Meibomian Gland Dysfunction: Intense Pulsed Light Therapy in Combination with Low-Level Light Therapy as Rescue Treatment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Procedures
2.2. Study Design
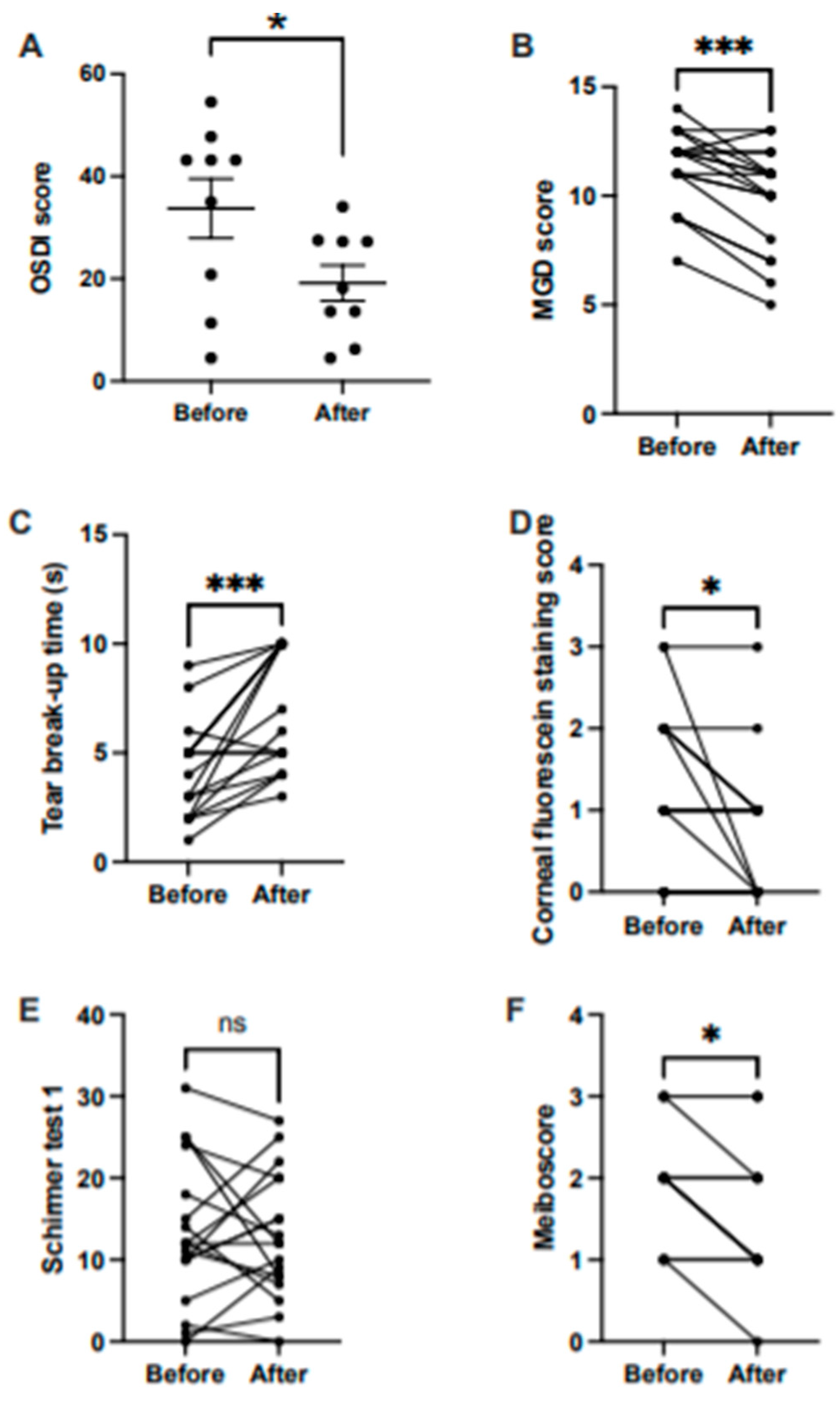
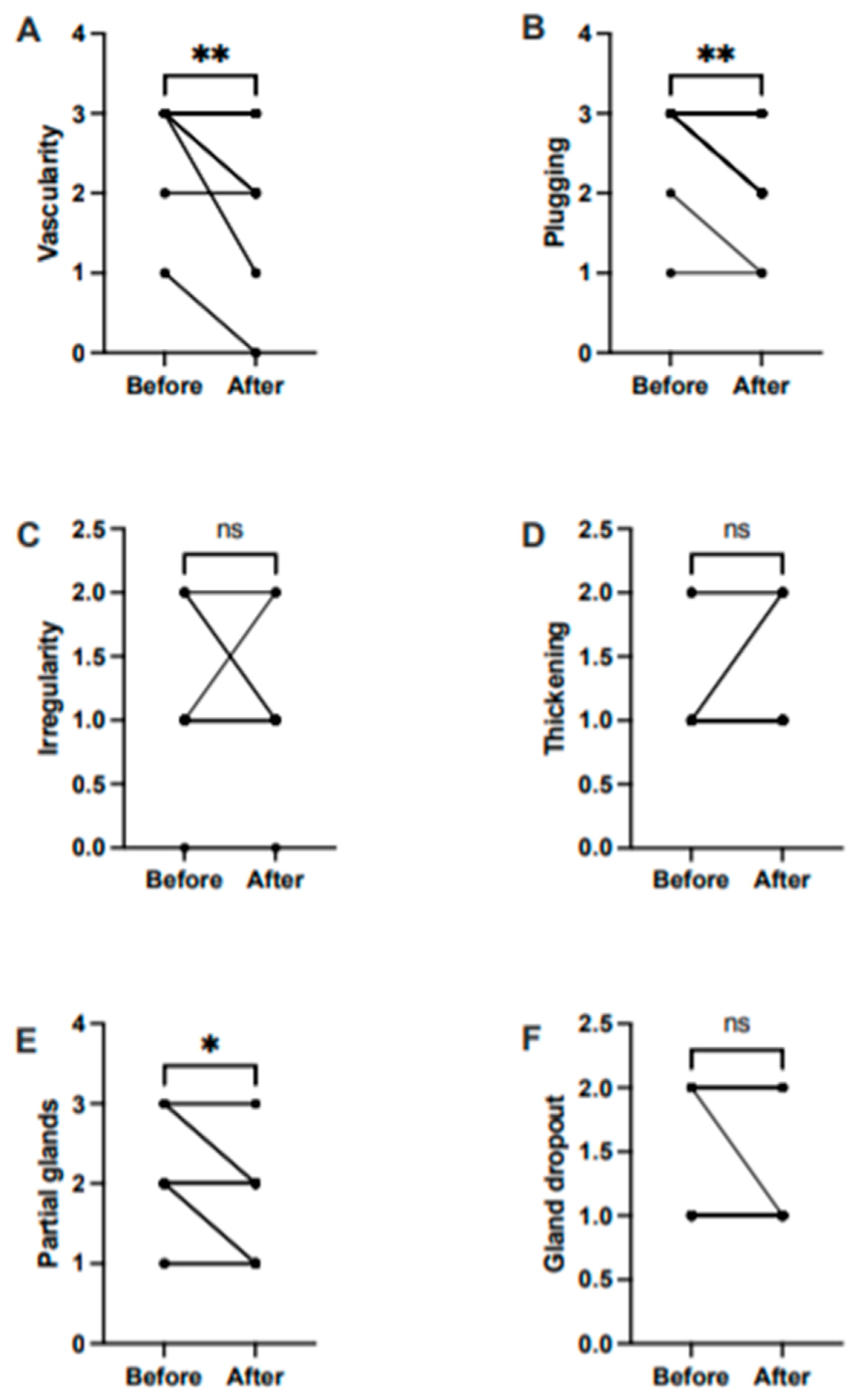
2.3. Outcomes
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lemp, M.A.; Crews, L.A.; Bron, A.J.; Foulks, G.N.; Sullivan, B.D. Distribution of aqueous-deficient and evaporative dry eye in a clinic-based patient cohort. Cornea 2012, 31, 472–478. [Google Scholar] [CrossRef]
- O’Neil, E.C.; Henderson, M.; Massaro-Giordano, M.; Bunya, V.Y. Advances in dry eye disease treatment. Curr. Opin. Ophthalmol. 2019, 30, 166–178. [Google Scholar] [CrossRef]
- Sabeti, S.; Kheirkhah, A.; Yin, J.; Dana, R. Management of meibomian gland dysfunction: A review. Surv. Ophthalmol. 2019, 65, 205–217. [Google Scholar] [CrossRef]
- Rong, B.; Tang, Y.; Liu, R.; Tu, P.; Qiao, J.; Song, W.; Yan, X. Long-term effects of intense pulsed light combined with meibomian gland expression in the treatment of meibomian gland dysfunction. Photomed. Laser Surg. 2018, 36, 562–567. [Google Scholar] [CrossRef]
- Wladis, E.J.; Aakalu, V.K.; Foster, J.A.; Freitag, S.K.; Sobel, R.K.; Tao, J.P.; Yen, M.T. Intense pulsed light for meibomian gland disease a report by the American Academy of Ophthalmology. Ophthalmology 2020, 127, 1227–1233. [Google Scholar] [CrossRef]
- Vora, G.K.; Gupta, P.K. Intense pulsed light therapy for the treatment of evaporative dry eye disease. Curr. Opin. Ophthalmol. 2015, 26, 314–318. [Google Scholar] [CrossRef]
- Kim, W.-S.; Calderhead, R.G. Is light-emitting diode phototherapy (LED-LLLT) really effective? Laser Ther. 2011, 20, 205–215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mahe, Y.F.; Cheniti, A.; Tacheau, C.; Antonelli, R.; Planard-Luong, L.; de Bernard, S.; Kanoun-Copy, L. Low-level light therapy downregulates scalp inflammatory biomarkers in men with androgenetic alopecia and boosts minoxidil 2% to bring a sustainable hair regrowth activity. Laser Surg. Med. 2021. Early View. [Google Scholar] [CrossRef] [PubMed]
- Stonecipher, K.; Abell, T.G.; Chotiner, B.; Chotiner, E.; Potvin, R. Combined low level light therapy and intense pulsed light therapy for the treatment of meibomian gland dysfunction. Clin. Ophthalmol. 2019, 13, 993–999. [Google Scholar] [CrossRef] [Green Version]
- Cote, S.; Zhang, A.C.; Ahmadzai, V.; Maleken, A.; Li, C.; Oppedisano, J.; Nair, K.; Busija, L.; Downie, L.E. Intense pulsed light (IPL) therapy for the treatment of meibomian gland dysfunction. Cochrane Database Syst. Rev. 2020, 3, CD013559. [Google Scholar] [CrossRef] [PubMed]
- Geerling, G.; Tauber, J.; Baudouin, C.; Goto, E.; Matsumoto, Y.; O’Brien, T.; Nichols, K.K. The international workshop on meibomian gland dysfunction: Report of the subcommittee on management and treatment of meibomian gland dysfunction. Invest. Ophthalmol. Vis. Sci. 2011, 52, 2050–2064. [Google Scholar] [CrossRef] [Green Version]
- Arita, R.; Minoura, I.; Morishige, N.; Shirakawa, R.; Fukuoka, S.; Asai, K.; Goto, T.; Imanaka, T.; Nakamura, M. Development of definitive and reliable grading scales for meibomian gland dysfunction. Am. J. Ophthalmol. 2016, 169, 125–137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prism Graphpad, 9th ed.; La Jolla, Graphpad Software Inc.: San Diego, CA, USA, 2021; Available online: www.graphpad.com (accessed on 28 April 2021).
- Tsubota, K. Short tear film breakup time–type dry eye. Invest. Ophthalmol. Vis. Sci. 2018, 59, DES64–DES70. [Google Scholar] [CrossRef] [PubMed]
- Toyos, R.; McGill, W.; Briscoe, D. Intense pulsed light treatment for dry eye disease due to meibomian gland dysfunction. A 3-year retrospective study. Photomed. Laser Surg. 2015, 33, 41–46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Albietz, J.M.; Schmid, K.L. Intense pulsed light treatment and meibomian gland expression for moderate to advanced meibomian gland dysfunction. Clin. Exp. Optom. 2018, 101, 23–33. [Google Scholar] [CrossRef] [Green Version]
- Mejía, L.F.; Gil, J.C.; Jaramillo, M. Terapia de luz pulsada intensa regulada: Un tratamiento complementario prometedor para la enfermedad de ojo seco. Arch. Soc. Española Oftalmol. 2019, 94, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.-J.; Lim, S.-C.; Kim, S.-G.; Kim, Y.-S.; Kang, S.-S.; Choi, S.-H.; Cho, Y.-S.; Bae, C.-S. Effect of low-level laser therapy on osteoarthropathy in rabbit. Vivo Athens Greece 2004, 18, 585–591. [Google Scholar]
- Mesquita-Ferrari, R.A.; Martins, M.D.; Silva, J.A.; Da Silva, T.D.; Piovesan, R.F.; Pavesi, V.C.S.; Bussadori, S.K.; Fernandes, K.P.S., Jr. Effects of low-level laser therapy on expression of TNF-α and TGF-β in skeletal muscle during the repair process. Laser Med. Sci. 2011, 26, 335–340. [Google Scholar] [CrossRef]
- Ji, Y.W.; Byun, Y.J.; Choi, W.; Jeong, E.; Kim, J.S.; Noh, H.; Lee, H.K. Neutralization of ocular surface TNF-α reduces ocular surface and lacrimal gland inflammation induced by in vivo dry eyea novel ocular surface TNF-α blocker for dry eye. Invest. Ophthalmol. Vis. Sci. 2013, 54, 7557–7566. [Google Scholar] [CrossRef] [Green Version]
- Aimbire, F.; Albertini, R.; Pacheco, M.T.T.; Castro-Faria-Neto, H.C.; Leonardo, P.S.L.M.; Iversen, V.V.; Bjordal, J.M. Low-level laser therapy induces dose-dependent reduction of TNF levels in acute inflammation. Photomed. Laser Surg. 2006, 24, 33–37. [Google Scholar] [CrossRef] [PubMed]
- Avci, P.; Gupta, A.; Sadasivam, M.; Vecchio, D.; Pam, Z.; Pam, N.; Hamblin, M.R. Low-level laser (light) therapy (LLLT) in skin: Stimulating, healing, restoring. Semin. Cutan. Med. Surg. 2013, 32, 41–52. [Google Scholar] [PubMed]
| Treatment | Number of Patients | Percentage |
|---|---|---|
| Eyelid hygiene with warm compresses | 9/9 | 100% |
| Artificial tears | 9/9 | 100% |
| Ongoing steroids | 1/9 | 11% |
| Past steroids * | 5/9 | 55% |
| Ongoing ciclosporine | 2/9 | 22% |
| Past ciclosporine * | 1/9 | 11% |
| Past fusidic acid * | 3/9 | 33% |
| Physiological serum | 2/9 | 22% |
| Autologous serum | 1/9 | 11% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Solomos, L.; Bouthour, W.; Malclès, A.; Thumann, G.; Massa, H. Meibomian Gland Dysfunction: Intense Pulsed Light Therapy in Combination with Low-Level Light Therapy as Rescue Treatment. Medicina 2021, 57, 619. https://0-doi-org.brum.beds.ac.uk/10.3390/medicina57060619
Solomos L, Bouthour W, Malclès A, Thumann G, Massa H. Meibomian Gland Dysfunction: Intense Pulsed Light Therapy in Combination with Low-Level Light Therapy as Rescue Treatment. Medicina. 2021; 57(6):619. https://0-doi-org.brum.beds.ac.uk/10.3390/medicina57060619
Chicago/Turabian StyleSolomos, Leonidas, Walid Bouthour, Ariane Malclès, Gabriele Thumann, and Horace Massa. 2021. "Meibomian Gland Dysfunction: Intense Pulsed Light Therapy in Combination with Low-Level Light Therapy as Rescue Treatment" Medicina 57, no. 6: 619. https://0-doi-org.brum.beds.ac.uk/10.3390/medicina57060619






