Antifibrotic and Anti-Inflammatory Actions of α-Melanocytic Hormone: New Roles for an Old Player
Abstract
:1. Introduction
2. Melanocortins
2.1. Ligands
2.2. Receptors
3. Anti-Inflammatory Effects of α-MSH
3.1. Fever and Multiple Organ Dysfunction Syndrome (MODS)
3.2. α-MSH and the Respiratory System
3.3. α-MSH and the Eye
3.4. α-MSH and Gastrointestinal System
3.5. α-MSH and Nervous System
3.6. α-MSH and Skin
3.7. α-MSH and the Musculoskeletal System
3.8. Other Anti-Inflammatory Actions
4. Mechanisms of Anti-Inflammatory Effects of α-MSH
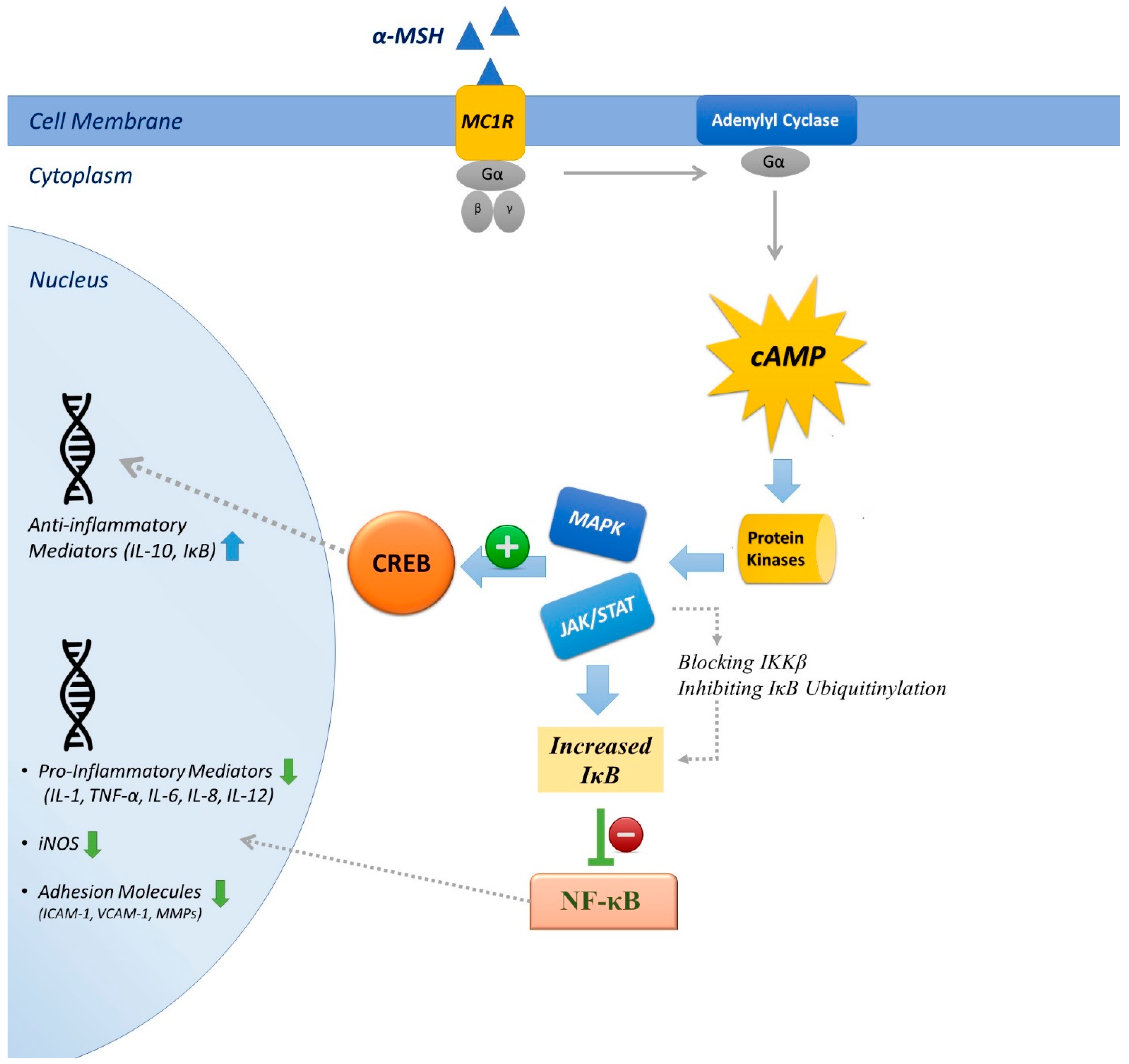
4.1. Inhibition of NF-κB
4.2. Suppression of Proinflammatory Cytokines
4.3. Inhibition of Adhesion Molecules
4.4. Suppression of Non-Cytokine Inflammatory Mediators
4.5. Induction of Regulatory T Cells (Tregs)
4.6. Promotion of Efferocytosis
5. α-MSH and Tissue Repair and Remodeling
6. Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tao, Y.X. Melanocortin receptors. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 2411–2413. [Google Scholar] [CrossRef] [PubMed]
- Gallo-Payet, N. 60 YEARS OF POMC: Adrenal and extra-adrenal functions of ACTH. J. Mol. Endocrinol. 2016, 56, T135–T156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hench, P.S.; Kendall, E.C.; Slocumb, C.H.; Polley, H.F. The effect of a hormone of the adrenal cortex (17-hydroxy-11-dehydrocorticosterone: Compound E) and of pituitary adrenocortical hormone in arthritis: Preliminary report. Ann. Rheum. Dis. 1949, 8, 97–104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, W.; Guo, D.Y.; Lin, Y.J.; Tao, Y.X. Melanocortin Regulation of Inflammation. Front. Endocrinol. 2019, 10, 683. [Google Scholar] [CrossRef]
- Smith, P.C.; Martínez, C.; Martínez, J.; McCulloch, C.A. Role of Fibroblast Populations in Periodontal Wound Healing and Tissue Remodeling. Front. Physiol. 2019, 10, 270. [Google Scholar] [CrossRef] [Green Version]
- Dong, J.; Ma, Q. Myofibroblasts and lung fibrosis induced by carbon nanotube exposure. Part. Fibre Toxicol. 2016, 13, 60. [Google Scholar] [CrossRef] [Green Version]
- Wójcik-Pszczoła, K.; Jakieła, B.; Plutecka, H.; Koczurkiewicz, P.; Madeja, Z.; Michalik, M.; Sanak, M. Connective tissue growth factor regulates transition of primary bronchial fibroblasts to myofibroblasts in asthmatic subjects. Cytokine 2018, 102, 187–190. [Google Scholar] [CrossRef]
- Toda, C.; Santoro, A.; Kim, J.D.; Diano, S. POMC Neurons: From Birth to Death. Annu. Rev. Physiol. 2017, 79, 209–236. [Google Scholar] [CrossRef] [Green Version]
- Zhu, M.; Xu, B.; Wang, M.; Liu, S.; Zhang, Y.; Zhang, C. Pharmacological modulation of MRAP2 protein on melanocortin receptors in the sea lamprey. Endocr. Connect. 2019, 8, 378–388. [Google Scholar] [CrossRef] [Green Version]
- Maddila, S.C.; Busch-Dienstfertig, M.; Stein, C. B Lymphocytes Express Pomc mRNA, Processing Enzymes and β-Endorphin in Painful Inflammation. J. Neuroimmune Pharmacol. 2017, 12, 180–186. [Google Scholar] [CrossRef]
- Chrétien, M.; Mbikay, M. 60 YEARS OF POMC: From the prohormone theory to pro-opiomelanocortin and to proprotein convertases (PCSK1 to PCSK9). J. Mol. Endocrinol. 2016, 56, T49–T62. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cawley, N.X.; Li, Z.; Loh, Y.P. 60 YEARS OF POMC: Biosynthesis, trafficking, and secretion of pro-opiomelanocortin-derived peptides. J. Mol. Endocrinol. 2016, 56, T77–T97. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lowry, P. 60 YEARS OF POMC: Purification and biological characterisation of melanotrophins and corticotrophins. J. Mol. Endocrinol. 2016, 56, T1–T12. [Google Scholar] [CrossRef] [Green Version]
- Winget, M.D.; Ericson, M.D.; Freeman, K.T.; Haskell-Luevano, C. Single Nucleotide Polymorphisms in the Melanocortin His-Phe-Arg-Trp Sequences Decrease Tetrapeptide Potency and Efficacy. ACS Med. Chem. Lett. 2020, 11, 272–277. [Google Scholar] [CrossRef]
- Mowlazadeh Haghighi, S.; Zhou, Y.; Dai, J.; Sawyer, J.R.; Hruby, V.J.; Cai, M. Replacement of Arg with Nle and modified D-Phe in the core sequence of MSHs, Ac-His-D-Phe-Arg-Trp-NH(2), leads to hMC1R selectivity and pigmentation. Eur. J. Med. Chem. 2018, 151, 815–823. [Google Scholar] [CrossRef]
- Ghaddhab, C.; Vuissoz, J.M.; Deladoëy, J. From Bioinactive ACTH to ACTH Antagonist: The Clinical Perspective. Front. Endocrinol. 2017, 8, 17. [Google Scholar] [CrossRef] [Green Version]
- Lindner, J.M.; Suhr, A.C.; Grimm, S.H.; Möhnle, P.; Vogeser, M.; Briegel, J. The dynamics of a serum steroid profile after stimulation with intravenous ACTH. J. Pharm Biomed. Anal. 2018, 151, 159–163. [Google Scholar] [CrossRef] [PubMed]
- Ruggiero, C.; Lalli, E. Impact of ACTH Signaling on Transcriptional Regulation of Steroidogenic Genes. Front. Endocrinol. 2016, 7, 24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lewis, A.E.; Aesoy, R.; Bakke, M. Role of EPAC in cAMP-Mediated Actions in Adrenocortical Cells. Front. Endocrinol. 2016, 7, 63. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Saarinen, A.M.; Campbell, L.E.; De Filippis, E.A.; Liu, J. Regulation of Lipolytic Response and Energy Balance by Melanocortin 2 Receptor Accessory Protein (MRAP) in Adipocytes. Diabetes 2018, 67, 222–234. [Google Scholar] [CrossRef]
- Betz, M.J.; Hatiboglu, N.; Mauracher, B.; Hadaschik, D.; Sauter, A.; Demmelmair, H.; Koletzko, B.; Beuschlein, F.; Slawik, M. Mc2 receptor knockdown modulates differentiation and lipid composition in adipocytes. Horm. Metab. Res. 2012, 44, 670–675. [Google Scholar] [CrossRef] [PubMed]
- Gioldasi, S.; Karvela, A.; Rojas-Gil, A.P.; Rodi, M.; de Lastic, A.L.; Thomas, I.; Spiliotis, B.E.; Mouzaki, A. Metabolic Association between Leptin and the Corticotropin Releasing Hormone. Endocr. Metab. Immune Disord. Drug Targets 2019, 19, 458–466. [Google Scholar] [CrossRef] [PubMed]
- Sadeghi, F.; Vahednia, E.; Naderi Meshkin, H.; Kerachian, M.A. The effect of adrenocorticotropic hormone on alpha-2-macroglobulin in osteoblasts derived from human mesenchymal stem cells. J. Cell. Mol. Med. 2020, 24, 4784–4790. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Belaya, Z.E.; Grebennikova, T.A.; Melnichenko, G.A.; Nikitin, A.G.; Solodovnikov, A.G.; Brovkina, O.I.; Grigoriev, A.U.; Rozhinskaya, L.Y.; Dedov, I.I. Effects of endogenous hypercortisolism on bone mRNA and microRNA expression in humans. Osteoporos. Int. 2018, 29, 211–221. [Google Scholar] [CrossRef]
- Belotti, E.M.; Amweg, A.N.; Matiller, V.; Varela, M.L.; Stassi, A.F.; Velázquez, M.M.L.; Ortega, H.H.; Rey, F.; Salvetti, N.R. Effects of adrenocorticotrophic hormone on the expression of matrix metalloproteinases and their inhibitors in the bovine ovary. Reprod. Fertil. Dev. 2020, 32, 748–762. [Google Scholar] [CrossRef]
- Tishevskaya, N.V.; Gevorkyan, N.M.; Kozlova, N.I. Sensitivity of T-Lymphocytes to Hormones of the Anterior Pituitary Gland. Uspekhi Fiziol. Nauk 2017, 48, 80–90. [Google Scholar]
- Si, J.; Ge, Y.; Zhuang, S.; Wang, L.J.; Chen, S.; Gong, R. Adrenocorticotropic hormone ameliorates acute kidney injury by steroidogenic-dependent and -independent mechanisms. Kidney Int. 2013, 83, 635–646. [Google Scholar] [CrossRef] [Green Version]
- Chen, S.; Zhu, B.; Yin, C.; Liu, W.; Han, C.; Chen, B.; Liu, T.; Li, X.; Chen, X.; Li, C.; et al. Palmitoylation-dependent activation of MC1R prevents melanomagenesis. Nature 2017, 549, 399–403. [Google Scholar] [CrossRef] [Green Version]
- Wang, M.; Zhi, D.; Wang, H.; Ru, Y.; Ren, H.; Wang, N.; Liu, Y.; Li, Y.; Li, H. TAT-HSA-α-MSH fusion protein with extended half-life inhibits tumor necrosis factor-α in brain inflammation of mice. Appl. Microbiol. Biotechnol. 2016, 100, 5353–5361. [Google Scholar] [CrossRef]
- Ramírez, D.; Saba, J.; Turati, J.; Carniglia, L.; Imsen, M.; Mohn, C.; Scimonelli, T.; Durand, D.; Caruso, C.; Lasaga, M. NDP-MSH reduces oxidative damage induced by palmitic acid in primary astrocytes. J. Neuroendocrinol. 2019, 31, e12673. [Google Scholar] [CrossRef]
- Zhang, C.; Chery, S.; Lazerson, A.; Altman, N.H.; Jackson, R.; Holt, G.; Campos, M.; Schally, A.V.; Mirsaeidi, M. Anti-inflammatory effects of α-MSH through p-CREB expression in sarcoidosis like granuloma model. Sci. Rep. 2020, 10, 7277. [Google Scholar] [CrossRef] [PubMed]
- Nieto, J.E.; Casanova, I.; Serna-Ojeda, J.C.; Graue-Hernández, E.O.; Quintana, G.; Salazar, A.; Jiménez-Martinez, M.C. Increased Expression of TLR4 in Circulating CD4+T Cells in Patients with Allergic Conjunctivitis and In Vitro Attenuation of Th2 Inflammatory Response by Alpha-MSH. Int. J. Mol. Sci. 2020, 21, 7861. [Google Scholar] [CrossRef] [PubMed]
- Can, V.C.; Locke, I.C.; Kaneva, M.K.; Kerrigan, M.J.P.; Merlino, F.; De Pascale, C.; Grieco, P.; Getting, S.J. Novel anti-inflammatory and chondroprotective effects of the human melanocortin MC1 receptor agonist BMS-470539 dihydrochloride and human melanocortin MC3 receptor agonist PG-990 on lipopolysaccharide activated chondrocytes. Eur. J. Pharmacol. 2020, 872, 172971. [Google Scholar] [CrossRef] [PubMed]
- Lužnik, Z.; Sun, Z.; Nakagawa, H.; Taylor, A.W.; Jurkunas, U.V.; Yin, J.; Dana, R. Association of α-Melanocyte-Stimulating Hormone With Corneal Endothelial Cell Survival During Oxidative Stress and Inflammation-Induced Cell Loss in Donor Tissue. JAMA Ophthalmol. 2020, 138, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Kadiri, J.J.; Thapa, K.; Kaipio, K.; Cai, M.; Hruby, V.J.; Rinne, P. Melanocortin 3 receptor activation with [D-Trp8]-γ-MSH suppresses inflammation in apolipoprotein E deficient mice. Eur. J. Pharmacol. 2020, 880, 173186. [Google Scholar] [CrossRef] [PubMed]
- Getting, S.J.; Allcock, G.H.; Flower, R.; Perretti, M. Natural and synthetic agonists of the melanocortin receptor type 3 possess anti-inflammatory properties. J. Leukoc. Biol. 2001, 69, 98–104. [Google Scholar]
- Gómez-SanMiguel, A.B.; Villanúa, M.; Martín, A.I.; López-Calderón, A. D-TRP(8)-γMSH Prevents the Effects of Endotoxin in Rat Skeletal Muscle Cells through TNFα/NF-KB Signalling Pathway. PLoS ONE 2016, 11, e0155645. [Google Scholar] [CrossRef]
- Gómez-SanMiguel, A.B.; Martín, A.I.; Nieto-Bona, M.P.; Fernández-Galaz, C.; Villanúa, M.; López-Calderón, A. The melanocortin receptor type 3 agonist d-Trp(8)-γMSH decreases inflammation and muscle wasting in arthritic rats. J. Cachexia Sarcopenia Muscle 2016, 7, 79–89. [Google Scholar] [CrossRef]
- Madeira, M.F.; Queiroz-Junior, C.M.; Montero-Melendez, T.; Werneck, S.M.; Corrêa, J.D.; Soriani, F.M.; Garlet, G.P.; Souza, D.G.; Teixeira, M.M.; Silva, T.A.; et al. Melanocortin agonism as a viable strategy to control alveolar bone loss induced by oral infection. FASEB J. 2016, 30, 4033–4041. [Google Scholar] [CrossRef] [Green Version]
- Dores, R.M.; Liang, L.; Davis, P.; Thomas, A.L.; Petko, B. 60 YEARS OF POMC: Melanocortin receptors: Evolution of ligand selectivity for melanocortin peptides. J. Mol. Endocrinol. 2016, 56, T119–T133. [Google Scholar] [CrossRef] [Green Version]
- Mountjoy, K.G.; Robbins, L.S.; Mortrud, M.T.; Cone, R.D. The cloning of a family of genes that encode the melanocortin receptors. Science 1992, 257, 1248–1251. [Google Scholar] [CrossRef]
- Chhajlani, V.; Wikberg, J.E. Molecular cloning and expression of the human melanocyte stimulating hormone receptor cDNA. FEBS Lett. 1992, 309, 417–420. [Google Scholar] [CrossRef] [Green Version]
- Cone, R.D. Studies on the physiological functions of the melanocortin system. Endocr. Rev. 2006, 27, 736–749. [Google Scholar] [CrossRef] [PubMed]
- Irani, B.G.; Haskell-Luevano, C. Feeding effects of melanocortin ligands—A historical perspective. Peptides 2005, 26, 1788–1799. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, C.; Liu, Y.; Liu, J.; Wang, H.Y.; Liu, A.F.; He, D.Q. Agouti signalling protein (ASIP) gene: Molecular cloning, sequence characterisation and tissue distribution in domestic goose. Br. Poult. Sci. 2016, 57, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Almathen, F.; Elbir, H.; Bahbahani, H.; Mwacharo, J.; Hanotte, O. Polymorphisms in MC1R and ASIP Genes are Associated with Coat Color Variation in the Arabian Camel. J. Hered. 2018, 109, 700–706. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Wikberg, J.E.; Chhajlani, V. Expression of melanocortin 1 receptor in periaqueductal gray matter. Neuroreport 1995, 6, 2193–2196. [Google Scholar] [CrossRef]
- Zhang, Y.; Fang, C.; Wang, R.E.; Wang, Y.; Guo, H.; Guo, C.; Zhao, L.; Li, S.; Li, X.; Schultz, P.G.; et al. A tumor-targeted immune checkpoint blocker. Proc. Natl. Acad. Sci. USA 2019, 116, 15889–15894. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.; Ju, W.; Tin, T.D.; Kim, J.; Lee, J.S.; Park, C.H.; Kwak, S.H. Effect of BMS-470539 on lipopolysaccharide-induced neutrophil activation. Korean J. Anesthesiol. 2020, 73, 151–157. [Google Scholar] [CrossRef] [Green Version]
- Jang, E.A.; Kim, J.Y.; Tin, T.D.; Song, J.A.; Lee, S.H.; Kwak, S.H. The effects of BMS-470539 on lipopolysaccharide-induced acute lung injury. Acute Crit. Care 2019, 34, 133–140. [Google Scholar] [CrossRef]
- Taherzadeh, S.; Sharma, S.; Chhajlani, V.; Gantz, I.; Rajora, N.; Demitri, M.T.; Kelly, L.; Zhao, H.; Ichiyama, T.; Catania, A.; et al. alpha-MSH and its receptors in regulation of tumor necrosis factor-alpha production by human monocyte/macrophages. Am. J. Physiol. 1999, 276, R1289–R1294. [Google Scholar] [CrossRef] [PubMed]
- Neumann Andersen, G.; Nagaeva, O.; Mandrika, I.; Petrovska, R.; Muceniece, R.; Mincheva-Nilsson, L.; Wikberg, J.E. MC(1) receptors are constitutively expressed on leucocyte subpopulations with antigen presenting and cytotoxic functions. Clin. Exp. Immunol. 2001, 126, 441–446. [Google Scholar] [CrossRef]
- Goldenberg, A.J.; Gehrand, A.L.; Waples, E.; Jablonski, M.; Hoeynck, B.; Raff, H. Effect of a melanocortin type 2 receptor (MC2R) antagonist on the corticosterone response to hypoxia and ACTH stimulation in the neonatal rat. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2018, 315, R128–R133. [Google Scholar] [CrossRef] [PubMed]
- Nensey, N.K.; Bodager, J.; Gehrand, A.L.; Raff, H. Effect of Novel Melanocortin Type 2 Receptor Antagonists on the Corticosterone Response to ACTH in the Neonatal Rat Adrenal Gland In Vivo and In Vitro. Front. Endocrinol 2016, 7, 23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grässel, S.; Opolka, A.; Anders, S.; Straub, R.H.; Grifka, J.; Luger, T.A.; Böhm, M. The melanocortin system in articular chondrocytes: Melanocortin receptors, pro-opiomelanocortin, precursor proteases, and a regulatory effect of alpha-melanocyte-stimulating hormone on proinflammatory cytokines and extracellular matrix components. Arthritis Rheum. 2009, 60, 3017–3027. [Google Scholar] [CrossRef]
- Böhm, M.; Grässel, S. Role of proopiomelanocortin-derived peptides and their receptors in the osteoarticular system: From basic to translational research. Endocr. Rev. 2012, 33, 623–651. [Google Scholar] [CrossRef] [Green Version]
- Roselli-Rehfuss, L.; Mountjoy, K.G.; Robbins, L.S.; Mortrud, M.T.; Low, M.J.; Tatro, J.B.; Entwistle, M.L.; Simerly, R.B.; Cone, R.D. Identification of a receptor for gamma melanotropin and other proopiomelanocortin peptides in the hypothalamus and limbic system. Proc. Natl. Acad. Sci. USA 1993, 90, 8856–8860. [Google Scholar] [CrossRef] [Green Version]
- Pei, H.; Patterson, C.M.; Sutton, A.K.; Burnett, K.H.; Myers, M.G., Jr.; Olson, D.P. Lateral Hypothalamic Mc3R-Expressing Neurons Modulate Locomotor Activity, Energy Expenditure, and Adiposity in Male Mice. Endocrinology 2019, 160, 343–358. [Google Scholar] [CrossRef] [Green Version]
- Butler, A.A.; Girardet, C.; Mavrikaki, M.; Trevaskis, J.L.; Macarthur, H.; Marks, D.L.; Farr, S.A. A Life without Hunger: The Ups (and Downs) to Modulating Melanocortin-3 Receptor Signaling. Front. Neurosci. 2017, 11, 128. [Google Scholar] [CrossRef] [Green Version]
- Eerola, K.; Virtanen, S.; Vähätalo, L.; Ailanen, L.; Cai, M.; Hruby, V.; Savontaus, M.; Savontaus, E. Hypothalamic γ-melanocyte stimulating hormone gene delivery reduces fat mass in male mice. J. Endocrinol. 2018, 239, 19–31. [Google Scholar] [CrossRef]
- Getting, S.J.; Riffo-Vasquez, Y.; Pitchford, S.; Kaneva, M.; Grieco, P.; Page, C.P.; Perretti, M.; Spina, D. A role for MC3R in modulating lung inflammation. Pulm. Pharmacol. Ther. 2008, 21, 866–873. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montero-Melendez, T.; Forfar, R.A.; Cook, J.M.; Jerman, J.C.; Taylor, D.L.; Perretti, M. Old drugs with new skills: Fenoprofen as an allosteric enhancer at melanocortin receptor 3. Cell Mol. Life Sci. 2017, 74, 1335–1345. [Google Scholar] [CrossRef] [Green Version]
- Juárez-Rebollar, D.; Rios, C.; Nava-Ruíz, C.; Méndez-Armenta, M. Metallothionein in Brain Disorders. Oxidative Med. Cell. Longev. 2017, 2017, 5828056. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kristensen, J.; Jonassen, T.E.; Rehling, M.; Tønnesen, E.; Sloth, E.; Nielsen, S.; Frøkiaer, J. The α-MSH analogue AP214 attenuates rise in pulmonary pressure and fall in ejection fraction in lipopolysaccharide-induced systemic inflammatory response syndrome in pigs. Clin. Physiol. Funct. Imaging 2011, 31, 54–60. [Google Scholar] [CrossRef]
- Montero-Melendez, T.; Patel, H.B.; Seed, M.; Nielsen, S.; Jonassen, T.E.; Perretti, M. The melanocortin agonist AP214 exerts anti-inflammatory and proresolving properties. Am. J. Pathol. 2011, 179, 259–269. [Google Scholar] [CrossRef]
- Montero-Melendez, T.; Gobbetti, T.; Cooray, S.N.; Jonassen, T.E.; Perretti, M. Biased agonism as a novel strategy to harness the proresolving properties of melanocortin receptors without eliciting melanogenic effects. J. Immunol. 2015, 194, 3381–3388. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Getting, S.J.; Gibbs, L.; Clark, A.J.; Flower, R.J.; Perretti, M. POMC gene-derived peptides activate melanocortin type 3 receptor on murine macrophages, suppress cytokine release, and inhibit neutrophil migration in acute experimental inflammation. J. Immunol. 1999, 162, 7446–7453. [Google Scholar]
- King, S.H.; Mayorov, A.V.; Balse-Srinivasan, P.; Hruby, V.J.; Vanderah, T.W.; Wessells, H. Melanocortin receptors, melanotropic peptides and penile erection. Curr. Top. Med. Chem. 2007, 7, 1098–1106. [Google Scholar]
- Gantz, I.; Miwa, H.; Konda, Y.; Shimoto, Y.; Tashiro, T.; Watson, S.J.; DelValle, J.; Yamada, T. Molecular cloning, expression, and gene localization of a fourth melanocortin receptor. J. Biol. Chem. 1993, 268, 15174–15179. [Google Scholar] [CrossRef]
- Gantz, I.; Fong, T.M. The melanocortin system. Am. J. Physiol. Endocrinol. Metab. 2003, 284, E468–E474. [Google Scholar] [CrossRef]
- Méquinion, M.; Foldi, C.J.; Andrews, Z.B. The Ghrelin-AgRP Neuron Nexus in Anorexia Nervosa: Implications for Metabolic and Behavioral Adaptations. Front. Nutr. 2019, 6, 190. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Morgan, D.A.; Cui, H.; Rahmouni, K. Activation of hypothalamic AgRP and POMC neurons evokes disparate sympathetic and cardiovascular responses. Am. J. Physiol. Circ. Physiol. 2020, 319, H1069–H1077. [Google Scholar] [CrossRef] [PubMed]
- Kishi, T.; Aschkenasi, C.J.; Lee, C.E.; Mountjoy, K.G.; Saper, C.B.; Elmquist, J.K. Expression of melanocortin 4 receptor mRNA in the central nervous system of the rat. J. Comp. Neurol. 2003, 457, 213–235. [Google Scholar] [CrossRef] [PubMed]
- Michael, N.J.; Caron, A.; Lee, C.E.; Castorena, C.M.; Lee, S.; Zigman, J.M.; Williams, K.W.; Elmquist, J.K. Melanocortin regulation of histaminergic neurons via perifornical lateral hypothalamic melanocortin 4 receptors. Mol. Metab. 2020, 35, 100956. [Google Scholar] [CrossRef]
- Demont, E.H.; Bailey, J.M.; Bit, R.A.; Brown, J.A.; Campbell, C.A.; Deeks, N.; Dowell, S.J.; Eldred, C.; Gaskin, P.; Gray, J.R.; et al. Discovery of Tetrahydropyrazolopyridine as Sphingosine 1-Phosphate Receptor 3 (S1P3)-Sparing S1P1 Agonists Active at Low Oral Doses. J. Med. Chem. 2016, 59, 1003–1020. [Google Scholar] [CrossRef]
- Muceniece, R.; Zvejniece, L.; Vilskersts, R.; Liepinsh, E.; Baumane, L.; Kalvinsh, I.; Wikberg, J.E.; Dambrova, M. Functional evaluation of THIQ, a melanocortin 4 receptor agonist, in models of food intake and inflammation. Basic Clin. Pharmacol. Toxicol. 2007, 101, 416–420. [Google Scholar] [CrossRef]
- Wessells, H.; Gralnek, D.; Dorr, R.; Hruby, V.J.; Hadley, M.E.; Levine, N. Effect of an alpha-melanocyte stimulating hormone analog on penile erection and sexual desire in men with organic erectile dysfunction. Urology 2000, 56, 641–646. [Google Scholar] [CrossRef]
- Molinoff, P.B.; Shadiack, A.M.; Earle, D.; Diamond, L.E.; Quon, C.Y. PT-141: A melanocortin agonist for the treatment of sexual dysfunction. Ann. N. Y. Acad. Sci. 2003, 994, 96–102. [Google Scholar] [CrossRef]
- Gantz, I.; Shimoto, Y.; Konda, Y.; Miwa, H.; Dickinson, C.J.; Yamada, T. Molecular cloning, expression, and characterization of a fifth melanocortin receptor. Biochem. Biophys. Res. Commun. 1994, 200, 1214–1220. [Google Scholar] [CrossRef] [Green Version]
- Xu, Y.; Guan, X.; Zhou, R.; Gong, R. Melanocortin 5 receptor signaling pathway in health and disease. Cell Mol. Life Sci. 2020, 77, 3831–3840. [Google Scholar] [CrossRef]
- Buggy, J.J. Binding of alpha-melanocyte-stimulating hormone to its G-protein-coupled receptor on B-lymphocytes activates the Jak/STAT pathway. Biochem. J. 1998, 331, 211–216. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taylor, A.; Namba, K. In vitro induction of CD25+ CD4+ regulatory T cells by the neuropeptide alpha-melanocyte stimulating hormone (alpha-MSH). Immunol. Cell Biol. 2001, 79, 358–367. [Google Scholar] [CrossRef] [PubMed]
- Trotta, M.C.; Maisto, R.; Alessio, N.; Hermenean, A.; D’Amico, M.; Di Filippo, C. The Melanocortin MC5R as a New Target for Treatment of High Glucose-Induced Hypertrophy of the Cardiac H9c2 Cells. Front. Physiol. 2018, 9, 1475. [Google Scholar] [CrossRef] [PubMed]
- Glyn, J.R.; Lipton, J.M. Hypothermic and antipyretic effects of centrally administered ACTH (1--24) and alpha-melanotropin. Peptides 1981, 2, 177–187. [Google Scholar] [CrossRef]
- Kandasamy, S.B.; Williams, B.A. Hypothermic and antipyretic effects of ACTH (1-24) and alpha-melanotropin in guinea-pigs. Neuropharmacology 1984, 23, 49–53. [Google Scholar] [CrossRef]
- Shih, S.T.; Lipton, J.M. Intravenous alpha-MSH reduces fever in the squirrel monkey. Peptides 1985, 6, 685–687. [Google Scholar] [CrossRef]
- Bitto, A.; Polito, F.; Altavilla, D.; Irrera, N.; Giuliani, D.; Ottani, A.; Minutoli, L.; Spaccapelo, L.; Galantucci, M.; Lodi, R.; et al. Melanocortins protect against multiple organ dysfunction syndrome in mice. Br. J. Pharmacol. 2011, 162, 917–928. [Google Scholar] [CrossRef] [Green Version]
- Giuliani, D.; Mioni, C.; Bazzani, C.; Zaffe, D.; Botticelli, A.R.; Capolongo, S.; Sabba, A.; Galantucci, M.; Iannone, A.; Grieco, P.; et al. Selective melanocortin MC4 receptor agonists reverse haemorrhagic shock and prevent multiple organ damage. Br. J. Pharmacol. 2007, 150, 595–603. [Google Scholar] [CrossRef] [Green Version]
- Ottani, A.; Neri, L.; Canalini, F.; Calevro, A.; Rossi, R.; Cappelli, G.; Ballestri, M.; Giuliani, D.; Guarini, S. Protective effects of the melanocortin analog NDP-α-MSH in rats undergoing cardiac arrest. Eur. J. Pharmacol. 2014, 745, 108–116. [Google Scholar] [CrossRef]
- Vecsernyes, M.; Szokol, M.; Bombicz, M.; Priksz, D.; Gesztelyi, R.; Fulop, G.A.; Varga, B.; Juhasz, B.; Haines, D.; Tosaki, A. Alpha-Melanocyte-stimulating Hormone Induces Vasodilation and Exerts Cardioprotection Through the Heme-Oxygenase Pathway in Rat Hearts. J. Cardiovasc. Pharmacol. 2017, 69, 286–297. [Google Scholar] [CrossRef] [Green Version]
- Qiang, X.; Liotta, A.S.; Shiloach, J.; Gutierrez, J.C.; Wang, H.; Ochani, M.; Ochani, K.; Yang, H.; Rabin, A.; LeRoith, D.; et al. New melanocortin-like peptide of E. coli can suppress inflammation via the mammalian melanocortin-1 receptor (MC1R): Possible endocrine-like function for microbes of the gut. NPJ Biofilms Microbiomes 2017, 3, 31. [Google Scholar] [CrossRef] [PubMed]
- Du, X.; Dong, B.; Li, C.; Zhang, F.; Ji, Y.; Zhang, J.; Yin, C. Dynamic changes of α-melanocyte-stimulating hormone levels in the serum of patients with craniocerebral trauma. Exp. Ther. Med. 2017, 14, 2511–2516. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patruno, S.; Garrido-Mesa, J.; Romano, M.; Perretti, M.; Montero-Melendez, T. Ligand Bias and Its Association With Pro-resolving Actions of Melanocortin Drugs. Front. Pharmacol. 2018, 9, 919. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lipton, J.M.; Ceriani, G.; Macaluso, A.; McCoy, D.; Carnes, K.; Biltz, J.; Catania, A. Antiinflammatory effects of the neuropeptide alpha-MSH in acute, chronic, and systemic inflammation. Ann. N. Y. Acad. Sci. 1994, 741, 137–148. [Google Scholar] [CrossRef]
- Deng, J.; Hu, X.; Yuen, P.S.; Star, R.A. Alpha-melanocyte-stimulating hormone inhibits lung injury after renal ischemia/reperfusion. Am. J. Respir. Crit. Care Med. 2004, 169, 749–756. [Google Scholar] [CrossRef]
- Miao, Y.L.; Deng, X.M.; Li, J.B.; Zhao, X.K.; Cao, J.M.; Tian, Y.P. Effect of alpha-melanocyte stimulating hormone on the apoptosis of the vascular endothelial cell of the lung in two-hit acute respiratory distress syndrome in rat. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue 2004, 16, 596–598. [Google Scholar]
- Colombo, G.; Gatti, S.; Sordi, A.; Turcatti, F.; Carlin, A.; Rossi, C.; Lonati, C.; Catania, A. Production and effects of alpha-melanocyte-stimulating hormone during acute lung injury. Shock 2007, 27, 326–333. [Google Scholar] [CrossRef]
- Raap, U.; Brzoska, T.; Sohl, S.; Päth, G.; Emmel, J.; Herz, U.; Braun, A.; Luger, T.; Renz, H. Alpha-melanocyte-stimulating hormone inhibits allergic airway inflammation. J. Immunol. 2003, 171, 353–359. [Google Scholar] [CrossRef] [Green Version]
- Webering, S.; Lunding, L.P.; Vock, C.; Schröder, A.; Gaede, K.I.; Herzmann, C.; Fehrenbach, H.; Wegmann, M. The alpha-melanocyte-stimulating hormone acts as a local immune homeostasis factor in experimental allergic asthma. Clin. Exp. Allergy 2019, 49, 1026–1039. [Google Scholar] [CrossRef]
- Böhm, M.; Luger, T. Are melanocortin peptides future therapeutics for cutaneous wound healing? Exp. Dermatol. 2019, 28, 219–224. [Google Scholar] [CrossRef] [Green Version]
- Land, S.C. Inhibition of cellular and systemic inflammation cues in human bronchial epithelial cells by melanocortin-related peptides: Mechanism of KPV action and a role for MC3R agonists. Int. J. Physiol. Pathophysiol. Pharmacol. 2012, 4, 59–73. [Google Scholar] [PubMed]
- Clemson, C.M.; Yost, J.; Taylor, A.W. The Role of Alpha-MSH as a Modulator of Ocular Immunobiology Exemplifies Mechanistic Differences between Melanocortins and Steroids. Ocul. Immunol. Inflamm. 2017, 25, 179–189. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, D.J.; Biros, D.J.; Taylor, A.W. Injection of an alpha-melanocyte stimulating hormone expression plasmid is effective in suppressing experimental autoimmune uveitis. Int. Immunopharmacol. 2009, 9, 1079–1086. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taylor, A.W.; Ng, T.F. Negative regulators that mediate ocular immune privilege. J. Leukoc. Biol. 2018, 103, 1179–1187. [Google Scholar] [CrossRef]
- Lee, D.J.; Taylor, A.W. Both MC5r and A2Ar are required for protective regulatory immunity in the spleen of post-experimental autoimmune uveitis in mice. J. Immunol. 2013, 191, 4103–4111. [Google Scholar] [CrossRef] [Green Version]
- Cai, S.; Yang, Q.; Hou, M.; Han, Q.; Zhang, H.; Wang, J.; Qi, C.; Bo, Q.; Ru, Y.; Yang, W.; et al. A-Melanocyte-Stimulating Hormone Protects Early Diabetic Retina from Blood-Retinal Barrier Breakdown and Vascular Leakage via MC4R. Cell Physiol. Biochem. 2018, 45, 505–522. [Google Scholar] [CrossRef]
- Zhang, L.; Dong, L.; Liu, X.; Jiang, Y.; Zhang, L.; Zhang, X.; Li, X.; Zhang, Y. α-Melanocyte-stimulating hormone protects retinal vascular endothelial cells from oxidative stress and apoptosis in a rat model of diabetes. PLoS ONE 2014, 9, e93433. [Google Scholar] [CrossRef]
- Zhang, Z.; Yao, K.; Ma, J.; Tang, X. Anti-inflammatory effect of the alpha-melanocyte stimulating hormone in animal eyes undergoing extracapsular lens extraction. Mol. Biol. 2011, 45, 272–281. [Google Scholar] [CrossRef]
- Laroui, H.; Dalmasso, G.; Nguyen, H.T.; Yan, Y.; Sitaraman, S.V.; Merlin, D. Drug-loaded nanoparticles targeted to the colon with polysaccharide hydrogel reduce colitis in a mouse model. Gastroenterology 2010, 138, 843–853.e2. [Google Scholar] [CrossRef]
- Zeng, M.; Shao, A.; Li, H.; Tang, Y.; Li, Q.; Guo, Z.; Wu, C.; Cheng, Y.; Tian, H.; Zhu, W.H. Peptide Receptor-Targeted Fluorescent Probe: Visualization and Discrimination between Chronic and Acute Ulcerative Colitis. ACS Appl. Mater. Interfaces 2017, 9, 13029–13036. [Google Scholar] [CrossRef]
- Váradi, J.; Harazin, A.; Fenyvesi, F.; Réti-Nagy, K.; Gogolák, P.; Vámosi, G.; Bácskay, I.; Fehér, P.; Ujhelyi, Z.; Vasvári, G.; et al. Alpha-Melanocyte Stimulating Hormone Protects against Cytokine-Induced Barrier Damage in Caco-2 Intestinal Epithelial Monolayers. PLoS ONE 2017, 12, e0170537. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wei, P.; Yang, Y.; Liu, Z.; Huang, J.; Gong, Y.; Sun, H. Oral Bifidobacterium longum expressing alpha-melanocyte-stimulating hormone to fight experimental colitis. Drug Deliv. 2016, 23, 2058–2064. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dalmasso, G.; Charrier-Hisamuddin, L.; Nguyen, H.T.; Yan, Y.; Sitaraman, S.; Merlin, D. PepT1-mediated tripeptide KPV uptake reduces intestinal inflammation. Gastroenterology 2008, 134, 166–178. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kannengiesser, K.; Maaser, C.; Heidemann, J.; Luegering, A.; Ross, M.; Brzoska, T.; Bohm, M.; Luger, T.A.; Domschke, W.; Kucharzik, T. Melanocortin-derived tripeptide KPV has anti-inflammatory potential in murine models of inflammatory bowel disease. Inflamm. Bowel Dis. 2008, 14, 324–331. [Google Scholar] [CrossRef] [PubMed]
- Xiao, B.; Xu, Z.; Viennois, E.; Zhang, Y.; Zhang, Z.; Zhang, M.; Han, M.K.; Kang, Y.; Merlin, D. Orally Targeted Delivery of Tripeptide KPV via Hyaluronic Acid-Functionalized Nanoparticles Efficiently Alleviates Ulcerative Colitis. Mol. Ther. 2017, 25, 1628–1640. [Google Scholar] [CrossRef] [Green Version]
- Peñate-Medina, T.; Damoah, C.; Benezra, M.; Will, O.; Kairemo, K.; Humbert, J.; Sebens, S.; Peñate-Medina, O. Alpha-MSH Targeted Liposomal Nanoparticle for Imaging in Inflammatory Bowel Disease (IBD). Curr. Pharm. Des. 2020, 26, 3840–3846. [Google Scholar] [CrossRef]
- Min, Y.; Han, D.; Fu, Z.; Wang, H.; Liu, L.; Tian, Y. α-MSH inhibits TNF-α-induced maturation of human dendritic cells in vitro through the up-regulation of ANXA1. Acta Biochim. Biophys. Sin. 2011, 43, 61–68. [Google Scholar] [CrossRef] [Green Version]
- Flores-Bastías, O.; Adriasola-Carrasco, A.; Karahanian, E. Activation of Melanocortin-4 Receptor Inhibits Both Neuroinflammation Induced by Early Exposure to Ethanol and Subsequent Voluntary Alcohol Intake in Adulthood in Animal Models: Is BDNF the Key Mediator? Front. Cell. Neurosci. 2020, 14, 5. [Google Scholar] [CrossRef]
- Flores-Bastías, O.; Gómez, G.I.; Orellana, J.A.; Karahanian, E. Activation of Melanocortin-4 Receptor by a Synthetic Agonist Inhibits Ethanolinduced Neuroinflammation in Rats. Curr. Pharm. Des. 2019, 25, 4799–4805. [Google Scholar] [CrossRef]
- Wu, X.; Fu, S.; Liu, Y.; Luo, H.; Li, F.; Wang, Y.; Gao, M.; Cheng, Y.; Xie, Z. NDP-MSH binding melanocortin-1 receptor ameliorates neuroinflammation and BBB disruption through CREB/Nr4a1/NF-κB pathway after intracerebral hemorrhage in mice. J. Neuroinflam. 2019, 16, 192. [Google Scholar] [CrossRef]
- Mykicki, N.; Herrmann, A.M.; Schwab, N.; Deenen, R.; Sparwasser, T.; Limmer, A.; Wachsmuth, L.; Klotz, L.; Köhrer, K.; Faber, C.; et al. Melanocortin-1 receptor activation is neuroprotective in mouse models of neuroinflammatory disease. Sci. Transl. Med. 2016, 8, 362ra146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carniglia, L.; Durand, D.; Caruso, C.; Lasaga, M. Effect of NDP-α-MSH on PPAR-γ and -β expression and anti-inflammatory cytokine release in rat astrocytes and microglia. PLoS ONE 2013, 8, e57313. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feng, G.; Feng, J.; Zhang, S.; Tong, Y.; Zhang, Q.; Yang, X.; Zhang, H. Altered levels of α-melanocyte stimulating hormone in cerebrospinal fluid and plasma of patients with traumatic brain injury. Brain Res. 2018, 1696, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Kamermans, A.; Verhoeven, T.; van Het Hof, B.; Koning, J.J.; Borghuis, L.; Witte, M.; van Horssen, J.; de Vries, H.E.; Rijnsburger, M. Setmelanotide, a Novel, Selective Melanocortin Receptor-4 Agonist Exerts Anti-inflammatory Actions in Astrocytes and Promotes an Anti-inflammatory Macrophage Phenotype. Front. Immunol. 2019, 10, 2312. [Google Scholar] [CrossRef]
- Chen, S.; Zhao, L.; Sherchan, P.; Ding, Y.; Yu, J.; Nowrangi, D.; Tang, J.; Xia, Y.; Zhang, J.H. Activation of melanocortin receptor 4 with RO27-3225 attenuates neuroinflammation through AMPK/JNK/p38 MAPK pathway after intracerebral hemorrhage in mice. J. Neuroinflamm. 2018, 15, 106. [Google Scholar] [CrossRef]
- Chen, S.; Zuo, Y.; Huang, L.; Sherchan, P.; Zhang, J.; Yu, Z.; Peng, J.; Zhang, J.; Zhao, L.; Doycheva, D.; et al. The MC(4) receptor agonist RO27-3225 inhibits NLRP1-dependent neuronal pyroptosis via the ASK1/JNK/p38 MAPK pathway in a mouse model of intracerebral haemorrhage. Br. J. Pharmacol. 2019, 176, 1341–1356. [Google Scholar] [CrossRef]
- Kleiner, S.; Braunstahl, G.J.; Rüdrich, U.; Gehring, M.; Eiz-Vesper, B.; Luger, T.A.; Steelant, B.; Seys, S.F.; Kapp, A.; Böhm, M.; et al. Regulation of melanocortin 1 receptor in allergic rhinitis in vitro and in vivo. Clin. Exp. Allergy 2016, 46, 1066–1074. [Google Scholar] [CrossRef]
- Etori, M.; Yonekubo, K.; Sato, E.; Mizukami, K.; Hirahara, K.; Karasuyama, H.; Maeda, H.; Yamashita, M. Melanocortin receptors 1 and 5 might mediate inhibitory effects of α-melanocyte-stimulating hormone on antigen-induced chronic allergic skin inflammation in IgE transgenic mice. J. Investig. Dermatol. 2012, 132, 1925–1927. [Google Scholar] [CrossRef] [Green Version]
- Andoh, T.; Akasaka, C.; Shimizu, K.; Lee, J.B.; Yoshihisa, Y.; Shimizu, T. Involvement of α-Melanocyte-Stimulating Hormone-Thromboxane A(2) System on Itching in Atopic Dermatitis. Am. J. Pathol. 2019, 189, 1775–1785. [Google Scholar] [CrossRef]
- Mehta, D.; Granstein, R.D. Immunoregulatory Effects of Neuropeptides on Endothelial Cells: Relevance to Dermatological Disorders. Dermatology 2019, 235, 175–186. [Google Scholar] [CrossRef]
- Capsoni, F.; Ongari, A.M.; Lonati, C.; Accetta, R.; Gatti, S.; Catania, A. α-Melanocyte-stimulating-hormone (α-MSH) modulates human chondrocyte activation induced by proinflammatory cytokines. BMC Musculoskelet. Disord. 2015, 16, 154. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montero-Melendez, T.; Nagano, A.; Chelala, C.; Filer, A.; Buckley, C.D.; Perretti, M. Therapeutic senescence via GPCR activation in synovial fibroblasts facilitates resolution of arthritis. Nat. Commun. 2020, 11, 745. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Yao, Z.; He, M.; Tian, C. Synovial Fluid α-MSH Levels are Inversely Correlated with Articular Cartilage Degeneration in Anterior Cruciate Ligament Deficient Knees. Clin. Lab. 2015, 61, 965–971. [Google Scholar] [CrossRef] [PubMed]
- Guan, J.; Li, Y.; Ding, L.B.; Liu, G.Y.; Zheng, X.F.; Xue, W.; Wang, H.J. Synovial fluid alpha-melanocyte-stimulating hormone may act as a protective biomarker for primary knee osteoarthritis. Discov. Med. 2019, 27, 17–26. [Google Scholar]
- Böhm, M.; Apel, M.; Lowin, T.; Lorenz, J.; Jenei-Lanzl, Z.; Capellino, S.; Dosoki, H.; Luger, T.A.; Straub, R.H.; Grässel, S. α-MSH modulates cell adhesion and inflammatory responses of synovial fibroblasts from osteoarthritis patients. Biochem. Pharmacol. 2016, 116, 89–99. [Google Scholar] [CrossRef]
- Liu, G.; Chen, Y.; Wang, G.; Niu, J. Decreased Synovial Fluid α-Melanocyte-Stimulating-Hormone (α-MSH) Levels Reflect Disease Severity in Patients with Posttraumatic Ankle Osteoarthritis. Clin. Lab. 2016, 62, 1491–1500. [Google Scholar] [CrossRef]
- Zaidi, M.; Sun, L.; Robinson, L.J.; Tourkova, I.L.; Liu, L.; Wang, Y.; Zhu, L.L.; Liu, X.; Li, J.; Peng, Y.; et al. ACTH protects against glucocorticoid-induced osteonecrosis of bone. Proc. Natl. Acad. Sci. USA 2010, 107, 8782–8787. [Google Scholar] [CrossRef] [Green Version]
- Mao, Z.; Liu, G.; Chen, J.J.; Liu, D.; Xu, M.P.; Zhao, C.; Yang, H.T.; Yue, Y.B. Serum α-melanocyte-stimulating hormone may act as a protective biomarker for non-traumatic osteonecrosis of the femoral head. Ann. Clin. Biochem. 2018, 55, 453–460. [Google Scholar] [CrossRef]
- Holloway, P.M.; Durrenberger, P.F.; Trutschl, M.; Cvek, U.; Cooper, D.; Orr, A.W.; Perretti, M.; Getting, S.J.; Gavins, F.N. Both MC1 and MC3 Receptors Provide Protection From Cerebral Ischemia-Reperfusion-Induced Neutrophil Recruitment. Arter. Thromb. Vasc. Biol. 2015, 35, 1936–1944. [Google Scholar] [CrossRef] [Green Version]
- Ottani, A.; Giuliani, D.; Neri, L.; Calevro, A.; Canalini, F.; Vandini, E.; Cainazzo, M.M.; Ruberto, I.A.; Barbieri, A.; Rossi, R.; et al. NDP-α-MSH attenuates heart and liver responses to myocardial reperfusion via the vagus nerve and JAK/ERK/STAT signaling. Eur. J. Pharmacol. 2015, 769, 22–32. [Google Scholar] [CrossRef]
- Hassoun, H.T.; Zou, L.; Moore, F.A.; Kozar, R.A.; Weisbrodt, N.W.; Kone, B.C. Alpha-melanocyte-stimulating hormone protects against mesenteric ischemia-reperfusion injury. Am. J. Physiol. Liver Physiol. 2002, 282, G1059–G1068. [Google Scholar] [CrossRef]
- Minutoli, L.; Bitto, A.; Squadrito, F.; Irrera, N.; Rinaldi, M.; Nicotina, P.A.; Arena, S.; Magno, C.; Marini, H.; Spaccapelo, L.; et al. Melanocortin 4 receptor activation protects against testicular ischemia-reperfusion injury by triggering the cholinergic antiinflammatory pathway. Endocrinology 2011, 152, 3852–3861. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Skott, M.; Nørregaard, R.; Birke-Sørensen, H.; Palmfeldt, J.; Kwon, T.H.; Jonassen, T.; Frøkiær, J.; Nielsen, S. Development of intestinal ischemia/reperfusion-induced acute kidney injury in rats with or without chronic kidney disease: Cytokine/chemokine response and effect of α-melanocyte-stimulating hormone. Kidney Res. Clin. Pract. 2014, 33, 79–88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nuutinen, S.; Ailanen, L.; Savontaus, E.; Rinne, P. Melanocortin overexpression limits diet-induced inflammation and atherosclerosis in LDLR(-/-) mice. J. Endocrinol. 2018, 236, 111–123. [Google Scholar] [CrossRef]
- Le Saux, G.; Plawinski, L.; Nlate, S.; Ripoche, J.; Buffeteau, T.; Durrieu, M.C. Beneficial Effect of Covalently Grafted α-MSH on Endothelial Release of Inflammatory Mediators for Applications in Implantable Devices. PLoS ONE 2016, 11, e0150706. [Google Scholar] [CrossRef]
- Gatti, S.; Colombo, G.; Buffa, R.; Turcatti, F.; Garofalo, L.; Carboni, N.; Ferla, L.; Fassati, L.R.; Lipton, J.M.; Catania, A. alpha-Melanocyte-stimulating hormone protects the allograft in experimental heart transplantation. Transplantation 2002, 74, 1678–1684. [Google Scholar] [CrossRef]
- Liu, G.; Li, M.; Saeed, M.; Xu, Y.; Ren, Q.; Sun, C. αMSH inhibits adipose inflammation via reducing FoxOs transcription and blocking Akt/JNK pathway in mice. Oncotarget 2017, 8, 47642–47654. [Google Scholar] [CrossRef] [Green Version]
- Manna, S.K.; Aggarwal, B.B. Alpha-melanocyte-stimulating hormone inhibits the nuclear transcription factor NF-kappa B activation induced by various inflammatory agents. J. Immunol. 1998, 161, 2873–2880. [Google Scholar]
- Gerlo, S.; Kooijman, R.; Beck, I.M.; Kolmus, K.; Spooren, A.; Haegeman, G. Cyclic AMP: A selective modulator of NF-κB action. Cell Mol. Life Sci. 2011, 68, 3823–3841. [Google Scholar] [CrossRef]
- Mandrika, I.; Muceniece, R.; Wikberg, J.E. Effects of melanocortin peptides on lipopolysaccharide/interferon-gamma-induced NF-kappaB DNA binding and nitric oxide production in macrophage-like RAW 264.7 cells: Evidence for dual mechanisms of action. Biochem. Pharmacol. 2001, 61, 613–621. [Google Scholar] [CrossRef]
- Delgado, R.; Carlin, A.; Airaghi, L.; Demitri, M.T.; Meda, L.; Galimberti, D.; Baron, P.; Lipton, J.M.; Catania, A. Melanocortin peptides inhibit production of proinflammatory cytokines and nitric oxide by activated microglia. J. Leukoc. Biol. 1998, 63, 740–745. [Google Scholar] [CrossRef] [PubMed]
- Galimberti, D.; Baron, P.; Meda, L.; Prat, E.; Scarpini, E.; Delgado, R.; Catania, A.; Lipton, J.M.; Scarlato, G. Alpha-MSH peptides inhibit production of nitric oxide and tumor necrosis factor-alpha by microglial cells activated with beta-amyloid and interferon gamma. Biochem. Biophys. Res. Commun. 1999, 263, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Mukaida, N.; Okamoto, S.; Ishikawa, Y.; Matsushima, K. Molecular mechanism of interleukin-8 gene expression. J. Leukoc. Biol. 1994, 56, 554–558. [Google Scholar] [CrossRef] [PubMed]
- Böhm, M.; Schulte, U.; Kalden, H.; Luger, T.A. Alpha-melanocyte-stimulating hormone modulates activation of NF-kappa B and AP-1 and secretion of interleukin-8 in human dermal fibroblasts. Ann. N. Y. Acad. Sci. 1999, 885, 277–286. [Google Scholar] [CrossRef]
- Tanaka, T.; Narazaki, M.; Kishimoto, T. IL-6 in inflammation, immunity, and disease. Cold Spring Harb. Perspect. Biol. 2014, 6, a016295. [Google Scholar] [CrossRef]
- Saraiva, M.; O’Garra, A. The regulation of IL-10 production by immune cells. Nat. Rev. Immunol. 2010, 10, 170–181. [Google Scholar] [CrossRef] [Green Version]
- Wen, A.Y.; Sakamoto, K.M.; Miller, L.S. The role of the transcription factor CREB in immune function. J. Immunol. 2010, 185, 6413–6419. [Google Scholar] [CrossRef] [Green Version]
- Kalden, D.H.; Scholzen, T.; Brzoska, T.; Luger, T.A. Mechanisms of the antiinflammatory effects of alpha-MSH. Role of transcription factor NF-kappa B and adhesion molecule expression. Ann. N. Y. Acad. Sci. 1999, 885, 254–261. [Google Scholar] [CrossRef]
- Scholzen, T.E.; Sunderkötter, C.; Kalden, D.H.; Brzoska, T.; Fastrich, M.; Fisbeck, T.; Armstrong, C.A.; Ansel, J.C.; Luger, T.A. Alpha-melanocyte stimulating hormone prevents lipopolysaccharide-induced vasculitis by down-regulating endothelial cell adhesion molecule expression. Endocrinology 2003, 144, 360–370. [Google Scholar] [CrossRef] [Green Version]
- Becher, E.; Mahnke, K.; Brzoska, T.; Kalden, D.H.; Grabbe, S.; Luger, T.A. Human peripheral blood-derived dendritic cells express functional melanocortin receptor MC-1R. Ann. N. Y. Acad. Sci. 1999, 885, 188–195. [Google Scholar] [CrossRef]
- Bonfiglio, V.; Camillieri, G.; Avitabile, T.; Leggio, G.M.; Drago, F. Effects of the COOH-terminal tripeptide alpha-MSH(11-13) on corneal epithelial wound healing: Role of nitric oxide. Exp. Eye Res. 2006, 83, 1366–1372. [Google Scholar] [CrossRef] [PubMed]
- Star, R.A.; Rajora, N.; Huang, J.; Stock, R.C.; Catania, A.; Lipton, J.M. Evidence of autocrine modulation of macrophage nitric oxide synthase by alpha-melanocyte-stimulating hormone. Proc. Natl. Acad. Sci. USA 1995, 92, 8016–8020. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rajora, N.; Ceriani, G.; Catania, A.; Star, R.A.; Murphy, M.T.; Lipton, J.M. alpha-MSH production, receptors, and influence on neopterin in a human monocyte/macrophage cell line. J. Leukoc. Biol. 1996, 59, 248–253. [Google Scholar] [CrossRef] [PubMed]
- Jung, E.J.; Han, D.J.; Chang, S.H.; Lim, D.G.; Wee, Y.M.; Kim, J.H.; Kim, Y.H.; Koo, S.K.; Choi, M.; Kim, S.C. Protective effect of alpha-melanocyte-stimulating hormone on pancreas islet cell against peripheral blood mononuclear cell-mediated cytotoxicity in vitro. Transplant. Proc. 2007, 39, 1604–1606. [Google Scholar] [CrossRef] [PubMed]
- Oktar, B.K.; Yüksel, M.; Alican, I. The role of cyclooxygenase inhibition in the effect of alpha-melanocyte-stimulating hormone on reactive oxygen species production by rat peritoneal neutrophils. Prostaglandins Leukot. Essent. Fat. Acids 2004, 71, 1–5. [Google Scholar] [CrossRef]
- Taylor, A.W.; Lee, D.J. The alpha-melanocyte stimulating hormone induces conversion of effector T cells into treg cells. J. Transplant. 2011, 2011, 246856. [Google Scholar] [CrossRef] [Green Version]
- Fang, J.; Han, D.; Hong, J.; Zhang, H.; Ying, Y.; Tian, Y.; Zhang, L.; Lin, J. SVα-MSH, a novel α-melanocyte stimulating hormone analog, ameliorates autoimmune encephalomyelitis through inhibiting autoreactive CD4(+) T cells activation. J. Neuroimmunol. 2014, 269, 9–19. [Google Scholar] [CrossRef]
- Namba, K.; Kitaichi, N.; Nishida, T.; Taylor, A.W. Induction of regulatory T cells by the immunomodulating cytokines alpha-melanocyte-stimulating hormone and transforming growth factor-beta2. J. Leukoc. Biol. 2002, 72, 946–952. [Google Scholar]
- Ng, T.F.; Kitaichi, N.; Taylor, A.W. In vitro generated autoimmune regulatory T cells enhance intravitreous allogeneic retinal graft survival. Investig. Opthalmol. Vis. Sci. 2007, 48, 5112–5117. [Google Scholar] [CrossRef]
- Nishida, T.; Miyata, S.; Itoh, Y.; Mizuki, N.; Ohgami, K.; Shiratori, K.; Ilieva, I.B.; Ohno, S.; Taylor, A.W. Anti-inflammatory effects of alpha-melanocyte-stimulating hormone against rat endotoxin-induced uveitis and the time course of inflammatory agents in aqueous humor. Int. Immunopharmacol. 2004, 4, 1059–1066. [Google Scholar] [CrossRef]
- Boada-Romero, E.; Martinez, J.; Heckmann, B.L.; Green, D.R. The clearance of dead cells by efferocytosis. Nat. Rev. Mol. Cell Biol. 2020, 21, 398–414. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N.; Savill, J. Resolution of inflammation: The beginning programs the end. Nat. Immunol. 2005, 6, 1191–1197. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Kim, T.J.; Peng, D.H.; Duan, D.; Gibbons, D.L.; Yamauchi, M.; Jackson, J.R.; Le Saux, C.J.; Calhoun, C.; Peters, J.; et al. Fibroblast-specific inhibition of TGF-β1 signaling attenuates lung and tumor fibrosis. J. Clin. Investig. 2017, 127, 3675–3688. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thannickal, V.J.; Lee, D.Y.; White, E.S.; Cui, Z.; Larios, J.M.; Chacon, R.; Horowitz, J.C.; Day, R.M.; Thomas, P.E. Myofibroblast differentiation by transforming growth factor-beta1 is dependent on cell adhesion and integrin signaling via focal adhesion kinase. J. Biol. Chem. 2003, 278, 12384–12389. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Böhm, M.; Raghunath, M.; Sunderkötter, C.; Schiller, M.; Ständer, S.; Brzoska, T.; Cauvet, T.; Schiöth, H.B.; Schwarz, T.; Luger, T.A. Collagen metabolism is a novel target of the neuropeptide alpha-melanocyte-stimulating hormone. J. Biol. Chem. 2004, 279, 6959–6966. [Google Scholar] [CrossRef] [Green Version]
- Hill, R.P.; Wheeler, P.; MacNeil, S.; Haycock, J.W. Alpha-melanocyte stimulating hormone cytoprotective biology in human dermal fibroblast cells. Peptides 2005, 26, 1150–1158. [Google Scholar] [CrossRef]
- Kokot, A.; Sindrilaru, A.; Schiller, M.; Sunderkötter, C.; Kerkhoff, C.; Eckes, B.; Scharffetter-Kochanek, K.; Luger, T.A.; Böhm, M. alpha-melanocyte-stimulating hormone suppresses bleomycin-induced collagen synthesis and reduces tissue fibrosis in a mouse model of scleroderma: Melanocortin peptides as a novel treatment strategy for scleroderma? Arthritis Rheum. 2009, 60, 592–603. [Google Scholar] [CrossRef]
- Böhm, M.; Stegemann, A. Bleomycin-induced fibrosis in MC1 signalling-deficient C57BL/6J-Mc1r(e/e) mice further supports a modulating role for melanocortins in collagen synthesis of the skin. Exp. Dermatol. 2014, 23, 431–433. [Google Scholar] [CrossRef]
- Luo, L.F.; Shi, Y.; Zhou, Q.; Xu, S.Z.; Lei, T.C. Insufficient expression of the melanocortin-1 receptor by human dermal fibroblasts contributes to excess collagen synthesis in keloid scars. Exp. Dermatol. 2013, 22, 764–766. [Google Scholar] [CrossRef]
- De Souza, K.S.; Cantaruti, T.A.; Azevedo, G.M., Jr.; Galdino, D.A.; Rodrigues, C.M.; Costa, R.A.; Vaz, N.M.; Carvalho, C.R. Improved cutaneous wound healing after intraperitoneal injection of alpha-melanocyte-stimulating hormone. Exp. Dermatol. 2015, 24, 198–203. [Google Scholar] [CrossRef]
- Lee, T.H.; Jawan, B.; Chou, W.Y.; Lu, C.N.; Wu, C.L.; Kuo, H.M.; Concejero, A.M.; Wang, C.H. Alpha-melanocyte-stimulating hormone gene therapy reverses carbon tetrachloride induced liver fibrosis in mice. J. Gene Med. 2006, 8, 764–772. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.H.; Lee, T.H.; Lu, C.N.; Chou, W.Y.; Hung, K.S.; Concejero, A.M.; Jawan, B. Electroporative alpha-MSH gene transfer attenuates thioacetamide-induced murine hepatic fibrosis by MMP and TIMP modulation. Gene Ther. 2006, 13, 1000–1009. [Google Scholar] [CrossRef] [PubMed]
- Lonati, C.; Carlin, A.; Leonardi, P.; Valenza, F.; Bosari, S.; Catania, A.; Gatti, S. Modulatory effects of NDP-MSH in the regenerating liver after partial hepatectomy in rats. Peptides 2013, 50, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Vándor, E.; Simon, G.; Anda, E.; Budavári, I. The effect of alpha-melanophor-stimulating hormone on liver regeneration and incorporation of amino acid in rats’ liver protein. Endokrinologie 1975, 66, 81–87. [Google Scholar] [PubMed]
- Xu, P.B.; Mao, Y.F.; Meng, H.B.; Tian, Y.P.; Deng, X.M. STY39, a novel alpha-melanocyte-stimulating hormone analogue, attenuates bleomycin-induced pulmonary inflammation and fibrosis in mice. Shock 2011, 35, 308–314. [Google Scholar] [CrossRef]
- Lee, S.Y.; Jo, S.K.; Cho, W.Y.; Kim, H.K.; Won, N.H. The effect of alpha-melanocyte-stimulating hormone on renal tubular cell apoptosis and tubulointerstitial fibrosis in cyclosporine A nephrotoxicity. Transplantation 2004, 78, 1756–1764. [Google Scholar] [CrossRef]
- Verhaagen, J.; Edwards, P.M.; Jennekens, F.G.; Schotman, P.; Gispen, W.H. Alpha-melanocyte-stimulating hormone stimulates the outgrowth of myelinated nerve fibers after peripheral nerve crush. Exp. Neurol. 1986, 92, 451–454. [Google Scholar] [CrossRef]
- Dekker, A.J. Effect of alpha-melanocyte-stimulating hormone on peripheral nerve regeneration in the rat: Histological aspects and comparison with the effect of gangliosides. Exp. Neurol. 1988, 99, 490–497. [Google Scholar] [CrossRef]
- Laquerriere, A.; Peulve, P.; Jin, O.; Tiollier, J.; Tardy, M.; Vaudry, H.; Hemet, J.; Tadie, M. Effect of basic fibroblast growth factor and alpha-melanocytic stimulating hormone on nerve regeneration through a collagen channel. Microsurgery 1994, 15, 203–210. [Google Scholar] [CrossRef]
- Plantinga, L.C.; Verhaagen, J.; Edwards, P.M.; Hali, M.; Brakkee, J.H.; Gispen, W.H. Pharmacological evidence for the involvement of endogenous alpha-MSH-like peptides in peripheral nerve regeneration. Peptides 1995, 16, 319–324. [Google Scholar] [CrossRef] [Green Version]
- Ter Laak, M.P.; Brakkee, J.H.; Adan, R.A.; Hamers, F.P.; Gispen, W.H. The potent melanocortin receptor agonist melanotan-II promotes peripheral nerve regeneration and has neuroprotective properties in the rat. Eur. J. Pharmacol. 2003, 462, 179–183. [Google Scholar] [CrossRef]
- Pavan, J.; Lukenda, A.; Stambuk, N.; Konjevoda, P.; Kastelan, S.; Curković, M. Effects of alpha-MSH on corneal epithelial lesions in rats. Coll. Antropol. 2012, 36, 1407–1411. [Google Scholar] [PubMed]
- Zhang, Z.; Ma, J.; Yao, K.; Yin, J. Alpha-melanocyte stimulating hormone suppresses the proliferation of human tenon’s capsule fibroblast proliferation induced by transforming growth factor beta 1. Mol. Biol. 2012, 46, 628–633. [Google Scholar] [CrossRef]

| Receptor | Tissue Distribution | Species | Agonist | Biologic Effects | Antagonist | Biologic Effects |
|---|---|---|---|---|---|---|
| MC1R | Present in melanocytes/skin | Human [41,42] | α-MSH, ACTH, β-MSH, γ-MSH | Pigmentation, anti-inflammatory [43,44] | Agouti | Suppresses melanine production [45,46] |
| Present in grey matter | Rat [47] | NDP-MSH | Anti-inflammatory [48,49,50] | |||
| BMS-470539 | ||||||
| Present in monocyte, macrophage (including alveolar), lymphocyte, neutrophil | Human, murine [51,52] | AP-1189 | ||||
| MC2R | Present in adrenal cortex; absent in liver, lung, thyroid and kidney | Rhesus macaque [41] | ACTH | Induce steroidogenesis [18] | GPS1574 | Inhibits steroidogenesis [53,54] |
| Present in chondrocyte and osteoblast | Human [55,56] | |||||
| MC3R | Present in brain, placenta; absent in adrenal, kidney, liver | Rat [57] | γ-MSH ≥ ACTH = β-MSH = α-MSH | Energy equilibrium, cardiovascular [58,59,60] | SHU-9119 | Inhibits anti-inflammatory effects of γMSH [36] |
| Present in lung | Murine [61] | MTII | Anti-inflammatory [61,62,63,64,65,66] | |||
| D-Trp8-γMSH | ||||||
| Present in macrophage and monocytes | Murine [51,52,55,56,57,67] | AP-214 | AVM-127 | inhibits α-MSH-induced penile erection [68] | ||
| AP-1189 (biased agonist) | ||||||
| MC4R | Present in brain; absent in lung, liver, kidney, adrenal | Rat/Canine [69] | α-MSH = ACTH > β-MSH > γ-MSH | Energy balance, erectile function, cardiovascular effects [43,70] | AgRP | Inhibitory cardiovascular effects, increases food intake [71,72] |
| Present in thalamus and hypothalamus | Rat [73] | THIQ | Anti-inflammatory, inhibits food intake [74,75,76] | ML00253764 | ||
| Ro27-3225 | ||||||
| PT-141 | Induces erection [77,78] | |||||
| MC5R | Present in lung, skeletal muscle, brain; absent in adrenal | Murine/Human [79] | α-MSH, ACTH, β-MSH, | Anti-inflammatory [80] | ||
| Present in B-lymphocytes | Mouse [81] | |||||
| Present in T-lymphocytes | Mouse [82] | PG-901 | Inhibits glucose uptake [83] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dinparastisaleh, R.; Mirsaeidi, M. Antifibrotic and Anti-Inflammatory Actions of α-Melanocytic Hormone: New Roles for an Old Player. Pharmaceuticals 2021, 14, 45. https://0-doi-org.brum.beds.ac.uk/10.3390/ph14010045
Dinparastisaleh R, Mirsaeidi M. Antifibrotic and Anti-Inflammatory Actions of α-Melanocytic Hormone: New Roles for an Old Player. Pharmaceuticals. 2021; 14(1):45. https://0-doi-org.brum.beds.ac.uk/10.3390/ph14010045
Chicago/Turabian StyleDinparastisaleh, Roshan, and Mehdi Mirsaeidi. 2021. "Antifibrotic and Anti-Inflammatory Actions of α-Melanocytic Hormone: New Roles for an Old Player" Pharmaceuticals 14, no. 1: 45. https://0-doi-org.brum.beds.ac.uk/10.3390/ph14010045





