Ceramide Metabolism Enzymes—Therapeutic Targets against Cancer
Abstract
:1. Introduction
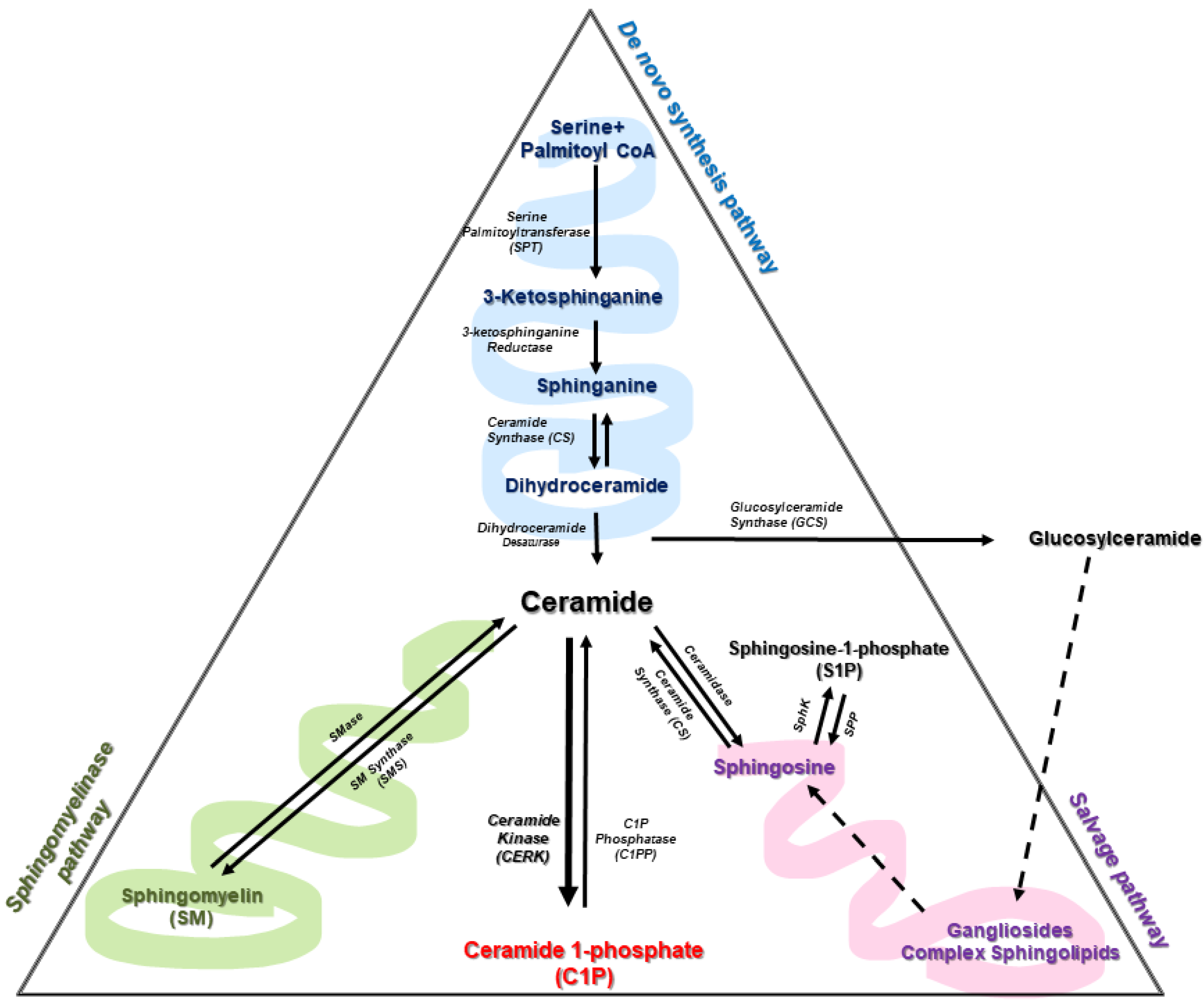
2. Sphingolipid Metabolism
3. Enzymes of Ceramide Metabolism
3.1. The De Novo Pathway of Ceramide Synthesis
3.1.1. Serine Palmitoyl Transferase (SPT)
3.1.2. Ceramide Synthase (CerS)
3.1.3. Dihydroceramide Desaturase (Des1, also Known as DEGS1)
3.2. Enzymes of the Sphingomyelinase Pathway
3.2.1. Sphingomyelinases (SMase)
3.2.2. Sphingomyelin Synthase (SMS)
3.3. Enzymes of the Salvage Pathway
3.3.1. Glucosylceramide Synthase (GCS)
3.3.2. Acid β-Glucosidase (β-GCase)
3.3.3. Ceramidase
3.4. Sphingosine Kinase (SphK)/Sphingosine 1-Phosphate Phosphate (SPP)
3.4.1. Sphingosine Kinase (SphK)
3.4.2. Sphingosine 1-Phosphate Phosphatase (SPP, also Known as SGPP)
3.5. Ceramide Kinase (Cerk), Phosphatases and CERT
4. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hannun, Y.A.; Loomis, C.R.; Merrill, A.H., Jr.; Bell, R.M. Sphingosine Inhibition of Protein Kinase C Activity and of Phorbol Dibutyrate Binding in Vitro and in Human Platelets. J. Biol. Chem. 1986, 261, 12604–12609. [Google Scholar] [CrossRef]
- Hannun, Y.A.; Obeid, L.M. Principles of Bioactive Lipid Signalling: Lessons from Sphingolipids. Nat. Rev. Mol. Cell Biol. 2008, 9, 139–150. [Google Scholar] [CrossRef]
- Gangoiti, P.; Camacho, L.; Arana, L.; Ouro, A.; Granado, M.H.; Brizuela, L.; Casas, J.; Fabrias, G.; Abad, J.L.; Delgado, A.; et al. Control of Metabolism and Signaling of Simple Bioactive Sphingolipids: Implications in Disease. Prog. Lipid Res. 2010, 49, 316–334. [Google Scholar] [CrossRef]
- Gomez-Larrauri, A.; Presa, N.; Dominguez-Herrera, A.; Ouro, A.; Trueba, M.; Gomez-Munoz, A. Role of Bioactive Sphingolipids in Physiology and Pathology. Essays Biochem. 2020, 64, 579–589. [Google Scholar] [CrossRef]
- Arana, L.; Gangoiti, P.; Ouro, A.; Trueba, M.; Gomez-Munoz, A.; Gómez-Muñoz, A. Ceramide and Ceramide 1-Phosphate in Health and Disease. Lipids Health Dis. 2010, 9, 15. [Google Scholar] [CrossRef] [Green Version]
- Gangoiti, P.; Granado, M.H.; Wei, S.; Kong, J.Y.; Steinbrecher, U.P.; Gómez-muñoz, A. Ceramide 1-Phosphate Stimulates Macrophage Proliferation through Activation of the PI3-Kinase/PKB, JNK and ERK1/2 Pathways. Cell. Signal. 2008, 20, 726–736. [Google Scholar] [CrossRef]
- Maceyka, M.; Payne, S.G.; Milstien, S.; Spiegel, S. Sphingosine Kinase, Sphingosine-1-Phosphate, and Apoptosis. Biochim. Biophys. Acta 2002, 1585, 193–201. [Google Scholar] [CrossRef]
- Taha, T.A.; Hannun, Y.A.; Obeid, L.M. Sphingosine Kinase: Biochemical and Cellular Regulation and Role in Disease. J. Biochem. Mol. Biol. 2006, 39, 113–131. [Google Scholar] [CrossRef]
- Simanshu, D.K.; Kamlekar, R.K.; Wijesinghe, D.S.; Zou, X.; Zhai, X.; Mishra, S.K.; Molotkovsky, J.G.; Malinina, L.; Hinchcliffe, E.H.; Chalfant, C.E.; et al. Non-Vesicular Trafficking by a Ceramide-1-Phosphate Transfer Protein Regulates Eicosanoids. Nature 2013, 500, 463–467. [Google Scholar] [CrossRef] [Green Version]
- Liang, H.; Yao, N.; Song, J.T.; Luo, S.; Lu, H.; Greenberg, J.T. Ceramides Modulate Programmed Cell Death in Plants. Genes Dev. 2003, 17, 2636–2641. [Google Scholar] [CrossRef] [Green Version]
- Boath, A.; Graf, C.; Lidome, E.; Ullrich, T.; Nussbaumer, P.; Bornancin, F. Regulation and Traffic of Ceramide 1-Phosphate Produced by Ceramide Kinase: Comparative Analysis to Glucosylceramide and Sphingomyelin. J. Biol. Chem. 2008, 283, 8517–8526. [Google Scholar] [CrossRef] [Green Version]
- Guan, X.L.; Mäser, P. Comparative Sphingolipidomics of Disease-Causing Trypanosomatids Reveal Unique Lifecycle- and Taxonomy-Specific Lipid Chemistries. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mielke, M.M.; Haughey, N.J.; Bandaru, V.V.R.; Zetterberg, H.; Blennow, K.; Andreasson, U.; Johnson, S.C.; Gleason, C.E.; Blazel, H.M.; Puglielli, L.; et al. Cerebrospinal Fluid Sphingolipids, β-Amyloid, and Tau in Adults at Risk for Alzheimer’s Disease. Neurobiol. Aging 2014, 35, 2486–2494. [Google Scholar] [CrossRef] [Green Version]
- van Kruining, D.; Luo, Q.; van Echten-Deckert, G.; Mielke, M.M.; Bowman, A.; Ellis, S.; Oliveira, T.G.; Martinez-Martinez, P. Sphingolipids as Prognostic Biomarkers of Neurodegeneration, Neuroinflammation, and Psychiatric Diseases and Their Emerging Role in Lipidomic Investigation Methods. Adv. Drug Deliv. Rev. 2020, 159, 232–244. [Google Scholar] [CrossRef]
- Griese, M.; Bonella, F.; Costabel, U.; de Blic, J.; Tran, N.-B.; Liebisch, G. Quantitative Lipidomics in Pulmonary Alveolar Proteinosis. Am. J. Respir. Crit. Care Med. 2019, 200, 881–887. [Google Scholar] [CrossRef]
- Kurek, K.; Mikłosz, A.; Łukaszuk, B.; Chabowski, A.; Górski, J.; Zendzian-Piotrowska, M. Inhibition of Ceramide de Novo Synthesis Ameliorates Diet Induced Skeletal Muscles Insulin Resistance. J. Diabetes Res. 2015, 2015. [Google Scholar] [CrossRef] [Green Version]
- Li, C.; Zhou, J.; Wang, A.; Li, N.; Chen, B.; Lei, M. Inhibition of Ceramide Synthesis Reverses Endothelial Dysfunction and Atherosclerosis in Streptozotocin-Induced Diabetic Rats. Diabetes Res. Clin. Pract. 2011, 93, 77–85. [Google Scholar] [CrossRef]
- Alberg, A.J.; Armeson, K.; Pierce, J.S.; Bielawski, J.; Bielawska, A.; Visvanathan, K.; Hill, E.G.; Ogretmen, B. Plasma Sphingolipids and Lung Cancer: A Population-Based, Nested Case-Control Study. Cancer Epidemiol. Biomark. Prev. 2013, 22, 1374–1382. [Google Scholar] [CrossRef] [Green Version]
- Ogretmen, B. Sphingolipid Metabolism in Cancer Signalling and Therapy. Nat. Rev. Cancer 2018, 18, 33–50. [Google Scholar] [CrossRef]
- Custodia, A.; Aramburu-Núñez, M.; Correa-Paz, C.; Posado-Fernández, A.; Gómez-Larrauri, A.; Castillo, J.; Gómez-Muñoz, A.; Sobrino, T.; Ouro, A. Ceramide Metabolism and Parkinson’s Disease-Therapeutic Targets. Biomolecules 2021, 11, 945. [Google Scholar] [CrossRef]
- Sheridan, M.; Ogretmen, B. The Role of Ceramide Metabolism and Signaling in the Regulation of Mitophagy and Cancer Therapy. Cancers 2021, 13, 2475. [Google Scholar] [CrossRef]
- Wilhelm, R.; Eckes, T.; Imre, G.; Kippenberger, S.; Meissner, M.; Thomas, D.; Trautmann, S.; Merlio, J.P.; Chevret, E.; Kaufmann, R.; et al. C6 Ceramide (D18: 1/6: 0) as a Novel Treatment of Cutaneous T Cell Lymphoma. Cancers 2021, 13, 270. [Google Scholar] [CrossRef]
- Xu, Y.; Pan, J.; Lin, Y.; Wu, Y.; Li, H.; Chen, Y. C6-Ceramide Induces Apoptosis in Lung Non-Small Cell Lung Cancer and Suppresses Brain Metastasis by Downregulating the PI3K/AKT/MTOR Signaling Pathway. Res. Sq. 2021. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, H.; Chen, T.; Wang, H.; Liang, X.; Zhang, Y.; Duan, J.; Qian, S.; Qiao, K.; Zhang, L.; et al. C24-Ceramide Drives Gallbladder Cancer Progression Through Directly Targeting Phosphatidylinositol 5-Phosphate 4-Kinase Type-2 Gamma to Facilitate Mammalian Target of Rapamycin Signaling Activation. Hepatology 2021, 73, 692–712. [Google Scholar] [CrossRef]
- Watters, R.J.; Kester, M.; Tran, M.A.; Loughran, T.P.; Liu, X. Development and Use of Ceramide Nanoliposomes in Cancer. Methods Enzymol. 2012, 508, 89–108. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Niu, Y.; Zhang, Z.; Gable, K.; Gupta, S.D.; Somashekarappa, N.; Han, G.; Zhao, H.; Myasnikov, A.G.; Kalathur, R.C.; et al. Structural Insights into the Regulation of Human Serine Palmitoyltransferase Complexes. Nat. Struct. Mol. Biol. 2021, 28, 240–248. [Google Scholar] [CrossRef]
- Kojima, T.; Asano, Y.; Kurasawa, O.; Hirata, Y.; Iwamura, N.; Wong, T.T.; Saito, B.; Tanaka, Y.; Arai, R.; Yonemori, K.; et al. Discovery of Novel Serine Palmitoyltransferase Inhibitors as Cancer Therapeutic Agents. Bioorg. Med. Chem. 2018, 26, 2452–2465. [Google Scholar] [CrossRef]
- Genin, M.J.; Gonzalez Valcarcel, I.C.; Holloway, W.G.; Lamar, J.; Mosior, M.; Hawkins, E.; Estridge, T.; Weidner, J.; Seng, T.; Yurek, D.; et al. Imidazopyridine and Pyrazolopiperidine Derivatives as Novel Inhibitors of Serine Palmitoyl Transferase. J. Med. Chem. 2016, 59, 5904–5910. [Google Scholar] [CrossRef]
- Pant, D.C.; Dorboz, I.; Schluter, A.; Fourcade, S.; Launay, N.; Joya, J.; Aguilera-Albesa, S.; Yoldi, M.E.; Casasnovas, C.; Willis, M.J.; et al. Loss of the Sphingolipid Desaturase DEGS1 Causes Hypomyelinating Leukodystrophy. J. Clin. Investig. 2019, 129, 1240–1256. [Google Scholar] [CrossRef] [PubMed]
- Karsai, G.; Kraft, F.; Haag, N.; Christoph Korenke, G.; Hänisch, B.; Othman, A.; Suriyanarayanan, S.; Steiner, R.; Knopp, C.; Mull, M.; et al. DEGS1-Associated Aberrant Sphingolipid Metabolism Impairs Nervous System Function in Humans. J. Clin. Investig. 2019, 129, 1229–1239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ohi, K.; Ursini, G.; Li, M.; Shin, J.H.; Ye, T.; Chen, Q.; Tao, R.; Kleinman, J.E.; Hyde, T.M.; Hashimoto, R.; et al. DEGS2 Polymorphism Associated with Cognition in Schizophrenia Is Associated with Gene Expression in Brain. Transl. Psychiatry 2015, 5, 550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Casasampere, M.; Ordoñez, Y.F.; Pou, A.; Casas, J. Inhibitors of Dihydroceramide Desaturase 1: Therapeutic Agents and Pharmacological Tools to Decipher the Role of Dihydroceramides in Cell Biology. Chem. Phys. Lipids 2016, 197, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Chaurasia, B.; Tippetts, T.S.; Monibas, R.M.; Liu, J.; Li, Y.; Wang, L.L.; Wilkerson, J.L.; Rufus Sweeney, C.; Pereira, R.F.; Sumida, D.H.; et al. Targeting a Ceramide Double Bond Improves Insulin Resistance and Hepatic Steatosis. Science 2019, 365, 386–392. [Google Scholar] [CrossRef] [PubMed]
- Jojima, K.; Edagawa, M.; Sawai, M.; Ohno, Y.; Kihara, A. Biosynthesis of the Anti-Lipid-Microdomain Sphingoid Base 4,14-Sphingadiene by the Ceramide Desaturase FADS3. FASEB J. 2020, 34, 3318–3335. [Google Scholar] [CrossRef] [PubMed]
- Karsai, G.; Lone, M.; Kutalik, Z.; Brenna, J.T.; Li, H.; Pan, D.; von Eckardstein, A.; Hornemann, T. FADS3 Is a Δ14Z Sphingoid Base Desaturase That Contributes to Gender Differences in the Human Plasma Sphingolipidome. J. Biol. Chem. 2020, 295, 1889–1897. [Google Scholar] [CrossRef] [Green Version]
- Cataldi, S.; Borrelli, A.; Ceccarini, M.R.; Nakashidze, I.; Codini, M.; Belov, O.; Ivanov, A.; Krasavin, E.; Ferri, I.; Conte, C.; et al. Acid and Neutral Sphingomyelinase Behavior in Radiation-Induced Liver Pyroptosis and in the Protective/Preventive Role of RMnSOD. Int. J. Mol. Sci. 2020, 21, 3281. [Google Scholar] [CrossRef]
- Goni, F.M.; Alonso, A. Sphingomyelinases: Enzymology and Membrane Activity. FEBS Lett. 2002, 531, 38–46. [Google Scholar] [CrossRef] [Green Version]
- Jenkins, R.W.; Canals, D.; Idkowiak-Baldys, J.; Simbari, F.; Roddy, P.; Perry, D.M.; Kitatani, K.; Luberto, C.; Hannun, Y.A. Regulated Secretion of Acid Sphingomyelinase: Implications for Selectivity of Ceramide Formation. J. Biol. Chem. 2010, 285, 35706–35718. [Google Scholar] [CrossRef] [Green Version]
- Becker, K.A.; Riethmuller, J.; Luth, A.; Doring, G.; Kleuser, B.; Gulbins, E. Acid Sphingomyelinase Inhibitors Normalize Pulmonary Ceramide and Inflammation in Cystic Fibrosis. Am. J. Respir. Cell Mol. Biol. 2010, 42, 716–724. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Muñoz, A.; Gangoiti, P.; Arana, L.; Ouro, A.; Rivera, I.G.; Ordoñez, M.; Trueba, M. New Insights on the Role of Ceramide 1-Phosphate in Inflammation. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2013, 1831, 1060–1066. [Google Scholar] [CrossRef]
- Morad, S.A.F.; Cabot, M.C. Ceramide-Orchestrated Signalling in Cancer Cells. Nat. Rev. Cancer 2012, 13, 51–65. [Google Scholar] [CrossRef]
- Kitatani, K.; Idkowiak-Baldys, J.; Hannun, Y.A. The Sphingolipid Salvage Pathway in Ceramide Metabolism and Signaling. Cell. Signal. 2008, 20, 1010–1018. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Levy, M.; Futerman, A.H. Mammalian Ceramide Synthases. IUBMB Life 2010, 62, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Verlekar, D.; Wei, S.J.; Cho, H.; Yang, S.; Kang, M.H. Ceramide Synthase-6 Confers Resistance to Chemotherapy by Binding to CD95/Fas in T-Cell Acute Lymphoblastic Leukemia. Cell Death Dis. 2018, 9, 1–14. [Google Scholar] [CrossRef]
- Park, J.W.; Park, W.J.; Futerman, A.H. Ceramide Synthases as Potential Targets for Therapeutic Intervention in Human Diseases. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2014, 1841, 671–681. [Google Scholar] [CrossRef]
- Kim, J.L.; Mestre, B.; Shin, S.-H.; Futerman, A.H. Ceramide Synthases: Reflections on the Impact of Dr. Lina M. Obeid. Cell. Signal. 2021, 82, 109958. [Google Scholar] [CrossRef] [PubMed]
- Raichur, S.; Wang, S.T.; Chan, P.W.; Li, Y.; Ching, J.; Chaurasia, B.; Dogra, S.; Öhman, M.K.; Takeda, K.; Sugii, S.; et al. CerS2 Haploinsufficiency Inhibits β-Oxidation and Confers Susceptibility to Diet-Induced Steatohepatitis and Insulin Resistance. Cell Metab. 2014, 20, 687–695. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Turpin, S.M.; Nicholls, H.T.; Willmes, D.M.; Mourier, A.; Brodesser, S.; Wunderlich, C.M.; Mauer, J.; Xu, E.; Hammerschmidt, P.; Brönneke, H.S.; et al. Obesity-Induced CerS6-Dependent C16:0 Ceramide Production Promotes Weight Gain and Glucose Intolerance. Cell Metab. 2014, 20, 678–686. [Google Scholar] [CrossRef] [Green Version]
- Coant, N.; Hannun, Y.A. Neutral Ceramidase: Advances in Mechanisms, Cell Regulation, and Roles in Cancer. Adv. Biol. Regul. 2019, 71, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Romiti, E.; Meacci, E.; Tani, M.; Nuti, F.; Farnararo, M.; Ito, M.; Bruni, P. Neutral/Alkaline and Acid Ceramidase Activities Are Actively Released by Murine Endothelial Cells. Biochem. Biophys. Res. Commun. 2000, 275, 746–751. [Google Scholar] [CrossRef]
- Gebai, A.; Gorelik, A.; Li, Z.; Illes, K.; Nagar, B. Structural Basis for the Activation of Acid Ceramidase. Nat. Commun. 2018, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.; Antwi Boasiako, P.; Mao, C. Alkaline Ceramidase Family: The First Two Decades. Cell. Signal. 2021, 78, 109860. [Google Scholar] [CrossRef]
- Saba, J.D. Fifty Years of Lyase and a Moment of Truth: Sphingosine Phosphate Lyase from Discovery to Disease. J. Lipid Res. 2019, 60, 456–463. [Google Scholar] [CrossRef] [Green Version]
- Boudker, O.; Futerman, A.H. Detection and Characterization of Ceramide-1-Phosphate Phosphatase Activity in Rat Liver Plasma Membrane. J. Biol. Chem. 1993, 268, 22150–22155. [Google Scholar] [CrossRef]
- Waggoner, D.W.; Gomez-Munoz, A.; Dewald, J.; Brindley, D.N. Phosphatidate Phosphohydrolase Catalyzes the Hydrolysis of Ceramide 1-Phosphate, Lysophosphatidate, and Sphingosine 1-Phosphate. J. Biol. Chem. 1996, 271, 16506–16509. [Google Scholar] [CrossRef] [Green Version]
- Campbell, K.N.; Tumlin, J.A. Protecting Podocytes: A Key Target for Therapy of Focal Segmental Glomerulosclerosis. Am. J. Nephrol. 2018, 47, 14–29. [Google Scholar] [CrossRef] [Green Version]
- Mallela, S.K.; Mitrofanova, A.; Merscher, S.; Fornoni, A. Regulation of the Amount of Ceramide-1-Phosphate Synthesized in Differentiated Human Podocytes. Biochim. Et Biophys. Acta Mol. Cell Biol. Lipids 2019, 1864, 158517. [Google Scholar] [CrossRef]
- Glaros, E.N.; Kim, W.S.; Wu, B.J.; Suarna, C.; Quinn, C.M.; Rye, K.A.; Stocker, R.; Jessup, W.; Garner, B. Inhibition of Atherosclerosis by the Serine Palmitoyl Transferase Inhibitor Myriocin Is Associated with Reduced Plasma Glycosphingolipid Concentration. Biochem. Pharmacol. 2007, 73, 1340–1346. [Google Scholar] [CrossRef]
- Kluepfel, D.; Bagli, J.; Baker, H.; Charest, M.P.; Kudelski, A.; Sehgal, S.N.; Vézina, C. Myriocin, a New Antifungal Antibiotic from Myriococcum Albomyces. J. Antibiot. 1972, 25, 109–115. [Google Scholar] [CrossRef] [Green Version]
- Riley, R.T.; Norred, W.P.; Wang, E.; Merrill, A.H. Alteration in Sphingolipid Metabolism: Bioassays for Fumonisin- and ISP-I-like Activity in Tissues, Cells and Other Matrices. Nat. Toxins 1999, 7, 407–414. [Google Scholar] [CrossRef]
- Lee, Y.S.; Choi, K.M.; Choi, M.H.; Ji, S.Y.; Lee, S.; Sin, D.M.; Oh, K.W.; Lee, Y.M.; Hong, J.T.; Yun, Y.P.; et al. Serine Palmitoyltransferase Inhibitor Myriocin Induces Growth Inhibition of B16F10 Melanoma Cells through G2/M Phase Arrest. Cell Prolif. 2011, 44, 320–329. [Google Scholar] [CrossRef] [PubMed]
- Sano, O.; Kazetani, K.; Adachi, R.; Kurasawa, O.; Kawamoto, T.; Iwata, H. Using a Biologically Annotated Library to Analyze the Anticancer Mechanism of Serine Palmitoyl Transferase (SPT) Inhibitors. FEBS Open Bio 2017, 7, 495–503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bernhart, E.; Damm, S.; Wintersperger, A.; Nusshold, C.; Brunner, A.M.; Plastira, I.; Rechberger, G.; Reicher, H.; Wadsack, C.; Zimmer, A.; et al. Interference with Distinct Steps of Sphingolipid Synthesis and Signaling Attenuates Proliferation of U87MG Glioma Cells. Biochem. Pharmacol. 2015, 96, 119–130. [Google Scholar] [CrossRef] [Green Version]
- Yaguchi, M.; Shibata, S.; Satomi, Y.; Hirayama, M.; Adachi, R.; Asano, Y.; Kojima, T.; Hirata, Y.; Mizutani, A.; Kiba, A.; et al. Antitumor Activity of a Novel and Orally Available Inhibitor of Serine Palmitoyltransferase. Biochem. Biophys. Res. Commun. 2017, 484, 493–500. [Google Scholar] [CrossRef]
- Maurer, B.J.; Metelitsa, L.S.; Seeger, R.C.; Cabot, M.C.; Patrick Reynolds, C. Increase of Ceramide and Induction of Mixed Apoptosis/Necrosis by N-(4- Hydroxyphenyl)-Retinamide in Neuroblastoma Cell Lines. J. Natl. Cancer Inst. 1999, 91, 1138–1146. [Google Scholar] [CrossRef]
- Villani, M.G.; Appierto, V.; Cavadini, E.; Bettiga, A.; Prinetti, A.; Clagett-Dame, M.; Curley, R.W.; Formelli, F. 4-Oxo-Fenretinide, a Recently Identified Fenretinide Metabolite, Induces Marked G2-M Cell Cycle Arrest and Apoptosis in Fenretinide-Sensitive and Fenretinide-Resistant Cell Lines. Cancer Res. 2006, 66, 3238–3247. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scarlatti, F.; Sala, G.; Somenzi, G.; Signorelli, P.; Sacchi, N.; Ghidoni, R. Resveratrol Induces Growth Inhibition and Apoptosis in Metastatic Breast Cancer Cells via de Novo Ceramide Signaling. FASEB J. 2003, 17, 2339–2341. [Google Scholar] [CrossRef]
- Hornemann, T.; Penno, A.; Rütti, M.F.; Ernst, D.; Kivrak-Pfiffner, F.; Rohrer, L.; von Eckardstein, A. The SPTLC3 Subunit of Serine Palmitoyltransferase Generates Short Chain Sphingoid Bases. J. Biol. Chem. 2009, 284, 26322–26330. [Google Scholar] [CrossRef] [Green Version]
- Lone, M.A.; Hülsmeier, A.J.; Saied, E.M.; Karsai, G.; Arenz, C.; von Eckardstein, A.; Hornemann, T. Subunit Composition of the Mammalian Serine-Palmitoyltransferase Defines the Spectrum of Straight and Methyl-Branched Long-Chain Bases. Proc. Natl. Acad. Sci. USA 2020, 117, 15591–15598. [Google Scholar] [CrossRef]
- Costa-Pinheiro, P.; Heher, A.; Raymond, M.H.; Jividen, K.; Shaw, J.J.; Paschal, B.M.; Walker, S.J.; Fox, T.E.; Kester, M. Role of SPTSSB-Regulated de Novo Sphingolipid Synthesis in Prostate Cancer Depends on Androgen Receptor Signaling. IScience 2020, 23, 101855. [Google Scholar] [CrossRef]
- Gable, K.; Slife, H.; Bacikova, D.; Monaghan, E.; Dunn, T.M. Tsc3p Is an 80-Amino Acid Protein Associated with Serine Palmitoyltransferase and Required for Optimal Enzyme Activity. J. Biol. Chem. 2000, 275, 7597–7603. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ren, J.; Saied, E.M.; Zhong, A.; Snider, J.; Ruiz, C.; Arenz, C.; Obeid, L.M.; Girnun, G.D.; Hannun, Y.A. Tsc3 Regulates SPT Amino Acid Choice in Saccharomyces Cerevisiae by Promoting Alanine in the Sphingolipid Pathway. J. Lipid Res. 2018, 59, 2126–2139. [Google Scholar] [CrossRef]
- Wang, Z.; Wen, L.; Zhu, F.; Wang, Y.; Xie, Q.; Chen, Z.; Li, Y. Overexpression of Ceramide Synthase 1 Increases C18-Ceramide and Leads to Lethal Autophagy in Human Glioma. Oncotarget 2017, 8, 104022–104036. [Google Scholar] [CrossRef]
- Moro, K.; Nagahashi, M.; Gabriel, E.; Takabe, K.; Wakai, T. Clinical Application of Ceramide in Cancer Treatment. Breast Cancer 2019, 26, 407–415. [Google Scholar] [CrossRef]
- Xia, Q.S.; Lu, F.E.; Wu, F.; Huang, Z.Y.; Dong, H.; Xu, L.J.; Gong, J. New Role for Ceramide in Hypoxia and Insulin Resistance. World J. Gastroenterol. 2020, 26, 2177–2186. [Google Scholar] [CrossRef]
- Crivelli, S.M.; Luo, Q.; Stevens, J.A.A.; Giovagnoni, C.; van Kruining, D.; Bode, G.; den Hoedt, S.; Hobo, B.; Scheithauer, A.-L.; Walter, J.; et al. CERTL Reduces C16 Ceramide, Amyloid-β Levels, and Inflammation in a Model of Alzheimer’s Disease. Alzheimer’s Res. Ther. 2021, 13, 45. [Google Scholar] [CrossRef]
- Turner, N.; Lim, X.Y.; Toop, H.D.; Osborne, B.; Brandon, A.E.; Taylor, E.N.; Fiveash, C.E.; Govindaraju, H.; Teo, J.D.; McEwen, H.P.; et al. A Selective Inhibitor of Ceramide Synthase 1 Reveals a Novel Role in Fat Metabolism. Nat. Commun. 2018, 9, 1–14. [Google Scholar] [CrossRef]
- Hartmann, D.; Lucks, J.; Fuchs, S.; Schiffmann, S.; Schreiber, Y.; Ferreirós, N.; Merkens, J.; Marschalek, R.; Geisslinger, G.; Grösch, S. Long Chain Ceramides and Very Long Chain Ceramides Have Opposite Effects on Human Breast and Colon Cancer Cell Growth. Int. J. Biochem. Cell Biol. 2012, 44, 620–628. [Google Scholar] [CrossRef] [PubMed]
- Schiffmann, S.; Hartmann, D.; Fuchs, S.; Birod, K.; Ferreirs, N.; Schreiber, Y.; Zivkovic, A.; Geisslinger, G.; Grösch, S.; Stark, H. Inhibitors of Specific Ceramide Synthases. Biochimie 2012, 94, 558–565. [Google Scholar] [CrossRef]
- Bose, R.; Verheij, M.; Haimovitz-friedman, A.; Scotto, K.; Fuks, Z.; Kolesnick, R. Ceramide Synthase Mediates Daunorubicin-Lnduced Apoptosis: An Alternative Mechanism for Generating Death Signals. Cell 1995, 82, 405–414. [Google Scholar] [CrossRef] [Green Version]
- Cabot, M.C.; Han, T.; Giuliano, A.E. The Multidrug Resistance Modulator SDZ PSC 833 Is a Potent Activator of Cellular Ceramide Formation. FEBS Lett. 1998, 431, 185–188. [Google Scholar] [CrossRef] [Green Version]
- Charles, A.G.; Han, T.Y.; Liu, Y.Y.; Hansen, N.; Giuliano, A.E.; Cabot, M.C. Taxol-Induced Ceramide Generation and Apoptosis in Human Breast Cancer Cells. Cancer Chemother. Pharmacol. 2001, 47, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Fekry, B.; Esmaeilniakooshkghazi, A.; Krupenko, S.A.; Krupenko, N.I. Ceramide Synthase 6 Is a Novel Target of Methotrexate Mediating Its Antiproliferative Effect in a P53-Dependent Manner. PLoS ONE 2016, 11, e0146618. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, P.; White-Gilbertson, S.; Beeson, G.; Beeson, C.; Ogretmen, B.; Norris, J.; Voelkel-Johnson, C. Ceramide Synthase 6 Maximizes P53 Function to Prevent Progeny Formation from Polyploid Giant Cancer Cells. Cancers 2021, 13, 2212. [Google Scholar] [CrossRef]
- Boppana, N.B.; Kodiha, M.; Stochaj, U.; Lin, H.S.; Haimovitz-Friedman, A.; Bielawska, A.; Bielawski, J.; Divine, G.W.; Boyd, J.A.; Korbelik, M.; et al. Ceramide Synthase Inhibitor Fumonisin B1 Inhibits Apoptotic Cell Death in SCC17B Human Head and Neck Squamous Carcinoma Cells after Pc4 Photosensitization. Photochem. Photobiol. Sci. 2014, 13, 1621–1627. [Google Scholar] [CrossRef] [Green Version]
- Holland, W.L.; Brozinick, J.T.; Wang, L.P.; Hawkins, E.D.; Sargent, K.M.; Liu, Y.; Narra, K.; Hoehn, K.L.; Knotts, T.A.; Siesky, A.; et al. Inhibition of Ceramide Synthesis Ameliorates Glucocorticoid-, Saturated-Fat-, and Obesity-Induced Insulin Resistance. Cell Metab. 2007, 5, 167–179. [Google Scholar] [CrossRef] [Green Version]
- Kraveka, J.M.; Li, L.; Szulc, Z.M.; Bielawski, J.; Ogretmen, B.; Hannun, Y.A.; Obeid, L.M.; Bielawska, A. Involvement of Dihydroceramide Desaturase in Cell Cycle Progression in Human Neuroblastoma Cells. J. Biol. Chem. 2007, 282, 16718–16728. [Google Scholar] [CrossRef] [Green Version]
- Casasampere, M.; Ordóñez, Y.F.; Casas, J.; Fabrias, G. Dihydroceramide Desaturase Inhibitors Induce Autophagy via Dihydroceramide-Dependent and Independent Mechanisms. Biochim. Biophys. Acta Gen. Subj. 2017, 1861, 264–275. [Google Scholar] [CrossRef]
- Hernández-Tiedra, S.; Fabriàs, G.; Dávila, D.; Salanueva, Í.J.; Casas, J.; Montes, L.R.; Antón, Z.; García-Taboada, E.; Salazar-Roa, M.; Lorente, M.; et al. Dihydroceramide Accumulation Mediates Cytotoxic Autophagy of Cancer Cells via Autolysosome Destabilization. Autophagy 2016, 12, 2213–2229. [Google Scholar] [CrossRef] [Green Version]
- Missiroli, S.; Bonora, M.; Patergnani, S.; Poletti, F.; Perrone, M.; Gafà, R.; Magri, E.; Raimondi, A.; Lanza, G.; Tacchetti, C.; et al. PML at Mitochondria-Associated Membranes Is Critical for the Repression of Autophagy and Cancer Development. Cell Rep. 2016, 16, 2415–2427. [Google Scholar] [CrossRef] [Green Version]
- Signorelli, P.; Munoz-Olaya, J.M.; Gagliostro, V.; Casas, J.; Ghidoni, R.; Fabriàs, G. Dihydroceramide Intracellular Increase in Response to Resveratrol Treatment Mediates Autophagy in Gastric Cancer Cells. Cancer Lett. 2009, 282, 238–243. [Google Scholar] [CrossRef]
- Ordóñez, Y.F.; González, J.; Bedia, C.; Casas, J.; Abad, J.L.; Delgado, A.; Fabrias, G. 3-Ketosphinganine Provokes the Accumulation of Dihydroshingolipids and Induces Autophagy in Cancer Cells. Mol. Biosyst. 2016, 12, 1166–1173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boppana, N.B.; Delor, J.S.; van Buren, E.; Bielawska, A.; Bielawski, J.; Pierce, J.S.; Korbelik, M.; Separovic, D. Enhanced Apoptotic Cancer Cell Killing after Foscan Photodynamic Therapy Combined with Fenretinide via de Novo Sphingolipid Biosynthesis Pathway. J. Photochem. Photobiol. B Biol. 2016, 159, 191–195. [Google Scholar] [CrossRef]
- Malhi, H.; Guicciardi, M.E.; Gores, G.J. Hepatocyte Death: A Clear and Present Danger. Physiol. Rev. 2010, 90, 1165–1194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clarke, C.J.; Cloessner, E.A.; Roddy, P.L.; Hannun, Y.A. Neutral Sphingomyelinase 2 (NSMase 2) Isthe Primary Neutral Sphingomyelinase Isoform Activated by Tumour Necrosis Factor-α in MCF-7 Cells. Biochem. J. 2011, 435, 381–390. [Google Scholar] [CrossRef] [Green Version]
- Shamseddine, A.; Clarke, C.; Carroll, B.; Airola, M.V.; Mohammed, S.; Rella, A.; Obeid, L.; Hannun, Y. P53-Dependent Upregulation of Neutral Sphingomyelinase-2: Role in Doxorubicin-Induced Growth Arrest. Cell Death Dis. 2015, 6, e1947. [Google Scholar] [CrossRef] [Green Version]
- Kachler, K.; Bailer, M.; Heim, L.; Schumacher, F.; Reichel, M.; Holzinger, C.D.; Trump, S.; Mittler, S.; Monti, J.; Trufa, D.I.; et al. Enhanced Acid Sphingomyelinase Activity Drives Immune Evasion and Tumor Growth in Non–Small Cell Lung Carcinoma. Cancer Res. 2017, 77, 5963–5976. [Google Scholar] [CrossRef] [Green Version]
- Carpinteiro, A.; Becker, K.A.; Japtok, L.; Hessler, G.; Keitsch, S.; Požgajovà, M.; Schmid, K.W.; Adams, C.; Müller, S.; Kleuser, B.; et al. Regulation of Hematogenous Tumor Metastasis by Acid Sphingomyelinase. EMBO Mol. Med. 2015, 7, 714–734. [Google Scholar] [CrossRef]
- Mizutani, N.; Omori, Y.; Kawamoto, Y.; Sobue, S.; Ichihara, M.; Suzuki, M.; Kyogashima, M.; Nakamura, M.; Tamiya-Koizumi, K.; Nozawa, Y.; et al. Resveratrol-Induced Transcriptional up-Regulation of ASMase (SMPD1) of Human Leukemia and Cancer Cells. Biochem. Biophys. Res. Commun. 2016, 470, 851–856. [Google Scholar] [CrossRef]
- Kornhuber, J.; Tripal, P.; Reichel, M.; Mühle, C.; Rhein, C.; Muehlbacher, M.; Groemer, T.W.; Gulbins, E. Functional Inhibitors of Acid Sphingomyelinase (FIASMAS): A Novel Pharmacological Group of Drugs with Broad Clinical Applications. Cell. Physiol. Biochem. 2010, 26, 9–20. [Google Scholar] [CrossRef]
- Carpinteiro, A.; Edwards, M.J.; Hoffmann, M.; Ahmad, S.A.; Fassbender, K.; Gulbins, E. Pharmacological Inhibition of Acid Sphingomyelinase Prevents Uptake of SARS-CoV-2 by Epithelial Cells. Cell Rep. Med. 2020, 1, 100142. [Google Scholar] [CrossRef]
- Schloer, S.; Brunotte, L.; Goretzko, J.; Mecate-Zambrano, A.; Korthals, N.; Gerke, V.; Ludwig, S.; Rescher, U. Targeting the Endolysosomal Host-SARS-CoV-2 Interface by Clinically Licensed Functional Inhibitors of Acid Sphingomyelinase (FIASMA) Including the Antidepressant Fluoxetine. Emerg. Microbes Infect. 2020, 9, 2245–2255. [Google Scholar] [CrossRef]
- le Corre, P.; Loas, G. Repurposing Functional Inhibitors of Acid Sphingomyelinase (Fiasmas): An Opportunity against SARS-CoV-2 Infection? J. Clin. Pharm. Ther. 2021, 1–7. [Google Scholar] [CrossRef]
- Horinouchi, K.; Erlich, S.; Perl, D.P.; Ferlinz, K.; Bisgaier, C.L.; Sandhoff, K.; Desnick, R.J.; Stewart, C.L.; Schuchman, E.H. Acid Sphingomyelinase Deficient Mice: A Model of Types A and B Niemann–Pick Disease. Nat. Genet. 1995, 10, 288–293. [Google Scholar] [CrossRef]
- Kuzu, O.F.; Gowda, R.; Noory, M.A.; Robertson, G.P. Modulating Cancer Cell Survival by Targeting Intracellular Cholesterol Transport. Br. J. Cancer 2017, 117, 513–524. [Google Scholar] [CrossRef] [Green Version]
- Naser, E.; Kadow, S.; Schumacher, F.; Mohamed, Z.H.; Kappe, C.; Hessler, G.; Pollmeier, B.; Kleuser, B.; Arenz, C.; Becker, K.A.; et al. Characterization of the Small Molecule ARC39, a Direct and Specific Inhibitor of Acid Sphingomyelinase in Vitro. J. Lipid Res. 2020, 61, 896–910. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prause, K.; Naseri, G.; Schumacher, F.; Kappe, C.; Kleuser, B.; Arenz, C. A Photocaged Inhibitor of Acid Sphingomyelinase. Chem. Commun. 2020, 56, 14885–14888. [Google Scholar] [CrossRef]
- Börtlein, C.; Schumacher, F.; Kleuser, B.; Dölken, L.; Avota, E. Role of Neutral Sphingomyelinase-2 (Nsm 2) in the Control of t Cell Plasma Membrane Lipid Composition and Cholesterol Homeostasis. Front. Cell Dev. Biol. 2019, 7, 226. [Google Scholar] [CrossRef] [Green Version]
- Mizrachi, A.; Ben-Aharon, I.; Li, H.; Bar-Joseph, H.; Bodden, C.; Hikri, E.; Popovtzer, A.; Shalgi, R.; Haimovitz-Friedman, A. Chemotherapy-Induced Acute Vascular Injury Involves Intracellular Generation of ROS via Activation of the Acid Sphingomyelinase Pathway. Cell. Signal. 2021, 82, 109969. [Google Scholar] [CrossRef]
- Rajagopal, C.; Harikumar, K.B. The Origin and Functions of Exosomes in Cancer. Front. Oncol. 2018, 8, 66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kunou, S.; Shimada, K.; Takai, M.; Sakamoto, A.; Aoki, T.; Hikita, T.; Kagaya, Y.; Iwamoto, E.; Sanada, M.; Shimada, S.; et al. Exosomes Secreted from Cancer-Associated Fibroblasts Elicit Anti-Pyrimidine Drug Resistance through Modulation of Its Transporter in Malignant Lymphoma. Oncogene 2021, 40, 3989–4003. [Google Scholar] [CrossRef]
- Li, S.; Yi, M.; Dong, B.; Jiao, Y.; Luo, S.; Wu, K. The Roles of Exosomes in Cancer Drug Resistance and Its Therapeutic Application. Clin. Transl. Med. 2020, 10, e257. [Google Scholar] [CrossRef]
- Kosaka, N.; Iguchi, H.; Hagiwara, K.; Yoshioka, Y.; Takeshita, F.; Ochiya, T. Neutral Sphingomyelinase 2 (NSMase2)-Dependent Exosomal Transfer of Angiogenic Micrornas Regulate Cancer Cell Metastasis. J. Biol. Chem. 2013, 288, 10849–10859. [Google Scholar] [CrossRef] [Green Version]
- Lin, M.; Liao, W.; Dong, M.; Zhu, R.; Xiao, J.; Sun, T.; Chen, Z.; Wu, B.; Jin, J. Exosomal Neutral Sphingomyelinase 1 Suppresses Hepatocellular Carcinoma via Decreasing the Ratio of Sphingomyelin/Ceramide. FEBS J. 2018, 285, 3835–3848. [Google Scholar] [CrossRef] [PubMed]
- Gurunathan, S.; Kang, M.H.; Jeyaraj, M.; Kim, J.H. Platinum Nanoparticles Enhance Exosome Release in Human Lung Epithelial Adenocarcinoma Cancer Cells (A549): Oxidative Stress and the Ceramide Pathway Are Key Players. Int. J. Nanomed. 2021, 16, 515–538. [Google Scholar] [CrossRef]
- Chen, Y.; Cao, Y. The Sphingomyelin Synthase Family: Proteins, Diseases, and Inhibitors. Biol. Chem. 2017, 398, 1319–1325. [Google Scholar] [CrossRef] [PubMed]
- Gulbins, A.; Schumacher, F.; Becker, K.A.; Wilker, B.; Soddemann, M.; Boldrin, F.; Müller, C.P.; Edwards, M.J.; Goodman, M.; Caldwell, C.C.; et al. Antidepressants Act by Inducing Autophagy Controlled by Sphingomyelin–Ceramide. Mol. Psychiatry 2018, 23, 2324–2346. [Google Scholar] [CrossRef] [Green Version]
- Barceló-Coblijn, G.; Martin, M.L.; de Almeida, R.F.M.; Noguera-Salvà, M.A.; Marcilla-Etxenike, A.; Guardiola-Serrano, F.; Lüth, A.; Kleuser, B.; Halver, J.E.; Escribá, P.v. Sphingomyelin and Sphingomyelin Synthase (SMS) in the Malignant Transformation of Glioma Cells and in 2-Hydroxyoleic Acid Therapy. Proc. Natl. Acad. Sci. USA 2011, 108, 19569–19574. [Google Scholar] [CrossRef] [Green Version]
- Deng, Y.; Hu, J.C.; He, S.H.; Lou, B.; Ding, T.B.; Yang, J.T.; Mo, M.G.; Ye, D.Y.; Zhou, L.; Jiang, X.C.; et al. Sphingomyelin Synthase 2 Facilitates M2-like Macrophage Polarization and Tumor Progression in a Mouse Model of Triple-Negative Breast Cancer. Acta Pharmacol. Sin. 2021, 42, 149–159. [Google Scholar] [CrossRef]
- Jing, F.; Jing, C.; Dai, X.; Zhou, G.; Di, S.; Bi, X.; Dai, T.; Qin, T.; Hong, L. Sphingomyelin Synthase 2 but Not Sphingomyelin Synthase 1 Is Upregulated in Ovarian Cancer and Involved in Migration, Growth and Survival via Different Mechanisms. Am. J. Transl. Res. 2021, 13, 4412–4421. [Google Scholar] [PubMed]
- Zheng, K.; Chen, Z.; Feng, H.; Chen, Y.; Zhang, C.; Yu, J.; Luo, Y.; Zhao, L.; Jiang, X.; Shi, F. Sphingomyelin Synthase 2 Promotes an Aggressive Breast Cancer Phenotype by Disrupting the Homoeostasis of Ceramide and Sphingomyelin. Cell Death Dis. 2019, 10, 1–11. [Google Scholar] [CrossRef]
- Bilal, F.; Montfort, A.; Gilhodes, J.; Garcia, V.; Riond, J.; Carpentier, S.; Filleron, T.; Colacios, C.; Levade, T.; Daher, A.; et al. Sphingomyelin Synthase 1 (SMS1) Downregulation Is Associated with Sphingolipid Reprogramming and a Worse Prognosis in Melanoma. Front. Pharmacol. 2019, 10, 443. [Google Scholar] [CrossRef] [Green Version]
- Song, M.; Zang, W.; Zhang, B.; Cao, J.; Yang, G. GCS Overexpression Is Associated with Multidrug Resistance of Human HCT-8 Colon Cancer Cells. J. Exp. Clin. Cancer Res. 2012, 31, 1–6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, J.; Wang, S.; Wang, C.; Kong, X.; Sun, P. Prognostic Value of Using Glucosylceramide Synthase and Cytochrome P450 Family 1 Subfamily A1 Expression Levels for Patients with Triple-negative Breast Cancer Following Neoadjuvant Chemotherapy. Exp. Ther. Med. 2021, 21, 1. [Google Scholar] [CrossRef]
- Lee, J.Y.; Nam, M.; Son, H.Y.; Hyun, K.; Jang, S.Y.; Kim, J.W.; Kim, M.W.; Jung, Y.; Jang, E.; Yoon, S.J.; et al. Polyunsaturated Fatty Acid Biosynthesis Pathway Determines Ferroptosis Sensitivity in Gastric Cancer. Proc. Natl. Acad. Sci. USA 2020, 117, 32433–32442. [Google Scholar] [CrossRef]
- Gouazé, V.; Liu, Y.Y.; Prickett, C.S.; Yu, J.Y.; Giuliano, A.E.; Cabot, M.C. Glucosylceramide Synthase Blockade Down-Regulates P-Glycoprotein and Resensitizes Multidrug-Resistant Breast Cancer Cells to Anticancer Drugs. Cancer Res. 2005, 65, 3861–3867. [Google Scholar] [CrossRef] [Green Version]
- Madigan, J.P.; Robey, R.W.; Poprawski, J.E.; Huang, H.; Clarke, C.J.; Gottesman, M.M.; Cabot, M.C.; Rosenberg, D.W. A Role for Ceramide Glycosylation in Resistance to Oxaliplatin in Colorectal Cancer. Exp. Cell Res. 2020, 388, 111860. [Google Scholar] [CrossRef]
- Snider, A.J.; Seeds, M.C.; Johnstone, L.; Snider, J.M.; Hallmark, B.; Dutta, R.; Franco, C.M.; Parks, J.S.; Bensen, J.T.; Broeckling, C.D.; et al. Identification of Plasma Glycosphingolipids as Potential Biomarkers for Prostate Cancer (PCA) Status. Biomolecules 2020, 10, 1393. [Google Scholar] [CrossRef]
- Hartwig, P.; Höglinger, D. The Glucosylceramide Synthase Inhibitor PDMP Causes Lysosomal Lipid Accumulation and MTOR Inactivation. Int. J. Mol. Sci. 2021, 22, 7065. [Google Scholar] [CrossRef]
- Ortega, R.A.; Wang, C.; Raymond, D.; Bryant, N.; Scherzer, C.R.; Thaler, A.; Alcalay, R.N.; West, A.B.; Mirelman, A.; Kuras, Y.; et al. Association of Dual LRRK2 G2019S and GBA Variations with Parkinson Disease Progression. JAMA Netw. Open 2021, 4, 215845. [Google Scholar] [CrossRef]
- Gan-Or, Z.; Liong, C.; Alcalay, R.N. GBA-Associated Parkinson’s Disease and Other Synucleinopathies. Curr. Neurol. Neurosci. Rep. 2018, 18, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Astudillo, L.; Therville, N.; Colacios, C.; Ségui, B.; Andrieu-Abadie, N.; Levade, T. Glucosylceramidases and Malignancies in Mammals. Biochimie 2016, 125, 267–280. [Google Scholar] [CrossRef] [PubMed]
- Sturchio, A.; Dwivedi, A.K.; Vizcarra, J.A.; Chirra, M.; Keeling, E.G.; Mata, I.F.; Kauffman, M.A.; Pandey, M.K.; Roviello, G.; Comi, C.; et al. Genetic Parkinsonisms and Cancer: A Systematic Review and Meta-Analysis. Rev. Neurosci. 2021, 32, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Xu, D.; Tong, X.; Shan, C. Inhibition of β-Glucosidase Overcomes Gastric Cancer Chemoresistance through Inducing Lysosomal Dysfunction. Clin. Res. Hepatol. Gastroenterol. 2021, 45, 101456. [Google Scholar] [CrossRef]
- Vijayan, Y.; Lankadasari, M.B.; Harikumar, K.B. Acid Ceramidase: A Novel Therapeutic Target in Cancer. Curr. Top. Med. Chem. 2019, 19, 1512–1520. [Google Scholar] [CrossRef]
- Saied, E.M.; Arenz, C. Inhibitors of Ceramidases. Chem. Phys. Lipids 2016, 197, 60–68. [Google Scholar] [CrossRef]
- Lai, M.; La Rocca, V.; Amato, R.; Freer, G.; Costa, M.; Spezia, P.G.; Quaranta, P.; Lombardo, G.; Piomelli, D.; Pistello, M. Ablation of Acid Ceramidase Impairs Autophagy and Mitochondria Activity in Melanoma Cells. Int. J. Mol. Sci. 2021, 22, 3247. [Google Scholar] [CrossRef]
- Lai, M.; Amato, R.; La Rocca, V.; Bilgin, M.; Freer, G.; Spezia, P.; Quaranta, P.; Piomelli, D.; Pistello, M. Acid Ceramidase Controls Apoptosis and Increases Autophagy in Human Melanoma Cells Treated with Doxorubicin. Sci. Rep. 2021, 11, 1–14. [Google Scholar] [CrossRef]
- Clifford, R.E.; Govindarajah, N.; Bowden, D.; Sutton, P.; Glenn, M.; Darvish-Damavandi, M.; Buczacki, S.; McDermott, U.; Szulc, Z.; Ogretmen, B.; et al. Targeting Acid Ceramidase to Improve the Radiosensitivity of Rectal Cancer. Cells 2020, 9, 2693. [Google Scholar] [CrossRef]
- White-Gilbertson, S.; Lu, P.; Jones, C.M.; Chiodini, S.; Hurley, D.; Das, A.; Delaney, J.R.; Norris, J.S.; Voelkel-Johnson, C. Tamoxifen Is a Candidate First-in-Class Inhibitor of Acid Ceramidase That Reduces Amitotic Division in Polyploid Giant Cancer Cells—Unrecognized Players in Tumorigenesis. Cancer Med. 2020, 9, 3142–3152. [Google Scholar] [CrossRef] [Green Version]
- Rothemund, M.; Bär, A.; Klatt, F.; Weidler, S.; Köhler, L.; Unverzagt, C.; Kuhn, C.D.; Schobert, R. N-Metallocenoylsphingosines as Targeted Ceramidase Inhibitors: Syntheses and Antitumoral Effects. Bioorg. Chem. 2020, 97, 103703. [Google Scholar] [CrossRef]
- Liu, B.; Xiao, J.; Dong, M.; Qiu, Z.; Jin, J. Human Alkaline Ceramidase 2 Promotes the Growth, Invasion, and Migration of Hepatocellular Carcinoma Cells via Sphingomyelin Phosphodiesterase Acid-like 3B. Cancer Sci. 2020, 111, 2259. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Huang, P.; Dai, H.; Li, Q.; Hu, L.; Peng, J.; Jiang, S.; Xu, Y.; Wu, Z.; Nie, H.; et al. TIMELESS Regulates Sphingolipid Metabolism and Tumor Cell Growth through Sp1/ACER2/S1P Axis in ER-Positive Breast Cancer. Cell Death Dis. 2020, 11, 1–14. [Google Scholar] [CrossRef]
- Yin, Y.; Xu, M.; Gao, J.; Li, M. Alkaline Ceramidase 3 Promotes Growth of Hepatocellular Carcinoma Cells via Regulating S1P/S1PR2/PI3K/AKT Signaling. Pathol. Res. Pract. 2018, 214, 1381–1387. [Google Scholar] [CrossRef]
- Steiner, R.; Saied, E.M.; Othman, A.; Arenz, C.; Maccarone, A.T.; Poad, B.L.J.; Blanksby, S.J.; von Eckardstein, A.; Hornemann, T. Elucidating the Chemical Structure of Native 1-Deoxysphingosine. J. Lipid Res. 2016, 57, 1194–1203. [Google Scholar] [CrossRef] [Green Version]
- Saied, E.M.; Le, T.L.S.; Hornemann, T.; Arenz, C. Synthesis and Characterization of Some Atypical Sphingoid Bases. Bioorg. Med. Chem. 2018, 26, 4047–4057. [Google Scholar] [CrossRef]
- Bielsa, N.; Casasampere, M.; Aseeri, M.; Casas, J.; Delgado, A.; Abad, J.L.; Fabriàs, G. Discovery of Deoxyceramide Analogs as Highly Selective ACER3 Inhibitors in Live Cells. Eur. J. Med. Chem. 2021, 216, 113296. [Google Scholar] [CrossRef]
- Vasiliauskaité-Brooks, I.; Healey, R.D.; Rochaix, P.; Saint-Paul, J.; Sounier, R.; Grison, C.; Waltrich-Augusto, T.; Fortier, M.; Hoh, F.; Saied, E.M.; et al. Structure of a Human Intramembrane Ceramidase Explains Enzymatic Dysfunction Found in Leukodystrophy. Nat. Commun. 2018, 9, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Sakamoto, W.; Coant, N.; Canals, D.; Obeid, L.M.; Hannun, Y.A. Functions of Neutral Ceramidase in the Golgi Apparatus. J. Lipid Res. 2018, 59, 2116–2125. [Google Scholar] [CrossRef] [Green Version]
- García-Barros, M.; Coant, N.; Kawamori, T.; Wada, M.; Snider, A.J.; Truman, J.-P.; Wu, B.X.; Furuya, H.; Clarke, C.J.; Bialkowska, A.B.; et al. Role of Neutral Ceramidase in Colon Cancer. FASEB J. 2016, 30, 4159. [Google Scholar] [CrossRef] [Green Version]
- Coant, N.; García-Barros, M.; Zhang, Q.; Obeid, L.M.; Hannun, Y.A. AKT as a Key Target for Growth Promoting Functions of Neutral Ceramidase in Colon Cancer Cells. Oncogene 2018, 37, 3852–3863. [Google Scholar] [CrossRef] [PubMed]
- Pfeiler, G.; Hudelist, G.; Wülfing, P.; Mattsson, B.; Königsberg, R.; Kubista, E.; Singer, C.F. Impact of AdipoR1 Expression on Breast Cancer Development. Gynecol. Oncol. 2010, 117, 134–138. [Google Scholar] [CrossRef]
- Ye, J.; Jiang, L.; Wu, C.; Liu, A.; Mao, S.; Ge, L. Three ADIPOR1 Polymorphisms and Cancer Risk: A Meta-Analysis of Case-Control Studies. PLoS ONE 2015, 10, e0127253. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kordafshari, M.; Nourian, M.; Mehrvar, N.; Jalaeikhoo, H.; Etemadi, A.; Khoshdel, A.R.; Idris, M.G.; Iravani, S.; Mehrvar, A. Expression of AdipoR1 and AdipoR2 and Serum Level of Adiponectin in Gastric Cancer. Gastrointest. Tumors 2020, 7, 103–109. [Google Scholar] [CrossRef]
- Bui, K.C.; Nguyen, M.L.T.; Barat, S.; Chen, X.; Bhuria, V.; Xing, J.; Nguyen, L.T.; Le, H.S.; Velavan, T.P.; Wilkens, L.; et al. Effect of AdipoR Agonist in Cholangiocarcinoma. J. Clin. Oncol. 2018, 36, 323. [Google Scholar] [CrossRef]
- Philp, L.K.; Rockstroh, A.; Lehman, M.; Sadowski, M.C.; Bartonicek, N.; Wade, J.D.; Otvos, L.; Nelson, C.C. Adiponectin Receptor Activation Inhibits Prostate Cancer Xenograft Growth. Endocr. Relat. Cancer 2020, 27, 711–729. [Google Scholar] [CrossRef]
- Akimoto, M.; Maruyama, R.; Kawabata, Y.; Tajima, Y.; Takenaga, K. Antidiabetic Adiponectin Receptor Agonist AdipoRon Suppresses Tumour Growth of Pancreatic Cancer by Inducing RIPK1/ERK-Dependent Necroptosis. Cell Death Dis. 2018, 9, 1–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vasiliauskaité-Brooks, I.; Sounier, R.; Rochaix, P.; Bellot, G.; Fortier, M.; Hoh, F.; de Colibus, L.; Bechara, C.; Saied, E.M.; Arenz, C.; et al. Structural Insights into Adiponectin Receptors Suggest Ceramidase Activity. Nature 2017, 544, 120–123. [Google Scholar] [CrossRef] [Green Version]
- Lee, M.J.; van Brocklyn, J.R.; Thangada, S.; Liu, C.H.; Hand, A.R.; Menzeleev, R.; Spiegel, S.; Hla, T. Sphingosine-1-Phosphate as a Ligand for the G Protein-Coupled Receptor EDG-1. Science 1998, 279, 1552–1555. [Google Scholar] [CrossRef] [PubMed]
- MacEyka, M.; Spiegel, S. Sphingolipid Metabolites in Inflammatory Disease. Nature 2014, 510, 58–67. [Google Scholar] [CrossRef] [Green Version]
- Ratajczak, M.Z.; Lee, H.; Wysoczynski, M.; Wan, W.; Marlicz, W.; Laughlin, M.J.; Kucia, M.; Janowska-Wieczorek, A.; Ratajczak, J. Novel insight into stem cell mobilization-plasma sphingosine-1-phosphate is a major chemoattractant that directs the egress of hematopoietic stem progenitor cells from the bone marrow and its level in peripheral blood increases during mobilization due to activation of complement cascade/membrane attack complex. Leukemia 2010, 24, 976–985. [Google Scholar]
- Spiegel, S.; Milstien, S. The Outs and the Ins of Sphingosine-1-Phosphate in Immunity. Nat. Rev. Immunol. 2011, 11, 403–415. [Google Scholar] [CrossRef]
- Fyrst, H.; Saba, J.D. An Update on Sphingosine-1-Phosphate and Other Sphingolipid Mediators. Nat. Chem. Biol. 2010, 6, 489–497. [Google Scholar] [CrossRef]
- Shida, D.; Takabe, K.; Kapitonov, D.; Milstien, S.; Spiegel, S. Targeting SphK1 as a New Strategy against Cancer. Curr. Drug Targets 2008, 9, 662–673. [Google Scholar] [CrossRef] [Green Version]
- Pyne, N.J.; Tonelli, F.; Lim, K.G.; Long, J.S.; Edwards, J.; Pyne, S. Sphingosine 1-Phosphate Signalling in Cancer. Biochem. Soc. Trans. 2012, 40, 94–100. [Google Scholar] [CrossRef]
- Peters, S.L.; Alewijnse, A.E. Sphingosine-1-Phosphate Signaling in the Cardiovascular System. Curr. Opin. Pharmacol. 2007, 7, 186–192. [Google Scholar] [CrossRef]
- Pyne, S.; Pyne, N.J. Sphingosine 1-Phosphate Signalling in Mammalian Cells. Biochem. J. 2000, 349, 385–402. [Google Scholar] [CrossRef]
- Donati, C.; Meacci, E.; Nuti, F.; Becciolini, L.; Farnararo, M.; Bruni, P. Sphingosine 1-Phosphate Regulates Myogenic Differentiation: A Major Role for S1P2 Receptor. FASEB J. 2005, 19, 449–451. [Google Scholar] [CrossRef]
- Bruni, P.; Donati, C. Pleiotropic Effects of Sphingolipids in Skeletal Muscle. Cell. Mol. Life Sci. 2008, 65, 3725–3736. [Google Scholar] [CrossRef]
- Maceyka, M.; Harikumar, K.B.; Milstien, S.; Spiegel, S. Sphingosine-1-Phosphate Signaling and Its Role in Disease. Trends Cell Biol. 2012, 22, 50–60. [Google Scholar] [CrossRef] [Green Version]
- Maceyka, M.; Rohrbach, T.; Milstien, S.; Spiegel, S. Role of Sphingosine Kinase 1 and Sphingosine-1-Phosphate Axis in Hepatocellular Carcinoma. Handb. Exp. Pharmacol. 2020, 259, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Hammad, S.M.; Crellin, H.G.; Wu, B.X.; Melton, J.; Anelli, V.; Obeid, L.M. Dual and Distinct Roles for Sphingosine Kinase 1 and Sphingosine 1 Phosphate in the Response to Inflammatory Stimuli in RAW Macrophages. Prostaglandins Other Lipid Mediat. 2008, 85, 107–114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Albinet, V.; Bats, M.L.; Huwiler, A.; Rochaix, P.; Chevreau, C.; Ségui, B.; Levade, T.; Andrieu-Abadie, N. Dual Role of Sphingosine Kinase-1 in Promoting the Differentiation of Dermal Fibroblasts and the Dissemination of Melanoma Cells. Br. Dent. J. 2014, 217, 3364–3373. [Google Scholar] [CrossRef]
- Simon, J.; Ouro, A.; Ala-Ibanibo, L.; Presa, N.; Delgado, T.C.; Martínez-Chantar, M.L. Sphingolipids in Non-Alcoholic Fatty Liver Disease and Hepatocellular Carcinoma: Ceramide Turnover. Int. J. Mol. Sci. 2020, 21, 40. [Google Scholar] [CrossRef] [Green Version]
- Schneider, G.; Sellers, Z.P.; Bujko, K.; Kakar, S.S.; Kucia, M.; Ratajczak, M.Z. Novel Pleiotropic Effects of Bioactive Phospholipids in Human Lung Cancer Metastasis. Oncotarget 2017, 8, 58247–58263. [Google Scholar] [CrossRef] [Green Version]
- Ushitora, Y.; Tashiro, H.; Ogawa, T.; Tanimoto, Y.; Kuroda, S.; Kobayashi, T.; Miyata, Y.; Itamoto, T.; Asahara, T.; Ohdan, H. Suppression of Hepatocellular Carcinoma Recurrence after Rat Liver Transplantation by FTY720, a Sphingosine-1-Phosphate Analog. Transplantation 2009, 88, 980–986. [Google Scholar] [CrossRef]
- Budkowska, M.; Ostrycharz, E.; Wojtowicz, A.; Marcinowska, Z.; Woźniak, J.; Ratajczak, M.Z.; Dołęgowska, B. A Circadian Rhythm in Both Complement Cascade (ComC) Activation and Sphingosine-1-Phosphate (S1P) Levels in Human Peripheral Blood Supports a Role for the ComC-S1P Axis in Circadian Changes in the Number of Stem Cells Circulating in Peripheral Blood. Stem Cell Rev. Rep. 2015, 14, 677–685. [Google Scholar] [CrossRef] [Green Version]
- Sukocheva, O.A.; Furuya, H.; Ng, M.L.; Friedemann, M.; Menschikowski, M.; Tarasov, V.V.; Chubarev, V.N.; Klochkov, S.G.; Neganova, M.E.; Mangoni, A.A.; et al. Sphingosine Kinase and Sphingosine-1-Phosphate Receptor Signaling Pathway in Inflammatory Gastrointestinal Disease and Cancers: A Novel Therapeutic Target. Pharmacol. Ther. 2020, 207, 107464. [Google Scholar] [CrossRef]
- Liu, H.; Toman, R.E.; Goparaju, S.K.; Maceyka, M.; Nava, V.E.; Sankala, H.; Payner, S.G.; Bektas, M.; Ishii, I.; Chun, J.; et al. Sphingosine Kinase Type 2 Is a Putative BH3-Only Protein That Induces Apoptosis. J. Biol. Chem. 2003, 278, 40330–40336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Igarashi, N.; Okada, T.; Hayashi, S.; Fujita, T.; Jahangeer, S.; Nakamura, S.I. Sphingosine Kinase 2 Is a Nuclear Protein and Inhibits DNA Synthesis. J. Biol. Chem. 2003, 278, 46832–46839. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Okada, T.; Ding, G.; Sonoda, H.; Kajimoto, T.; Haga, Y.; Khosrowbeygi, A.; Gao, S.; Miwa, N.; Jahangeer, S.; Nakamura, S.I. Involvement of N-Terminal-Extended Form of Sphingosine Kinase 2 in Serum-Dependent Regulation of Cell Proliferation and Apoptosis. J. Biol. Chem. 2005, 280, 36318–36325. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sankala, H.M.; Hait, N.C.; Paugh, S.W.; Shida, D.; Lépine, S.; Elmore, L.W.; Dent, P.; Milstien, S.; Spiegel, S. Involvement of Sphingosine Kinase 2 in P53-Independent Induction of P21 by the Chemotherapeutic Drug Doxorubicin. Cancer Res. 2007, 67, 10466–10474. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, D.; Zhu, H.; Wang, C.; Zhao, W.; Liu, G.; Bao, G.; Cui, D.; Fan, J.; Wang, F.; Jin, H.; et al. SphK2 Over-Expression Promotes Osteosarcoma Cell Growth. Oncotarget 2017, 8, 105525–105535. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Brooklyn, J.R.; Jackson, C.A.; Pearl, D.K.; Kotur, M.S.; Snyder, P.J.; Prior, T.W. Sphingosine Kinase-1 Expression Correlates with Poor Survival of Patients with Glioblastoma Multiforme: Roles of Sphingosine Kinase Isoforms in Growth of Glioblastoma Cell Lines. J. Neuropathol. Exp. Neurol. 2005, 64, 695–705. [Google Scholar] [CrossRef]
- Yang, J.; Yang, C.; Zhang, S.; Mei, Z.; Shi, M.; Sun, S.; Shi, L.; Wang, Z.; Wang, Y.; Li, Z.; et al. ABC294640, a Sphingosine Kinase 2 Inhibitor, Enhances the Antitumor Effects of TRAIL in Non-Small Cell Lung Cancer. Cancer Biol. Ther. 2015, 16, 1194–1204. [Google Scholar] [CrossRef] [Green Version]
- Beider, K.; Rosenberg, E.; Bitner, H.; Shimoni, A.; Leiba, M.; Koren-Michowitz, M.; Ribakovsky, E.; Klein, S.; Olam, D.; Weiss, L.; et al. The Sphingosine-1-Phosphate Modulator FTY720 Targets Multiple Myeloma via the CXCR4/CXCL12 Pathway. Clinical Cancer Res. 2017, 23, 1733–1747. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- White, C.; Alshaker, H.; Cooper, C.; Winkler, M.; Pchejetski, D. The Emerging Role of FTY720 (Fingolimod) in Cancer Treatment. Oncotarget 2016, 7, 23106–23127. [Google Scholar] [CrossRef] [Green Version]
- Tonelli, F.; Lim, K.G.; Loveridge, C.; Long, J.; Pitson, S.M.; Tigyi, G.; Bittman, R.; Pyne, S.; Pyne, N.J. FTY720 and (S)-FTY720 Vinylphosphonate Inhibit Sphingosine Kinase 1 and Promote Its Proteasomal Degradation in Human Pulmonary Artery Smooth Muscle, Breast Cancer and Androgen-Independent Prostate Cancer Cells. Cell. Signal. 2010, 22, 1536–1542. [Google Scholar] [CrossRef]
- Ouro, A.; Arana, L.; Gangoiti, P.; Rivera, I.G.; Ordoñez, M.; Trueba, M.; Lankalapalli, R.S.; Bittman, R.; Gomez-Muñoz, A. Ceramide 1-Phosphate Stimulates Glucose Uptake in Macrophages. Cell. Signal. 2013, 25, 786–795. [Google Scholar] [CrossRef] [Green Version]
- Halmer, R.; Walter, S.; Faßbender, K. Sphingolipids: Important Players in Multiple Sclerosis. Cell. Physiol. Biochem. 2014, 34, 111–118. [Google Scholar] [CrossRef]
- Brinkmann, V. FTY720 (Fingolimod) in Multiple Sclerosis: Therapeutic Effects in the Immune and the Central Nervous System. Br. J. Pharmacol. 2009, 158, 1173–1182. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Copland, D.A.; Liu, J.; Schewitz-Bowers, L.P.; Brinkmann, V.; Anderson, K.; Nicholson, L.B.; Dick, A.D. Therapeutic Dosing of Fingolimod (FTY720) Prevents Cell Infiltration, Rapidly Suppresses Ocular Inflammation, and Maintains the Blood-Ocular Barrier. Am. J. Pathol. 2012, 180, 672–681. [Google Scholar] [CrossRef] [Green Version]
- Epstein, D.J.; Dunn, J.; Deresinski, S. Infectious Complications of Multiple Sclerosis Therapies: Implications for Screening, Prophylaxis, and Management. Open Forum Infect. Dis. 2018, 5, ofy174. [Google Scholar] [CrossRef] [Green Version]
- Kapitonov, D.; Allegood, J.C.; Mitchell, C.; Hait, N.C.; Almenara, J.A.; Adams, J.K.; Zipkin, R.E.; Dent, P.; Kordula, T.; Milstien, S.; et al. Targeting Sphingosine Kinase 1 Inhibits Akt Signaling, Induces Apoptosis, and Suppresses Growth of Human Glioblastoma Cells and Xenografts. Cancer Res. 2009, 69, 6915–6923. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, K.; Pan, Q.; Gao, Y.; Yang, X.; Wang, S.; Peppelenbosch, M.P.; Kong, X. DMS Triggers Apoptosis Associated with the Inhibition of SPHK1/NF-ΚB Activation and Increase in Intracellular Ca2+ Concentration in Human Cancer Cells. Int. J. Mol. Med. 2014, 33, 17–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hii, L.W.; Chung, F.F.L.; Mai, C.W.; Yee, Z.Y.; Chan, H.H.; Raja, V.J.; Dephoure, N.E.; Pyne, N.J.; Pyne, S.; Leong, C.-O. Sphingosine Kinase 1 Regulates the Survival of Breast Cancer Stem Cells and Non-Stem Breast Cancer Cells by Suppression of STAT1. Cells 2020, 9, 886. [Google Scholar] [CrossRef] [Green Version]
- Trayssac, M.; Clarke, C.J.; Stith, J.L.; Snider, J.M.; Newen, N.; Gault, C.R.; Hannun, Y.A.; Obeid, L.M. Targeting Sphingosine Kinase 1 (SK1) Enhances Oncogene-Induced Senescence through Ceramide Synthase 2 (CerS2)-Mediated Generation of Very-Long-Chain Ceramides. Cell Death Dis. 2021, 12, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Cartier, A.; Hla, T. Sphingosine 1-Phosphate: Lipid Signaling in Pathology and Therapy. Science 2019, 366. [Google Scholar] [CrossRef] [PubMed]
- Pyne, N.J.; Pyne, S. Recent Advances in the Role of Sphingosine 1-phosphate in Cancer. FEBS Lett. 2020, 594, 3583–3601. [Google Scholar] [CrossRef]
- Pyne, S.; Kong, K.C.; Darroch, P.I. Lysophosphatidic Acid and Sphingosine 1-Phosphate Biology: The Role of Lipid Phosphate Phosphatases. Semin. Cell Dev. Biol. 2004, 15, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Brindley, D.N.; English, D.; Pilquil, C.; Buri, K.; Ling, Z.C. Lipid Phosphate Phosphatases Regulate Signal Transduction through Glycerolipids and Sphingolipids. Biochim. Biophys. Acta 2002, 1582, 33–44. [Google Scholar] [CrossRef]
- Mandala, S.M. Sphingosine-1-Phosphate Phosphatases. Prostaglandins Other Lipid Mediat. 2001, 64, 143–156. [Google Scholar] [CrossRef]
- Allende, M.L.; Sipe, L.M.; Tuymetova, G.; Wilson-Henjum, K.L.; Chen, W.; Proia, R.L. Sphingosine-1-Phosphate Phosphatase 1 Regulates Keratinocyte Differentiation and Epidermal Homeostasis. J. Biol. Chem. 2013, 288, 18381–18391. [Google Scholar] [CrossRef] [Green Version]
- Huang, W.; Liang, J.; Nagahashi, M.; Avni, D.; Yamada, A.; Maceyka, M.; Wolen, A.R.; Kordula, T.; Milstien, S.; Takabe, K.; et al. Sphingosine-1-phosphate Phosphatase 2 Promotes Disruption of Mucosal Integrity, and Contributes to Ulcerative Colitis in Mice and Humans. FASEB J. 2016, 30, 2945–2958. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taguchi, Y.; Allende, M.L.; Mizukami, H.; Cook, E.K.; Gavrilova, O.; Tuymetova, G.; Clarke, B.A.; Chen, W.; Olivera, A.; Proia, R.L. Sphingosine-1-Phosphate Phosphatase 2 Regulates Pancreatic Islet-Cell Endoplasmic Reticulum Stress and Proliferation. J. Biol. Chem. 2016, 291, 12029–12038. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, X.Y.; Li, L.; Wang, X.H.; Wen, X.Z.; Ji, K.; Ye, L.; Cai, J.; Jiang, W.G.; Ji, J.F. Inhibition of Sphingosine-1-Phosphate Phosphatase 1 Promotes Cancer Cells Migration in Gastric Cancer: Clinical Implications. Oncol. Rep. 2015, 34, 1977–1987. [Google Scholar] [CrossRef] [PubMed]
- le Stunff, H.; Mikami, A.; Giussani, P.; Hobson, J.P.; Jolly, P.S.; Milstien, S.; Spiegel, S. Role of Sphingosine-1-Phosphate Phosphatase 1 in Epidermal Growth Factor-Induced Chemotaxis. J. Biol. Chem. 2004, 279, 34290–34297. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bao, Y.; Chen, Z.; Guo, Y.; Feng, Y.; Li, Z.; Han, W.; Wang, J.; Zhao, W.; Jiao, Y.; Li, K.; et al. Tumor Suppressor MicroRNA-27a in Colorectal Carcinogenesis and Progression by Targeting SGPP1 and Smad2. PLoS ONE 2014, 9, e105991. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hannun, Y.A.; Obeid, L.M. Sphingolipids and Their Metabolism in Physiology and Disease. Nat. Rev. Mol. Cell Biol. 2018, 19, 175–191. [Google Scholar] [CrossRef]
- Pulkoski-Gross, M.J.; Donaldson, J.C.; Obeid, L.M. Sphingosine-1-Phosphate Metabolism: A Structural Perspective. Crit. Rev. Biochem. Mol. Biol. 2015, 50, 298–313. [Google Scholar] [CrossRef] [Green Version]
- Pyne, N.J.; Pyne, S. Sphingosine 1-Phosphate and Cancer. Nat. Rev. Cancer 2010, 10, 489. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gomez-Munoz, A.; Duffy, P.A.; Martin, A.; O’Brien, L.; Byun, H.S.; Bittman, R.; Brindley, D.N. Short-Chain Ceramide-1-Phosphates Are Novel Stimulators of DNA Synthesis and Cell Division: Antagonism by Cell-Permeable Ceramides. Mol. Pharmacol. 1995, 47, 833–839. [Google Scholar] [PubMed]
- Gomez-Munoz, A.; Frago, L.M.; Alvarez, L.; Varela-Nieto, I. Stimulation of DNA Synthesis by Natural Ceramide 1-Phosphate. Biochem. J. 1997, 325, 435–440. [Google Scholar] [CrossRef] [Green Version]
- Arana, L.; Gangoiti, P.; Ouro, A.; Rivera, I.G.; Ordoñez, M.; Trueba, M.; Lankalapalli, R.S.; Bittman, R.; Gomez-Muñoz, A. Generation of Reactive Oxygen Species (ROS) Is a Key Factor for Stimulation of Macrophage Proliferation by Ceramide 1-Phosphate. Exp. Cell Res. 2012, 318, 350–360. [Google Scholar] [CrossRef]
- Gangoiti, P.; Bernacchioni, C.; Donati, C.; Cencetti, F.; Ouro, A.; Gómez-Muñoz, A.; Bruni, P. Ceramide 1-Phosphate Stimulates Proliferation of C2C12 Myoblasts. Biochimie 2012, 94, 597–607. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ouro, A.; Arana, L.; Riazy, M.; Zhang, P.; Gomez-Larrauri, A.; Steinbrecher, U.; Duronio, V.; Gomez-Muñoz, A. Vascular Endothelial Growth Factor Mediates Ceramide 1-Phosphate-Stimulated Macrophage Proliferation. Exp. Cell Res. 2017, 361, 277–283. [Google Scholar] [CrossRef]
- Gómez-Muñoz, A.; Kong, J.Y.; Salh, B.; Steinbrecher, U.P. Ceramide-1-Phosphate Blocks Apoptosis through Inhibition of Acid Sphingomyelinase in Macrophages. J. Lipid Res. 2004, 45, 99–105. [Google Scholar] [CrossRef] [Green Version]
- Gomez-Munoz, A.; Kong, J.Y.; Parhar, K.; Wang, S.W.; Gangoiti, P.; Gonzalez, M.; Eivemark, S.; Salh, B.; Duronio, V.; Steinbrecher, U.P. Ceramide-1-Phosphate Promotes Cell Survival through Activation of the Phosphatidylinositol 3-Kinase/Protein Kinase B Pathway. FEBS Lett. 2005, 579, 3744–3750. [Google Scholar] [CrossRef] [Green Version]
- Gangoiti, P.; Granado, M.H.; Arana, L.; Ouro, A.; Gómez-Muñoz, A. Involvement of Nitric Oxide in the Promotion of Cell Survival by Ceramide 1-Phosphate. FEBS Lett. 2008, 582, 2263–2269. [Google Scholar] [CrossRef] [Green Version]
- Granado, M.H.; Gangoiti, P.; Ouro, A.; Arana, L.; Gómez-Muñoz, A. Ceramide 1-Phosphate Inhibits Serine Palmitoyltransferase and Blocks Apoptosis in Alveolar Macrophages. Biochim. Biophys. Acta 2009, 1791, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Pascuali, N.; Scotti, L.; di Pietro, M.; Oubiña, G.; Bas, D.; May, M.; Gómez Muñoz, A.; Cuasnicú, P.S.; Cohen, D.J.; Tesone, M.; et al. Ceramide-1-Phosphate Has Protective Properties against Cyclophosphamide-Induced Ovarian Damage in a Mice Model of Premature Ovarian Failure. Hum. Reprod. 2018, 33, 844–859. [Google Scholar] [CrossRef]
- Gomez-Larrauri, A.; Ouro, A.; Trueba, M.; Gomez-Muñoz, A. Regulation of Cell Growth, Survival and Migration by Ceramide 1-Phosphate-Implications in Lung Cancer Progression and Inflammation. Cell. Signal. 2021, 83, 109980. [Google Scholar] [CrossRef]
- Gangoiti, P.; Granado, M.H.; Arana, L.; Ouro, A.; Gomez-Muñoz, A. Activation of Protein Kinase C-Alpha Is Essential for Stimulation of Cell Proliferation by Ceramide 1-Phosphate. FEBS Lett. 2010, 584, 517–524. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gomez-Munoz, A.; Kong, J.; Salh, B.; Steinbrecher, U.P. Sphingosine-1-Phosphate Inhibits Acid Sphingomyelinase and Blocks Apoptosis in Macrophages. FEBS Lett. 2003, 539, 56–60. [Google Scholar] [CrossRef] [Green Version]
- Newcomb, B.; Rhein, C.; Mileva, I.; Ahmad, R.; Clarke, C.J.; Snider, J.; Obeid, L.M.; Hannun, Y.A. Identification of an Acid Sphingomyelinase Ceramide Kinase Pathway in the Regulation of the Chemokine CCL5. J. Lipid Res. 2018, 59, 1219–1229. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitra, P.; Maceyka, M.; Payne, S.G.; Lamour, N.; Milstien, S.; Chalfant, C.E.; Spiegel, S. Ceramide Kinase Regulates Growth and Survival of A549 Human Lung Adenocarcinoma Cells. FEBS Lett. 2007, 581, 735–740. [Google Scholar] [CrossRef] [PubMed]
- Schwalm, S.; Erhardt, M.; Römer, I.; Pfeilschifter, J.; Zangemeister-Wittke, U.; Huwiler, A. Ceramide Kinase Is Upregulated in Metastatic Breast Cancer Cells and Contributes to Migration and Invasion by Activation of PI 3-Kinase and Akt. Int. J. Mol. Sci. 2020, 21, 1396. [Google Scholar] [CrossRef] [Green Version]
- Pastukhov, O.; Schwalm, S.; Zangemeister-Wittke, U.; Fabbro, D.; Bornancin, F.; Japtok, L.; Kleuser, B.; Pfeilschifter, J.; Huwiler, A. The Ceramide Kinase Inhibitor NVP-231 Inhibits Breast and Lung Cancer Cell Proliferation by Inducing M Phase Arrest and Subsequent Cell Death. Br. J. Pharmacol. 2014, 171, 5829–5844. [Google Scholar] [CrossRef] [Green Version]
- Payne, A.W.; Pant, D.K.; Pan, T.C.; Chodosh, L.A. Ceramide Kinase Promotes Tumor Cell Survival and Mammary Tumor Recurrence. Cancer Res. 2014, 74, 6352–6363. [Google Scholar] [CrossRef] [Green Version]
- Lamour, N.; Chalfant, C. Ceramide Kinase and the Ceramide-1-Phosphate/CPLA2α; Interaction as a Therapeutic Target. Curr. Drug Targets 2008, 9, 674–682. [Google Scholar] [CrossRef]
- Berwick, M.L.; Dudley, B.A.; Maus, K.; Chalfant, C.E. The Role of Ceramide 1-Phosphate in Inflammation, Cellular Proliferation, and Wound Healing. Adv. Exp. Med. Biol. 2019, 1159, 65–77. [Google Scholar] [CrossRef]
- Hoeferlin, L.A.; Wijesinghe, D.S.; Chalfant, C.E. The Role of Ceramide-1-Phosphate in Biological Functions. Handb. Exp. Pharmacol. 2013, 153–166. [Google Scholar] [CrossRef] [Green Version]
- Baudiß, K.; de Paula Vieira, R.; Cicko, S.; Ayata, K.; Hossfeld, M.; Ehrat, N.; Gómez-Muñoz, A.; Eltzschig, H.K.; Idzko, M. C1P Attenuates Lipopolysaccharide-Induced Acute Lung Injury by Preventing NF-ΚB Activation in Neutrophils. J. Immunol. 2016, 196, 2319–2326. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baudiß, K.; Ayata, C.K.; Lazar, Z.; Cicko, S.; Beckert, J.; Meyer, A.; Zech, A.; Vieira, R.P.; Bittman, R.; Gómez-Muñoz, A.; et al. Ceramide-1-Phosphate Inhibits Cigarette Smoke-Induced Airway Inflammation. Eur. Respir. J. 2015, 45, 1669–1680. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gomez-Larrauri, A.; Trueba, M.; Gomez-Muñoz, A. Potential of Ceramide 1-Phosphate as a Novel Therapeutic Agent in Pulmonary Inflammation. Expert Rev. Clin. Pharmacol. 2016, 9, 629–631. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Presa, N.; Gomez-Larrauri, A.; Dominguez-Herrera, A.; Trueba, M.; Gomez-Muñoz, A. Novel Signaling Aspects of Ceramide 1-Phosphate. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2020, 1865. [Google Scholar] [CrossRef]
- Samaha, D.; Hamdo, H.H.; Wilde, M.; Prause, K.; Arenz, C. Sphingolipid-Transporting Proteins as Cancer Therapeutic Targets. Int. J. Mol. Sci. 2019, 20, 3554. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yasuda, S.; Kitagawa, H.; Ueno, M.; Ishitani, H.; Fukasawa, M.; Nishijima, M.; Kobayashi, S.; Hanada, K. A Novel Inhibitor of Ceramide Trafficking from the Endoplasmic Reticulum to the Site of Sphingomyelin Synthesis. J. Biol. Chem. 2001, 276, 43994–44002. [Google Scholar] [CrossRef] [Green Version]
- Saied, E.M.; Diederich, S.; Arenz, C. Facile Synthesis of the CERT Inhibitor HPA-12 and Some Novel Derivatives. Chem. Asian J. 2014, 9, 2092–2094. [Google Scholar] [CrossRef]
- Berkeš, D.; Daïch, A.; Santos, C.; Ballereau, S.; Génisson, Y. Chemistry and Biology of HPAs: A Family of Ceramide Trafficking Inhibitors. Chem. A Eur. J. 2016, 22, 17514–17525. [Google Scholar] [CrossRef]
- Nakao, N.; Ueno, M.; Sakai, S.; Egawa, D.; Hanzawa, H.; Kawasaki, S.; Kumagai, K.; Suzuki, M.; Kobayashi, S.; Hanada, K. Natural Ligand-Nonmimetic Inhibitors of the Lipid-Transfer Protein CERT. Commun. Chem. 2019, 2, 1–11. [Google Scholar] [CrossRef]
- Samaha, D.; Hamdo, H.H.; Cong, X.; Schumacher, F.; Banhart, S.; Aglar, Ö.; Möller, H.M.; Heuer, D.; Kleuser, B.; Saied, E.M.; et al. Liposomal FRET Assay Identifies Potent Drug-Like Inhibitors of the Ceramide Transport Protein (CERT). Chem. A Eur. J. 2020, 26, 16616–16621. [Google Scholar] [CrossRef] [PubMed]
- Castellan, T.; Santos, C.; Rodriguez, F.; Lepage, M.L.; Liang, Y.; Bodlenner, A.; Compain, P.; Génisson, Y.; Dehoux, C.; Ballereau, S. N,O-Dialkyl Deoxynojirimycin Derivatives as CERT START Domain Ligands. Bioorg. Med. Chem. Lett. 2020, 30, 126796. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.J.X.; Roylance, R.; Sander, J.; Gorman, P.; Endesfelder, D.; Kschischo, M.; Jones, N.P.; East, P.; Nicke, B.; Spassieva, S.; et al. CERT Depletion Predicts Chemotherapy Benefit and Mediates Cytotoxic and Polyploid-Specific Cancer Cell Death through Autophagy Induction. J. Pathol. 2012, 226, 482–494. [Google Scholar] [CrossRef] [Green Version]
- Sonoda, Y.; Yamamoto, D.; Sakurai, S.; Hasegawa, M.; Aizu-Yokota, E.; Momoi, T.; Kasahara, T. FTY720, a Novel Immunosuppressive Agent, Induces Apoptosis in Human Glioma Cells. Biochem. Biophys. Res. Commun. 2001, 281, 282–288. [Google Scholar] [CrossRef]
- Zheng, T.; Meng, X.; Wang, J.B.; Chen, X.; Yin, D.; Liang, Y.; Song, X.; Pan, S.; Jiang, H.; Liu, L. PTEN- and P53-Mediated Apoptosis and Cell Cycle Arrest by FTY720 in Gastric Cancer Cells and Nude Mice. J. Cell. Biochem. 2010, 111, 218–228. [Google Scholar] [CrossRef]
- Zhang, N.; Qi, Y.; Wadham, C.; Wang, L.; Warren, A.; Di, W.; Xia, P. FTY720 Induces Necrotic Cell Death and Autophagy in Ovarian Cancer Cells: A Protective Role of Autophagy. Autophagy 2010, 6, 1157–1167. [Google Scholar] [CrossRef] [Green Version]
- Azuma, H.; Takahara, S.; Horie, S.; Muto, S.; Otsuki, Y.; Katsuoka, Y. Induction of Apoptosis in Human Bladder Cancer Cells In Vitro and In Vivo Caused by FTY720 Treatment. J. Urol. 2003, 169, 2372–2377. [Google Scholar] [CrossRef] [PubMed]
- Yasui, H.; Hideshima, T.; Raje, N.; Roccaro, A.M.; Shiraishi, N.; Kumar, S.; Hamasaki, M.; Ishitsuka, K.; Tai, Y.T.; Podar, K.; et al. FTY720 Induces Apoptosis in Multiple Myeloma Cells and Overcomes Drug Resistance. Cancer Res. 2005, 65, 7478–7484. [Google Scholar] [CrossRef] [Green Version]
- Min, K.-J.; Kwon, T.K. Induction of Lysosomal Membrane Permeabilization Is a Major Event of FTY720-Mediated Non-Apoptotic Cell Death in Human Glioma Cells. Cancers 2020, 12, 3388. [Google Scholar] [CrossRef]
- Li, C.X.; Yang, X.X.; Wang, H.W.; Li, X.C.; Ng, K.T.-P.; Lo, C.M.; Man, K. FTY720 Suppresses Liver Tumor Growth and Metastasis by Reducing Circulating Regulating T Cells and Enhancing the Anti-Tumor Effect of Rapamycin. Oncotargets Ther. 2020, 13, 4743–4754. [Google Scholar] [CrossRef] [PubMed]
- Salinas, N.R.A.; Oshima, C.T.F.; Cury, P.M.; Cordeiro, J.A.; Bueno, V. FTY720 and Lung Tumor Development. Int. Immunopharmacol. 2009, 9, 689–693. [Google Scholar] [CrossRef]
- Suzuki, S.; Li, X.K.; Enosawa, S.; Shinomiya, T. A New Immunosuppressant, FTY720, Induces Bcl-2-Associated Apoptotic Cell Death in Human Lymphocytes. Immunology 1996, 89, 518–523. [Google Scholar] [CrossRef] [PubMed]
- Yoshino, T.; Tabunoki, H.; Sugiyama, S.; Ishii, K.; Kim, S.U.; Satoh, J.I. Non-Phosphorylated FTY720 Induces Apoptosis of Human Microglia by Activating SREBP2. Cell. Mol. Neurobiol. 2011, 31, 1009–1020. [Google Scholar] [CrossRef]
- Pchejetski, D.; Bohler, T.; Brizuela, L.; Sauer, L.; Doumerc, N.; Golzio, M.; Salunkhe, V.; Teissié, J.; Malavaud, B.; Waxman, J.; et al. FTY720 (Fingolimod) Sensitizes Prostate Cancer Cells to Radiotherapy by Inhibition of Sphingosine Kinase-1. Cancer Res. 2010, 70, 8651–8661. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hait, N.C.; Avni, D.; Yamada, A.; Nagahashi, M.; Aoyagi, T.; Aoki, H.; Dumur, C.I.; Zelenko, Z.; Gallagher, E.J.; Leroith, D.; et al. The Phosphorylated Prodrug FTY720 Is a Histone Deacetylase Inhibitor That Reactivates ERα Expression and Enhances Hormonal Therapy for Breast Cancer. Oncogenesis 2015, 4, e156. [Google Scholar] [CrossRef] [PubMed]
- di Pardo, A.; Maglione, V. Sphingolipid Metabolism: A New Therapeutic Opportunity for Brain Degenerative Disorders. Front. Neurosci. 2018, 12, 249. [Google Scholar] [CrossRef]
- Chatzakos, V.; Rundlöf, A.K.; Ahmed, D.; de Verdier, P.J.; Flygare, J. Inhibition of Sphingosine Kinase 1 Enhances Cytotoxicity, Ceramide Levels and ROS Formation in Liver Cancer Cells Treated with Selenite. Biochem. Pharmacol. 2012, 84, 712–721. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, C.; Hu, L.; He, Y.; Shi, Z.; Tang, S.; Chen, Y. Abnormal Expression of MiR-21 and MiR-95 in Cancer Stem-like Cells Is Associated with Radioresistance of Lung Cancer. Cancer Investig. 2015, 33, 165–171. [Google Scholar] [CrossRef]
- Huang, X.; Taeb, S.; Jahangiri, S.; Emmenegger, U.; Tran, E.; Bruce, J.; Mesci, A.; Korpela, E.; Vesprini, D.; Wong, C.S.; et al. MiRNA-95 Mediates Radioresistance in Tumors by Targeting the Sphingolipid Phosphatase SGPP1. Cancer Res. 2013, 73, 6972–6986. [Google Scholar] [CrossRef] [Green Version]

| Targeted Enzyme | Effector (Function) | Tissue/Cell Type | Ref. |
|---|---|---|---|
| SPT | Myriocin (inhibitor) | B16F10 melanoma cells; Human lung adenocarcinoma cell line (HCC4006); Human glioblastoma cells (U87MG) Breast cancer cells | [19,61,62,63] |
| Tetrahydropyrazolopyridine; 3-phenylpiperidine (inhibitors) | PL-21 cells derived from an acute promyelocytic leukemia; Human lung adenocarcinoma cell line (HCC4006) | [27,64] | |
| Fenretinide (activator) | Ovarian; Breast and neuroblastoma tumor cells | [65,66] | |
| Taxol (activator) | Human breast cancer cells | [82] | |
| Resveratrol (activator) | Metastatic breast cancer cells | [67] | |
| Anti-androgens (activator) | Prostate cancer | [70] | |
| CerS | Anthracyclines, Vinblastine (Activators) | Liver | [80,81] |
| Taxol (activator) | Human breast cancer cells | [82] | |
| Antifolate methotrexate (indirect activator) | A549 lung adenocarcinoma cells | [83] | |
| P053 (inhibitor) | HEK 293 cells | [77] | |
| ST1058, ST1060, ST1072, ST1074 | HCT-106 and HeLa cells | [79] | |
| Des1 | Resveratrol (inhibitor) | Gastric cancer HGC27 cells; T98G and U87MG glioblastoma cell lines | [88,91] |
| γ-tocotrienol, phenoxodiol, celecoxib (inhibitors) | T98G and U87MG glioblastoma cell lines | [88] | |
| Dideuterated analog of 3-keotsphinganine (inhibitor) | HGC27, T98G and U87MG cancer cells | [92] | |
| Fenretinide combined with Foscan (inhibitor) | SCC19 human head and neck squamous cell carcinoma | [93] | |
| GT11 (inhibitor) | Human U87MG glioma cell line | [89] | |
| Tetrahydrocannabinol (THC) by indirect mechanism inhibition | |||
| SMase | Gamma radiation (activator) | Liver | [36] |
| GW4869 (inhibitor) | Lung from human melanoma cells | [113] | |
| siRNA (inhibitor) | Adenocarcinoma cell line A549 | [97] | |
| Doxorubicin (activator) | Breast cancer cells | [96] | |
| Resveratrol (activator) | K562 and HCT116 cell lines | [99] | |
| Perphenazine and fluphenazine (inhibitor) | Xenografted tumor growth | [105] | |
| ARC39 (inhibitor) | L929, HepG2 and B16F10 cell lines | [106] | |
| GCS | PDMP | HeLa cell line | [129] |
| β-GCase | siRNA | Gastric cancer | [134] |
| Ceramidase | LCL521 (inhibitor) | HT29, HCT116 and LIM1215 cell lines | [139] |
| siRNA | |||
| Tamoxifen (inhibitor) | Polyploid giant cancer cells | [140,244] | |
| N-metallocenoylsphingosine (inhibitor) | High-5 insect cells | [141] | |
| D-erythro-MAPP (inhibitor) | Hepatocellular carcinoma cells | [142] | |
| Deoxy-sphingolipids (inhibitor) | Several cancer cells | [145,147] | |
| C6-urea-ceramide (inhibitor) | Colon cancer cells | [150,151] | |
| SMS | D609 (inhibitor) | Hippocampal neurons and glioblastoma | [117,118] |
| SMS siRNA | Breast cancer | [119,121] | |
| 15w (inhibitor) | [119] | ||
| SphK1 | SK1 siRNA | U-1242 and U-87MG glioblastoma cell lines | [184] |
| SK1-I (BML-258) (inhibitor) | Human glioblastoma cells and xenografts | [194] | |
| FTY-720 (Degradation inhibition) | Lymphocytes, microglia, myeloma, leukaemia, prostate, lung, liver, breast, colon, gastric, bladder, renal glioma and ovarian cancers | [186,187,245,246,247,248,249,250,251,252,253,254,255,256,257] | |
| SphK-/- mouse models | Murine melanoma cells | [173] | |
| N, N-Dimethyl-D-erythro-sphingosine (DMS) (inhibitor) | Human lung cancer | [195] | |
| FTY-720 and PF453 | Breast cancer cells | [196] | |
| PF453 | MCF10A cell line | [197] | |
| SK1-II | Liver cancer cells | [258] | |
| SphK2 | Sphk2 siRNA | U-1242 and U-87MG glioblastoma cell lines and HCT116 colon carcinoma cells | [182,184] |
| ABC294640 (inhibitor) | non-small cell lung cancer | [185] | |
| SPP | miR95 and miR21(inhibition) | Lung cancer | [259,260] |
| miR-27a (inhibition) | Colorectal cancer | [208] | |
| CerK | siRNA | A549 human lung adenocarcinoma cells | [226] |
| NVP-231 (inhibitor) | MCF-7 and NCI-H358 human lung cancer cell lines | [228] | |
| CERT | HPA-12 family (inhibitors) | Chinese hamster ovary cells, HeLa cells | [238,239,240] |
| (1S, 2R)-HPCB-5 | HeLa cells | [241] | |
| Lomitapide, Clevidipine, Fluralaner and Eltrombopag (inhibitors) | HeLa cells | [242] | |
| N,O-Dialkyl deoxynojirimycin | in vitro | [243] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gomez-Larrauri, A.; Das Adhikari, U.; Aramburu-Nuñez, M.; Custodia, A.; Ouro, A. Ceramide Metabolism Enzymes—Therapeutic Targets against Cancer. Medicina 2021, 57, 729. https://0-doi-org.brum.beds.ac.uk/10.3390/medicina57070729
Gomez-Larrauri A, Das Adhikari U, Aramburu-Nuñez M, Custodia A, Ouro A. Ceramide Metabolism Enzymes—Therapeutic Targets against Cancer. Medicina. 2021; 57(7):729. https://0-doi-org.brum.beds.ac.uk/10.3390/medicina57070729
Chicago/Turabian StyleGomez-Larrauri, Ana, Upasana Das Adhikari, Marta Aramburu-Nuñez, Antía Custodia, and Alberto Ouro. 2021. "Ceramide Metabolism Enzymes—Therapeutic Targets against Cancer" Medicina 57, no. 7: 729. https://0-doi-org.brum.beds.ac.uk/10.3390/medicina57070729







