Interaction Mechanism between Antibiotics and Humic Acid by UV-Vis Spectrometry
Abstract
:1. Introduction
2. Materials and Methods
2.1. Instruments and Chemicals
2.2. Preparation of Humic Acid
2.3. Experimental Methods
2.3.1. UV-Vis Spectroscopy Experiments of Antibiotic Interactions with Humic Acid
2.3.2. Fluorescence Quenching Experiments of Humic Acid and Antibiotics
2.3.3. Environmental Factors Impact on the Experiment
3. Results and Discussion
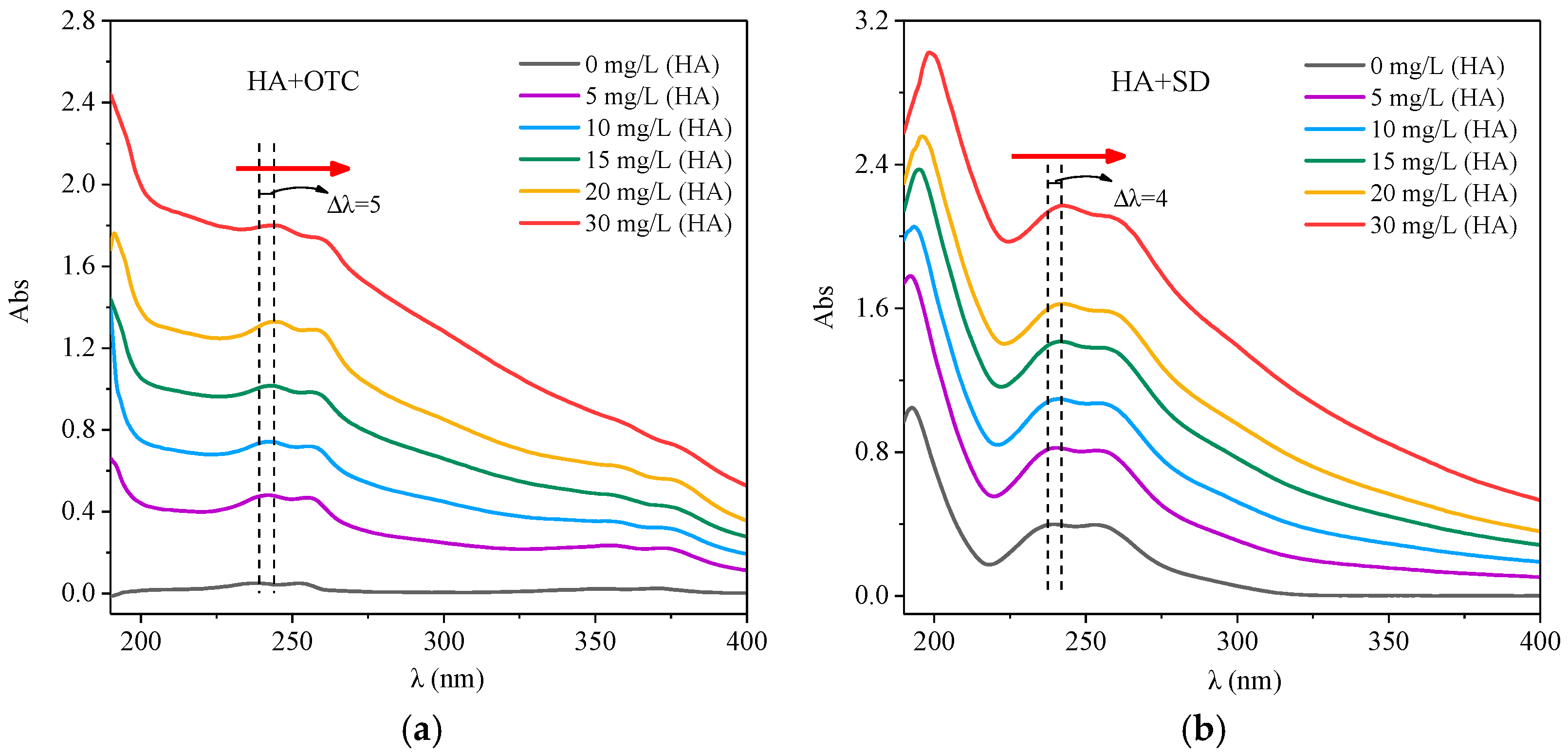
3.1. UV-Visible Spectroscopy Analysis of Antibiotics and HA
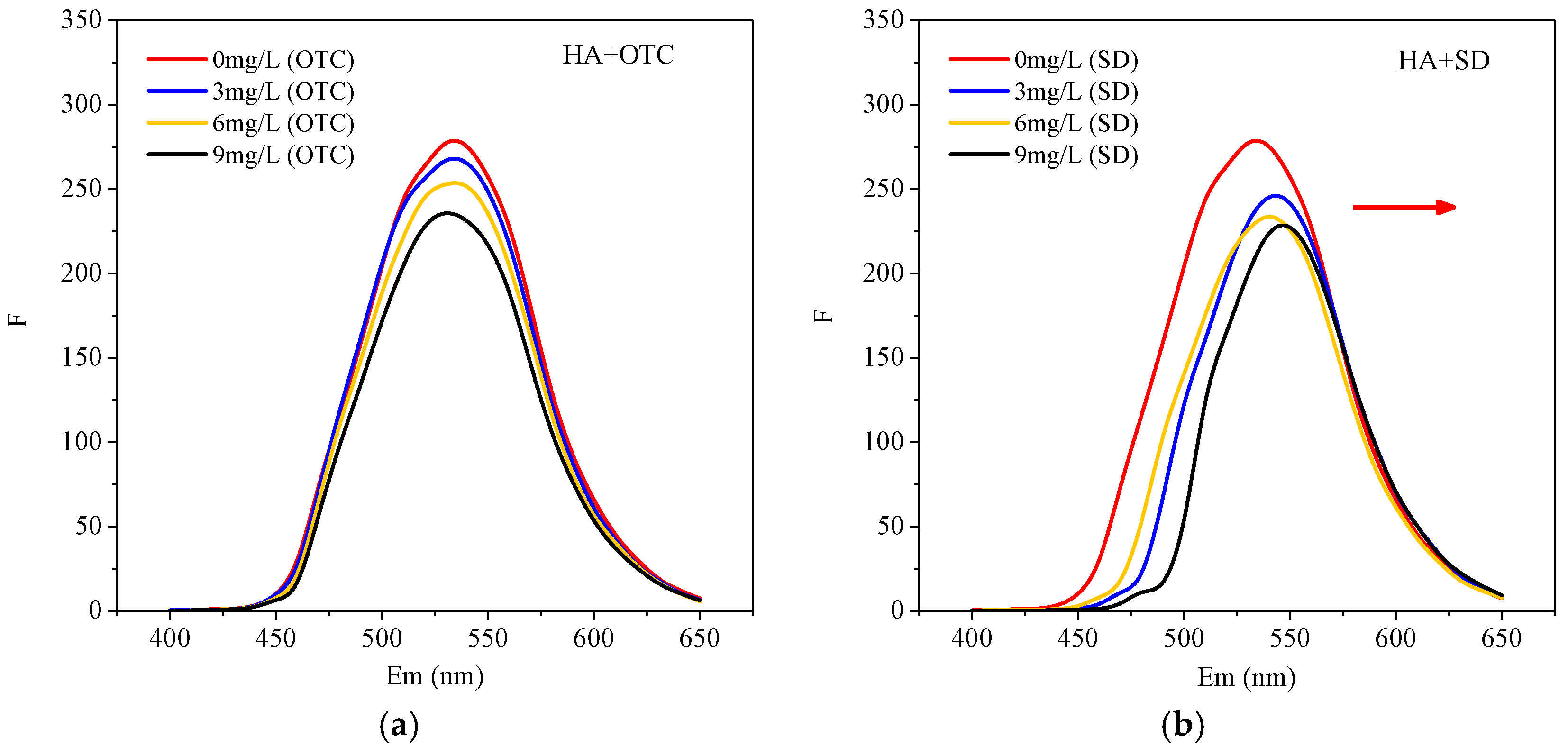
3.2. Fluorescence Evidence of Interactions between the Different Antibiotics and HA
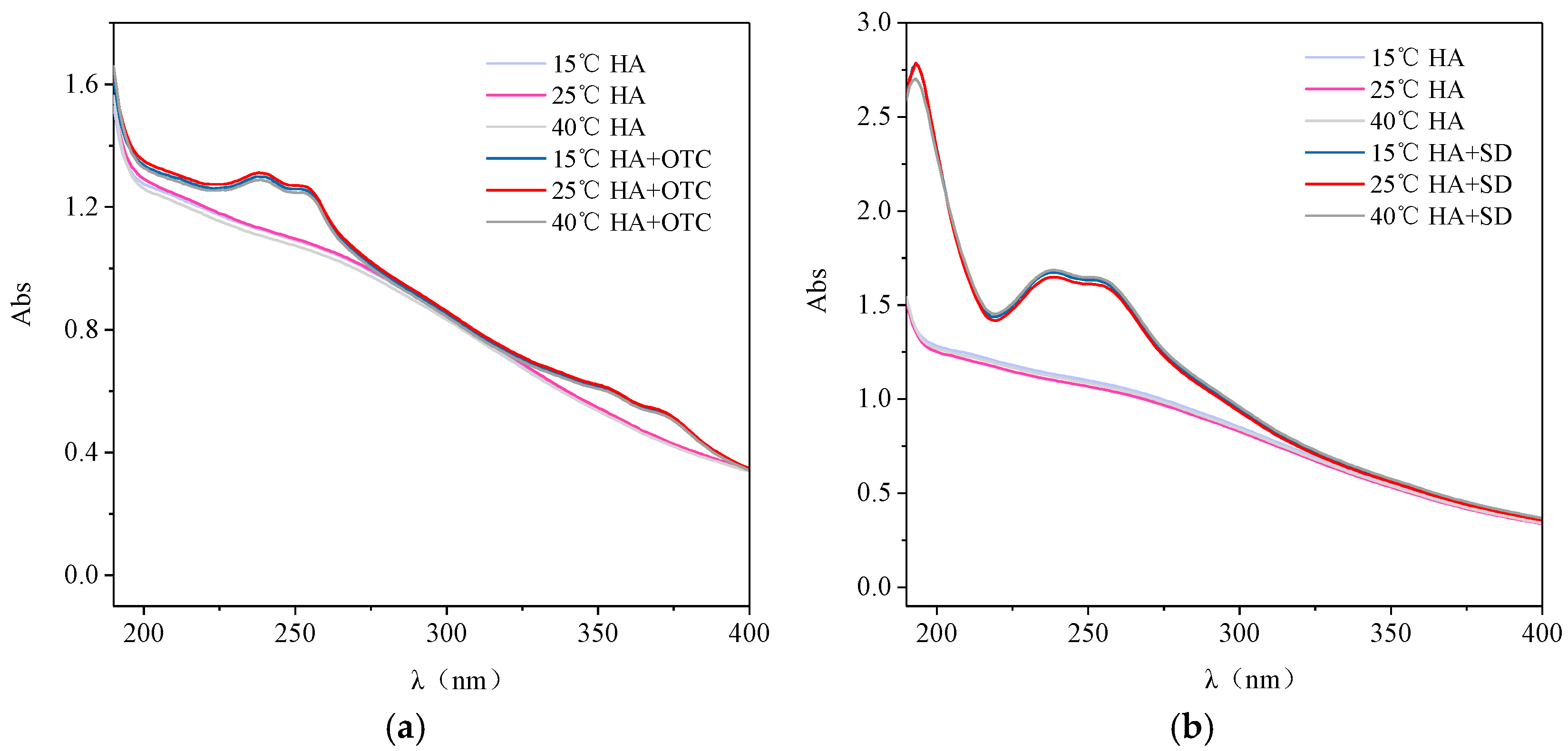
3.3. The Effect of Temperature on the Action of Antibiotics and HA
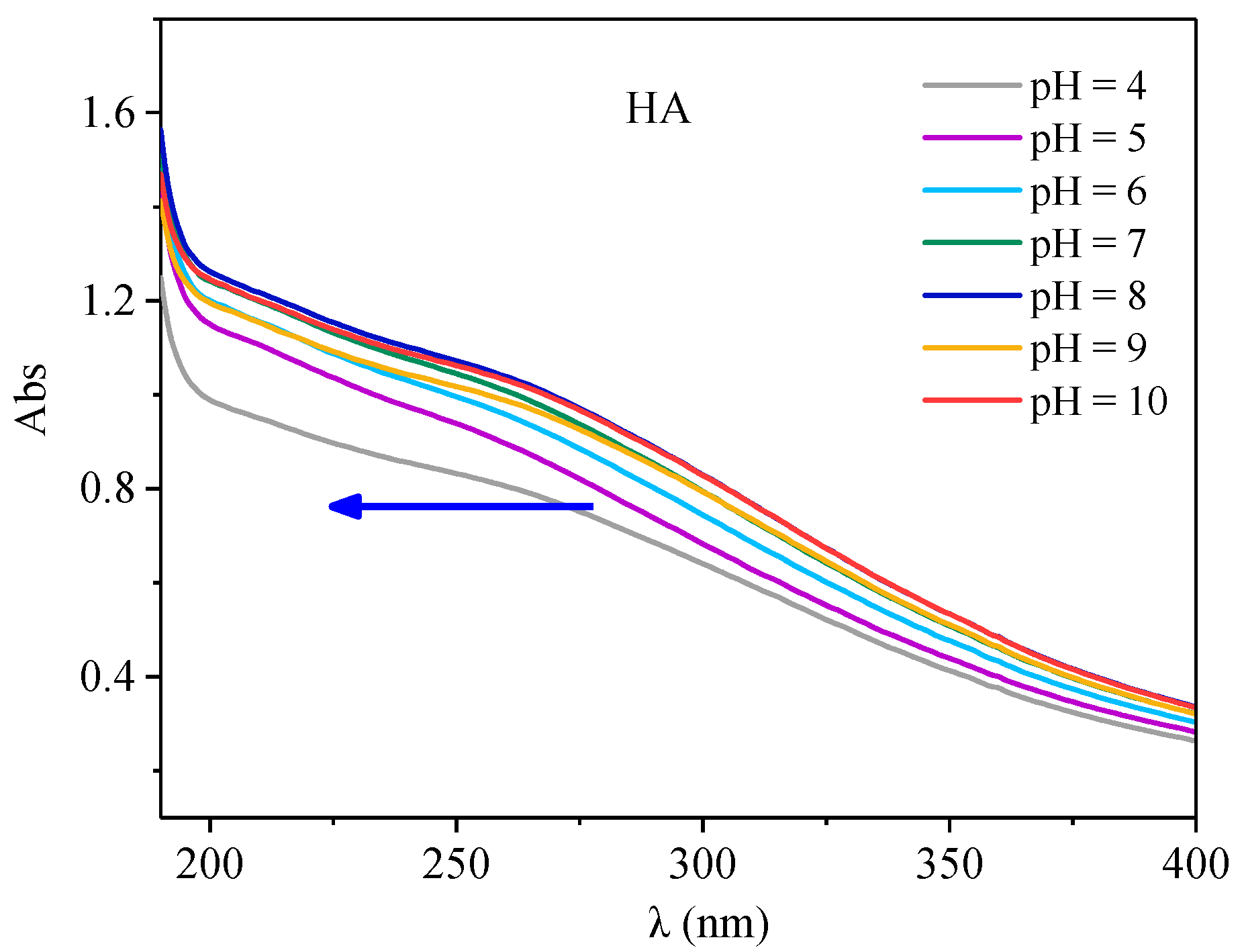
3.4. Effect of PH on Antibiotic and HA Action
3.4.1. Effect of PH on HA UV-Vis Spectra
3.4.2. Effect of pH on the Interaction between Two Antibiotics and HA
3.4.3. Effect of pH on E4/E6 Values
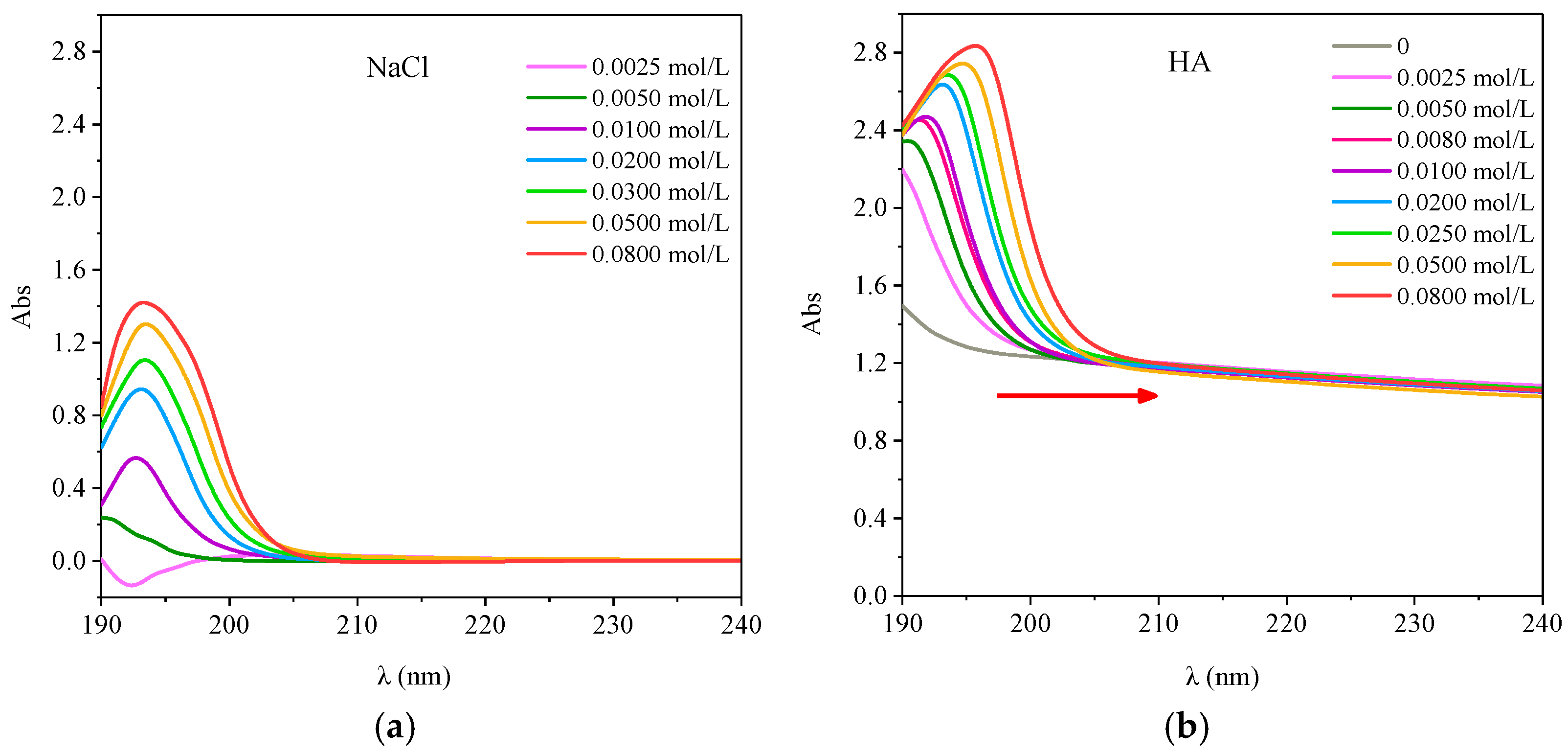
3.5. Effect of Ionic Strength on Antibiotics and HA
3.5.1. Effect of Ionic Strength on UV-Visible Spectra of HA
3.5.2. Effect of Ionic Strength on the Interaction between the Two Antibiotics and HA
3.5.3. Effect of Ionic Strength on E4/E6 Values
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Islas-Espinoza, M.; Wexler, M.; Bond, P.L. Soil Bacterial Consortia and Previous Exposure Enhance the Biodegradation of Sulfonamides from Pig Manure. Microb. Ecol. 2012, 64, 140–151. [Google Scholar] [CrossRef] [PubMed]
- Juan-García, A.; Font, G.; Picó, Y. Simultaneous determination of different classes of antibiotics in fish and livestock by CE-MS. Electrophoresis 2010, 28, 4180–4191. [Google Scholar] [CrossRef] [PubMed]
- Nicolaou, K.C.; Boddy, C.N.; Bräse, S.; Winssinger, N. Chemistry, Biology, and Medicine of the Glycopeptide Antibiotics. Angew. Chem. Int. Ed. 1999, 30, 2096–2152. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, X.H.; Huang, Y.; Wang, H. Comprehensive evaluation of pharmaceuticals and personal care products (PPCPs) in typical highly urbanized regions across China. Environ. Pollut. 2015, 204, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Sarmah, A.K.; Meyer, M.T.; Boxall, A.B. A global perspective on the use, sales, exposure pathways, occurrence, fate and effects of veterinary antibiotics (VAs) in the environment. Chemosphere 2006, 65, 725–759. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, R.; Ternes, T.; Haberer, K.; Kratz, K.L. Occurrence of antibiotics in the aquatic environment. Sci. Total Environ. 1999, 225, 109–118. [Google Scholar] [CrossRef]
- Petrović, M.; Hernando, M.D.; Díazcruz, M.S.; Barceló, D. Liquid chromatography-tandem mass spectrometry for the analysis of pharmaceutical residues in environmental samples: A review. J. Chromatogr. A 2005, 1067, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Sacher, F.; Lange, F.T.; Brauch, H.J.; Blankenhorn, I. Pharmaceuticals in groundwaters analytical methods and results of a monitoring program in Baden-Wurttemberg, Germany. J. Chromatogr. A 2001, 938, 199–210. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, J.; Zhong, Z.; Guo, C.; Li, L.; He, Y.; Fan, W.; Chen, Y. Degradation of sulfonamides antibiotics in lake water and sediment. Environ. Sci. Pollut. Res. Int. 2013, 20, 2372–2380. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Dong, Y.H.; Wang, H. Residues of veterinary antibiotics in manures from feedlot livestock in eight provinces of China. Sci. Total Environ. 2010, 408, 1069–1075. [Google Scholar] [CrossRef] [PubMed]
- Wu, N.; Qiao, M.; Zhang, B.; Cheng, W.-D.; Zhu, Y.-G. Abundance and Diversity of Tetracycline Resistance Genes in Soils Adjacent to Representative Swine Feedlots in China. Environ. Sci. Technol. 2010, 44, 6933–6939. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Z.J.; Qi, R.H.; Long, J.; Fan, F.F.; Fan, F.L.; Liang, Y.C. Influence of particle size on the adsorption of oxytetracycline on black and red soils. J. Agro-Environ. Sci. 2010, 29, 2357–2362. [Google Scholar]
- Bing, L.; Tong, Z. Biodegradation and adsorption of antibiotics in the activated sludge process. Environ. Sci. Technol. 2010, 44, 3468–3473. [Google Scholar]
- Chen, W.R.; Huang, C.H. Adsorption and transformation of tetracycline antibiotics with aluminum oxide. Chemosphere 2010, 79, 779–785. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Yang, S.; Fang, J.; Wang, Z.; Chen, Y.; Zhang, D.; Yang, C. Characterizing the Interaction between Antibiotics and Humic Acid by Fluorescence Quenching Method. Int. J. Env. Res. Public Health 2018, 15, 1458. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Xiang, L.; MO, C.H.; Li, W.Y.; Cai, Q.Y.; Huang, X.P.; Wu, X.L.; Li, H. Migration Behavior and Influence Factors of Quinolone Antibiotics in Soil. J. Agro-Environ. Sci. 2014, 33, 1345–1350. [Google Scholar]
- Blackwell, P.A.; Kay, P.; Ashauer, R.; Boxall, A.B.A. Effects of agricultural conditions on the leaching behaviour of veterinary antibiotics in soils. Chemosphere 2009, 75, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Carmosini, N.; Lee, L.S. Ciprofloxacin sorption by dissolved organic carbon from reference and bio-waste materials. Chemosphere 2009, 77, 813–820. [Google Scholar] [CrossRef] [PubMed]
- Ling, W.; Sun, R.; Gao, X.; Xu, R.; Li, H. Low-molecular-weight organic acids enhance desorption of polycyclic aromatic hydrocarbons from soil. Eur. J. Soil Sci. 2015, 66, 339–347. [Google Scholar] [CrossRef]
- Amir, S.; Jouraiphy, A.; Meddich, A.; El, G.M.; Winterton, P.; Hafidi, M. Structural study of humic acids during composting of activated sludge-green waste: Elemental analysis, FTIR and 13C NMR. J. Hazard. Mater. 2010, 177, 524–529. [Google Scholar] [CrossRef] [PubMed]
- Sikora, F.J.; Stevenson, F.J. Silver complexation by humic substances: Conditional stability constants and nature of reactive sites. Geoderma 1988, 42, 353–363. [Google Scholar] [CrossRef]
- Carballeira, J.L.; Antelo, J.M.; Arce, F. Analysis of the Cu2+-soil fulvic acid complexation by anodic stripping voltammetry using an electrostatic model. Environ. Sci. Technol. 2000, 34, 4969–4973. [Google Scholar] [CrossRef]
- Ramos, M.A.; Fiol, S.; López, R.; Antelo, J.M.; Arce, F. Analysis of the effect of pH on Cu2+-fulvic acid complexation using a simple electostatic model. Environ. Sci. Technol. 2002, 36, 3109–3113. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Lam, K.C.; Dawson, R.W.; Liu, W.X.; Tao, S. The effect of pH, ion strength and reactant content on the complexation of Cu by various natural organic ligands from water and soil in Hong Kong. Chemosphere 2004, 54, 507–514. [Google Scholar] [CrossRef] [PubMed]
- Christl, I.; Milne, C.J.; Kinniburgh, D.G.; Kretzschmar, R. Relating ion binding by fulvic and humic acids to chemical composition and molecular size. 2. Metal binding. Environ. Sci. Technol. 2001, 35, 2512–2517. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Senesi, N.; Schnitzer, M. Information Provided on Humic Substances by E4/E6 Ratios. Soil Sci. Soc. Am. J. 1977, 41, 352–358. [Google Scholar] [CrossRef]
- Yang, Y.; Lan, Y.; Jin, P.; Wang, X. Effects of pH, ionic strength and heavy metal ions on UV spectras of humic acid. Chinese J. Environ. Eng. 2015, 9, 5336–5340. [Google Scholar]
- Xu, Y.; Zhanf, H.; Kan, L.; Zhou, Q.; Luo, M.; Chen, B. Investigation on the Interaction of Tetrabutyltin Compound and Humic Acids by Spectroscopy. GuangZhou Chem. Indus. 2010, 38, 179–181. [Google Scholar]
- Sato, O.; Kumada, K. The chemical nature of the green fraction of P type humic acid. Soil Sci. Plant Nutr. 1967, 13, 121–122. [Google Scholar] [CrossRef]
- Fu, L.; Liu, X.F.; Zhou, Q.X.; Zhang, J.X.; Dong, J.Y.; Wang, J.F. Characterization of the interactions of human serum albumin (HSA), gatifloxacin, and metronidazole using spectroscopic and electrochemical methods. J. Lumin. 2014, 149, 208–214. [Google Scholar] [CrossRef]
- Jiang, A.W.; Duan, Y.Q.; Yuan, W.G.; Wang, X.Z. Study of Interaction between m-Nitroaniline and Bovine Serum Albumin Using Fluorospectrphotometry. PTCA 2015, 51, 200–204. [Google Scholar]
- Xu, H.C.; Yu, G.H.; Yang, L.Y.; Jiang, H.L. Combination of two-dimensional correlation spectroscopy and parallel factor analysis to characterize the binding of heavy metals with DOM in lake sediments. J. Hazard. Mater. 2013, 263, 412–421. [Google Scholar] [CrossRef] [PubMed]
- Evans, D.; Dillon, P. Complexation between Hg(II) and Dissolved Organic Matter in Stream Waters: An Application of Fluorescence Spectroscopy. Biogeochemistry 2004, 71, 339–351. [Google Scholar]
- Hernández, D.; Plaza, C.; Senesi, N.; Polo, A. Detection of Copper(II) and Zinc(II) binding to humic acids from pig slurry and amended soils by fluorescence spectroscopy. Environ. Pollut. 2006, 143, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Xi, B.; Xie, S.; Ma, W.; Yu, H.; He, X. Study on fluorescence spectra and UV-vis spectra of dissolved organic matter collected from sediment pore water in Wuliangsuhai Lake. Chin. J. Environ. Eng. 2012, 6, 440–444. [Google Scholar]
- Yang, Y.; Lan, Y.; Jin, P.; Xu, H. Characteristic and influential factors of humic acid complexed with Cd2+. Environ. Chem. 2017, 36, 1198–1203. [Google Scholar]
- Tian, C.; Wang, J.; Yuan, X.; Liu, G. Extraction and ultraviolet absorption spectra of humic acid in marine sediment. Mar. Environ. Sci. 2012, 31, 821–823. [Google Scholar]
- Grassi, M.; Rosa, M. Humic acids of different origin as modifiers of cadmium-ion chemistry: A spectroscopic approach to structural properties and reactivity. Inorg. Chim. Acta 2010, 363, 495–503. [Google Scholar] [CrossRef]
- Liu, J.; Wang, J.; Chen, Y.; Lippold, H.; Lippmannpipke, J. Comparative characterization of two natural humic acids in the Pearl River Basin, China and their environmental implications. J. Environ. Sci 2010, 22, 1695–1702. [Google Scholar] [CrossRef]
- Gondar, D.; Lopez, R.; Fiol, S.; Antelo, J.M.; Arce, F. Characterization and acid–base properties of fulvic and humic acids isolated from two horizons of an ombrotrophic peat bog. Geoderma 2005, 126, 367–374. [Google Scholar] [CrossRef]
- Lan, Y.Q. Study on Characteristic of Heavy Metals and Humic Acid in the Water Environment. Master’s Thesis, Xi’an University of Architecture and Technology, Xi’an, China, 2011. [Google Scholar]

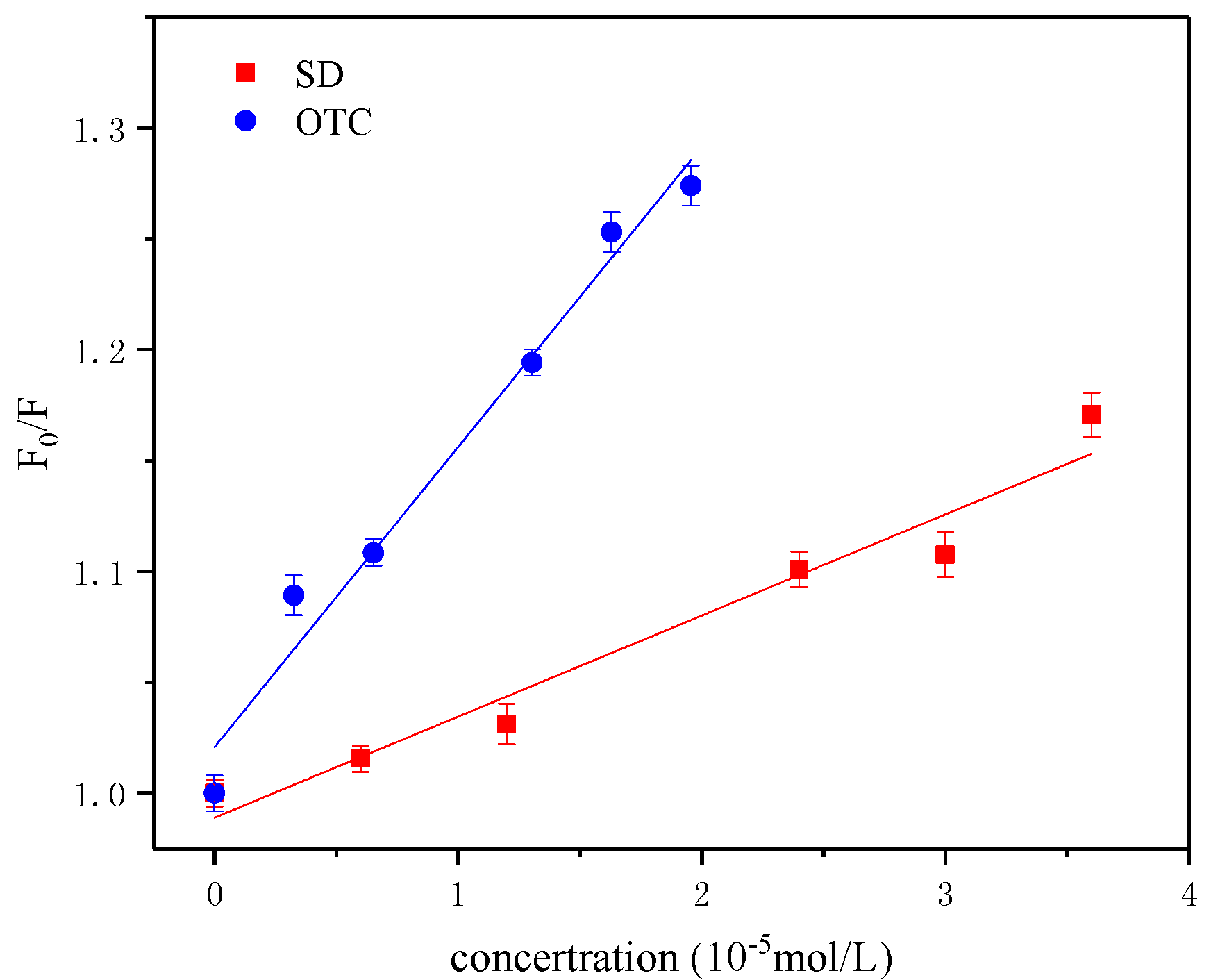





| Quencher | Ksv (103·L·mol−1) | Kq (1011·L·mol−1·s−1) | Kb (103·L·mol−1) | n |
|---|---|---|---|---|
| OTC | 9.811 | 9.811 | 7.183 | 1.207 |
| SD | 5.271 | 5.271 | 3.123 | 0.997 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yuan, X.; Yang, S.; Fang, J.; Wang, X.; Ma, H.; Wang, Z.; Wang, R.; Zhao, Y. Interaction Mechanism between Antibiotics and Humic Acid by UV-Vis Spectrometry. Int. J. Environ. Res. Public Health 2018, 15, 1911. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph15091911
Yuan X, Yang S, Fang J, Wang X, Ma H, Wang Z, Wang R, Zhao Y. Interaction Mechanism between Antibiotics and Humic Acid by UV-Vis Spectrometry. International Journal of Environmental Research and Public Health. 2018; 15(9):1911. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph15091911
Chicago/Turabian StyleYuan, Xiaoyu, Shengke Yang, Jie Fang, Xueli Wang, Haizhen Ma, Zongzhou Wang, Runze Wang, and Yaqian Zhao. 2018. "Interaction Mechanism between Antibiotics and Humic Acid by UV-Vis Spectrometry" International Journal of Environmental Research and Public Health 15, no. 9: 1911. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph15091911





