1. Introduction
Heavy metal stress is defined as any heavy metal pollutant that is potentially unfavorable to crops’ metabolism, growth, or development [
1]. With the development of industry, some heavy metal pollutants have caused farmland disturbance in developing countries, typically cadmium (Cd), copper (Cu), zinc (Zn), lead (Pb), and others [
2]. Cd stress is potentially unfavorable to rice metabolism, growth, or development, which is one of the critical food pollution problems in China [
3]. The adsorption of Cd in rice is higher than that of other pollutants; thus, Cd pollution in rice is relatively obvious [
4]. High concentrations of Cd pollution can have a negative effect on vegetation [
5]. Cd is absorbed by rice, causing rice pollution, which in turn harms the environment and the health of animals and plants [
6]. Therefore, detecting Cd pollution plays a crucial role in assessing crop quality, environmental quality, and human health.
Heavy metal pollution can lead to a change in vegetation color and morphogenesis, leaf yellowing, vegetation branching, defoliation, and even death [
7,
8,
9,
10,
11]. Studies have shown that Cd stress has a similar effect. Cd stress causes a reduction in rice growth; excessive Cd in the growth reduces rice seed germination, causea leaf chlorosis and necrosis; and photosynthesis decreases in rice, which further results in decreasing chlorophyll (Chl), leaf area index (LAI), and changes in average leaf angle (ALA) [
12,
13,
14]. Such pollution can also destroy the internal structure of vegetation leaves. Zhu et al. [
15] found that with an increase in copper content, the mesophyll cells shrink, the internal structure of leaves appears disordered, and the leaf structure parameter (N) increases. The spectral reflectance of vegetation changes considerably because of the remarkable changes in the various biochemical and biophysical parameters of vegetation.
Traditionally, assessment of Cd stress has often been conducted through soil testing, crop tissue analysis, and long-term field trials in sequential steps with increasing high cost. Remote sensing technology provides an efficient and economical means for assessing crop Cd stress over large areas [
16]. Many studies have shown that remote sensing has been used for monitoring crops in controlled laboratory conditions or specific field environments under heavy metal stress [
9,
17,
18]. Vegetation spectral indices are among the common methods that can characterize the variation in vegetation in response to heavy metal stress [
19]. For example, spectral indices, such as normalized difference vegetation index, modified chlorophyll absorption ratio index, structure insensitive pigment index, enhanced vegetation index, red-edge chlorophyll index (Clre), red-edge position (REP), and normalized red-edge differences (NDRE), are commonly used to detect crops under heavy metal stress [
20,
21,
22,
23,
24]. Kooistra et al. [
25] used REP and Cd in soil for regression analysis, and found that REP had a good correlation with Cd content. Likewise, many spectral signal enhancement methods, such as principal component regression, independent component analysis, wavelet transform, and spectral absorption depth, have been used to extract the spectral characteristics of stressed vegetation [
26,
27,
28]. Many studies have also applied partial least square regression, multiple linear regression, and artificial neural network methods to establish an empirical relationship between spectral reflectance data and vegetation biochemical and biophysical parameters; this relationship is used to identify heavy metal stress in vegetation [
19,
29,
30,
31,
32]. Liu et al. [
33] used a back propagation neural-network model to estimate chlorophyll concentration in rice under Cd stress.
Heavy metal stress under laboratory conditions or specific field-scale environments (e.g., nearby abandoned mines) can cause considerable changes in vegetation, such as obvious leaf yellowing and leaf curls [
12,
13,
34]. Thus, different stress levels in vegetation are easy to detect. Spectral indices that are sensitive to vegetation biochemical and biophysical parameters have been established on the basis of substantial field measurement data in experiments under heavy metal pollution in laborious or field scale. These field-scale approaches lack universality and cannot be used for large-scale information monitoring [
27,
32]. Detecting the level of pollutants in “real-world” ecosystems is relatively difficult, and how to use biochemical parameters, biophysical parameters, or spectral wavelengths in vegetation in response to heavy metal remains less explored. Li et al. [
35] constructed the hyperspectral index based on subtle changes in canopy chlorophyll, and used Hyperion images to monitor canopy chlorophyll under As stress on a large scale. Liu et al. [
36] integrated physiological function variability with spatio-temporal stability using multi-temporal thermal remote sensing for regional monitoring of heavy metal stress. To further assess large-area heavy metal concentrations in crops, the establishment of new vegetation spectral indices is needed based on the selection of sensitive wavelengths by integrating measurement data with simulation data.
Vegetation radiative transfer models (RTMs) have recently been widely used for vegetation parameter detection [
37,
38,
39,
40], and many people have used vegetation RTMs to develop spectral indices. Zhou et al. [
41] used the PROSPECT-5 model to establish a new spectral index for estimating the ratio of carotenoids to Chl content to monitor the physiological and phenological conditions of crops. Pasqualotto et al. [
42] utilized the PROSAIL model constructs with two spectral indices that accurately estimate the water content of the canopy. Among many available vegetation radiative transfer models, this study selected the PROSAIL model, which is coupled by leaf and canopy radiative transfer model. The model was chosen because of its availability, accuracy, and simplicity [
39]. The PROSAIL model is a physical model that provides a thorough understanding of biological and biochemical processes, analyzes signals from remote sensing data, allows input, and generates rich simulation datasets [
40]. The PROSAIL can not only simulate the spectral characteristics of different crop types under different physiological conditions, but can also compensate for the deficiency of measured data in field experiments (e.g., insufficient data, lack of key data, uncontrollable environmental parameters, and unrepeatable experiments) [
39,
43,
44]. In addition, spectral wavelengths that are sensitive to specific biochemical and biophysical parameters are determined, and spectral indices that are sensitive to different pollution types in farmland can be explored. The spectral indices developed by PROSAIL model have good universality and can be used on a large scale in similar crop type research [
39].
Satellite remote sensing images provide an efficient, economical, and large-scale monitoring method to verify the application of a spectral index developed by vegetation RTMs and monitor Cd stress in farmland on a large scale. Xiao et al. [
45] developed NDII as the land surface water index based on the SPOT-4 satellite image and used it to estimate water stress coefficient. Muramatsu et al. [
46] constructed a new index and applied it to Landsat satellite images for land-cover classification. Sentinel-2 satellite remote sensing images have a wide application in distinguishing different crop types, monitoring plant stress, and retrieving biophysical parameters, such as LAI, leaf Chl content, and leaf water content (Cw) [
47,
48,
49,
50,
51,
52]. They contain three red-edge bands that provide key information for monitoring the vegetation state. Several studies have confirmed that when plants are subjected to heavy metal stress, the red-edge parameters of plant spectra change [
25,
53,
54]. Therefore, Sentinel-2 can obtain more information about plant stress changes than SPOT and Landsat satellite images.
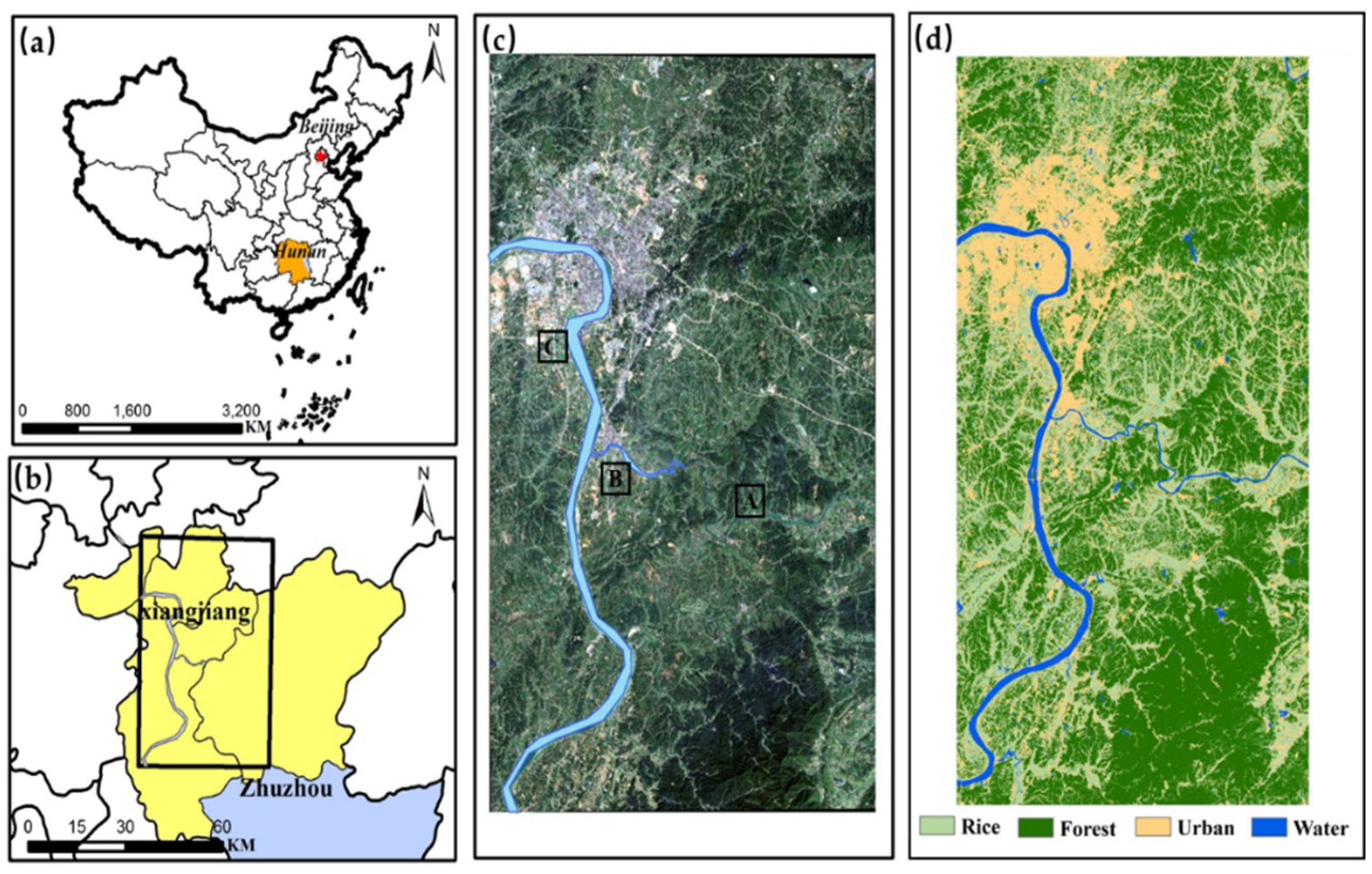
This study focuses on the development of a spectral index that can accurately monitor different Cd stress levels in rice on a large scale. The contributions of this study are as follows: (1) A new Cd stress-sensitive spectral index is established using the PROSAIL model, which can explore the sensitive wavelength bands and biochemical components of rice in response to Cd stress; (2) the common spectral indices and the newly developed spectral index are compared using field-measured and simulated data; (3) the newly developed spectral index is upscaled to Sentinel-2 remote sensing images to detect Cd stress levels in rice on a large scale.
5. Discussion
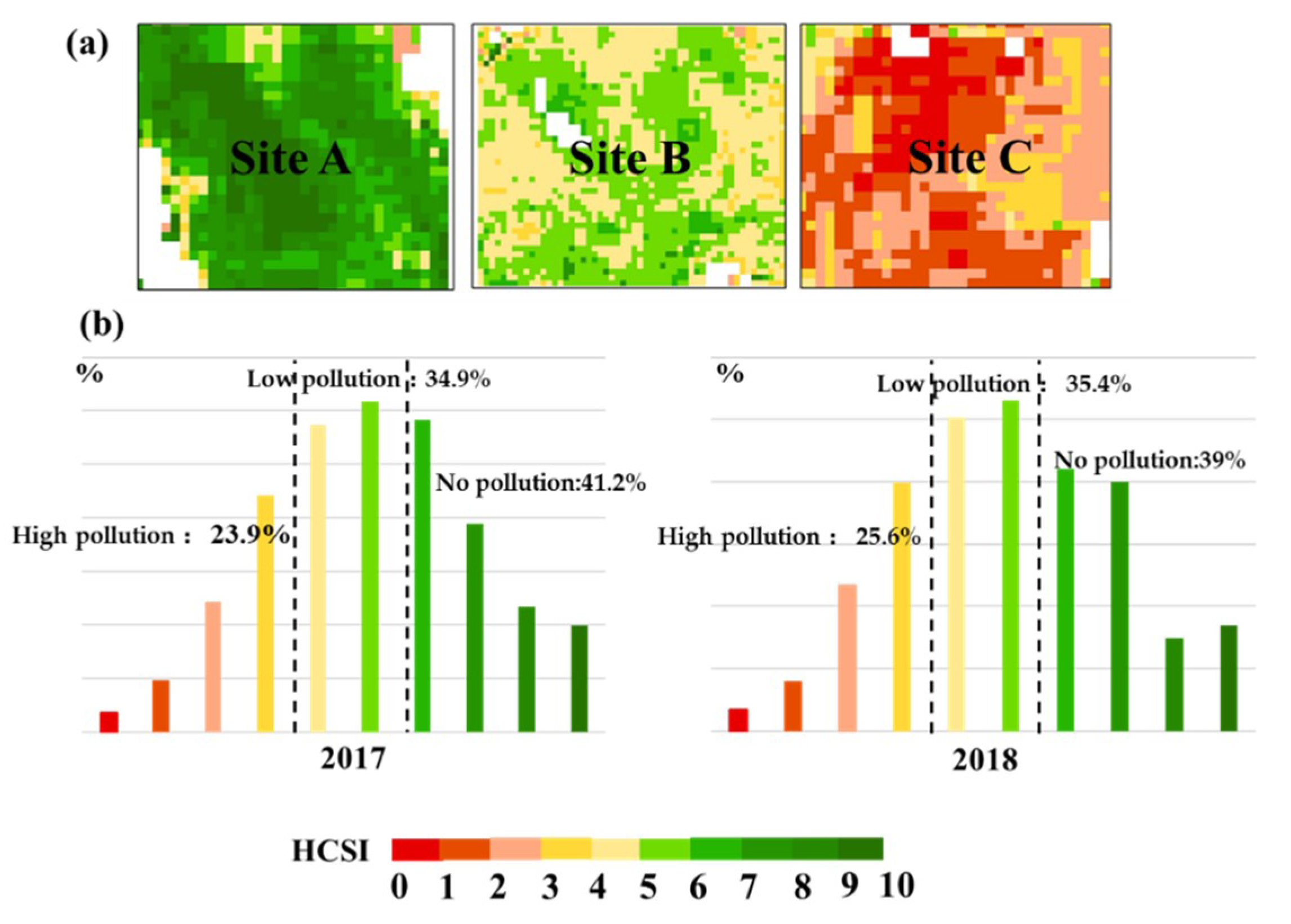
The main purpose of this study was to develop a new spectral index that can accurately detect different Cd stress levels in rice on a large scale. For that purpose, the radiative transfer model PROSAIL combined with measured data was used. The analysis found that chlorophyll was the most sensitive biochemical parameter to Cd stress in the farmland ecosystem. The new spectral index HCSI was developed based on chlorophyll. In comparison with the common spectral index, the HCSI can discriminate different stress levels more clearly. In addition, two years of Sentinel-2A remote sensing image data were used to predict the distribution of Cd stress, and the application of the new index has strong regional adaptability.
The satisfactory results obtained, on the one hand, differ from empirical approaches requiring several field measurement datasets for modeling to detect heavy metal stress [
24,
35,
75], as using the vegetation radiative transfer model requires only a few field measurement biophysical and biochemical data to the range settings of the model parameters. In addition, because the vegetation radiative transfer model is generally applicable in different situations with good universality, the results can be easily applied to remote sensing data and, compared with research that generally uses the field-measured data or ASD spectral data for detecting heavy metal stress [
27,
32,
76,
77], it can be applied on a larger scale. On the other hand, the spectral characteristics and mechanism of rice under Cd stress in the natural farmland ecosystem were studied and analyzed, and the typical biophysical and biochemical characteristics of rice leaf and canopy scale were selected for discussion. The development of the index was to improve the ability to detect Cd stress by reducing the interference of other parameters for the sensitive parameter [
78,
79]. However, because of the complicated factors, a further investigation of the biophysical and biochemical parameter changes of rice under Cd stress needs more controlled strict experiments.
This study was carried out under the condition of known stress factor beforehand, and field collection was conducted on the nutritional status and heavy metal content status of rice, excluding the interference of some factors. Under a condition of known environmental Cd pollution, the Cd stress can be well detected by using HCSI. However, the impact on crops in the natural farmland ecosystem is complicated, and how to differentiate Cd stress from environmental stress, such as pest stress and temperature stress, is still a problem that needs further discussion. Two years of remote sensing data were used to predict the distribution of Cd stress; the spatial distribution of the pollution levels are similar in the two years’ image, and the area percentages of rice with the same degree of pollution in two years are similar, which indicates that rice under Cd stress is spatio-temporally stable. However, environmental factors are usually abrupt and do not last continuously [
26,
80]. Therefore, a study that combines spectral and temporal information and uses long time-series remote sensing data will help to distinguish the Cd stress from these environmental factors.