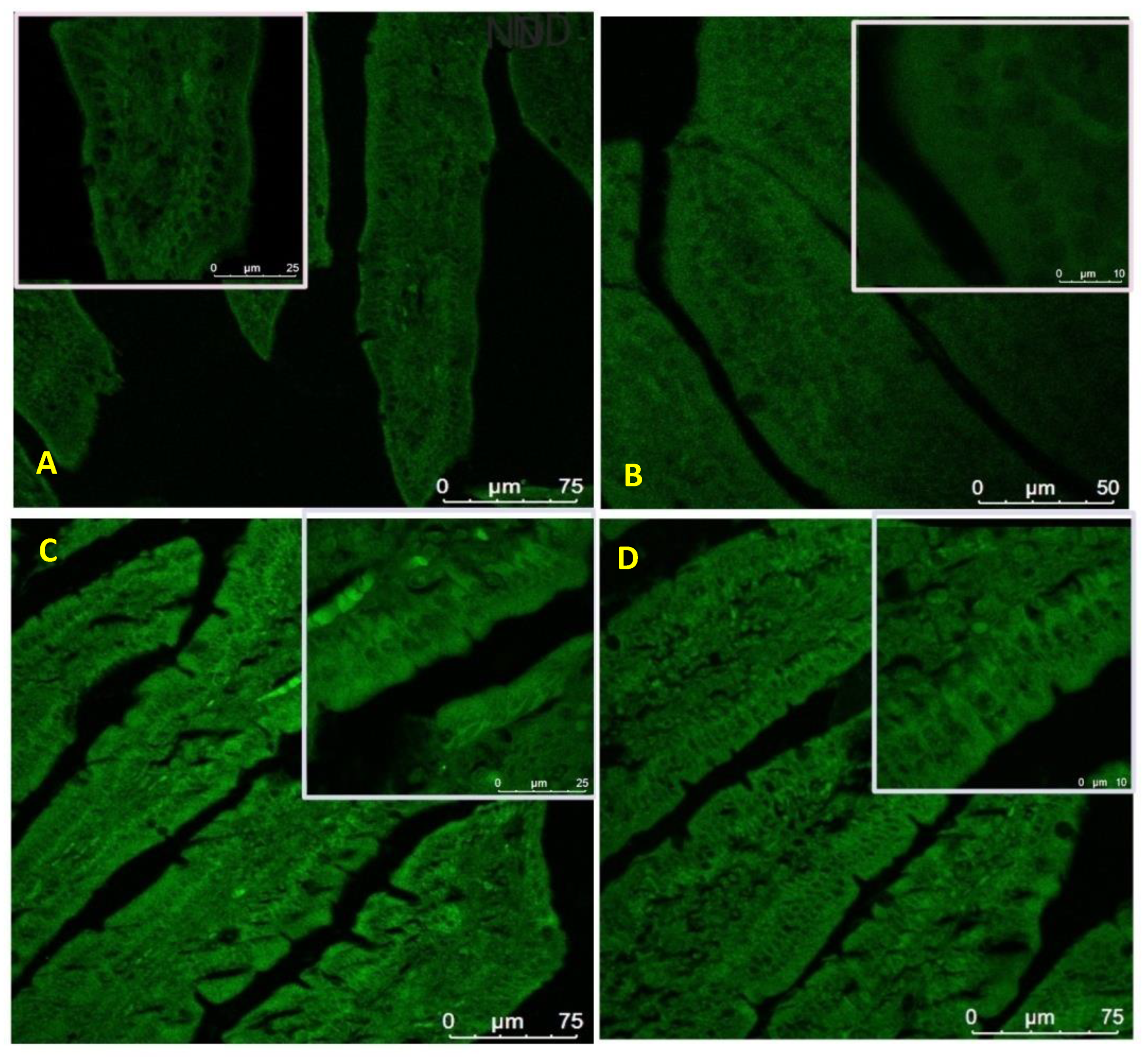
3.10. Expression of Nesfatin-1/NUCB-2 in the Small Intestine
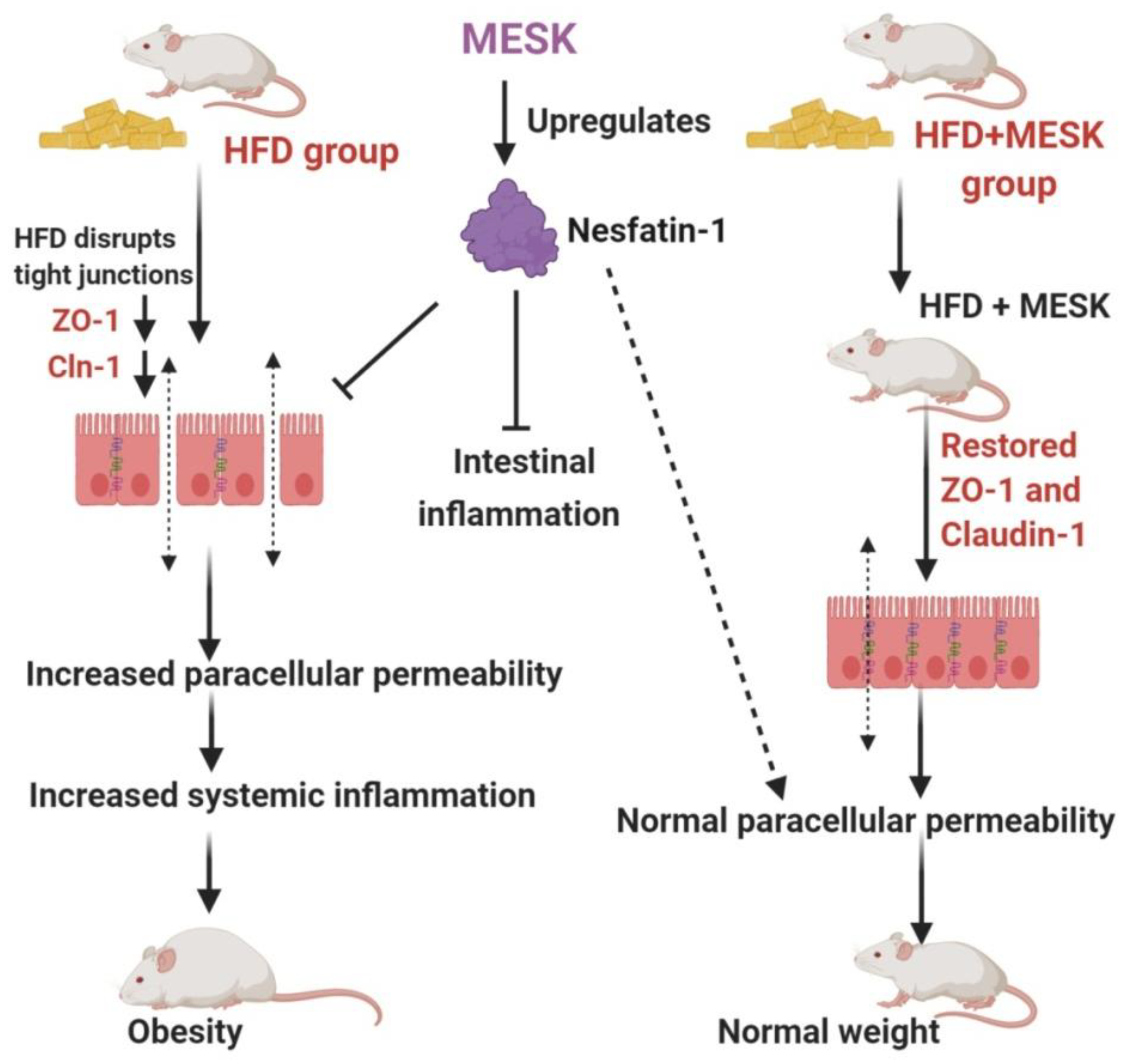
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BSA | Bovine Serum Albumin |
| EAT | Epididymal Adipose Tissue |
| EDTA | Ethylenediamine Tetra Acetic acid |
| FITC-dextran | Fluorescein Iso-Thio-Cyanate Dextran |
| GLP-1 | Glucagon-Like Peptide-1 |
| HB | Hepatocyte Ballooning |
| HFD | High Fat Diet |
| HPV | Hepatic Portal Vein |
| LPS | Lipopolysaccharides |
| MESK | 50% Ethanolic Extract of Mangifera indica seed kernel |
| NAFLD | Non-Alcoholic Fatty Liver Disease |
| NUCB-2 | Nucleobindin-2 |
| PYY | Peptide YY |
| TLR | Toll-Like Receptor |
| ZO-1 | Zonula occludens-1 |
References
- Turner, J.R. Intestinal mucosal barrier function in health & disease. Nat. Rev. Immunol. 2009, 9, 799–809. [Google Scholar] [PubMed]
- Musso, G.; Gambino, R.; Cassader, M. Gut microbiota as a regulator of energy homeostasis and ectopic fat deposition: Mechanisms and implications for metabolic disorders. Curr. Opin. Lipidol. 2010, 21, 76–83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ott, B.; Skurk, T.; Hastreiter, L.; Lagkouvardos, I.; Fischer, S.; Büttner, J.; Kellerer, T.; Clavel, T.; Rychlik, M.; Haller, D.; et al. Effect of caloric restriction on gut permeability, inflammation markers, and fecal microbiota in obese women. Sci. Rep. 2017, 7, 11955. [Google Scholar] [CrossRef] [PubMed]
- Araújo, J.R.; Tomas, J.; Brenner, C.; Sansonetti, P.J. Impact of high-fat diet on the intestinal microbiota and small intestinal physiology before and after the onset of obesity. Biochimie 2017, 141, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Yang, X.J. Effects of a high fat diet on intestinal microbiota and gastrointestinal diseases. World J. Gastroenterol. 2016, 22, 8905–8909. [Google Scholar] [CrossRef]
- Sarwar, R.; Pierce, N.; Koppe, S. Obesity and non-alcoholic fatty liver disease: Current perspectives. Diabet. Metab. Syndr. Obes. 2018, 11, 533–542. [Google Scholar] [CrossRef] [Green Version]
- Picchi, M.G.; de Mattos, A.M.; Barbosa, M.R.; Duarte, C.P.; de Azevedo, M.; Portari, G.V.; Jordão, A.A. A high-fat diet as a model of fatty liver disease in rats. Acta Cir. Bras. 2011, 26, 25–30. [Google Scholar] [CrossRef] [Green Version]
- Onyekwere, C.A.; Ogbera, A.O.; Samaila, A.A.; Balogun, B.O.; Abdulkareem, F.B. Non-alcoholic fatty liver disease: Synopsis of current developments. Niger. J. Clin. Pract. 2015, 18, 703–712. [Google Scholar] [CrossRef] [Green Version]
- Poeta, M.; Pierri, L.; Vajro, P. Gut-Liver axis derangement in non-alcoholic fatty liver disease. Children 2017, 4, 66. [Google Scholar] [CrossRef] [Green Version]
- Filippatos, T.D.; Derdemezis, C.S.; Gazi, I.F.; Nakou, E.S.; Mikhailidis, D.P.; Elisaf, M.S. Orlistat-associated adverse effects and drug interactions: A critical review. Drug Saf. 2008, 31, 53–65. [Google Scholar] [CrossRef]
- de Freitas Junior, L.M.; de Almeida, E.B., Jr. Medicinal plants for the treatment of obesity: Ethnopharmacological approach and chemical and biological studies. Am. J. Transl. Res. 2017, 9, 2050–2064. [Google Scholar] [PubMed]
- Lauricella, M.; Emanuele, S.; Calvaruso, G.; Giuliano, M.; D’Anneo, A. Multifaceted health benefits of Mangifera indica L. (Mango): The inestimable value of orchards recently planted in sicilian rural areas. Nutrients 2017, 9, 525. [Google Scholar] [CrossRef] [PubMed]
- Kankanampati, K.; Mishra, R.; Pushkala, K.; Velayudam, D.; Kapur, S. Ethanolic extract of Mangiferaindica seed kernel ameliorates visceral fat via improvement in lipid metabolism in high fat diet inducedobese mice. J. Pharm. Res. 2012, 5, 4974–4978. [Google Scholar]
- Gurjar, S.; Pal, A.; Kapur, S. Triphalaand its constituents ameliorate visceral adiposity in high fat diet –induced obese mice. Altern. Ther. Health Med. 2012, 18, 38–45. [Google Scholar] [PubMed]
- Numonov, S.R.; Qureshi, M.N.; Aisa, H.A. Development of HPLC protocol and simultaneous quantification of four free flavonoids from dracocephalum heterophyllum benth. Int. J. Anal. Chem. 2015, 2015, 503139. [Google Scholar] [CrossRef] [Green Version]
- Segnani, C.; Ippolito, C.; Antonioli, L.; Pellegrini, C.; Blandizzi, C.; Dolfi, A.; Bernardini, N. Histochemical detection of collagen fibers by sirius red/fast green is more sensitive than van gieson or sirius red alone in normal and inflamed rat colon. PLoS ONE 2015, 10, e0144630. [Google Scholar] [CrossRef]
- Keira, S.M.; Ferreira, L.M.; Gragnani, A.; Duarte, I.S.; Barbosa, J. Experimental model for collagen estimation in cell culture. Acta Cir. Bras. 2004, 19, 17–22. [Google Scholar] [CrossRef] [Green Version]
- Woting, A.; Blaut, M. Small intestinal permeability and gut-transit time determined with low and high molecular weight fluorescein isothiocyanate-dextrans in C3H mice. Nutrients 2018, 10, 685. [Google Scholar] [CrossRef] [Green Version]
- Gumber, S.; Nusrat, A.; Villinger, F. Immunohistological characterization of intercellular junction proteins in rhesus macaque intestine. Exp. Toxicol. Pathol. 2014, 66, 437–444. [Google Scholar] [CrossRef] [Green Version]
- Al Obeed, O.A.; Alkhayal, K.A.; Al Sheikh, A.; Zubaidi, A.M.; Vaali-Mohammed, M.A.; Boushey, R.; Mckerrow, J.H.; Abdulla, M.H. Increased expression of tumor necrosis factor-α is associated with advanced colorectal cancer stages. World J. Gastroenterol. 2014, 20, 18390–18396. [Google Scholar] [CrossRef]
- Mujawdiya, P.K.; Kapur, S. Screening of antioxidant and α-glucosidase inhibitory activities of Indian medicinal plants. Curr. Enzym. Inhib. 2020, 16, 145–154. [Google Scholar] [CrossRef]
- Caldwell, S.; Ikura, Y.; Dias, D.; Isomoto, K.; Yabu, A.; Moskaluk, C.; Pramoonjago, P.; Simmons, W.; Scruggs, H.; Rosenbaum, N.; et al. Hepatocellular ballooning in NASH. J. Hepatol. 2010, 53, 719–723. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ding, S.; Lund, P.K. Role of intestinal inflammation as an early event in obesity and insulin resistance. Curr. Opin. Clin. Nutr. Metab. Care 2011, 14, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Bischoff, S.C.; Barbara, G.; Buurman, W.; Ockhuizen, T.; Schulzke, J.D.; Serino, M.; Tilg, H.; Watson, A.; Wells, J.M. Intestinal permeability–A new target for disease prevention and therapy. BMC Gastroenterol. 2014, 14, 189. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cani, P.D.; Amar, J.; Iglesias, M.A.; Poggi, M.; Knauf, C.; Bastelica, D.; Neyrinck, A.M.; Fava, F.; Tuohy, K.M.; Chabo, C.; et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 2007, 56, 1761–1772. [Google Scholar] [CrossRef] [Green Version]
- Alessio, F. Gut permeability, obesity, and metabolic disorders: Who is the chicken and who is the egg? Am. J. Clin. Nutr. 2017, 105, 3–4. [Google Scholar] [CrossRef] [Green Version]
- Genser, L.; Aguanno, D.; Soula, H.A.; Liping, D.; Laurence, T.; Karen, A.; Salem, J.E.; Vaillant, J.E.; Oppert, J.M.; Laugerette, F.; et al. Increased jejunal permeability in human obesity is revealed by a lipid challenge and is linked to inflammation and type 2 diabetes. J. Pathol. 2018, 246, 217–230. [Google Scholar] [CrossRef]
- Paola, B.; Ignazio, C.; Vincenza, D.L.; Andrea, B.; Massimo, P.; Giorgio, P.; Diego, M. Increased intestinal permeability in obese mice: New evidence in the pathogenesis of nonalcoholic steatohepatitis. Am. J. Physiol. Gastrointest. Liver Physiol. 2007, 292, G518–G525. [Google Scholar]
- Sambolin-Escobales, L.; Nazario, M.; Torres-Aguiar, R.; Cruz, M.L.; Yamamura, Y.; Appleyard, C.B.; Chompre, G. High fat diet induces decreased levels of colonic tight junctions and inflammatory cytokine expression in sprague dawley rats. FASEB J. 2016, 30. [Google Scholar] [CrossRef]
- Sung, Y.Y.; Kim, S.H.; Yoo, B.W.; Kim, H.K. The nutritional composition and anti-obesity effects of an herbal mixed extract containing Allium fistulosum and Viola mandshurica in high-fat-diet-induced obese mice. BMC Complement. Altern. Med. 2015, 15, 370. [Google Scholar] [CrossRef] [Green Version]
- Ohashi, T.; Nakade, Y.; Ibusuki, M.; Kitano, R.; Yamauchi, T.; Kimoto, S.; Inoue, T.; Kobayashi, Y.; Sumida, Y.; Ito, K.; et al. Conophylline inhibits high fat diet-induced non-alcoholic fatty liver disease in mice. PLoS ONE 2019, 14, e0210068. [Google Scholar] [CrossRef] [PubMed]
- Ipsen, D.H.; Lykkesfeldt, J.; Tveden-Nyborg, P. Molecular mechanisms of hepatic lipid accumulation in non-alcoholic fatty liver disease. Cell Mol. Life Sci. 2018, 75, 3313–3327. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lackner, C. Hepatocellular ballooning in nonalcoholic steatohepatitis: The pathologist’s perspective. Expert Rev. Gastroenterol. Hepatol. 2011, 5, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Vermette, D.; Hu, P.; Canarie, M.F.; Funaro, M.; Glover, J.; Pierce, R.W. Tight junction structure, function, and assessment in the critically ill: A systematic review. Intensive Care Med. Exp. 2018, 6, 37. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Jiang, Z.; Han, S.; Li, L.Y.; Qiao, F.; Zhang, M.L.; Du, Z.Y. Inhibition of intestinal lipases alleviates the adverse effects caused by high-fat diet in Nile tilapia. Fish Physiol. Biochem. 2020, 46, 111–123. [Google Scholar] [CrossRef]
- Ayada, C.; Toru, Ü.; Korkut, Y. Nesfatin-1 and its effects on different systems. Hippokratia 2015, 19, 4–10. [Google Scholar]
- Psilopanagioti, A.; Nikou, S.; Papadaki, H. Nucleobindin-2/nesfatin-1 in the human hypothalamus is reduced in obese subjects and colocalizes with oxytocin, vasopressin, melanin-concentrating hormone, and cocaine- and amphetamine-regulated transcript. Neuroendocrinology 2019, 108, 190–200. [Google Scholar] [CrossRef]
- Sun, S.; Yang, H. Tissue-specific localization NUCB2/nesfatin-1 in the liver and heart of mouse fetus. Dev. Reprod. 2018, 22, 331–339. [Google Scholar] [CrossRef] [Green Version]
- Chaolu, H.; Asakawa, A.; Ushikai, M.; Li, Y.X.; Cheng, K.C.; Li, J.B.; Zoshiki, T.; Terashi, M.; Tanaka, C.; Atsuchi, K.; et al. Effect of exercise and high-fat diet on plasma adiponectin and nesfatin levels in mice. Exp. Ther. Med. 2011, 2, 369–373. [Google Scholar] [CrossRef]
- Zhang, A.Q.; Li, X.L.; Jiang, C.Y.; Lin, L.; Shi, R.H.; Chen, J.D.; Oomura, Y. Expression of nesfatin-1/NUCB2 in rodent digestive system. World J. Gastroenterol. 2010, 16, 1735–1741. [Google Scholar] [CrossRef]
- Cerit, K.K.; Turkan, K.; Damla, Y.; Eyuboglu, I.P.; Sirvanci, S.; Akkiprik, M.; Iksu, B.; Dagli, E.T.; Yegan, B.C. Nesfatin-1 ameliorates oxidative bowel injury in rats with necrotizing enterocolitis: The role of the microbiota composition and claudin-3 expression. J. Pediatric Surg. 2020. [Google Scholar] [CrossRef]
- Olszanecka-Glinianowicz, M.; Dąbrowski, P.; Kocełak, P.; Janowska, J.; Smertka, M.; Jonderko, K.; Chudek, J. Long-term inhibition of intestinal lipase by orlistat improves release of gut hormones increasing satiety in obese women. Pharmacol. Rep. 2013, 65, 666–671. [Google Scholar] [CrossRef]
- Tian, Z.B.; Deng, R.J.; Sun, G.R.; Wei, L.Z.; Kong, X.J.; Ding, X.L.; Jing, X.; Zhang, C.P.; Ge, Y.L. Expression of gastrointestinal nesfatin-1 and gastric emptying in ventromedial hypothalamic nucleus and ventrolateral hypothalamic nucleus-lesioned rats. World J. Gastroenterol. 2014, 20, 6897–6905. [Google Scholar] [CrossRef] [PubMed]
- Kolgazi, M.; Ozdemir-Kumral, Z.N.; Cantali-Ozturk, C.; Demirci, E.K.; Yuksel, M.; Sirvanci, S.; Yegen, B.C. Anti-inflammatory effects of nesfatin-1 on acetic acid-induced gastric ulcer in rats: Involvement of cyclo-oxygenase pathway. J. Physiol. Pharmacol. 2017, 68, 765–777. [Google Scholar] [PubMed]
- Ma, T.Y.; Iwamoto, G.K.; Hoa, N.T.; Akotia, V.; Pedram, A.; Boivin, M.A.; Said, H.M. TNF-alpha-induced increase in intestinal epithelial tight junction permeability requires NF-kappa B activation. Am. J. Physiol. Gastrointest. Liver Physiol. 2004, 286, G367–G376. [Google Scholar] [CrossRef] [Green Version]
- Al-Sadi, R.; Guo, S.; Ye, D.; Ma, T.Y. TNF-α modulation of intestinal epithelial tight junction barrier is regulated by ERK1/2 activation of Elk-1. Am. J. Pathol. 2013, 183, 1871–1884. [Google Scholar] [CrossRef] [Green Version]








© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mujawdiya, P.K.; Sharma, P.; Sharad, S.; Kapur, S. Reversal of Increase in Intestinal Permeability by Mangifera indica Seed Kernel Extract in High-Fat Diet-Induced Obese Mice. Pharmaceuticals 2020, 13, 190. https://0-doi-org.brum.beds.ac.uk/10.3390/ph13080190
Mujawdiya PK, Sharma P, Sharad S, Kapur S. Reversal of Increase in Intestinal Permeability by Mangifera indica Seed Kernel Extract in High-Fat Diet-Induced Obese Mice. Pharmaceuticals. 2020; 13(8):190. https://0-doi-org.brum.beds.ac.uk/10.3390/ph13080190
Chicago/Turabian StyleMujawdiya, Pavan Kumar, Pravesh Sharma, Shashwat Sharad, and Suman Kapur. 2020. "Reversal of Increase in Intestinal Permeability by Mangifera indica Seed Kernel Extract in High-Fat Diet-Induced Obese Mice" Pharmaceuticals 13, no. 8: 190. https://0-doi-org.brum.beds.ac.uk/10.3390/ph13080190