Patterns of Bacillary Dysentery in China, 2005–2010
Abstract
:1. Introduction
2. Materials and Methods
2.1. Data Collection
2.2. Ethical Statement
2.3. Definition
2.4. Methodology
2.4.1. Analysis on Geographic and Temporal Patterns
2.4.2. Identification of Endemically High-Risk Regions and Their Ecological Drivers
2.4.3. Exploration of Disparities of Urban and Rural Incidence
2.4.4. Meta-Analysis on Prevalence of Shigella Species
3. Results
3.1. Geographic and Temporal Patterns

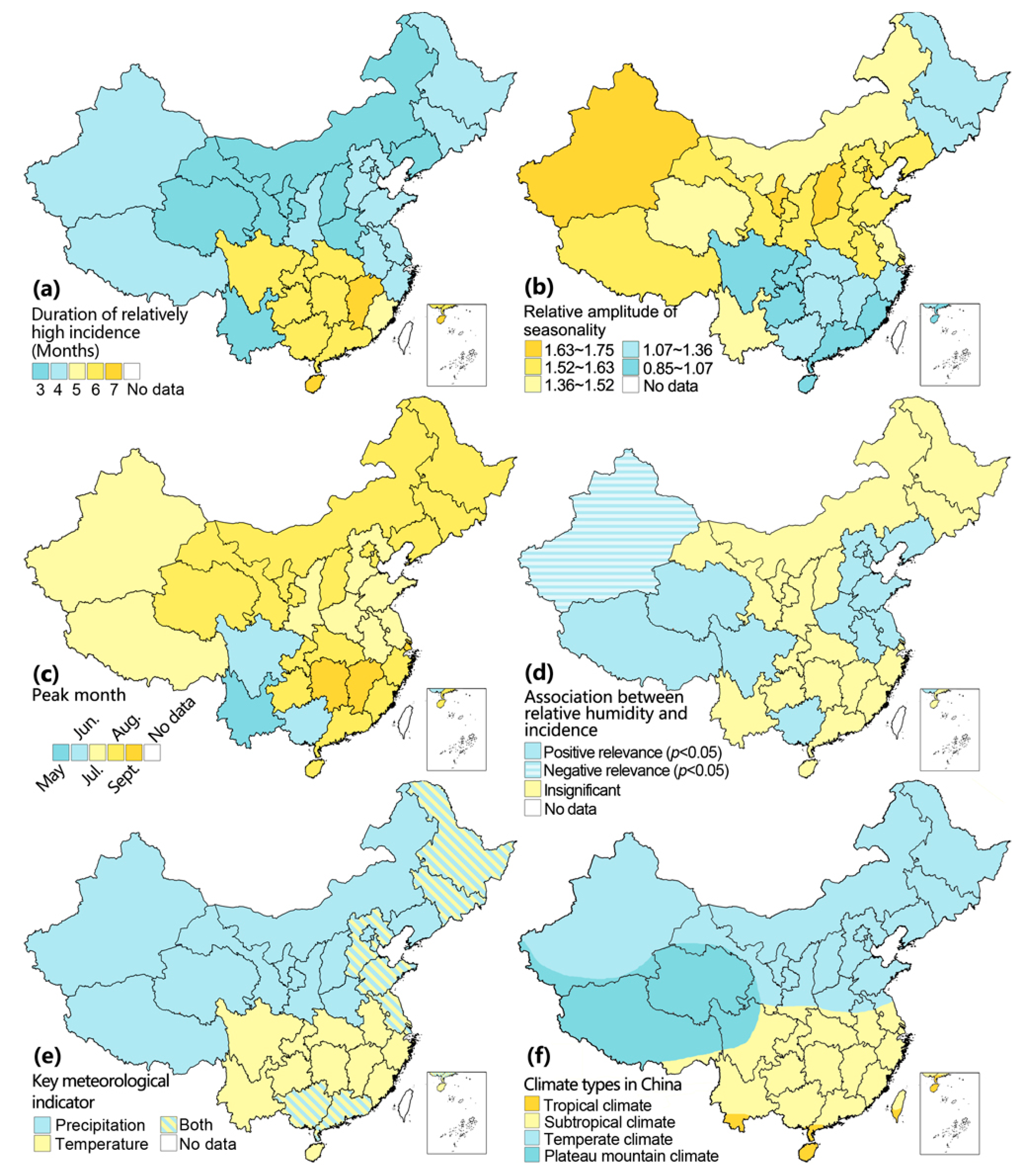
3.2. Environmental Drivers in High-Risk Regions

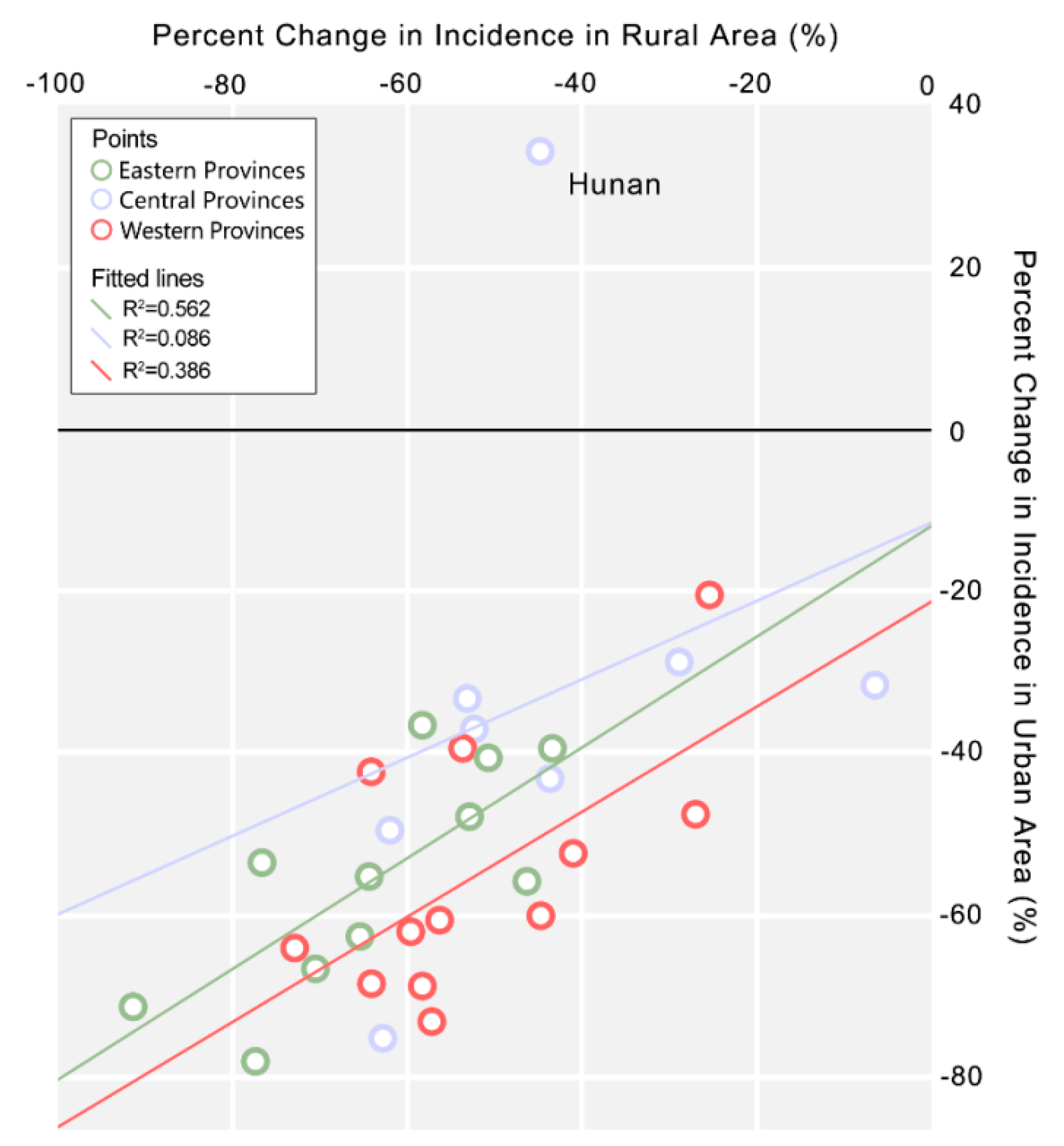
3.3. Disparity of Urban and Rural Incidence
| Urbanization Level | Bacillary Dysentery Incidence | ||
|---|---|---|---|
| Urban > Rural a (n = 165) | Urban < Rural b (n = 52) | Variable Trend c (n = 56) | |
| Median (%) (Q1, Q3) | 49.40 (40.00, 62.15) | 38.95 (34.03, 48.30) | 42.15 (35.28, 56.15) |
| Ranked Top 100 (n (%)) | 77 (46.67) | 7 (13.46) | 16 (28.57) |
| Ranked 200+ d (n (%)) | 29 (17.58) | 24 (46.15) | 19 (33.93) |
3.4. Prevalence of Shigella Species

4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CDC | Center for Disease Control and Prevention |
| NIDR | National Notifiable Infectious Disease Reporting System |
| DEM | Digital Elevation Model |
| CGIAR-CSI | The Consultative Group for International Agricultural Research Consortium for Spatial Information |
| NGCC | National Geomatics Center of China |
| GRP | Gross Regional Product |
References
- Kotloff, K.L.; Winickoff, J.P.; Ivanoff, B.; Clemens, J.D.; Swerdlow, D.L.; Sansonetti, P.J.; Sansonetti, P.J.; Adak, G.K.; Levine, M.M. Global burden of Shigella infections: Implications for vaccine development and implementation of control strategies. Bull. World Health Organ. 1999, 77, 651–666. [Google Scholar] [PubMed]
- Von Seidlein, L.; Kim, D.R.; Ali, M.; Lee, H.; Wang, X.; Thiem, V.D.; Canh, D.G.; Chaicumpa, W.; Agtini, M.D.; Hossain, A.; et al. A multicentre study of Shigella diarrhoea in six Asian countries: Disease burden, clinical manifestations, and microbiology. PLoS Med. 2006. [Google Scholar] [CrossRef] [PubMed]
- DuPont, H.L.; Levine, M.M.; Hornick, R.B.; Formal, S.B. Inoculum size in shigellosis and implications for expected mode of transmission. J. Infect. Dis. 1989, 159, 1126–1128. [Google Scholar] [CrossRef] [PubMed]
- Carlton, E.J.; Liang, S.; McDowell, J.Z.; Li, H.; Luo, W.; Remais, J.V. Regional disparities in the burden of disease attributable to unsafe water and poor sanitation in China. Bull. World Health Organ. 2012, 90, 578–587. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Hu, W.; Zhang, Y.; Wang, X.; Tong, S.; Zhou, M. Spatiotemporal pattern of bacillary dysentery in China from 1990 to 2009: What is the driver behind? PLoS ONE 2014. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Tao, F.; Xiao, D.; Lee, H.; Deen, J.; Gong, J.; Zhao, Y.; Zhou, W.; Li, W.; Shen, B.; et al. Trend and disease burden of bacillary dysentery in China (1991–2000). Bull. World Health Organ. 2006, 84, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Guan, P.; Guo, J.; Wang, P.; Zhou, B. Investigating the effects of climate variations on bacillary dysentery incidence in northeast China using ridge regression and hierarchical cluster analysis. BMC Infect. Dis. 2008. [Google Scholar] [CrossRef] [PubMed]
- Chompook, P.; Samosornsuk, S.; von Seidlein, L.; Jitsanguansuk, S.; Sirima, N.; Sudjai, S.; Mangjit, P.; Kim, D.R.; Wheeler, J.G.; Todd, J.; et al. Estimating the burden of bacillary dysentery in Thailand: 36-month population-based surveillance study. Bull. World Health Organ. 2005, 83, 739–746. [Google Scholar] [PubMed]
- Zhang, Y.; Bi, P.; Hiller, J.E. Weather and the transmission of bacillary dysentery in Jinan, northern China: A time-series analysis. Public Health Rep. 2008, 123, 61–66. [Google Scholar] [PubMed]
- Checkley, W.; Epstein, L.D.; Gilman, R.H.; Figueroa, D.; Cama, R.I.; Patz, J.A.; Black, R.E. Effects of EI Niño and ambient temperature on hospital admissions for diarrhoeal diseases in Peruvian children. Lancet 2000, 355, 442–450. [Google Scholar] [CrossRef]
- Singh, R.B.; Hales, S.; Wet, N.; Raj, R.; Hearnden, M.; Weinstein, P. The influence of climate variation and change on diarrheal disease in the Pacific Islands. Environ. Health Perspect. 2001, 109, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Patrick, M.E.; Christiansen, L.E.; Wainø, M.; Ethelberg, S.; Madsen, H.; Wegener, H.C. Effects of climate on incidence of Campylobacter spp. in humans and prevalence in broiler flocks in Denmark. Appl. Environ. Microbiol. 2004, 70, 7474–7480. [Google Scholar] [CrossRef] [PubMed]
- Kovats, R.S.; Edwards, S.J.; Hajat, S.; Armstrong, B.G.; Ebi, K.L.; Menne, B. The effects of temperature on food poisoning: A time-series analysis of salmonellosis in ten European countries. Epidemiol. Infect. 2004, 132, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Hall, G.V.; D’Souza, R.M.; Kirk, M.D. Foodborne disease in the New Millennium: Out of the frying pan and into the fire? Med. J. Aust. 2002, 177, 614–618. [Google Scholar] [PubMed]
- D'Souza, R.M.; Becker, N.G.; Hall, G.; Moodie, K.B. Does ambient temperature affect foodborne disease? Epidemiology 2004, 15, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Bi, P.; Hiller, J.E.; Sun, Y.; Ryan, P. Climate variations and bacillary dysentery in northern and southern cities of China. J. Infect. 2007, 55, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Wang, L.; Sun, W.; Hou, X.; Yang, H.; Sun, L.; Xu, S.; Sun, Q.; Zhang, J.; Song, H.; Lin, H. Identifying high-risk areas of bacillary dysentery and associated meteorological factors in Wuhan, China. Sci. Rep. 2013. [Google Scholar] [CrossRef] [PubMed]
- Jia, L.; Li, X.; Liu, G. Analysis of the association between meteorological factors and incidence of dysentery in Beijing. Modern Prev. Med. 2007, 34, 2470–2471. [Google Scholar]
- Kelly-Hope, L.A.; Alonso, W.J.; Thiem, V.D.; Anh, D.D.; Canh, D.G.; Lee, H.; Smith, D.L.; Miller, M.A. Geographical distribution and risk factors associated with enteric diseases in Vietnam. Am. J. Trop. Med. Hyg. 2007, 76, 706–712. [Google Scholar] [PubMed]
- Gupta, A.; Polyak, C.S.; Bishop, R.D.; Sobel, J.; Mintz, E.D. Laboratory-confirmed shigellosis in the United States, 1989–2002: Epidemiologic trends and patterns. Clin. Infect. Dis. 2004, 38, 1372–1377. [Google Scholar] [CrossRef] [PubMed]
- Ranjbar, R.; Dallal, M.S.; Pourshafie, M.R.; Aslani, M.M.; Majdzadeh, R. Serogroup distribution of Shigella in Tehran. Iran. J. Public Health 2004, 33, 32–35. [Google Scholar]
- Seol, S.Y.; Kim, Y.T.; Jeong, Y.S.; Oh, J.Y.; Kang, H.Y.; Moon, D.C.; Kim, J.; Lee, Y.C.; Cho, D.T.; Lee, J.C. Molecular characterization of antimicrobial resistance in Shigella sonnei isolates in Korea. J. Med. Microbiol. 2006, 55, 871–877. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Wang, Y.; Li, C.; Tung, S.K.; Chiou, C. Epidemiology and evolution of genotype and antimicrobial resistance of an imported Shigella sonnei clone circulating in central Taiwan. Diagn. Microbiol. Infect. Dis. 2007, 58, 469–475. [Google Scholar]
- Vinh, H.; Nhu, N.T.; Nga, T.V.; Duy, P.T.; Campbell, J.I.; Hoang, N.V.; Boni, M.F.; My, P.V.T.; Parry, C.; Nga, T.T.T.; et al. A changing picture of bacillary dysentery in southern Vietnam: Shifting species dominance, antimicrobial susceptibility and clinical presentation. BMC Infect. Dis. 2009. [Google Scholar] [CrossRef] [PubMed]
- Qu, M.; Deng, Y.; Zhang, X.; Liu, G.; Huang, Y.; Lin, C.; Li, J.; Yan, H.; Li, X.; Jia, L.; et al. Etiology of acute diarrhea due to enteropathogenic bacteria in Beijing, China. J. Infect. 2012, 65, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Das, S.K.; Ahmed, S.; Ferdous, F.; Farzana, F.D.; Chisti, M.J.; Leung, D.T.; Malek, M.A.; Talukder, K.A.; Bardhan, P.K.; Salam, M.A.; et al. Changing emergence of Shigella sero-groups in Bangladesh: Observation from four different diarrheal disease hospitals. PLoS ONE 2013. [Google Scholar] [CrossRef] [PubMed]
- Ji, G.Q.; Shi, J.X.; Zhang, W.Z.; Ma, Y.X.; Zhang, S.J. Epidemiological characteristics of bacillary dysentery in Shunyi District, Beijing from 1997 to 2008. Cap. J. Public Health 2009, 3, 214–215. [Google Scholar]
- Tang, F.; Cheng, Y.; Bao, C.; Hu, J.; Liu, W.; Liang, Q.; Wu, Y.; Norris, J.; Peng, Z.; Yu, R.; et al. Spatio-temporal trends and risk factors for Shigella from 2001 to 2011 in Jiangsu Province, People’s Republic of China. PLoS ONE 2014. [Google Scholar] [CrossRef] [PubMed]
- Chang, Z.; Lu, S.; Chen, L.; Jin, Q.; Yang, J. Causative species and serotypes of bacillary dysentery in mainland China: Systematic review and meta-analysis. PLoS ONE 2012. [Google Scholar] [CrossRef]
- Yang, G.; Stroup, D.F.; Thacker, S.B. National public health surveillance in China: Implications for public health in China and the United States. Biomed. Environ. Sci. 1997, 10, 1–13. [Google Scholar] [PubMed]
- Sui, J.; Zhang, J.; Sun, J.; Chang, Z.; Zhang, W.; Wang, Z. Surveillance of bacillary dysentery in China, 2009. Dis. Surveill. 2010, 25, 947–950. [Google Scholar]
- DerSimonian, R.; Laird, N. Meta-analysis in clinical trials. Control. Clin. Trials 1986, 7, 177–188. [Google Scholar] [CrossRef]
- Abrams, K.R.; Jones, D.R.; Jones, D.R.; Sheldon, T.A.; Song, F. General fixed effect model—The inverse variance-weighted method. In Methods for Meta-Analysis in Medical Research; J. Wiley: Chichester, England, UK, 2000; pp. 58–62. [Google Scholar]
- Wang, J.A.; Zuo, W. Geographic Atlas of China; SinoMaps Press: Beijing, China, 2010; pp. 34–35. [Google Scholar]
- Liu, J.; Zhang, X.; Wang, J.; Du, X.; Zeng, J.; Xu, J. Contrast analysis on infectious diseases in Tibet and backland. Acta Acad. Med. Militaris Tertiae 2005, 27, 2303–2304. [Google Scholar]
- Li, K. Analysis on 1954 Bacillary dysentery cases reported in Ningnan county from 1997 to 2006. J. Prevent. Med. Inf. 2007, 23, 579–581. [Google Scholar]
- Li, A. Analysis on the epidemic trend of legal type A and B contagious diseases in Jiuquan during 2004 to 2010. J. Med. Pest Control 2012, 28, 623–625. [Google Scholar]
- Zhao, H.; Gu, X.; Zhang, Q.; Song, X. Evaluation on the benefit of water improvement in Kashi, Xinjiang. Chin. J. Public Health 1998, 14, 285–286. [Google Scholar]
- Aishan, N. The existed problem and solution on safe drinking water project in rural and pastoral areas of Xinjiang. J. Hyd. Eng. 2005, 4, 661–663. [Google Scholar]
- Gao, T.; Liu, G.; Li, X.; Jia, L.; Liu, Y.; Tang, Y. Analysis about epidemic situation of dysentery near upon fourteen years in Beijing. Chin. J. Prevent. Med. 2007, 41, 54–57. [Google Scholar]
- Liu, S.; Wang, L.; Wang, X.; Zhang, C.; Guo, Q.; Zhou, M.; Ma, J. Evaluation on management and quality of communicable diseases network direct reporting in China. Dis. Surveill. 2009, 26, 392–397. [Google Scholar]
- Nie, C.; Li, H.; Yang, L.; Zhong, G.; Zhang, L. Socio-economic factors of bacillary dysentery based on spatial correlation analysis in Guangxi Province, China. PLoS ONE 2014. [Google Scholar] [CrossRef] [PubMed]
- Herikstad, H.; Yang, S.; van Gilder, T.J.; Vugia, D.; Hadler, J.; Blake, P.; Deneen, V.; Shiferaw, B.; Angulo, F.J.; et al. A population-based estimate of the burden of diarrhoeal illness in the United States: FoodNet, 1996–1997. Epidemiol. Infect. 2002, 129, 9–17. [Google Scholar] [PubMed]
- Hall, G.V.; Kirk, M.D.; Ashbolt, R.; Stafford, R.; Lalor, K. Frequency of infectious gastrointestinal illness in Australia, 2002: Regional, seasonal and demographic variation. Epidemiol. Infect. 2006, 134, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Simonsen, J.; Frisch, M.; Ethelberg, S. Socioeconomic risk factors for bacterial gastrointestinal infections. Epidemiology 2008, 19, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Mead, P.S.; Slutsker, L.; Dietz, V.; McCaig, L.F.; Bresee, J.S.; Shapiro, C.; Griffin, P.M.; Tauxe, R.V. Food-related illness and death in the United States. Emerg. Infect. Dis. 1999, 5, 607–625. [Google Scholar] [CrossRef] [PubMed]
- Scallan, E.; Hoekstra, R.M.; Angulo, F.J.; Tauxe, R.V.; Widdowson, M.A.; Roy, S.L.; Jones, J.L.; Griffin, P.M. Foodborne illness acquired in the United States—Major pathogens. Emerg. Infect. Dis. 2011, 17, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Xiao, G.; Xu, C.; Wang, J.; Yang, D.; Wang, L. Spatial-temporal pattern and risk factor analysis of bacillary dysentery in the Beijing-Tianjin-Tangshan urban region of China. BMC Public Health 2014. [Google Scholar] [CrossRef] [PubMed]
- Bagamboula, C.F.; Uyttendaele, M.; Debevere, J. Growth and survival of Shigella sonnei and S. flexneri in minimal processed vegetables packed under equilibrium modified atmosphere and stored at 7 °C and 12 °C. Food Microbiol. 2002, 19, 529–536. [Google Scholar] [CrossRef]
- Zhou, Y.; Ma, L.; Xu, W.; Zhang, H. A case-control study of bacillary dysentery in Dongcheng District Beijing City. Chin. J. Public Health 2007, 23, 1393–1394. [Google Scholar]
- Jia, L.; Dou, X.; Wu, X.; Wu, J.; Li, X.; Wang, Q.; He, X.; Xu, W.; Sui, J. Control study of risk factors of bacillary dysentery cases in Beijing. Dis. Surveill. 2007, 22, 597–598. [Google Scholar]
- Hu, X.; Cook, S.; Salazar, M.A. Internal migration and health in China. Lancet 2008, 372, 1717–1719. [Google Scholar] [CrossRef]
- Gong, P.; Liang, S.; Carlton, E.J.; Jiang, Q.; Wu, J.; Wang, L.; Remais, J.V. Urbanization and health in China. Lancet 2012, 379, 843–852. [Google Scholar] [CrossRef]
- Zhong, X.; Zhang, Q. Epidemiological analysis on bacterial dysentery in Dongguan city in 2003. J. Trop. Med. 2005, 5, 512–513. [Google Scholar]
- Shi, W.; Shen, H.; Cui, J.; Xi, Y.; Su, H.; Yu, L. Epidemiological analysis on bacillary dysentery during 2005 and 2009, in Minhang District, Shanghai. Shanghai J. Prevent. Med. 2011, 23, 419–420. [Google Scholar]
- Chompook, P.; Todd, J.; Wheeler, J.G.; Von Seidlein, L.; Clemens, J.; Chaicumpa, W. Risk factors for shigellosis in Thailand. Int. J. Infect. Dis. 2006, 10, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.R.; Ali, M.; Thiem, V.D.; Park, J.K.; Von Seidlein, L.; Clemens, J. Geographic analysis of shigellosis in Vietnam. Health Place 2008, 14, 755–767. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Mauzerall, D.L.; Zhu, T.; Liang, S.; Ezzati, M.; Remais, J.V. Environmental health in China: Progress towards clean air and safe water. Lancet 2010, 375, 1110–1119. [Google Scholar] [CrossRef]
- Yu, H.; Chang, Z.; Zhang, L.; Zhang, J.; Li, Z.; Xu, J.; Ran, L. Analysis on the status of Shigella spp. Antimicrobial resistance through data from the National Shigellosis Surveillance System in China, in 2005. Chin. J. Epidemiol. 2007, 28, 370–373. [Google Scholar]
- Ram, P.K.; Crump, J.A.; Gupta, S.K.; Miller, M.A.; Mintz, E.D. Part II. Analysis of data gaps pertaining to Shigella infections in low and medium human development index countries, 1984–2005. Epidemiol. Infect. 2008, 136, 577–603. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Luo, Y.; Li, J.; Lin, L.; Ma, Y.; Hu, C.; Jin, S.; Ran, L.; Cui, S. Wide dissemination of multidrug-resistant Shigella isolates in China. J. Antimicrob. Chemother. 2011, 66, 2527–2535. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, H.; Si, Y.; Wang, X.; Gong, P. Patterns of Bacillary Dysentery in China, 2005–2010. Int. J. Environ. Res. Public Health 2016, 13, 164. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph13020164
Zhang H, Si Y, Wang X, Gong P. Patterns of Bacillary Dysentery in China, 2005–2010. International Journal of Environmental Research and Public Health. 2016; 13(2):164. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph13020164
Chicago/Turabian StyleZhang, Han, Yali Si, Xiaofeng Wang, and Peng Gong. 2016. "Patterns of Bacillary Dysentery in China, 2005–2010" International Journal of Environmental Research and Public Health 13, no. 2: 164. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph13020164






