Patterns of Smoking Behaviour in Low-Income Pregnant Women: A Cohort Study of Differential Effects on Infant Birth Weight
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Setting
2.2. Study Population
2.3. Data Collection and Trial Management
2.4. Assessment
- Sustained quitters: initial quitters who continued to not smoke across the first three visits (QQQ)
- Continued smokers: initial smokers who continued to smoke across the first three visits (SSS)
- Initial smokers who had quit at V3 (SSQ/SQQ)
- Any other quit attempt during V1 or V2 (QQS/QSQ/QSS/SQS)
2.5. Statistical Analysis
2.6. Ethical Approval
3. Results
3.1. Multivariable Analysis
3.2. Multivariable Analysis
3.3. Household SHS
3.4. Gender
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Butler, C.C.; Rollnick, S.; Cohen, D.; Bachmann, M.; Russell, I.; Stott, N. Motivational consulting versus brief advice for smokers in general practice: A randomized trial. Br. J. Gen. Pract. 1999, 49, 611–616. [Google Scholar]
- Lindley, A.A.; Becker, S.; Gray, R.H.; Herman, A.A. Effect of continuing or stopping smoking during pregnancy on infant birth weight, crown-heel length, head circumference, ponderal index, and brain: Body weight ratio. Am. J. Epidemiol. 2000, 152, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Jaddoe, V.W.; Verburg, B.O.; De Ridder, M.; Hofman, A.; Mackenbach, J.P.; Moll, H.A.; Steegers, E.A.; Witteman, J.C. Maternal smoking and fetal growth characteristics in different periods of pregnancy. Am. J. Epidemiol. 2007, 165, 1207–1215. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Health and Human Services. The Health Consequences of Smoking. A Report of the Surgeon General. Chapter 5: Reproductive Effects; US Department of Health and Human Services: Atlanta, GA, USA, 2004.
- Cnattingius, S. The epidemiology of smoking during pregnancy: Smoking prevalence, maternal characteristics, and pregnancy outcomes. Nicotine Tob. Res. 2004, 6, S125–S140. [Google Scholar] [CrossRef] [PubMed]
- Salihu, H.M.; Wilson, R.E. Epidemiology of prenatal smoking and perinatal outcomes. Early Hum. Dev. 2007, 83, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Andersen, M.R.; Simonsen, U.; Uldbjerg, N.; Aalkjaer, C.; Stender, S. Smoking cessation early in pregnancy and birth weight, length, head circumference, and endothelial nitric oxide synthase activity in umbilical and chorionic vessels: An observational study of healthy singleton pregnancies. Circulation 2009, 119, 857–864. [Google Scholar] [CrossRef] [PubMed]
- Hiscock, R.; Bauld, L.; Amos, A.; Fidler, J.A.; Munafò, M. Socioeconomic status and smoking: A review. Ann. N. Y. Acad. Sci. 2012, 1248, 107–123. [Google Scholar] [CrossRef] [PubMed]
- Morgan, K.; McGee, H.; Watson, D.; Perry, I.; Barry, M.; Shelley, E.; Harrington, J.; Molcho, M.; Layte, R.; Tully, N.; et al. SLAN 2007: Survey of Lifestyle, Attitudes & Nutrition in Ireland: Main Report; Department of Health and Children: Dublin, Ireland, 2008. [Google Scholar]
- Friel, S.; Nic Gabhainn, S.; Kelleher, C. The National Health and Lifestyle Surveys: Survey of Lifestyle, Attitudes and Nutrition (SLAN) & the Irish Health Behaviour in School-Aged Children Survey (HBSC); Department of Health and Children: Dublin, Ireland, 1999.
- Kelleher, C.; Friel, S.; Nic Gabhainn, S.; Corrigan, H.; Nolan, G.; Sixsmith, J.; Walsh, O.; Cooke, M. The National Health and Lifestyle Surveys: Survey of Lifestyle, Attitudes and Nutrition (SLAN) & the Irish Health Behaviour in School-Aged Children Survey (HBSC); Centre for Health Promotion Studies: Galway, Ireland; Department of Health and Children: Dublin, Ireland, 2003.
- Murrin, C.; Segonds-Pichon, A.; Fallon, U.B.; Hannon, F.; Bury, G.; Loftus, B.G.; Murphy, A.W.; Morrison, J.J.; Daly, S.; Kelleher, C.C. Self-reported pre-pregnancy maternal body mass index and infant birth-weight. Ir. Med. J. 2007, 100, S20–S23. [Google Scholar]
- Butler, N.R.; Goldstein, H.; Ross, E.M. Cigarette smoking in pregnancy: Its influence on birth weight and perinatal mortality. Br. Med. J. 1972, 2, 127–130. [Google Scholar] [CrossRef] [PubMed]
- Ward, C.; Lewis, S.; Coleman, T. Prevalence of maternal smoking and environmental tobacco smoke exposure during pregnancy and impact on birth weight: Retrospective study using millennium cohort. BMC Public Health 2007, 7, 81. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Health and Human Services. The Health Consequences of Smoking—50 Years of Progress. A Report of the Surgeon General; U.S. Department of Health and Human Services: Atlanta, GA, USA, 2014.
- England, L.J.; Kendrick, J.S.; Gargiullo, P.M.; Zahniser, S.C.; Hannon, W.H. Measures of maternal tobacco exposure and infant birth weight at term. Am. J. Epidemiol. 2001, 153, 954–960. [Google Scholar] [CrossRef] [PubMed]
- Windham, G.C.; Hopkins, B.; Fenster, L.; Swan, S.H. Prenatal active or passive tobacco smoke exposure and the risk of preterm delivery or low birth weight. Epidemiology 2000, 11, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Misra, D.P.; Nguyen, R.H.N. Environmental tobacco smoke and low birth weight: A hazard in the workplace? Environ Health Perspect. Suppl. 1999, 107, 897. [Google Scholar] [CrossRef]
- Kharrazi, M.; DeLorenze, G.N.; Kaufman, F.L.; Eskenazi, B.; Bernert, J.T.; Graham, S.; Pearl, M.; Pirkle, J. Environmental tobacco smoke and pregnancy outcome. Epidemiology 2004, 15, 660–670. [Google Scholar] [CrossRef] [PubMed]
- Jaddoe, V.W.V.; Troe, E.-J.W.M.; Hofman, A.; Mackenbach, J.P.; Moll, H.A.; Steegers, E.A.P.; Witteman, J.C.M. Active and passive maternal smoking during pregnancy and the risks of low birthweight and preterm birth: The generation R study. Paediatr. Perinat. Epidemiol. 2008, 22, 162–171. [Google Scholar] [CrossRef] [PubMed]
- MacArthur, C.; Knox, E.G. Smoking in pregnancy: Effects of stopping at different stages. Br. J. Obstet. Gynaecol. 1988, 95, 551–555. [Google Scholar] [CrossRef] [PubMed]
- McCowan, L.M.; Dekker, G.A.; Chan, E.; Stewart, A.; Chappell, L.C.; Hunter, M.; Moss-Morris, R.; North, R.A. Spontaneous preterm birth and small for gestational age infants in women who stop smoking early in pregnancy: Prospective cohort study. Br. Med. J. 2009, 338, b1081. [Google Scholar] [CrossRef] [PubMed]
- Li, C.Q.; Windsor, R.A.; Perkins, L.; Goldenberg, R.L.; Lowe, J.B. The impact on infant birth weight and gestational age of cotinine-validated smoking reduction during pregnancy. JAMA 1993, 269, 1519–1524. [Google Scholar] [CrossRef] [PubMed]
- Pickett, K.E.; Wakschlag, L.S.; Dai, L.; Leventhal, B.L. Fluctuations of maternal smoking during pregnancy. Obstet. Gynecol. 2003, 101, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Rowa-Dewar, N.; Lumsdaine, C.; Amos, A. Protecting children from smoke exposure in disadvantaged homes. Nicotine Tob. Res. 2015, 17, 496–501. [Google Scholar] [CrossRef] [PubMed]
- Moore, G.F.; Currie, D.; Gilmore, G.; Holliday, J.C.; Moore, L. Socioeconomic inequalities in childhood exposure to secondhand smoke before and after smoke-free legislation in three UK countries. J. Public Health 2012, 34, 599–608. [Google Scholar] [CrossRef] [PubMed]
- Secker-Walker, R.H.; Vacek, P.M.; Flynn, B.S.; Mead, P.B. Estimated gains in birth weight associated with reductions in smoking during pregnancy. J. Reprod. Med. 1998, 43, 967–974. [Google Scholar] [PubMed]
- England, L.J.; Kendrick, J.S.; Wilson, H.G.; Merritt, R.K.; Gargiullo, P.M.; Zahniser, S.C. Effects of smoking reduction during pregnancy on the birth weight of term infants. Am. J. Epidemiol. 2001, 154, 694–701. [Google Scholar] [CrossRef] [PubMed]
- Lumley, J.; Chamberlain, C.; Dowswell, T.; Oliver, S.; Oakley, L.; Watson, L. Interventions for Promoting Smoking Cessation during Pregnancy. Available online: http://0-onlinelibrary-wiley-com.brum.beds.ac.uk/doi/10.1002/14651858.CD001055.pub4/abstract (accessed on 4 August 2016).
- Hayes, C.B.; Collins, C.; O’Carroll, H.; Wyse, E.; Gunning, M.; Geary, M.; Kelleher, C.C. Effectiveness of motivational interviewing in influencing smoking cessation in pregnant and postpartum disadvantaged women. Nicotine Tob. Res. 2013, 15, 969–977. [Google Scholar] [CrossRef] [PubMed]
- Geary, M. Clinical Report; Rotunda Hospital: Dublin, Ireland, 2004. [Google Scholar]
- Melvin, C.L.; Tucker, P. Measurement and definition for smoking cessation intervention research: The smoke-free families experience. Tob. Control 2000, 9 (Suppl. S3), iii87–iii90. [Google Scholar] [CrossRef] [PubMed]
- West, R.; Hajek, P.; Stead, L.; Stapleton, J. Outcome criteria in smoking cessation trials: Proposal for a common standard. Addiction 2005, 100, 299–303. [Google Scholar] [CrossRef] [PubMed]
- Abrahamsson, A.; Ejlertsson, G. Smoking patterns during pregnancy: Differences in socioeconomic and health-related variables. Eur. J. Public Health 2000, 10, 208–213. [Google Scholar] [CrossRef]
- Cope, G.; Nayyar, P.; Holder, R.; Gibbons, J.; Bunce, R. A simple near-patient test for nicotine and its metabolites in urine to assess smoking habit. Clin. Chim. Acta 1996, 256, 135–149. [Google Scholar] [CrossRef]
- IBM Corp. IBM SPSS Statistics for Windows, Version 20; IBM Corp.: Armonk, NY, USA, 2011. [Google Scholar]
- Benjamin-Garner, R.; Stotts, A. Impact of smoking exposure change on infant birth weight among a cohort of women in a prenatal smoking cessation study. Nicotine Tob. Res. 2013, 15, 685–692. [Google Scholar] [CrossRef] [PubMed]
- Phillips, R.; Amos, A.; Ritchie, D.; Cunningham-Burley, S.; Martin, C. Smoking in the home after the smoke-free legislation in scotland: Qualitative study. Br. Med. J. 2007, 335, 553. [Google Scholar] [CrossRef] [PubMed]
- Leonardi-Bee, J.; Smyth, A.; Britton, J.; Coleman, T. Environmental tobacco smoke and fetal health: Systematic review and meta-analysis. Arch. Dis. Child. Fetal Neonatal Ed. 2008, 93, F351–F361. [Google Scholar] [CrossRef] [PubMed]
- Salmasi, G.; Grady, R.; Jones, J.; McDonald, S.D.; On behalf of the Knowledge Synthesis Group. Environmental tobacco smoke exposure and perinatal outcomes: A systematic review and meta-analyses. Acta Obstet. Gynecol. Scand. 2010, 89, 423–441. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; He, X.C.; Cui, H.M.; Zhang, C.; Zhang, H.H.; Dang, Y.; Han, X.D.; Chen, Y.; Tang, Z.F.; Zhang, H.R.; et al. Passive smoking and preterm birth in urban china editorial comment. Obstet. Gynecol. Surv. 2014, 69, 647–649. [Google Scholar] [CrossRef]
- Cui, H.; Gong, T.T.; Liu, C.X.; Wu, Q.J. Associations between passive maternal smoking during pregnancy and preterm birth: Evidence from a meta-analysis of observational studies. PLoS ONE 2016, 11, e0147848. [Google Scholar] [CrossRef] [PubMed]
- Voigt, M.; Hermanussen, M.; Wittwer-Backofen, U.; Fusch, C.; Hesse, V. Sex-specific differences in birth weight due to maternal smoking during pregnancy. Eur. J. Pediatr. 2006, 165, 757–761. [Google Scholar] [CrossRef] [PubMed]
- Zarén, B.; Lindmark, G.; Bakketeig, L. Maternal smoking affects fetal growth more in the male fetus. Paediatr. Perinat. Epidemiol. 2000, 14, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Tayie, F.A.; Powell, C. Sex differences in the association between prenatal smoking and decreased birthweight, and intensive health care of the neonate. Behav. Med. 2012, 38, 138–142. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, M.B.; Hurrell, R.F. Nutritional iron deficiency. Lancet 2007, 370, 511–520. [Google Scholar] [CrossRef]
- Krafft, A. Iron supplementation in pregnancy. Br. Med. J. 2013, 347, f4399. [Google Scholar] [CrossRef] [PubMed] [Green Version]
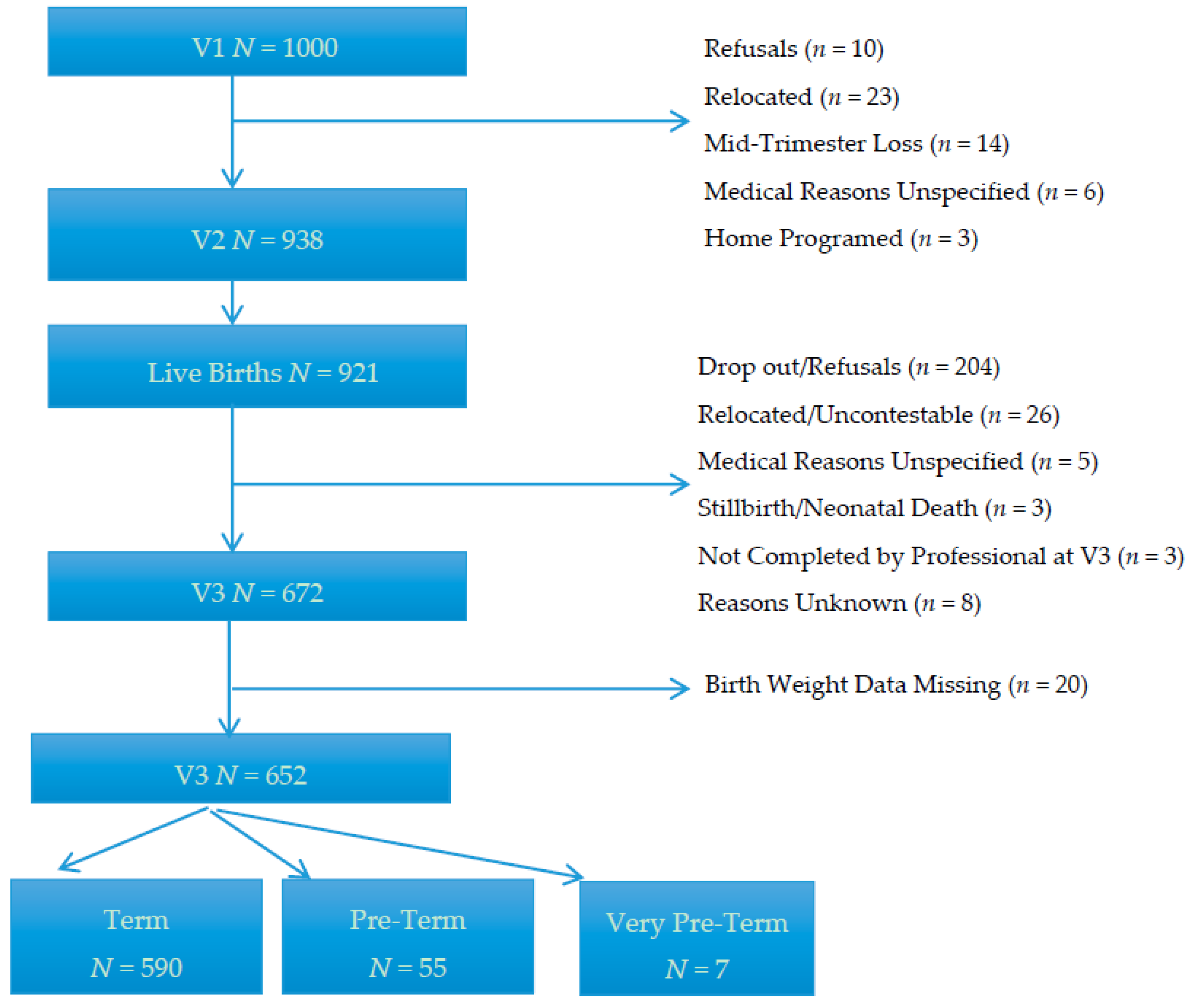
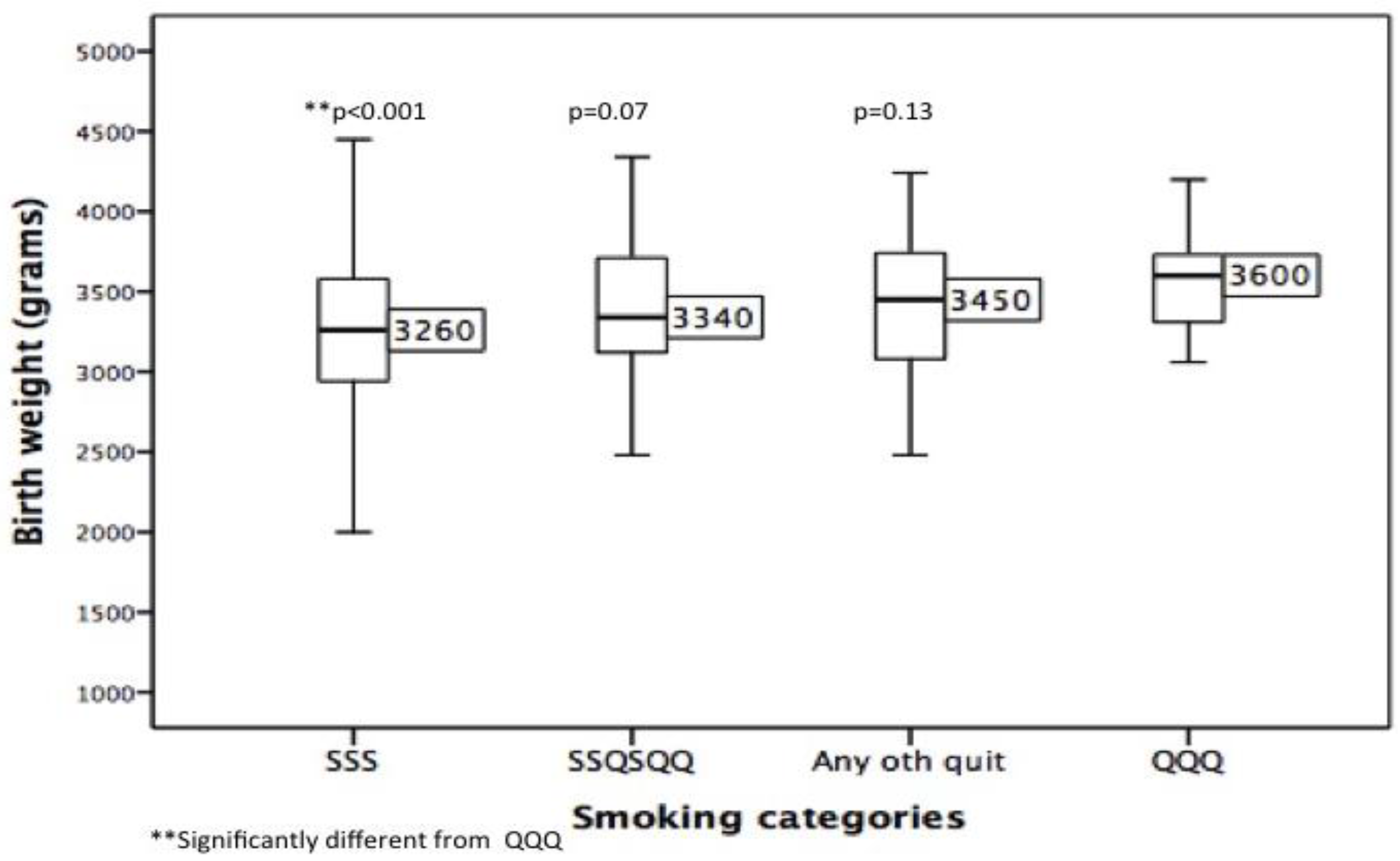
| Factors | N | % | Median | Minimum–Maximum | IQR a |
|---|---|---|---|---|---|
| Demographic | - | - | - | - | - |
| Maternal Age | 654 | - | 26.0 | 16–42 | 9 |
| First Pregnancy | 225 | 34.4 | - | - | - |
| General Medical Services Card | 363 | 55.5 | - | - | - |
| Single Parenthood | 236 | 36.1 | - | - | - |
| Number of Children Living in the Same House | - | - | - | - | - |
| One Child | 227 | 34.7 | - | - | - |
| Two Children | 137 | 20.9 | - | - | - |
| Three Children | 63 | 9.6 | - | - | - |
| Four or More Children | 38 | 5.8 | - | - | - |
| Smoking | - | - | - | - | - |
| Number of Years Smoking | 654 | - | 10 | 1–26 | 9 |
| Smoking Status at First Ante-Natal Visit (V1) | 654 | - | - | - | - |
| I smoke now | 127 | 19.5 | - | - | - |
| I smoke now but have cut down since becoming pregnant | 449 | 68.5 | - | - | - |
| I have stopped smoking since I thought I might be pregnant | 78 | 12.0 | - | - | - |
| Number of Cigarettes Smoked/Day at V1 | 576 | - | 10 | 1–60 | 9 |
| Partner Smoking at V1 (y/n) | 411 | 62.8 | - | - | - |
| Number of Cigarettes Smoked/Day by Partner at V1 | 383 | - | 15 | 1–60 | 10 |
| Number Of Cigarettes Smoked/Day at V2 | 565 | - | 10 | 1–60 | 10 |
| Number Of People Living in House (Other than Self or Partner) Who Smoke | 217 | - | - | - | - |
| One Additional Person | 121 | 18.5 | - | - | - |
| Two Additional Persons | 58 | 8.9 | - | - | - |
| Three Additional Persons | 18 | 2.8 | - | - | - |
| Four or More Additional Persons | 20 | 3.1 | - | - | - |
| Smoking Patterns across Three Visits | 638 | - | - | - | - |
| SSS b | 502 | 76.8 | - | - | - |
| QQQ c | 46 | 7.0 | - | - | - |
| SSQ/SQQ d | 52 | 8.0 | - | - | - |
| Any other Quit Attempt | 40 | 6.1 | - | - | - |
| Infant | - | - | - | - | - |
| Birth Weight | 654 | - | 3260 | 800–4800 | 680 |
| Baby Gender | 617 | 94.3 | - | - | |
| Males | 306 | 49.6 | - | - | - |
| Females | 311 | 50.4 | - | - | - |
| Single vs. Multiple | 617 | 94.3 | - | - | - |
| Singleton | 610 | 98.9 | - | - | - |
| Twin | 7 | 1.1 | - | - | - |
| Gestational Age at Birth | 635 | - | - | 26–42 | 2 |
| Variable | All | Pre Term < 37 Weeks | Term ≥ 37 Weeks | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | Beta | 95% CI b | p * | N | Beta | 95% CI b | p * | N | Beta | 95% CI b,* | p * | ||
| Smoking Categories across Three Visits | 579 | - | - | - | 35 | - | - | - | 544 | - | - | - | |
| QQQ | 42 | 288.0 | 153.1–423.0 | <0.001 * | 5 | 67.2 | −272.8–407.2 | 0.70 | 37 | 327.4 | 183.2–471.7 | <0.001 * | |
| S/Q | 88 | 146.9 | 49.5–244.1 | 0.003 * | 3 | 181.6 | −236.5–599.8 | 0.40 | 85 | 146.2 | 46.5–245.9 | 0.004 * | |
| SSS (reference) | 449 | - | - | - | 27 | - | - | - | 422 | - | - | - | |
| Additional Home Smokers | Four or More Home Smokers | 22 | −170.5 | −361.4–20.5 | 0.08 | 2 | −43.6 | −547.0–459.8 | 0.87 | 20 | −186.8 | −387.8–14.2 | 0.07 |
| Three Home Smokers | 40 | 58.9 | −89.7–207.4 | 0.44 | 2 | −280.4 | −806.6–245.7 | 0.30 | 38 | 82.3 | −71.4–236.0 | 0.29 | |
| Two Home Smokers | 81 | −93.4 | −208.5–21.7 | 0.11 | 7 | −387.6 | −723.1–(−52.1) | 0.02 * | 74 | −74.2 | −194.8–46.5 | 0.23 | |
| One Home Smoker | 288 | −25.0 | −109.3–59.2 | 0.56 | 13 | −374.2 | −646.4–(102.0) | 0.007 * | 275 | −7.2 | −94.7–80.3 | 0.87 | |
| No Home Smokers (reference) | 148 | - | - | - | 11 | - | - | - | 137 | - | - | - | |
| Baby Gender | Females | 294 | −155.1 | −224.6–(−85.7) | <0.001 * | 18 | −59.6 | −293.4–174.2 | 0.62 | 276 | −159.0 | −231.0–(−87.0) | <0.001 * |
| Males (reference) | 285 | - | - | - | 17 | - | - | - | 268 | - | - | - | |
| Gestational Age at Birth | 579 | 161.2 | - | <0.001 * | 35 | 124.5 | 56.9–192.2 | <0.001 * | 544 | 149.4 | 120.9–177.8 | <0.001 * | |
| Variable | All | Pre Term < 37 Weeks | Term ≥ 37 Weeks | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | Beta | 95% CI b | p * | N | Beta | 95% CI b | p * | N | Beta | 95% CI b,* | p * | ||
| Average number cigarettes Smoked at V1 and V2 (CiggsAv) | 590 | −8.4 | −11.7–(−5.2) | <0.001 * | 35 | −1.7 | −12.0–8.5 | 0.74 | 555 | −8.9 | −12.4–(−5.5) | <0.001 * | |
| Additional Home Smokers | Four or More Home Smokers | 22 | −180.3 | −369.5–8.8 | 0.06 | 2 | −73.1 | −575.6–429.3 | 0.78 | 20 | −195.4 | −394.5–3.7 | 0.05 |
| Three Home Smokers | 41 | 55.8 | −90.3–201.8 | 0.45 | 2 | −301.3 | −825.6–223.0 | 0.26 | 39 | 77.5 | −73.4–228.4 | 0.31 | |
| Two Home Smokers | 84 | −102.1 | −214.9–10.7 | 0.08 | 7 | −417.2 | −747.0–(−87.5) | 0.01 * | 77 | −80.7 | −198.8–37.5 | 0.18 | |
| One Home Smoker | 291 | −24.7 | −107.7–58.3 | 0.56 | 13 | −368.3 | −643.1–(−93.4) | 0.009 * | 278 | −7.3 | −93.5–78.8 | 0.87 | |
| No Home Smokers (reference) | 152 | - | - | - | 11 | - | - | - | 141 | - | - | - | |
| Baby Gender | Females | 296 | −141.5 | −09.9–(−73.2) | <0.001 * | 18 | −59.7 | −295.9–176.4 | 0.62 | 278 | −142.5 | −213.4–(−71.7) | <0.001 * |
| Males (reference) | 294 | - | - | - | 17 | - | - | - | 277 | - | - | - | |
| Gestational Age at Birth | 590 | 159.9 | 141.1–178.8 | <0.001 * | 35 | 119.9 | 54.3–185.4 | <0.001 * | 555 | 150.0 | 121.7–178.2 | <0.001 * | |
| Model | Males | Females | |||||||
|---|---|---|---|---|---|---|---|---|---|
| N | Beta | 95% CI a | p * | N | Beta | 95% CI a | p * | ||
| MODEL 1 | - | - | - | - | - | - | - | - | |
| Smoking Categories across Three Visits | QQQ | 13 | 346.7 | 100.6–592.7 | 0.006 * | 24 | 323.5 | 148.6–498.4 | <0.001 * |
| S/Q | 42 | 66.7 | −78.1–211.6 | 0.37 | 43 | 218.0 | 80.9–355.1 | 0.002 | |
| SSS (reference) | 213 | - | - | - | 209 | - | - | - | |
| Additional Home Smokers | Four or More Home Smokers | 10 | −248.0 | −539.2–43.2 | 0.095 | 10 | −137.3 | −413.0–138.4 | 0.33 |
| Three Home Smokers | 22 | 58.3 | −153.7–270.4 | 0.59 | 16 | 119.7 | −105.3–344.7 | 0.30 | |
| Two home smokers | 36 | −26.8 | −205.0–151.5 | 0.77 | 38 | −104.6 | −269.0–59.8 | 0.21 | |
| One home smoker | 134 | −26.9 | −156.7–102.9 | 0.69 | 141 | 15.4 | −102.8–133.6 | 0.80 | |
| No Home Smokers (reference) | 66 | - | - | - | 71 | - | - | - | |
| Gestational Age at Birth | 268 | 154.1 | 113.4–194.8 | <0.001 * | 276 | 143.3 | 103.0–183.5 | <0.001 * | |
| MODEL 2 | - | - | - | - | - | - | - | - | |
| Average number Cigarettes Smoked at V1 and V2 | 277 | −9.5 | −14.3–(−4.7) | 0.000 | 278 | −8.1 | −13.0–(−3.2) | 0.001 | |
| Additional Home Smokers | Four or More Home Smokers | 10 | −261.9 | −547.2–23.4 | 0.072 | 10 | −128.8 | −405.9–148.2 | 0.36 |
| Three Home Smokers | 23 | 65.3 | −138.6–269.1 | 0.53 | 16 | 96.5 | −131.1–324.2 | 0.41 | |
| Two Home Smokers | 39 | −45.2 | −215.8–125.4 | 0.60 | 38 | −118.3 | −284.4–47.7 | 0.16 | |
| One Home Smoker | 136 | −18.8 | −144.2–106.7 | 0.77 | 142 | 4.7 | −114.3–123.8 | 0.94 | |
| No Home Smokers (reference) | 69 | - | - | - | 72 | - | - | - | |
| Gestational Age at Birth | 277 | 153.5 | 113.9–193.2 | <0.001 * | 278 | 146.9 | 106.2–187.6 | <0.001 * | |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hayes, C.; Kearney, M.; O’Carroll, H.; Zgaga, L.; Geary, M.; Kelleher, C. Patterns of Smoking Behaviour in Low-Income Pregnant Women: A Cohort Study of Differential Effects on Infant Birth Weight. Int. J. Environ. Res. Public Health 2016, 13, 1060. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph13111060
Hayes C, Kearney M, O’Carroll H, Zgaga L, Geary M, Kelleher C. Patterns of Smoking Behaviour in Low-Income Pregnant Women: A Cohort Study of Differential Effects on Infant Birth Weight. International Journal of Environmental Research and Public Health. 2016; 13(11):1060. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph13111060
Chicago/Turabian StyleHayes, Catherine, Morgan Kearney, Helen O’Carroll, Lina Zgaga, Michael Geary, and Cecily Kelleher. 2016. "Patterns of Smoking Behaviour in Low-Income Pregnant Women: A Cohort Study of Differential Effects on Infant Birth Weight" International Journal of Environmental Research and Public Health 13, no. 11: 1060. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph13111060






