GC-MS Chemical Characterization and In Vitro Evaluation of Antioxidant and Toxic Effects Using Drosophila melanogaster Model of the Essential Oil of Lantana montevidensis (Spreng) Briq.
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Extraction of L. montevidensis Leaf Essential Oil
2.3. Composition and Identification of the Constituents of the Essential Oil
2.4. Reagents
2.5. Antioxidant Activity in Chemical Model
2.5.1. Antioxidant Capacity in Chemical Model: 1,1-Diphenyl-2-picrylhydrazyl (DPPH) Radical Scavenging Activity
2.5.2. Fe2+ Chelating Activity of L. montevidensis Leaf Essential Oil
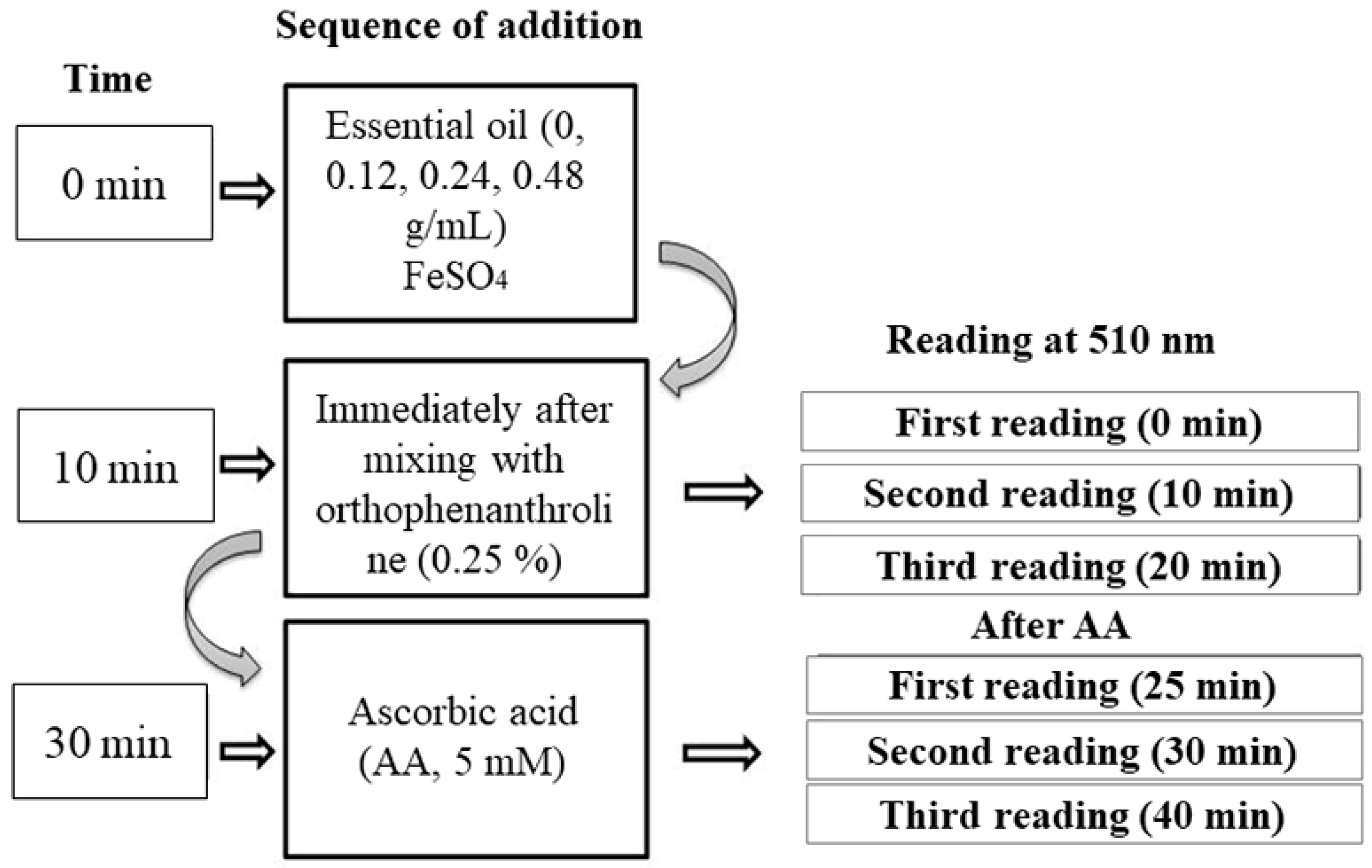
2.5.3. Fe3+ Reducing Power of L. montevidensis Leaf Essential Oil
2.6. Biological Assays with Drosophila Melanogaster
Fumigant Toxicity
2.7. Determination of Total Thiol and Non-Protein Thiols (NPSH)
2.8. Assessment of Cell Viability
2.9. Determination of Iron Levels
2.10. Measurement of Malondialdehyde (MDA)
2.11. Statistical Analysis
3. Results
3.1. Chemical Characterization of L. montevidensis Leaf Essential Oil
3.2. Antioxidant Activity
3.2.1. Scavenging Activity of the Essential Oil from L. montevidensis on DPPH Radical
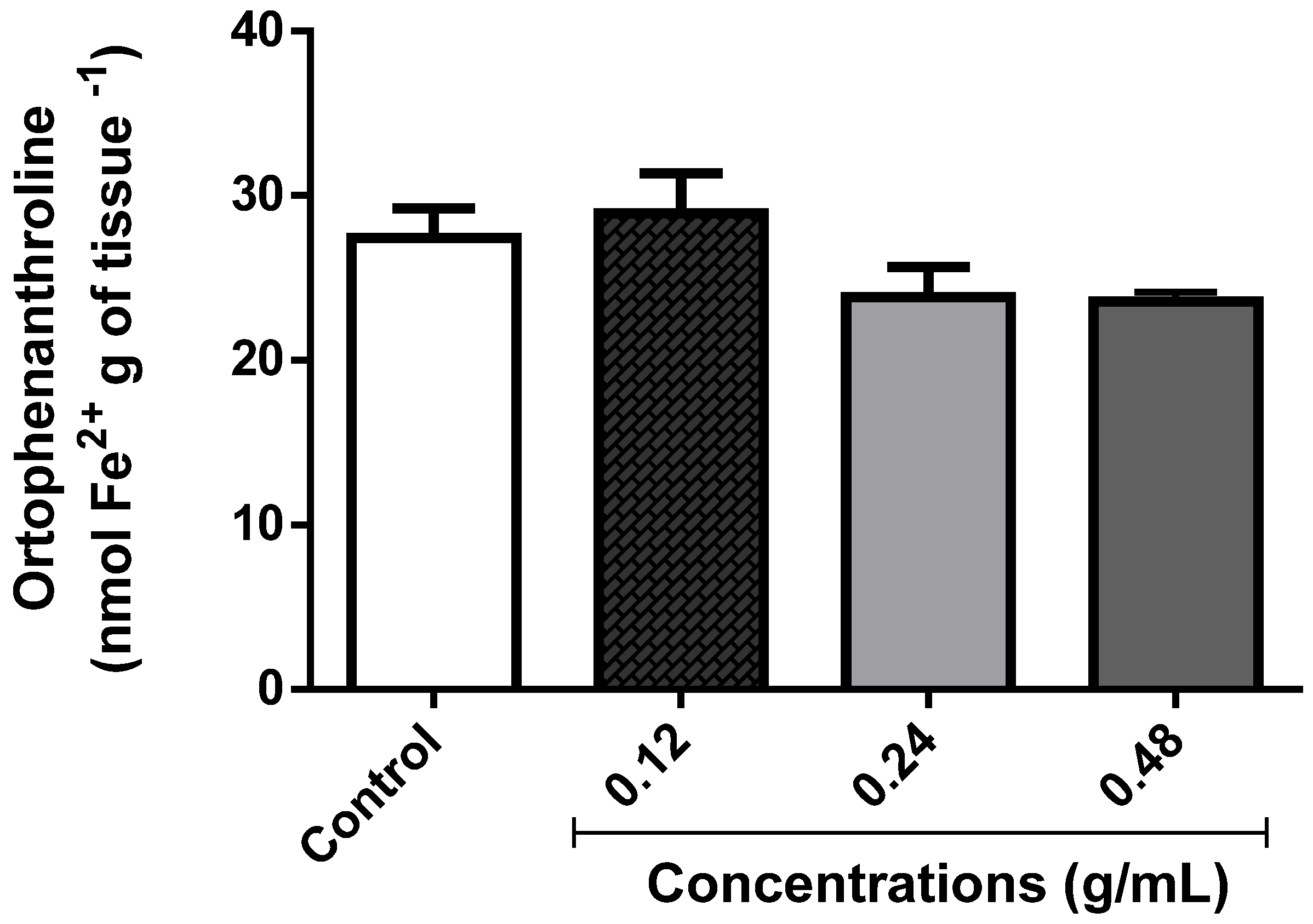
3.2.2. Fe2+ Chelation or Oxidation Potential of L. montevidensis Leaf Essential Oil
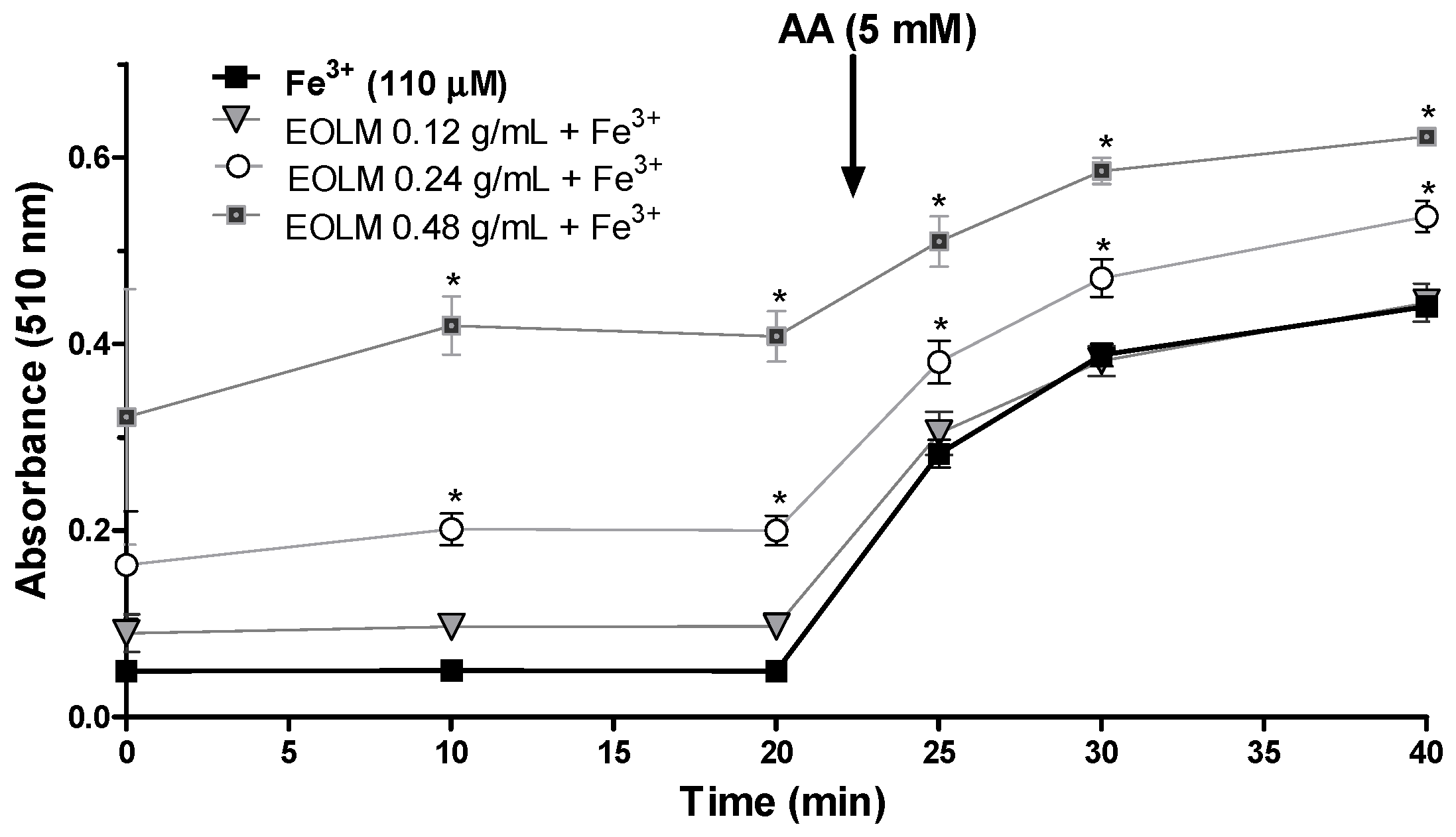
3.2.3. Fe3+ Reducing Properties of L. montevidensis Leaf Essential Oil
3.3. Biological Assays with Drosophila Melanogaster
3.3.1. Fumigant Activity of Leaf Essential Oil L. montevidensis
3.3.2. Quantification of Total Thiol and Non-Protein Thiols (GSH and NPSH)
3.3.3. Cell Viability of D. melanogaster Supernatant
3.3.4. Effect of EOLM on Fe2+ Content
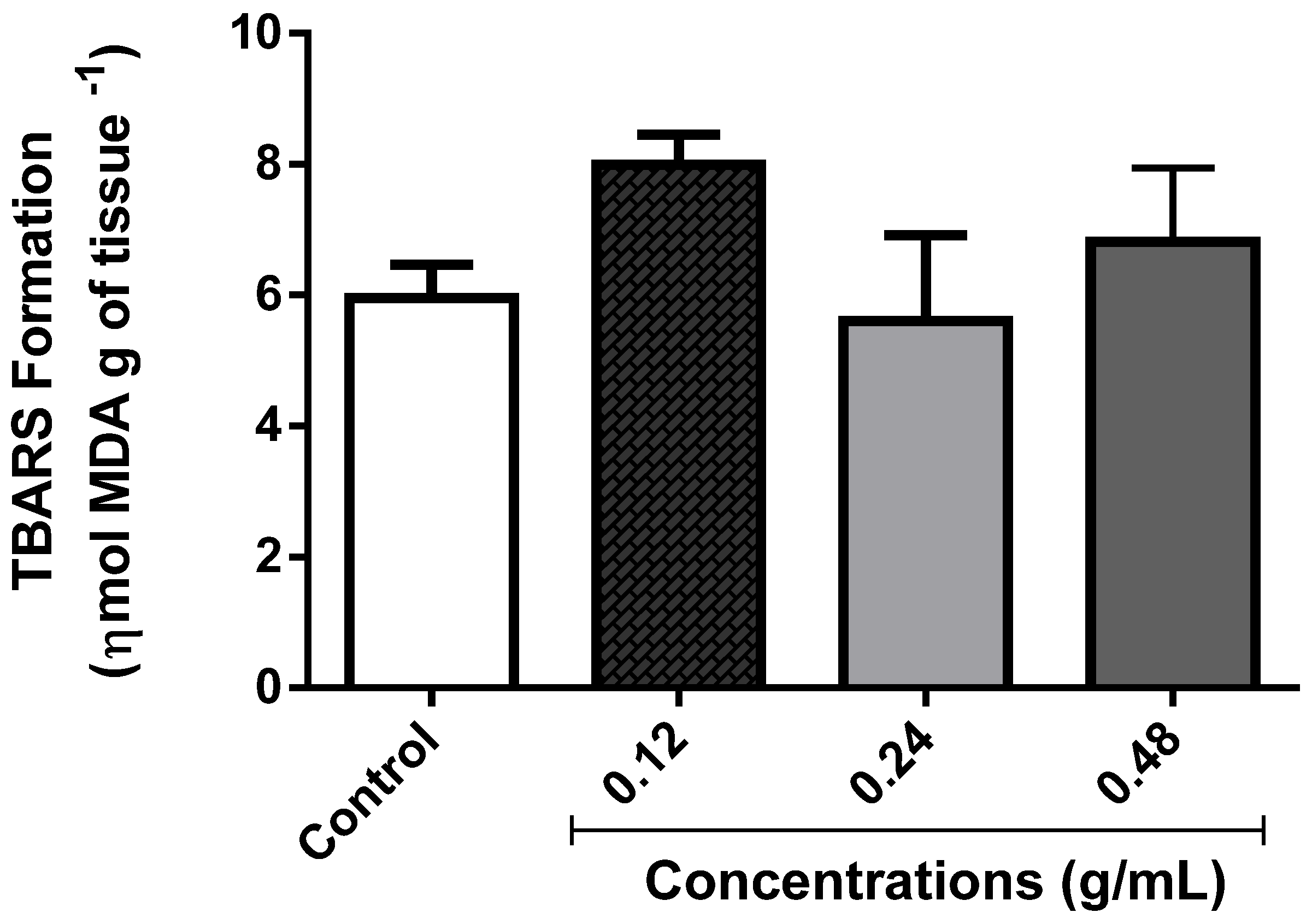
3.3.5. Determination of Lipid Peroxidation
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hua, H.; Xing, F.; Selvara, J.N.; Wang, Y.; Zhao, Y.; Zhou, L.; Liu, X.; Liu, Y. Inhibitory effect of essential oils on Aspergillus ochraceus growth and ochratoxin a production. PLoS ONE 2014, 9, e108285. [Google Scholar] [CrossRef] [PubMed]
- Kamdem, J.P.; Abolaji, A.O.; Elekofehinti, O.O.; Omotuyi, I.O.; Ibrahim, M.; Hassan, W.; Barbosa, N.V.; Souza, D.O.; Rocha, J.B.T. Therapeutic Potential of Plant Extracts and Phytochemicals Against Brain Ischemia-Reperfusion Injury: A Review. Nat. Prod. J. 2016, 6, 250–284. [Google Scholar] [CrossRef]
- Fang, Y.Z.; Yang, S.; Wu, G. Freeradicals, antioxidants, andnutrition. Nutrition 2002, 18, 872–879. [Google Scholar] [PubMed]
- Rao, K.M.; Shashidhara, L.S. Human APC sequesters β-catenin even in the absence of GSK-3β in a Drosophila model. Oncogene 2008, 27, 2488–2493. [Google Scholar] [CrossRef] [PubMed]
- Chikezie, P.C.; Uwakwe, A.A. Membrane stability of sickle erythrocytes incubated in essential oil of three medicinal plants: Anacardium occidentale, Psidium guajava, and Terminalia catappa. Pharmacogn. Mag. 2011, 7, 121. [Google Scholar] [CrossRef] [PubMed]
- De Beer, D.; Joubert, E.; Gelderblom, W.C.A.; Manley, M. Phenolic compounds: A review of their possible role as in vivo antioxidants of wine. S. Afr. J. Enol. Vitic. 2017, 23, 48–61. [Google Scholar] [CrossRef]
- Bekhit, A.E.D.A.; Cheng, V.J.; McConnell, M.; Zhao, J.H.; Sedcole, R.; Harrison, R. Antioxidant activities, sensory and anti-influenza activity of grape skin tea infusion. Food Chem. 2011, 129, 837–845. [Google Scholar]
- Vasilescu, I.; Eremia, S.A.; Albu, C.; Radoi, A.; Litescu, S.C.; Radu, G.L. Determination of the antiradical properties of olive oils using an electrochemical method based on DPPH radical. Food Chem. 2015, 166, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Barros, L.M.; Duarte, A.E.; Waczuk, E.P.; Roversi, K.; da Cunha, F.A.B.; Rolon, M.; Coronel, C.; Gomez, M.C.V.; de Menezes, I.R.A.; da Costa, J.G.M.; et al. Safety assessment and antioxidant activity of Lantana montevidensis leaves: Contribution to its phytochemical and pharmacological activity. EXCLI J. 2017, 16, 566–582. [Google Scholar] [PubMed]
- Scarfiotti, C.; Fabris, F.; Cestaro, B.; Giuliani, A. Free radicals, atherosclerosis, ageing, and related dysmetabolic pathologies: Pathological and clinical aspects. Eur. J. Cancer Prev. 1997, 6, 31–36. [Google Scholar] [CrossRef]
- Garcia, G.R.; Goodale, B.C.; Wiley, M.W.; La Du, J.K.; Hendrix, D.A.; Tanguay, R.L. In vivo characterization of an AHR-dependent long non-coding RNA required for proper Sox9b expression. Mol. Pharmacol. 2017, 91, 609–619. [Google Scholar] [CrossRef] [PubMed]
- Carmona-Jiménez, Y.; García-Moreno, M.V.; Igartuburu, J.M.; Barroso, C.G. Simplification of the DPPH assay for estimating the antioxidant activity of wine and wine by-products. Food Chem. 2014, 165, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Musa, K.H.; Abdullah, A.; Kuswandi, B.; Hidayat, M.A. A novel high throughput method based on the DPPH dry reagent array for 2 determination of antioxidant activity. Food Chem. 2013, 141, 4102–4106. [Google Scholar] [CrossRef] [PubMed]
- Sacchetti, G.; Maietti, S.; Muzzoli, M.; Scaglianti, M.; Manfredini, S.; Radice, M.; Bruni, R. Comparative evaluation of 11 essential oils of different origin as functional antioxidants, antiradicals and antimicrobials in foods. Food Chem. 2005, 91, 621–632. [Google Scholar] [CrossRef]
- Yassa, N.; Masoomi, F.; Rankouhi, S.R.; Hadjiakhoondi, A. Chemical composition and antioxidant activity of the extract and essential oil of Rosa damascena from Iran, population of Guilan. DARU J. Pharm. Sci. 2015, 17, 175–180. [Google Scholar]
- Xie, Y.J.; Wang, K.; Huang, Q.Y.; Lei, C.L. Evaluation toxicity of monoterpenes to subterranean termite, Reticulitermes chinensis Snyder. Ind. Crops Prod. 2014, 53, 163–166. [Google Scholar] [CrossRef]
- Neahoua-Bougherra, H.H.; Bedini, S.; Cosci, F.; Flamini, G.; Belhamel, K.; Conti, B. Enhancing the insecticidal efficacy of inert dusts against stored food insect pest by the combined action with essential oils. IOBC-WPRS Bull. 2015, 111, 31–38. [Google Scholar]
- Mansoura, S.A.; El-Sharkawy, A.Z.; Abdel-Hamid, N.A. Toxicity of essential plant oils, in comparison with conventional insecticides, against the desert locust, Schistocerca gregaria (Forskål). Ind. Crops Prod. 2015, 63, 92–99. [Google Scholar] [CrossRef]
- Peixoto, M.G.; Bacci, L.; Blank, A.F.; Araújo, A.P.A.; Alves, P.B.; Silva, J.H.S.; Santos, A.A.; Oliveira, A.P.; da Costa, A.S.; Blank, M.F.A. Toxicity and repellency of essential oils of Lippia alba chemotypes and their major monoterpenes against stored grain insects. Ind. Crops Prod. 2015, 71, 31–36. [Google Scholar] [CrossRef]
- Zhang, Z.; Yang, T.; Zhang, Y.; Wang, L.; Xie, Y. Fumigant toxicity of monoterpenes against fruitfly, Drosophila melanogaster. Ind. Crops Prod. 2016, 81, 147–151. [Google Scholar] [CrossRef]
- Nagao, T.; Abe, F.; Kinjo, J.; Okabe, H. Antiproliferative constituents in plants. 10 flavones from the leaves of Lantana montevidensis BRIQ. and consideration of structure activity relationship. Biol. Pharm. Bull. 2002, 25, 875–879. [Google Scholar] [CrossRef]
- Barreto, F.S.; Sousa, E.O.; Rodrigues, F.F.G.; Costa, J.G.M.; Campos, A.R. Antibacterial Activity of Lantana camara Linn Lantana montevidensis Briq extracts from Cariri-Ceara, Brazil. J. Young Pharm. 2010, 2, 42–44. [Google Scholar] [CrossRef]
- Sousa, E.O.; Almeida, T.S.; Rodrigues, F.F.; Campos, A.R.; Lima, S.G.; Costa, J.G. Lantana montevidensis Briq improves the aminoglycoside activity against multiresistant Escherichia coli and Staphylococcus aureus. Indian J. Pharmacol. 2011, 43, 180–182. [Google Scholar]
- Sousa, E.O.; Barreto, F.S.; Rodrigues, F.F.; Campos, A.R.; Costa, J.G. Chemical composition of the essential oils of Lantana camara L. and Lantana montevidensis Briq. and their synergistic antibiotic effects on aminoglycosides. J. Essent. Oil Res. 2012, 24, 447–452. [Google Scholar] [CrossRef]
- Sousa, E.O.; Rodrigues, F.F.G.; Campos, A.R.; Lima, S.G.; da Costa, J.G.M. Chemical composition and synergistic interaction between aminoglycosides antibiotics and essential oil of Lantana montevidensis Briq. Nat. Prod. Res. 2013, 27, 942–945. [Google Scholar] [CrossRef]
- Nichols, C.D. Drosophila melanogaster neurobiology, neuropharmacology, and how the fly can inform central nervous system drug discovery. Pharmacol. Ther. 2006, 112, 677–700. [Google Scholar] [CrossRef]
- Pandey, U.B.; Nichols, C.D. Human disease models in Drosophila melanogaster and the role of the fly in therapeutic drug discovery. Pharmacol. Rev. 2011, 63, 411–436. [Google Scholar] [CrossRef]
- Feany, M.B.; Bender, W.W. A Drosophila model of Parkinson’s disease. Nature 2000, 404, 394–398. [Google Scholar] [CrossRef]
- Tiwari, S.; Gondhalekar, A.D.; Mann, R.S.; Scharf, M.E.; Stelinski, L.L. Characterization of five CYP4 genes from Asian citrus psyllid and their expression levels in Candidatus Liberibacter asiaticus-infected and uninfected psyllids. Insect Mol. Biol. 2011, 20, 733–744. [Google Scholar] [CrossRef]
- Zolfaghari Emameh, R.; Syrjänen, L.; Barker, H.; Supuran, C.T.; Parkkila, S. Drosophila melanogaster: A model organism for controlling Dipteran vectors and pests. J. Enzyme Inhib. Med. Chem. 2015, 30, 505–513. [Google Scholar] [CrossRef]
- Panchal, K.; Tiwari, A.K. Drosophila melanogaster “a potential model organism” for identification of pharmacological properties of plants/plant-derived components. Biomed. Pharmacother. 2017, 89, 1331–1345. [Google Scholar] [CrossRef]
- Matos, F.J.A. Introduction to Experimental Phytochemistry, 2nd ed.; UFC: Fortaleza, Brazil, 2002. [Google Scholar]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry; Allured Publishing Corporation: Carol Stream, IL, USA, 2007. [Google Scholar]
- Kamdem, J.P.; Stefanello, S.T.; Boligon, A.A.; Wagner, C.; Kade, I.J.; Pereira, R.P.; Preste, A.S.; Roos, D.H.; Waczuk, E.P.; Appel, A.S.; et al. In vitro antioxidant activity of stem bark of Trichilia catigua Adr. Juss. Acta Pharm. 2012, 62, 371–382. [Google Scholar] [CrossRef] [PubMed]
- Dinis, T.C.P.; Madeira, V.M.C.; Almeida, M.L.M. Action of phenolic derivates (acetoaminophen, salycilate and 5-aminosalycilate) as inhibitors of membrane lipid peroxidation and as peroxyl radical scavengers. Arch. Biochem. Biophys. 1994, 315, 161–169. [Google Scholar] [CrossRef]
- Kamdem, J.P.; Adeniran, A.; Boligon, A.A.; Klimaczewsk, C.V.; Elekofehinti, O.O.; Hassan, W.; Ibrahim, M.; Waczuk, E.P.; Meinerz, D.F.; Athayde, M.L. Antioxidant activity, genotoxicity and cytotoxicity evaluation of lemon balm (Melissa officinalis L.) ethanolic extract: Its potential role in neuroprotection. Ind. Crops Prod. 2013, 51, 26–34. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Klimaczewsk, C.V.; Saraiva, R.A.; Roos, D.H.; Boligon, A.; Athayde, M.L.; Kamdem, J.P.; Barbosa, N.V.; Rocha, J.B.T. Antioxidant activity of Peumus boldus extract and alkaloid boldine against damage induced by Fe (II)–citrate in rat liver mitochondria in vitro. Ind. Crops Prod. 2014, 54, 240–247. [Google Scholar] [CrossRef]
- Barbosa Filho, V.M.; Waczuk, E.P.; Kamdem, J.P.; Abolaji, A.O.; Lacerda, S.R.; da Costa, J.G.; de Menezes, I.R.; Boligon, A.A.; Athayde, M.L.; da Rocha, J.B.; et al. Phytochemical constituents, antioxidant activity, cytotoxicity and osmotic fragility effects of Caju (Anacardium microcarpum). Ind. Crops Prod. 2014, 55, 280–288. [Google Scholar] [CrossRef]
- Hossain, M.A.; Shah, M.D. A study on the total phenols content and antioxidant activity of essential oil and different solvent extracts of endemic plant Merremia borneensis. Arab. J. Chem. 2015, 8, 66–71. [Google Scholar] [CrossRef]
- Siddique, Y.H.; Mujtaba, S.F.; Jyoti, S.; Naz, F. GC-MS analysis of Eucalyptus citriodora leaf extract and its role on the dietary supplementation in transgenic Drosophila model of Parkinson’s disease. Food Chem. Toxicol. 2013, 55, 29–35. [Google Scholar] [CrossRef]
- Liu, Q.F.; Lee, J.H.; Kim, Y.M.; Lee, S.; Hong, Y.K.; Hwang, S.; Oh, Y.; Lee, K.; Yun, H.S.; Lee, I.S.; et al. In vivo screening of traditional medicinal plants for neuroprotective activity against Aβ42 cytotoxicity by using Drosophila models of Alzheimer’s disease. Biol. Pharm. Bull. 2015, 38, 1891–1901. [Google Scholar] [CrossRef] [PubMed]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef]
- Guilhon de Castro, H.; Borges de Moura Perini, V.; Rodrigues dos Santos, G.; Castro Alves Barros Leal, T. Avaliação do teor e composição do óleo essencial de Cymbopogon nardus (L.) em diferentes épocas de colheita. Revista Ciência Agronômica, 2010; 41, 308–314. [Google Scholar]
- Bezerra, J.W.A.; Rodrigues, F.C.; Costa, A.R.; Boligon, A.A.; da Rocha, J.B.T.; Barros, L.M. Estudo químico-biológico do óleo essencial de Lantana montevidensis (chumbinho) (Spreng.) Briq. (Verbenaceae) contra Drosophila melanogaster. Rev. Bras. Plantas Med. 2017, 22, 1–13. [Google Scholar]
- Charles, D.J.; Simon, J.E. Comparison of extraction methods for the rapid determination of essential oil content and composition of basil. J. Am. Soc. Hortic. Sci. 1990, 115, 458–462. [Google Scholar] [CrossRef]
- Jorge, S.S.A.; Nardes, P.R.B.; Guarim, N.G.; Macedo, M. O uso medicinal da arnica, Brickelia brasiliensis (Spreng.) Robinson (Asteraceae). Revista Saúde e Ambiente 1998, 1, 107–121. [Google Scholar]
- Facanali, R.; Campos, M.M.S.; Pocius, O.; Ming, L.C.; Soares-Scott, M.D.; Marques, M.O.M. Biologia reprodutiva de populações de Ocimum selloi Benth. Rev. Bras. Plantas Med. 2009, 11, 141–146. [Google Scholar] [CrossRef]
- Passos, G.F.; Fernandes, E.S.; da Cunha, F.M.; Ferreira, J.; Pianowski, L.F.; Campos, M.M.; Calixto, J.B. Anti-inflammatory and anti-allergic properties of the essential oil and active compounds from Cordia verbenacea. J. Ethnopharmacol. 2007, 110, 323–333. [Google Scholar] [CrossRef]
- Marinho, D.F.; Oliveira, E.C.P.D.; Araújo, J.A.D.S.; Pinto, I.F.; Lima, H.S.D.; Moraes, W.P.; Ambrósio, C.E.; Morini, A.C. Evaluation of ultrasonic transmission of Copaifera duckei Dwyer herbal gel. Pesqui. Veterinária Bras. 2017, 37, 516–520. [Google Scholar] [CrossRef]
- Martins, A.O.; Rodrigues, L.B.; Cesário, F.R.; de Oliveira, M.R.; Tintino, C.D.; e Castro, F.F.; Alcântara, I.S.; Fernandes, M.N.; de Albuquerque, T.R.; da Silva, M.S.; et al. Anti-edematogenic and anti-inflammatory activity of the essential oil from Croton rhamnifolioides leaves and its major constituent 1, 8-cineole (eucalyptol). Biomed. Pharmacother. 2017, 96, 384–395. [Google Scholar] [CrossRef]
- Steliopoulos, P.; Wüst, M.; Adam, K.P.; Mosandl, A. Biosynthesis of the sesquiterpene germacrene D in Solidago canadensis: 13C and 2H labeling studies. Phytochemistry 2002, 60, 13–20. [Google Scholar] [CrossRef]
- Frankel, E.N.; Meyer, A.S. The problem of using one- dimensional methods to evaluate multifunctional food and biological antioxidants. J. Sci. Food Agric. 2000, 80, 1925–1941. [Google Scholar] [CrossRef]
- Siddhuraju, P.; Becker, K. Antioxidant properties of various solvent extracts of total phenolic constituents from three different agroclimatic origins of drumstick tree (Moringa oleifera Lam.) leaves. J. Agric. Food Chem. 2003, 51, 2144–2155. [Google Scholar] [CrossRef]
- Siddhuraju, P.; Becker, K. The antioxidant and free radical scavenging activities of processed cowpea (Vignaunguiculata (L.) Walp.) seed extracts. Food Chem. 2007, 101, 10–19. [Google Scholar] [CrossRef]
- Pavela, R.; Benelli, G. Essential oils as ecofriendly biopesticides? Challenges and constraints. Trends Plant Sci. 2016, 21, 1000–1007. [Google Scholar] [CrossRef]
- Pistón, M.; Machado, I.; Branco, C.S.; Cesio, V.; Heinzen, H.; Ribeiro, D.; Fernandes, E.; Chisté, R.C.; Freitas, M. Infusion, decoction and hydroalcoholic extracts of leaves from artichoke (Cynara cardunculus L. subsp. cardunculus) are effective scavengers of physiologically relevant ROS and RNS. Food Res. Int. 2014, 64, 150–156. [Google Scholar]
- Gupta, D.K.; Palma, J.M.; Corpas, F.J. Reactive Oxygen Species and Oxidative Damage in Plants under Stress; Springer: Heidelberg, Germany, 2015; pp. 1–22. [Google Scholar]
- Wang, J.; Hu, S.; Nie, S.; Yu, Q.; Xie, M. Reviews on mechanisms of in vitro antioxidant activity of polysaccharides. Oxidative Med. Cell. Longev. 2016, 2016. [Google Scholar] [CrossRef]
- Mittler, R. ROS are good. Trends Plant Sci. 2017, 22, 11–19. [Google Scholar] [CrossRef]
- Oliveira, F.; Rocha, S.; Fernandes, R. Iron metabolism: from health to disease. J. Clin. Lab. Anal. 2014, 28, 210–218. [Google Scholar] [CrossRef]
- Connor, J.R.; Malecki, E.A.; Cable, E.E.; Isom, H.C. The lipophilic iron compound TMH-ferrocene [(3, 5, 5-trimethylhexanoyl) ferrocene] increases iron concentrations, neuronal l-ferritin, and heme oxygenase in brains of BALB/c mice. Biol. Trace Elem. Res. 2002, 86, 73–84. [Google Scholar] [CrossRef]
- Rooney, J.P. The role of thiols, dithiols, nutritional factors and interacting ligands in the toxicology of mercury. Toxicology 2007, 234, 145–156. [Google Scholar] [CrossRef]
- Ferreira, I.C.; Abreu, R. Oxidative stress, antioxidants and phytochemicals. Bioanálise 2007, 4, 32–39. [Google Scholar]
- Ghorbel, I.; Khemakhem, M.; Boudawara, O.; Marrekchi, R.; Jamoussi, K.; Amar, R.B.; Boudawara, T.; Zeghal, N.; Kamoun, N.G. Effects of dietary extra virgin olive oil and its fractions on antioxidant status and DNA damage in the heart of rats co-exposed to aluminum and acrylamide. Food Funct. 2015, 6, 3098–3108. [Google Scholar] [CrossRef]




| Compounds | RI a | RI b | Oil Composition (%) L. montevidensis |
|---|---|---|---|
| α-Pinene | 939 | 937 | nd |
| Camphene | 953 | 951 | 0.11 |
| Sabinene | 976 | 675 | 0.29 |
| β-Pinene | 980 | 983 | nd |
| Myrcene | 991 | 990 | nd |
| α-Terpinene | 1018 | 1015 | nd |
| p-Cymene | 1026 | 1026 | nd |
| (Z)-β-Ocimene | 1040 | 1037 | nd |
| (E)-β-Ocimene | 1050 | 1054 | nd |
| γ-Terpinene | 1062 | 1061 | nd |
| Terpinolene | 1088 | 1079 | 1.67 |
| cis-Linalool oxide | 1074 | 1074 | 3.86 |
| Linalool | 1098 | 1199 | 1.09 |
| Camphor | 1143 | 1141 | 0.43 |
| Terpin-4-ol | 1177 | 1174 | nd |
| α-Terpineol | 1189 | 1193 | nd |
| t-Sabinene hydrate | 1254 | 1257 | 0.71 |
| α-Copaene | 1376 | 1376 | 5.98 |
| β-Elemene | 1391 | 1389 | 0.84 |
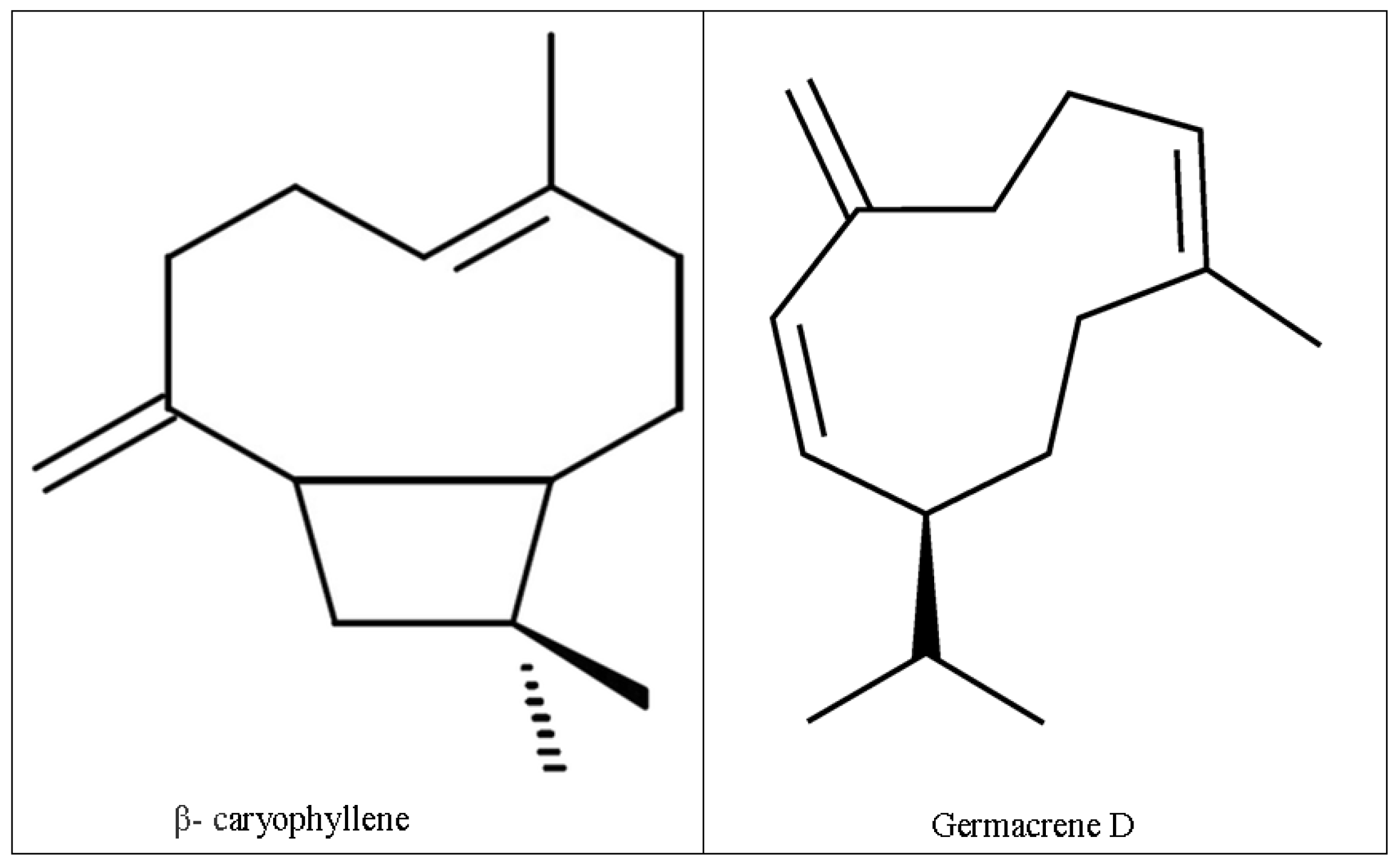
| β-Caryophyllene | 1404 | 1401 | 28.15 |
| (E)-Caryophyllene | 1418 | 1423 | 2.49 |
| Aromandendrene-allo | 1461 | 1460 | 1.12 |
| α-Humulene | 1454 | 1451 | 5.81 |
| Germacrene D | 1480 | 1480 | 31.27 |
| Valencene | 1491 | 1489 | nd |
| Biciclogermacrene | 1494 | 1497 | 6.04 |
| Cubebol | 1514 | 1518 | nd |
| δ-Cadinene | 1513 | 1509 | nd |
| α-Cadidene | 1538 | 1538 | 3.84 |
| Spathulenol | 1576 | 1573 | 0.96 |
| Caryophyllene oxide | 1581 | 1585 | 5.07 |
| Total identified (%) | 99.73 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Oliveira, M.R.C.; Barros, L.M.; Duarte, A.E.; Gabriely de Lima Silva, M.; da Silva, B.A.F.; Oliveira Brito Pereira Bezerra, A.; Oliveira Tintino, C.D.M.; Afonso Pereira de Oliveira, V.; Boligon, A.A.; Kamdem, J.P.; et al. GC-MS Chemical Characterization and In Vitro Evaluation of Antioxidant and Toxic Effects Using Drosophila melanogaster Model of the Essential Oil of Lantana montevidensis (Spreng) Briq. Medicina 2019, 55, 194. https://0-doi-org.brum.beds.ac.uk/10.3390/medicina55050194
de Oliveira MRC, Barros LM, Duarte AE, Gabriely de Lima Silva M, da Silva BAF, Oliveira Brito Pereira Bezerra A, Oliveira Tintino CDM, Afonso Pereira de Oliveira V, Boligon AA, Kamdem JP, et al. GC-MS Chemical Characterization and In Vitro Evaluation of Antioxidant and Toxic Effects Using Drosophila melanogaster Model of the Essential Oil of Lantana montevidensis (Spreng) Briq. Medicina. 2019; 55(5):194. https://0-doi-org.brum.beds.ac.uk/10.3390/medicina55050194
Chicago/Turabian Stylede Oliveira, Maria Rayane Correia, Luiz Marivando Barros, Antônia Eliene Duarte, Maria Gabriely de Lima Silva, Bruno Anderson Fernandes da Silva, Anita Oliveira Brito Pereira Bezerra, Cícera Datiane Morais Oliveira Tintino, Victor Afonso Pereira de Oliveira, Aline Augusti Boligon, Jean Paul Kamdem, and et al. 2019. "GC-MS Chemical Characterization and In Vitro Evaluation of Antioxidant and Toxic Effects Using Drosophila melanogaster Model of the Essential Oil of Lantana montevidensis (Spreng) Briq." Medicina 55, no. 5: 194. https://0-doi-org.brum.beds.ac.uk/10.3390/medicina55050194






