Functional Characterization of the Oxantel-Sensitive Acetylcholine Receptor from Trichuris muris
Abstract
:1. Introduction
2. Results
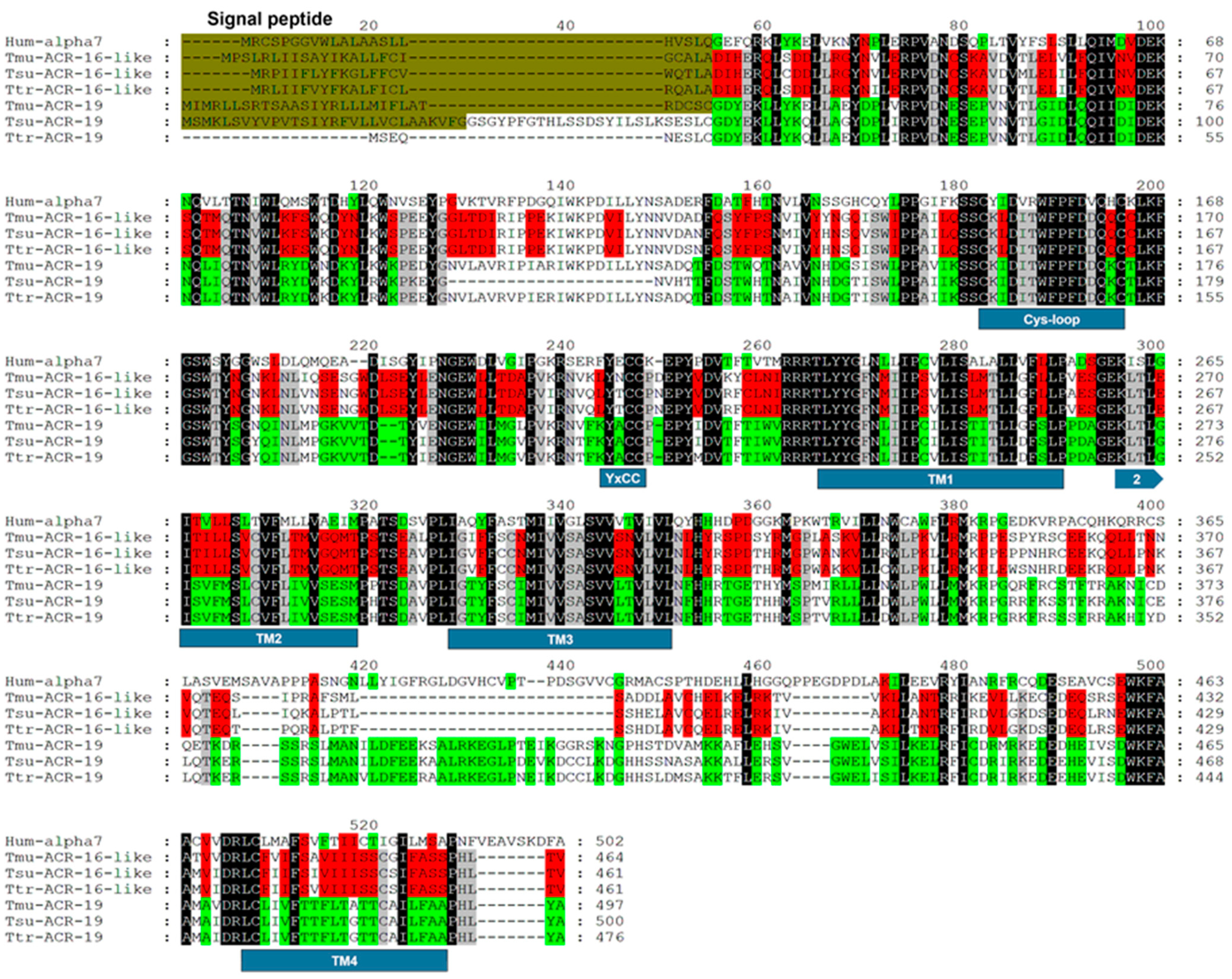
2.1. Identification of ACR-16-Like and ACR-19 Sequences from Trichuris muris
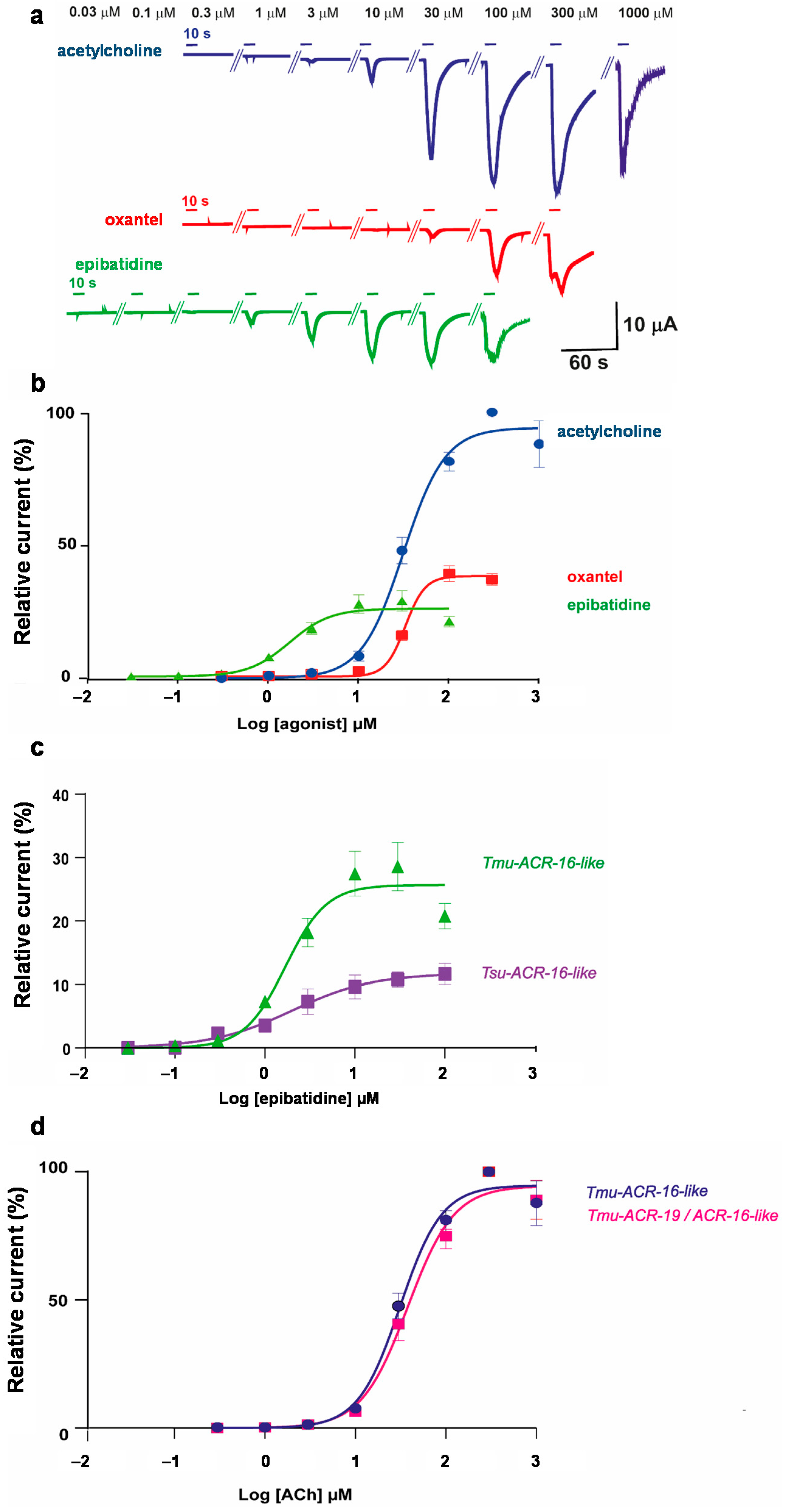
2.2. The T. muris ACR-16-Like Subunit Forms a Functional Acetylcholine Receptor in Xenopus laevis Oocytes
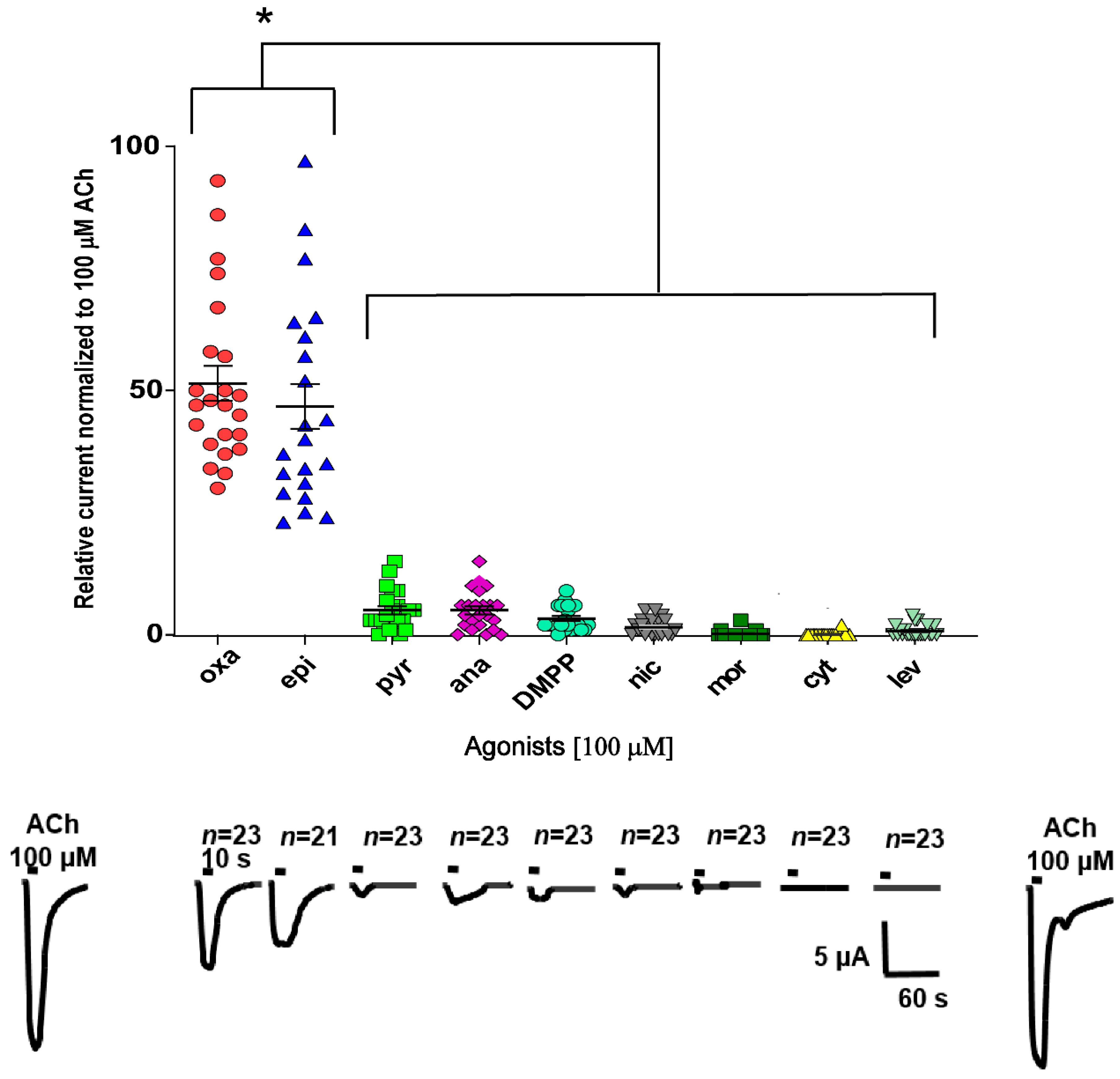
2.3. Characterization of ACh, Oxantel, and Epibatidine Effects on T. muris and T. suis ACR-16-Like AChRs
3. Discussion
4. Materials and Methods
4.1. Ethic Statement
4.2. Experimental Animals and Infections
4.3. Drugs
4.4. Sequence Analysis and Alignment
4.5. Cloning of the Coding Sequence of the ACR-16-Like and ACR-19 Subunits from Trichuris muris
4.6. Electrophysiological Experiments in Xenopus laevis Oocytes
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- International Helminth Genomes Consortium. Comparative genomics of the major parasitic worms. Nat. Genet. 2019, 51, 163–174. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- James, S.L.; Abate, D.; Abate, K.H.; Abay, S.M.; Abbafati, C.; Abbasi, N.; Abbastabar, H.; Abd-Allah, F.; Abdela, J.; Abdelalim, A.; et al. Global, regional, and national incidence, prevalence, and years lived with disability for 354 Diseases and Injuries for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1789–1858. [Google Scholar] [CrossRef] [Green Version]
- Stephenson, L.S.; Latham, M.C.; Ottesen, E.A. Malnutrition and parasitic helminth infections. Parasitology 2000, 121, S23–S38. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hall, A.; Hewitt, G.; Tuffrey, V.; Silva, N. A review and meta-analysis of the impact of intestinal worms on child growth and nutrition. Matern. Child Nutr. 2008, 4 (Suppl. 1), 118–236. [Google Scholar] [CrossRef] [Green Version]
- Bundy, D.A.; Cooper, E.S. Trichuris and trichuriasis in humans. Adv. Parasitol. 1989, 28, 107–173. [Google Scholar] [PubMed]
- World Health Organization. Helminths Control in School-Age Children—A Guide for Managers of Control Programmes, 2nd ed.; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Keiser, J.; Utzinger, J. Efficacy of current drugs against soil-transmitted helminth infections: Systematic review and meta-analysis. JAMA 2008, 299, 1937–1948. [Google Scholar] [CrossRef]
- Belizario, V.Y.; Amarillo, M.E.; Leon, W.U.; Reyes, A.E.; Bugayong, M.G.; Macatangay, B.J.C. A comparison of the efficacy of single doses of albendazole, ivermectin, and diethylcarbamazine alone or in combinations against Ascaris and Trichuris spp. Bull. World Health Organ. 2003, 81, 35–42. [Google Scholar] [PubMed]
- Albonico, M.; Bickle, Q.; Ramsan, M.; Montresor, A.; Savioli, L.; Taylor, M. Efficacy of mebendazole and levamisole alone or in combination against intestinal nematode infections after repeated targeted mebendazole treatment in Zanzibar. Bull. World Health Organ. 2003, 81, 343–352. [Google Scholar]
- Steinmann, P.; Utzinger, J.; Du, Z.; Jiang, J.; Chen, J.; Hattendorf, J.; Zhou, H.; Zhou, X.N. Efficacy of single-dose and triple-dose albendazole and mebendazole against soil-transmitted helminths and Taenia spp.: A randomized controlled trial. PLoS ONE 2011, 6, e25003. [Google Scholar] [CrossRef]
- Knopp, S.; Mohammed, K.A.; Speich, B.; Hattendorf, J.; Khamis, I.S.; Khamis, A.N.; Stothard, J.R.; Rollinson, D.; Marti, H.; Utzinger, J. Albendazole and mebendazole administered alone or in combination with ivermectin against Trichuris trichiura: A randomized controlled trial. Clin. Infect. Dis. 2010, 51, 1420–1428. [Google Scholar] [CrossRef] [Green Version]
- Olsen, A.; Namwanje, H.; Nejsum, P.; Roepstorff, A.; Thamsborg, S.M. Albendazole and mebendazole have low efficacy against Trichuris trichiura in school-age children in Kabale District, Uganda. Trans. R. Soc. Trop. Med. Hyg. 2009, 103, 443–446. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McFarland, J.W.; Howes, H.L. Novel anthelmintic agents. 6. Pyrantel analogs with activity against whipworm. J. Med. Chem. 1972, 15, 365–368. [Google Scholar] [CrossRef] [PubMed]
- Sheehan, D.J.; Sheehan, S.M.; Marchiondo, A.A. Discovery and Chemistry of Pyrantel, Morantel and Oxantel. Pyrantel Parasiticide Therapy in Humans and Domestic Animals, 1st ed.; Marchiondo, A.A., Ed.; Academic Press: Cambridge, MA, USA, 2016; pp. 1–19. [Google Scholar]
- Lee, E.L.; Iyngkaran, N.; Grieve, A.W.; Robinson, M.J.; Dissanaike, A.S. Therapeutic evaluation of oxantel pamoate (1, 4, 5, 6-tetrahydro-1-methyl-2-[trans-3-hydroxystyryl] pyrimidine pamoate) in severe Trichuris trichiura infection. Am. J. Trop. Med. Hyg. 1976, 25, 563–567. [Google Scholar] [CrossRef]
- Garcia, E.G. Treatment for trichuriasis with oxantel. Am. J. Trop. Med. Hyg. 1976, 25, 914–915. [Google Scholar] [CrossRef]
- Speich, B.; Ame, S.M.; Ali, S.M.; Alles, R.; Huwyler, J.; Hattendorf, J.; Utzinger, J.; Albonico, M.; Keiser, J. Oxantel pamoate-albendazole for Trichuris trichiura infection. N. Engl. J. Med. 2014, 370, 610–620. [Google Scholar] [CrossRef] [PubMed]
- Moser, W.; Ali, S.M.; Ame, S.M.; Speich, B.; Puchkov, M.; Huwyler, J.; Albonico, M.; Hattendorf, J.; Keiser, J. Efficacy and safety of oxantel pamoate in school-aged children infected with Trichuris trichiura on Pemba Island, Tanzania: A parallel, randomised, controlled, dose-ranging study. Lancet Infect. Dis. 2016, 16, 53–60. [Google Scholar] [CrossRef]
- Tritten, L.; Silbereisen, A.; Keiser, J. In vitro and in vivo efficacy of monepantel (AAD 1566) against laboratory models of human intestinal nematode infections. PLoS Negl. Trop. Dis. 2011, 5, e1457. [Google Scholar] [CrossRef] [PubMed]
- Keiser, J.; Tritten, L.; Silbereisen, A.; Speich, B.; Adelfio, R.; Vargas, M. Activity of oxantel pamoate monotherapy and combination chemotherapy against Trichuris muris and hookworms: Revival of an old drug. PLoS Negl. Trop. Dis. 2013, 7, e2119. [Google Scholar] [CrossRef] [Green Version]
- Karpstein, T.; Pasche, V.; Häberli, C.; Scandale, I.; Neodo, A.; Keiser, J. Evaluation of emodepside in laboratory models of human intestinal nematode and schistosome infections. Parasit. Vectors 2019, 12, 226. [Google Scholar] [CrossRef]
- Keiser, J.; Tritten, L.; Adelfio, R.; Vargas, M. Effect of combinations of marketed human anthelmintic drugs against Trichuris muris in vitro and in vivo. Parasit. Vectors 2012, 5, 292. [Google Scholar] [CrossRef] [Green Version]
- Klementowicz, J.E.; Travis, M.A.; Grencis, R.K. Trichuris muris: A model of gastrointestinal parasite infection. Semin. Immunopathol. 2012, 34, 815–828. [Google Scholar] [CrossRef] [Green Version]
- Beer, R.J.S. Studies on the biology of the life-cycle of Trichuris suis Schrank, 1788. Parasitology 1973, 67, 253–262. [Google Scholar] [CrossRef]
- Kringel, H.; Roepstorff, A. Trichuris suis population dynamics following a primary experimental infection. Veter. Parasitol. 2006, 139, 132–139. [Google Scholar] [CrossRef]
- Martin, R.J.; Clark, C.L.; Trailovic, S.M.; Robertson, A.P. Oxantel is an N-type (methyridine and nicotine) agonist not an L-type (levamisole and pyrantel) agonist: Classification of cholinergic anthelmintics in Ascaris. Int. J. Parasitol. 2004, 34, 1083–1090. [Google Scholar] [CrossRef]
- Wolstenholme, A.J.; Neveu, C. The interactions of anthelmintic drugs with nicotinic receptors in parasitic nematodes. Emerg. Top. Life Sci. 2017, 1, 667–673. [Google Scholar] [PubMed]
- Albuquerque, E.X.; Pereira, E.F.R.; Alkondon, M.; Rogers, S.W. Mammalian nicotinic acetylcholine receptors: From structure to function. Physiol. Rev. 2009, 89, 73–120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holden-Dye, L.; Joyner, M.; O’Connor, V.; Walker, R.J. Nicotinic acetylcholine receptors: A comparison of the nAChRs of Caenorhabditis elegans and parasitic nematodes. Parasitol. Int. 2013, 62, 606–615. [Google Scholar] [CrossRef] [PubMed]
- Hansen, T.V.A.; Cirera, S.; Neveu, C.; Courtot, E.; Charvet, C.L.; Calloe, K.; Klaerke, D.A.; Martin, R.J. The narrow-spectrum anthelmintic oxantel is a potent agonist of a novel acetylcholine receptor subtype in whipworms. PLoS Pathog. 2021, 17, e1008982. [Google Scholar] [CrossRef] [PubMed]
- Qian, H.; Martin, R.J.; Robertson, A.P. Pharmacology of N-, L-, and B-subtypes of nematode nAChR resolved at the single-channel level in Ascaris suum. FASEB J. 2006, 20, 2606–2608. [Google Scholar] [CrossRef] [Green Version]
- Courtot, E.; Charvet, C.L.; Beech, R.N.; Harmache, A.; Wolstenholme, A.J.; Holden-Dye, L.; O’Connor, V.; Peineau, N.; Woods, D.J.; Neveu, C. Functional characterization of a novel class of morantel-sensitive acetylcholine receptors in nematodes. PLoS Pathog. 2015, 11, e1005267. [Google Scholar] [CrossRef] [Green Version]
- Jones, A.K.; Sattelle, D.B. Functional genomics of the nicotinic acetylcholine receptor gene family of the nematode, Caenorhabditis elegans. Bioessays 2004, 26, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Brown, L.A.; Jones, A.K.; Buckingham, S.D.; Mee, C.J.; Sattelle, D.B. Contributions from Caenorhabditis elegans functional genetics to antiparasitic drug target identification and validation: Nicotinic acetylcholine receptors, a case study. Int. J. Parasitol. 2006, 36, 617–624. [Google Scholar] [CrossRef] [PubMed]
- Williamson, S.M.; Walsh, T.K.; Wolstenholme, A.J. The cys-loop ligand-gated ion channel gene family of Brugia malayi and Trichinella spiralis: A comparison with Caenorhabditis elegans. Invert. Neurosci. 2007, 7, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Ballivet, M.; Alliod, C.; Bertrand, S.; Bertrand, D. Nicotinic acetylcholine receptors in the nematode Caenorhabditis elegans. J. Mol. Biol. 1996, 258, 261–269. [Google Scholar] [CrossRef]
- Abongwa, M.; Buxton, S.K.; Courtot, E.; Charvet, C.L.; Neveu, C.; McCoy, C.J.; Verma, S.; Robertson, A.P.; Martin, R.J. Pharmacological profile of Ascaris suum ACR-16, a new homomeric nicotinic acetylcholine receptor widely distributed in Ascaris tissues. Br. J. Pharmacol. 2016, 173, 2463–2477. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaji, M.D.; Geary, T.G.; Beech, R.N. A Functional Comparison of homopentameric nicotinic acetylcholine receptors (ACR-16) receptors from Necator americanus and Ancylostoma ceylanicum. Front. Mol. Neurosci. 2020, 13, 601102. [Google Scholar] [CrossRef]
- Boulin, T.; Gielen, M.; Richmond, J.E.; Williams, D.C.; Paoletti, P.; Bessereau, J.L. Eight genes are required for functional reconstitution of the Caenorhabditis elegans levamisole-sensitive acetylcholine receptor. Proc. Natl. Acad. Sci. USA 2008, 105, 18590–18595. [Google Scholar] [CrossRef] [Green Version]
- Charvet, C.L.; Guégnard, F.; Courtot, E.; Cortet, J.; Neveu, C. Nicotine-sensitive acetylcholine receptors are relevant pharmacological targets for the control of multidrug resistant parasitic nematodes. Int. J. Parasitol. Drugs Drug Resist. 2018, 8, 540–549. [Google Scholar] [CrossRef]
- Choudhary, S.; Marjianović, D.S.; Wong, C.R.; Zhang, X.; Abongwa, M.; Coats, J.R.; Trailović, S.M.; Martin, R.J.; Robertson, A.P. Menthol acts as a positive allosteric modulator on nematode levamisole sensitive nicotinic acetylcholine receptors. Int. J. Parasitol. Drugs Drug Resist. 2019, 9, 44–53. [Google Scholar] [CrossRef]
- Palmeirim, M.S.; Specht, S.; Scandale, I.; Gander-Meisterernst, I.; Chabicovsky, M.; Keiser, J. Preclinical and clinical characteristics of the trichuricidal drug oxantel pamoate and clinical development plans: A review. Drugs 2021, 81, 907–921. [Google Scholar] [CrossRef]
- Raymond, V.; Mongan, N.P.; Sattelle, D.B. Anthelmintic actions on homomer-forming nicotinic acetylcholine receptor subunits: Chicken α7 and ACR-16 from the nematode Caenorhabditis elegans. Neuroscience 2000, 101, 785–791. [Google Scholar] [CrossRef]
- Howes, H.L., Jr. trans-1,4,5,6-Tetrahydro-2-(3-hydroxystyryl)-1-methyl pyrimidine (CP-14,445), A New antiwhipworm agent. Proc. Soc. Exp. Biol. Med. 1972, 139, 394–398. [Google Scholar] [CrossRef]
- Cowan, N.; Meier, C.; Neodo, A.; Keiser, J. Exposure of Heligmosomoides polygyrus and Trichuris muris to albendazole, albendazole sulfoxide, mebendazole and oxantel pamoate in vitro and in vivo to elucidate the pathway of drug entry into these gastrointestinal nematodes. Int. J. Parasitol. Drugs Drug Resist. 2017, 7, 159–173. [Google Scholar] [CrossRef]
- Cowan, N.; Vargas, M.; Keiser, J. In vitro and in vivo drug interaction study for two lead combinations oxantel pamoate plus albendazole and albendazole plus mebendazole for the treatment of human soil-transmitted helminthiasis. Antimicrob. Agents Chemother. 2016, 60, 6127–6133. [Google Scholar] [CrossRef] [Green Version]
- Spande, T.F.; Garraffo, H.M.; Yeh, H.J.C.; Pu, Q.L.; Pannell, L.K.; Daly, J.W. A new class of alkaloids from a dendrobatid poison frog: A structure for alkaloid 251F. J. Nat. Prod. 1992, 55, 707–722. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Sestito, S.; Rapposelli, S.; Peron, G.; Calina, D.; Sharifi-Rad, M.; Sharopov, F.; Martins, N.; Sharifi-Rad, J. Epibatidine: A Promising Natural Alkaloid in Health. Biomolecules 2018, 9, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sullivan, J.P.; Decker, M.W.; Brioni, J.D.; Donnelly-Roberts, D.; Anderson, D.J.; Bannon, A.W.; Kang, C.H.; Adams, P.; Piattoni-Kaplan, M.; Buckley, M.J. (+/−)-Epibatidine elicits a diversity of in vitro and in vivo effects mediated by nicotinic acetylcholine receptors. J. Pharmacol. Exp. Ther. 1994, 271, 624–631. [Google Scholar] [PubMed]
- Bartos, M.; Rayes, D.; Bouzat, C. Molecular determinants of pyrantel selectivity in nicotinic receptors. Mol. Pharmacol. 2006, 70, 1307–1318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rayes, D.; De Rosa, M.J.; Bartos, M.; Bouzat, C. Molecular basis of the differential sensitivity of nematode and mammalian muscle to the anthelmintic agent levamisole. J. Biol. Chem. 2004, 279, 36372–36381. [Google Scholar] [CrossRef] [Green Version]
- Buxton, S.K.; Charvet, C.L.; Neveu, C.; Cabaret, J.; Cortet, J.; Peineau, N.; Abongwa, M.; Courtot, E.; Robertson, A.P.; Martin, R.J. Investigation of acetylcholine receptor diversity in a nematode parasite leads to characterization of tribendimidine- and derquantel-sensitive nAChRs. PLoS Pathog. 2014, 10, e1003870. [Google Scholar] [CrossRef] [Green Version]
- Rauthan, M.; Gong, J.; Liu, J.; Li, Z.; Wescott, S.A.; Liu, J.; Xu, X.Z.S. MicroRNA Regulation of nAChR Expression and Nicotine-Dependent Behavior in C. elegans. Cell Rep. 2017, 21, 1434–1441. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jones, A.K.; Davis, P.; Hodgkin, J.; Sattelle, D.B. The nicotinic acetylcholine receptor gene family of the nematode Caenorhabditis elegans: An update on nomenclature. Invert. Neurosci. 2007, 7, 129–131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Millar, N.S. RIC-3: A nicotinic acetylcholine receptor chaperone. Br. J. Pharmacol. 2008, 153 (Suppl. 1), S177–S183. [Google Scholar] [CrossRef] [Green Version]
- Choudhary, S.; Buxton, S.K.; Puttachary, S.; Verma, S.; Mair, G.R.; McCoy, C.J.; Reaves, B.J.; Wolstenholme, A.J.; Martin, R.J.; Robertson, A.P. EAT-18 is an essential auxiliary protein interacting with the non-alpha nAChR subunit EAT-2 to form a functional receptor. PLoS Pathog. 2020, 16, e1008396. [Google Scholar] [CrossRef] [PubMed]
- Boulin, T.; Rapti, G.; Briseño-Roa, L.; Stigloher, C.; Richmond, J.E.; Paoletti, P.; Bessereau, J.L. Positive modulation of a Cys-loop acetylcholine receptor by an auxiliary transmembrane subunit. Nat. Neurosci. 2012, 15, 1374–1381. [Google Scholar] [CrossRef]
- Wakelin, D. Acquired immunity to Trichuris muris in the albino laboratory mouse. Parasitology 1967, 57, 515–524. [Google Scholar] [CrossRef] [PubMed]
- Altschul, S.F.; Madden, T.L.; Schäffer, A.A.; Zhang, J.; Zhang, Z.; Miller, W.; Lipman, D.J. Gapped BLAST and PSI-BLAST: A new generation of protein database search programs. Nucleic Acids Res. 1997, 25, 3389–3402. [Google Scholar] [CrossRef] [Green Version]
- Petersen, T.N.; Brunak, S.; Von Heijne, G.; Nielsen, H. SignalP 4.0: Discriminating signal peptides from transmembrane regions. Nat. Methods. 2011, 8, 785–786. [Google Scholar] [CrossRef]
- Krogh, A.; Larsson, B.; Von Heijne, G.; Sonnhammer, E.L. Predicting transmembrane protein topology with a hidden Markov model: Application to complete genomes. J. Mol. Biol. 2001, 305, 567–580. [Google Scholar] [CrossRef] [Green Version]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef] [Green Version]
- Miledi, R.; Parker, I. Chloride current induced by injection of calcium into Xenopus oocytes. J. Physiol. 1984, 357, 173–183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hansen, T.V.A.; Grencis, R.K.; Issouf, M.; Neveu, C.; Charvet, C.L. Functional Characterization of the Oxantel-Sensitive Acetylcholine Receptor from Trichuris muris. Pharmaceuticals 2021, 14, 698. https://0-doi-org.brum.beds.ac.uk/10.3390/ph14070698
Hansen TVA, Grencis RK, Issouf M, Neveu C, Charvet CL. Functional Characterization of the Oxantel-Sensitive Acetylcholine Receptor from Trichuris muris. Pharmaceuticals. 2021; 14(7):698. https://0-doi-org.brum.beds.ac.uk/10.3390/ph14070698
Chicago/Turabian StyleHansen, Tina V. A., Richard K. Grencis, Mohamed Issouf, Cédric Neveu, and Claude L. Charvet. 2021. "Functional Characterization of the Oxantel-Sensitive Acetylcholine Receptor from Trichuris muris" Pharmaceuticals 14, no. 7: 698. https://0-doi-org.brum.beds.ac.uk/10.3390/ph14070698