Evolution of the Speciation and Mobility of Pb, Zn and Cd in Relation to Transport Processes in a Mining Environment
Abstract
:1. Introduction
2. Materials and Methods
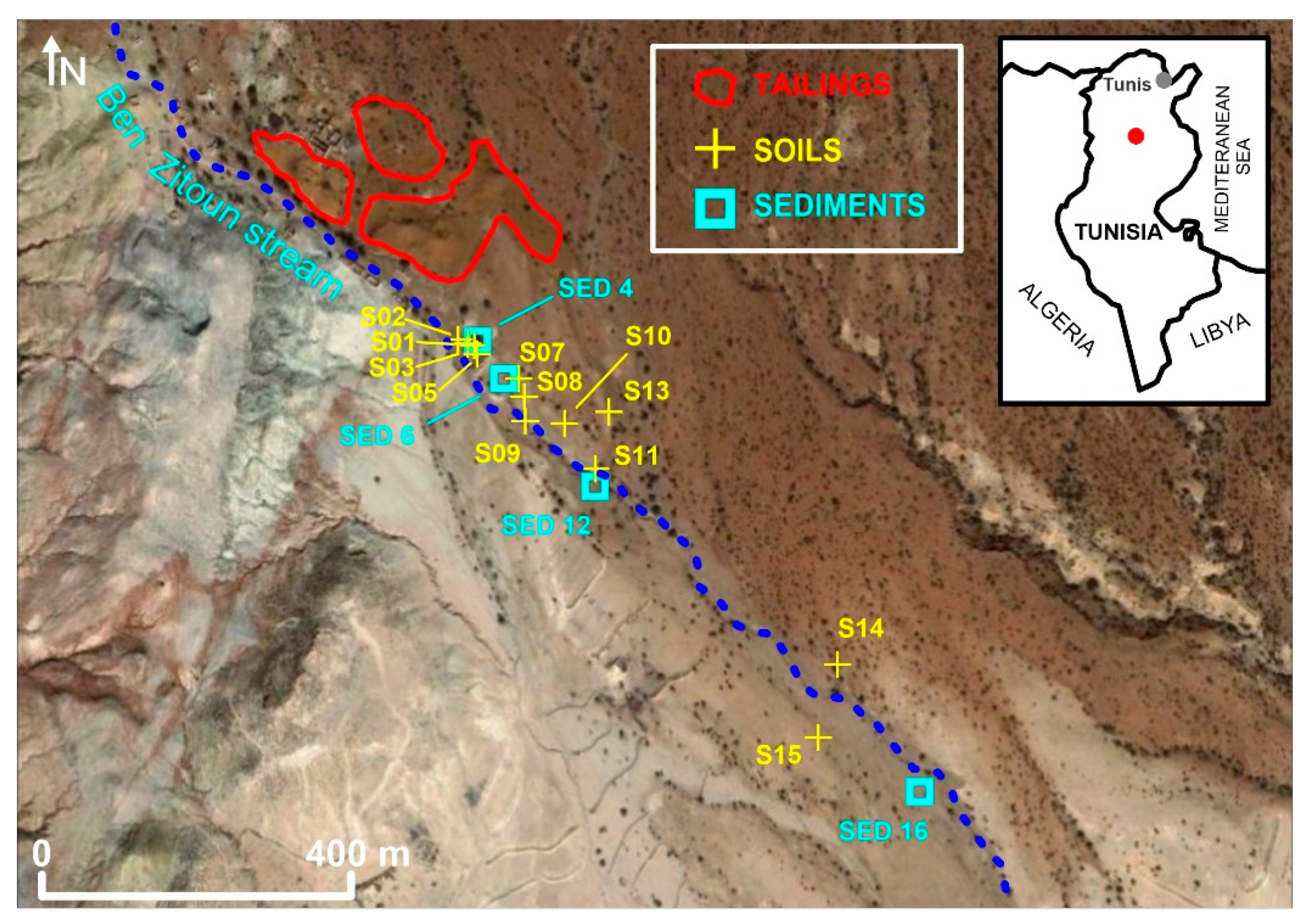
2.1. Study Area
2.2. Sample Preparation and Analysis
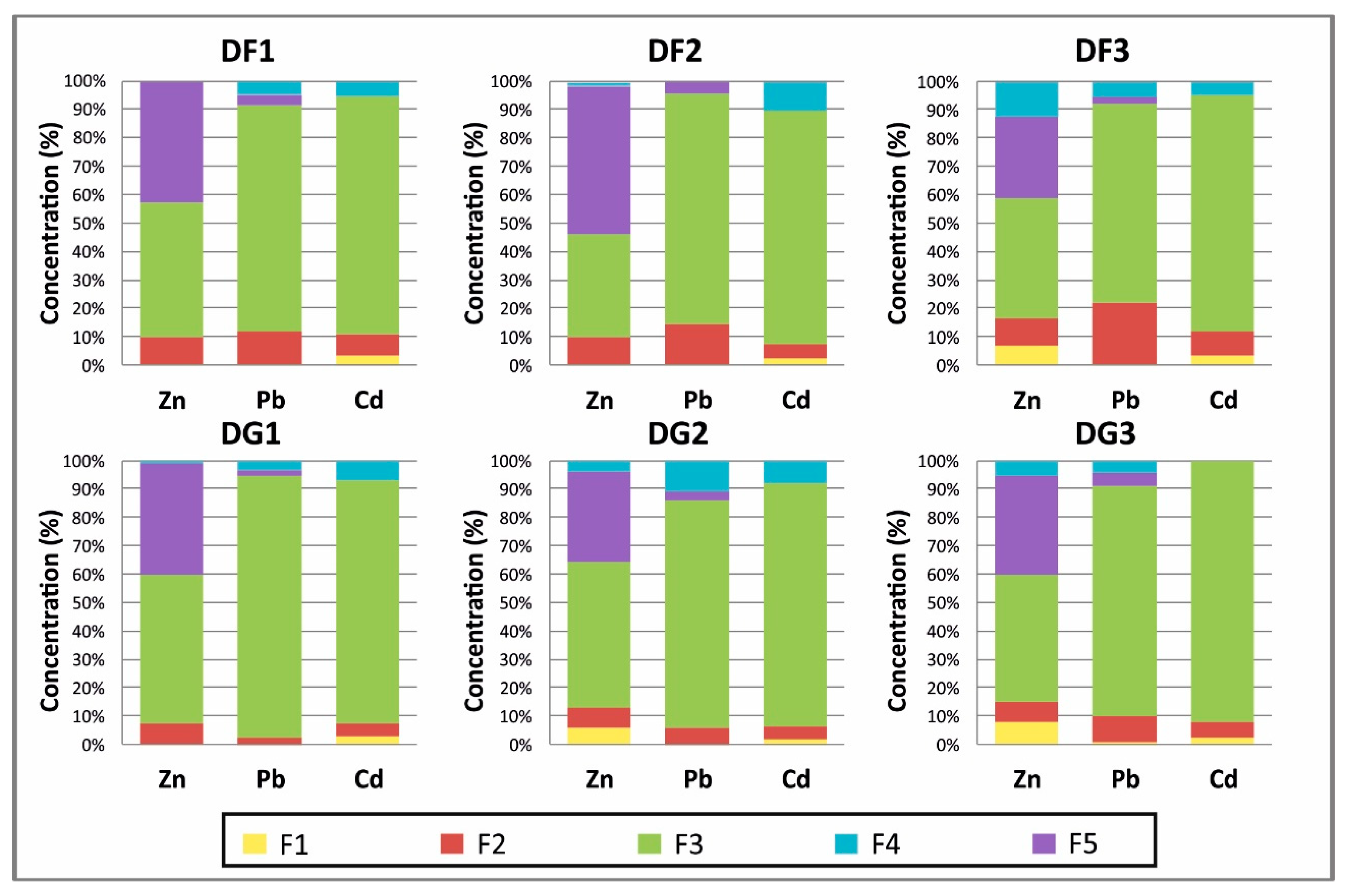
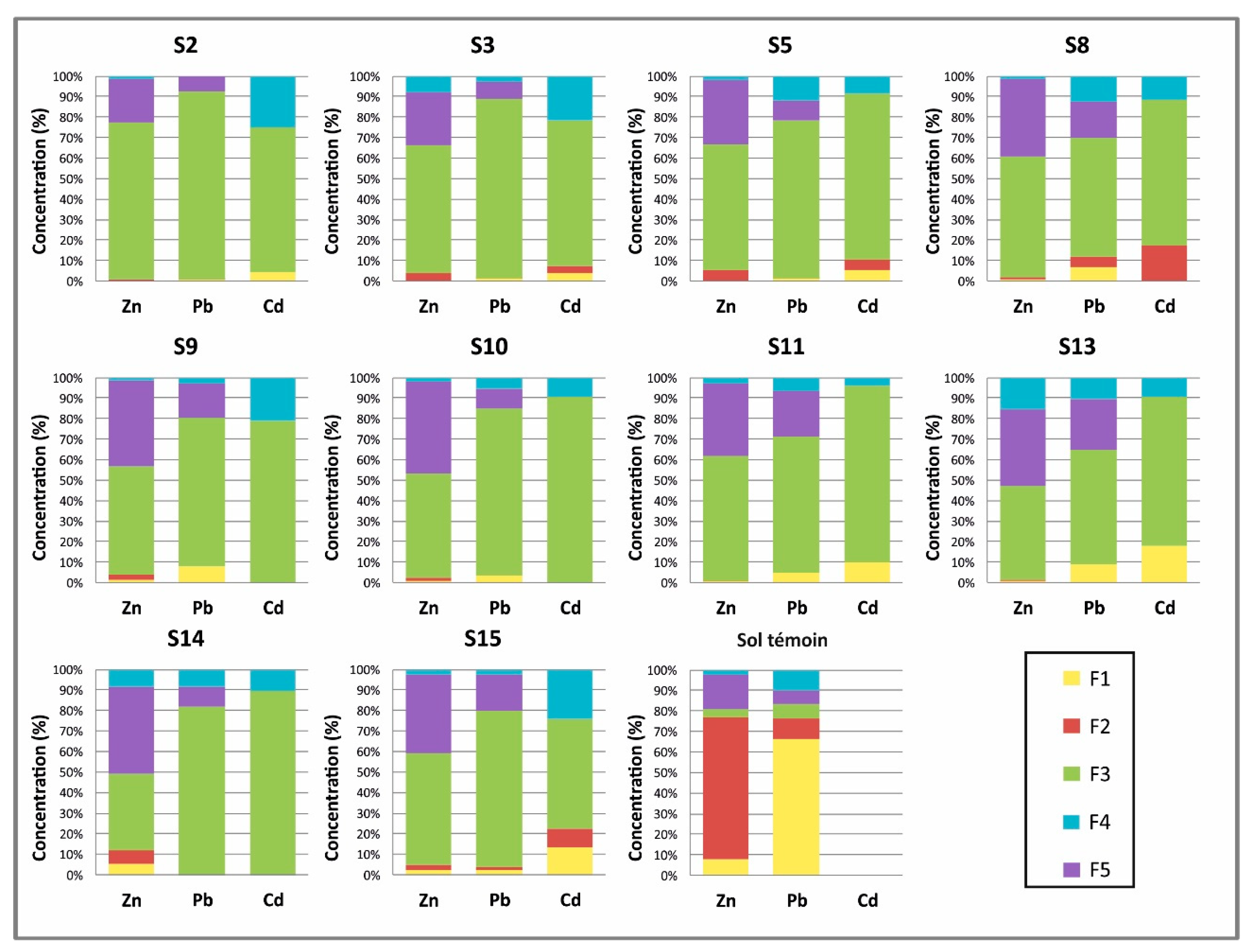
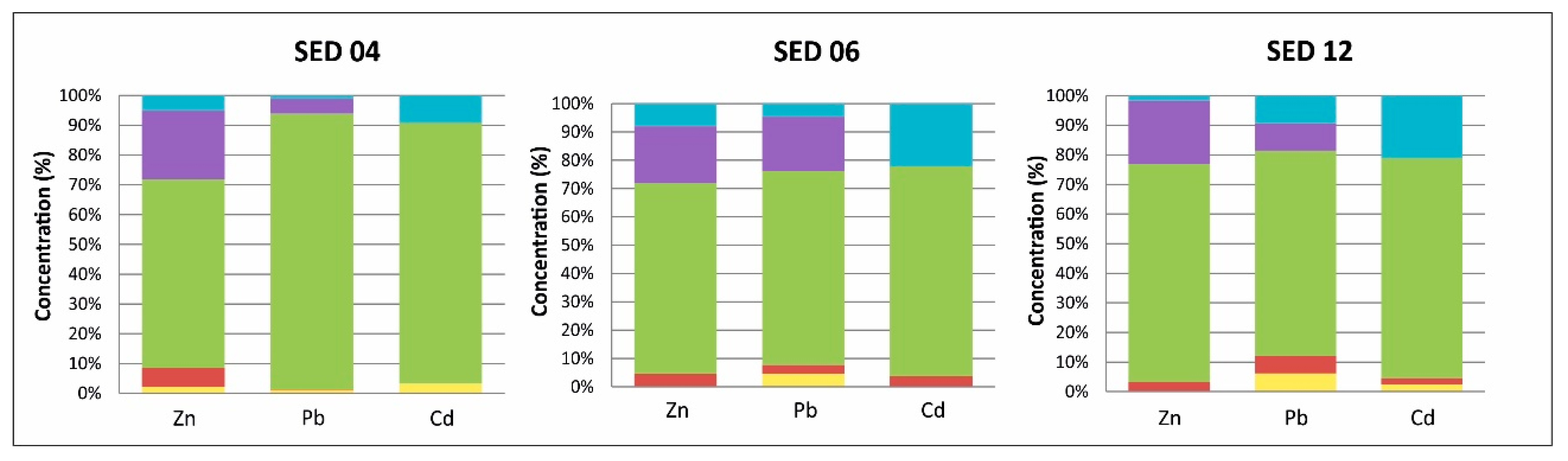
- F1: This fraction contains the metals that are considered to be adsorbed and not easily mobilized by ion exchange. A volume of 40 mL of magnesium chloride (1 M MgCl2) was added to 1 g of soil sample that had been dried in the open air and sieved at 0.63 mm (pH = 7). The mixture was stirred for one hour and then centrifuged at 3000 rpm for 20 min. The supernatant was recovered and filtered through a cellulose acetate membrane with a porosity of 0.45 μm and then stored prior to analysis by Inductively Coupled Plasma Optical Emisión Spectrometry (ICP-OES). The residue was stored for the remainder of the protocol.
- F2: This fraction corresponds to the acid-soluble fraction, and it combines the precipitated or coprecipitated metals with the natural carbonates, which dissolve on decreasing the pH. Besides, 8 mL of CH3COONa (1 M) at pH = 5 (adjusted with CH3COOH (1 M)) was added to the residue from step 1. The mixture was shaken for 5 h. The extract was recovered by the same procedure as in the previous step.
- F3: This fraction was obtained from the residue from the carbonated fraction (F2) by treatment with 20 mL of hydroxylamine chloride (0.04 M) in 25% acetic acid at 96 °C for 6 h.
- F4: This fraction was obtained by treatment of the residue from the preceding extraction with 3 mL of HNO3 (0.02 M) and 5 mL of H2O2 (30%) (pH = 2 with HNO3) at 85 °C for 2 h, 3 mL H2O2 (pH = 2 with HNO3) at 85 °C for 3 h and 5 mL CH3OONH4 in 20% (v/v) HNO3 diluted to 20 mL, with shaking for 30 min.
- F5: The residual fraction corresponds to recalcitrant or refractory minerals, usually with three metal ores present in the deposit, and this was obtained by adding hydrofluoric acid, hydrochloric acid and nitric acid to the residue from the preceding extraction. The extract was recovered by the same procedure as in the previous step.
3. Results and Discussion
3.1. Sequential Extraction Rsults
3.1.1. Tailings Results
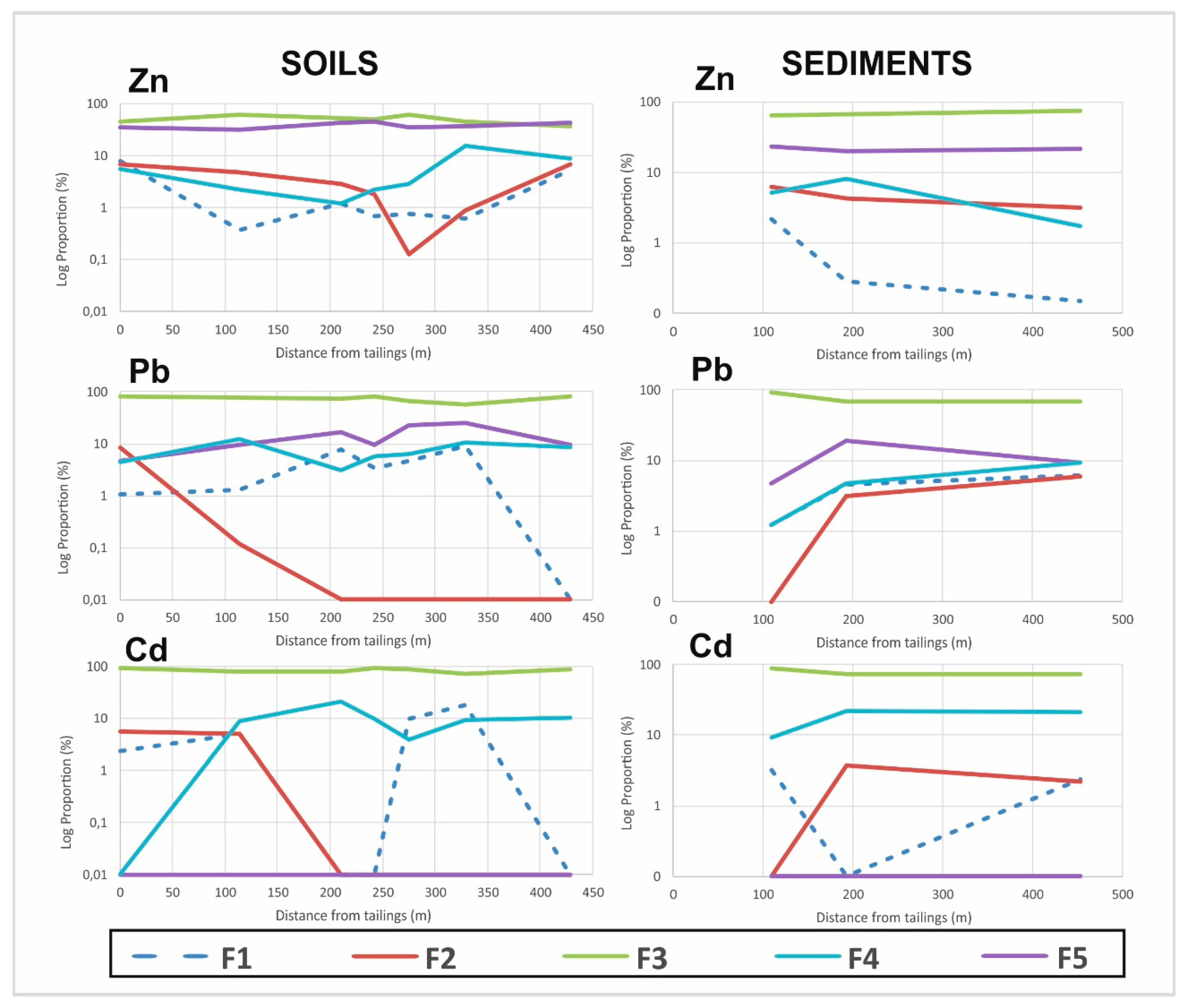
3.1.2. Soil Samples
3.1.3. Sediment Samples
3.1.4. Comparison between Tailings, Soils and Sediments Fractionations
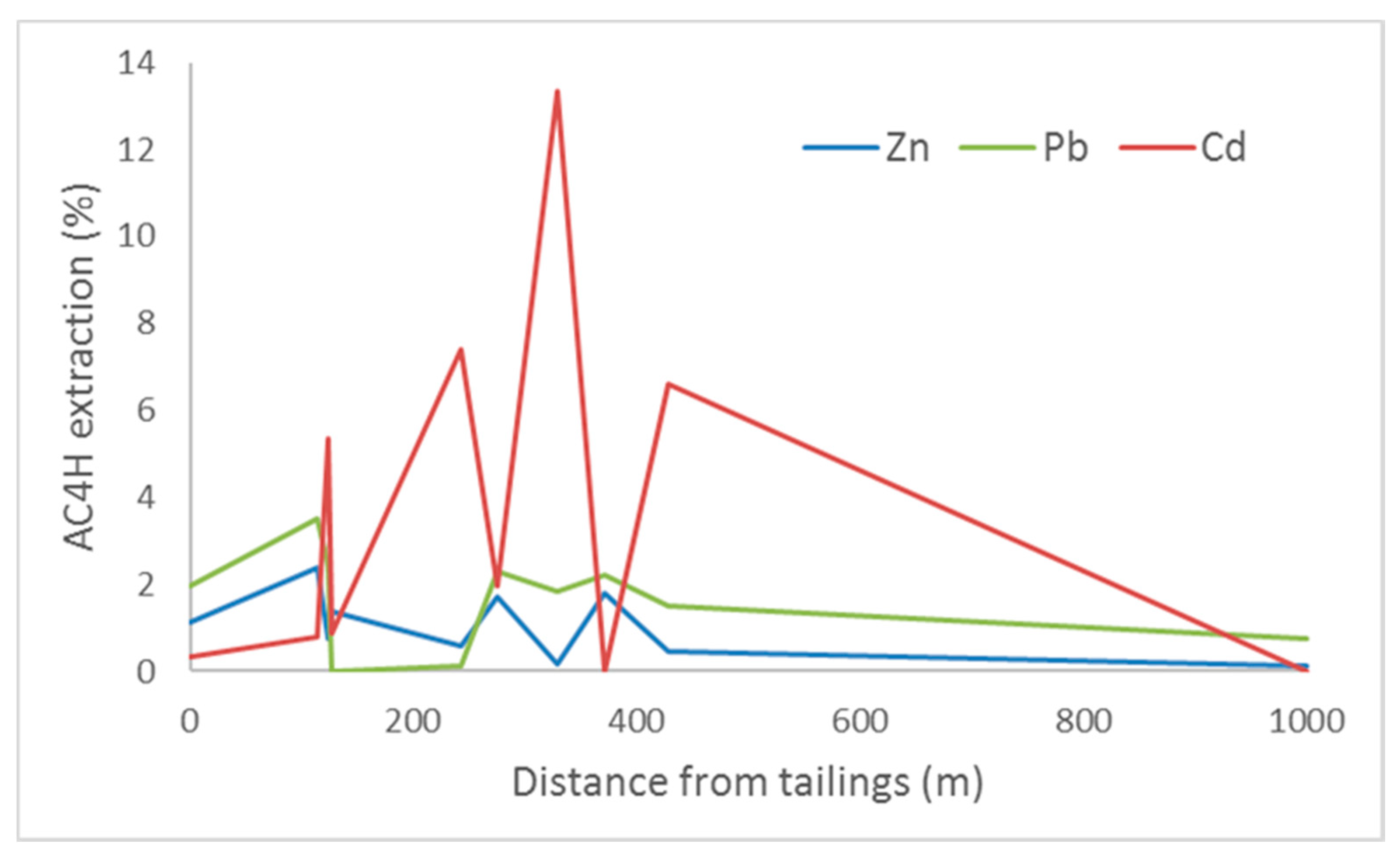
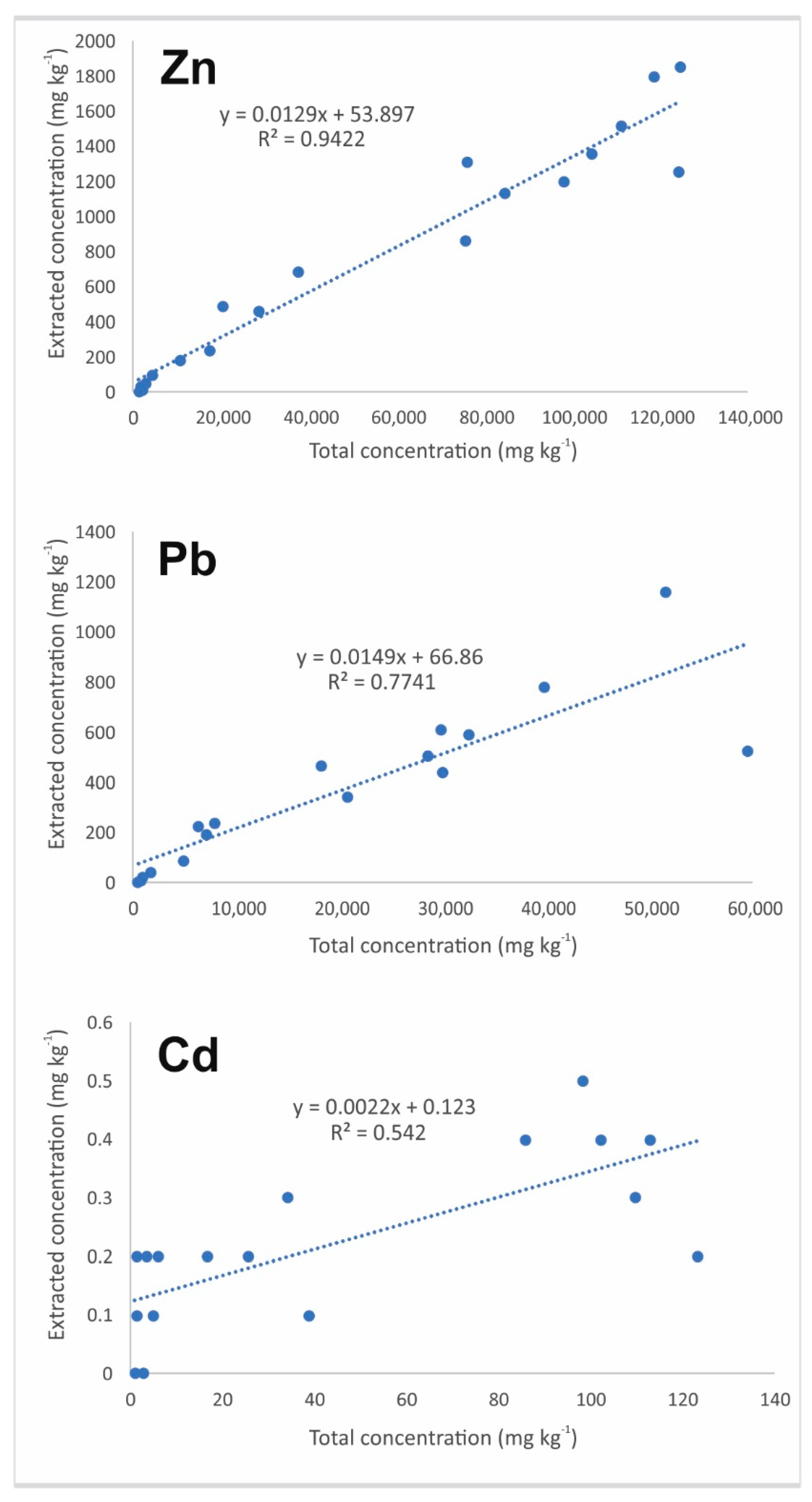
3.2. Selective Extractions
3.2.1. Tailings Results
3.2.2. Soil Results
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ma, L.; Xiao, T.; Ning, Z.; Liu, Y.; Chen, H.; Peng, J. Pollution and health risk assessment of toxic metal (loid) s in soils under different land use in sulphide mineralized areas. Sci. Total Environ 2020, 724, 138176. [Google Scholar] [CrossRef] [PubMed]
- Baltas, H.; Sirin, M.; Gökbayrak, E.; Ozcelik, A.E. A case study on pollution and a human health risk assessment of heavy metals in agricultural soils around Sinop province, Turkey. Chemosphere 2020, 241, 125015. [Google Scholar] [CrossRef] [PubMed]
- Hang, X.; Wang, H.; Zhou, J.; Ma, C.; Du, C.; Chen, X. Risk assessment of potentially toxic element pollution in soils and rice (Oryza sativa) in a typical area of the Yangtze River Delta. Environ. Pollut. 2009, 157, 2542–2549. [Google Scholar] [CrossRef] [PubMed]
- Naikoo, M.I.; Dar, M.I.; Khan, F.A.; Raghib, F.; Rajakaruna, N. Trophic transfer and bioaccumulation of lead along soil–plant–aphid–ladybird food chain. Environ. Sci. Pollut. Res. 2019, 26, 23460–23470. [Google Scholar] [CrossRef] [PubMed]
- Dar, M.I.; Green, I.D.; Khan, F.A. Trace metal contamination: Transfer and fate in food chains of terrestrial invertebrates. Food Webs 2019, 20, e00116. [Google Scholar] [CrossRef]
- Fernández-Martínez, R.; Esbrí, J.M.; Higueras, P.; Rucandio, I. Comparison of mercury distribution and mobility in soils affected by anthropogenic pollution around chloralkali plants and ancient mining sites. Sci. Total Environ. 2019, 671, 1066–1076. [Google Scholar] [CrossRef]
- García-Ordiales, E.; Flor-Blanco, G.; Roqueñí, N.; Covelli, S.; Cienfuegos, P.; Álvarez, R.; Fontolan, G.; Loredo, J. Anthropocene footprint in the Nalón estuarine sediments (northern Spain). Mar. Geol. 2020, 424, 106167. [Google Scholar] [CrossRef]
- Huang, Y.; Wang, L.; Wang, W.; Li, T.; He, Z.; Yang, X. Current status of agricultural soil pollution by heavy metals in China: A meta-analysis. Sci. Total Environ. 2019, 651, 3034–3042. [Google Scholar] [CrossRef]
- Szwalec, A.; Mundała, P.; Kędzior, R.; Pawlik, J. Monitoring and assessment of cadmium, lead, zinc and copper concentrations in arable roadside soils in terms of different traffic conditions. Environ. Monit. Assess. 2020, 192, 155. [Google Scholar] [CrossRef] [Green Version]
- Luo, X.; Bing, H.; Luo, Z.; Wang, Y.; Jin, L. Impacts of atmospheric particulate matter pollution on environmental biogeochemistry of trace metals in soil-plant system: A review. Environ. Pollut. 2019, 255, 113138. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Karmoker, J.; Pal, B.K.; Luo, C.; Zhao, M. Trace metals contamination in different compartments of the Sundarbans mangrove: A review. Mar. Pollut. Bull. 2019, 148, 47–60. [Google Scholar] [CrossRef] [PubMed]
- Silveira, M.L.; Alleoni, L.R.F.; O’connor, G.A.; Chang, A.C. Heavy metal sequential extraction methods—A modification for tropical soils. Chemosphere 2006, 64, 1929–1938. [Google Scholar] [CrossRef] [PubMed]
- Elmayel, I.; Esbri, J.M.; García-Ordiales, E.; Elouaer, Z.; Garcia-Noguero, E.M.; Bouzid, J.; Campos, H.A.; Higueras, P.L. Biogeochemical assessment of the impact of Zn mining activity in the area of the Jebal Trozza mine, Central Tunisia. Environ. Geochem. Health 2020. [Google Scholar] [CrossRef] [PubMed]
- García-Ordiales, E.; Covelli, S.; Esbrí, J.M.; Loredo, J.; Higueras, P.L. Sequential extraction procedure as a tool to investigate PTHE geochemistry and potential geoavailability of dam sediments (Almadén mining district, Spain). Catena 2016, 147, 394–403. [Google Scholar] [CrossRef]
- Buccolieri, A.; Buccolieri, G.; Dell’Atti, A.; Strisciullo, G.; Gagliano-Candela, R. Monitoring of total and bioavailable heavy metals concentration in agricultural soils. Environ. Monit. Assess. 2010, 168, 547–560. [Google Scholar] [CrossRef]
- Tessier, A.; Campbell, P.G.C.; Bisson, M. Sequential Extraction Procedure for the Speciation of Particulate Trace Metals. Anal. Chem. 1979, 51, 844–851. [Google Scholar] [CrossRef]
- Rauret, G.; López-Sánchez, J.F.; Sahuquillo, A.; Rubio, R.; Davidson, C.M.; Ure, A.; Quevauviller, P. Improvement of the BCR three step sequential extraction procedure prior to the certification of new sediment and soil reference materials. J. Environ. Monit. 1999, 1, 57–61. [Google Scholar] [CrossRef]
- Rosado, D.; Usero, J.; Morillo, J. Ability of 3 extraction methods (BCR, Tessier and protease K) to estimate bioavailable metals in sediments from Huelva estuary (Southwestern Spain). Mar. Pollut. Bull. 2016, 102, 65–71. [Google Scholar] [CrossRef]
- Kheboian, C.; Bauer, C.F. Accuracy of Selective Extraction Procedures for Metal Speciation in Model Aquatic Sediments. Anal. Chem. 1987, 59, 1417–1423. [Google Scholar] [CrossRef]
- Bloom, N.S.; Preus, E.; Katon, J.; Hiltner, M. Selective extractions to assess the biogeochemically relevant fractionation of inorganic mercury in sediments and soils. Anal. Chim. Acta 2003, 479, 233–248. [Google Scholar] [CrossRef]
- Favas, P.J.; Pratas, J.; Gomes, M.E.P.; Cala, V. Selective chemical extraction of heavy metals in tailings and soils contaminated by mining activity: Environmental implications. J. Geochem. Explor. 2011, 111, 160–171. [Google Scholar] [CrossRef]
- Shuman, L.M. Fractionation method for soil microelements. Soil Sci. 1985, 140, 11–22. [Google Scholar] [CrossRef]
- Ure, A.M. Single extraction schemes for soil analysis and related applications. Sci. Total Environ. 1996, 178, 3–10. [Google Scholar] [CrossRef]
- Pardo, R.; Helena, B.A.; Cazurro, C.; Guerra, C.; Deban, L. Application of two- and three-way principal component analysis to the interpretation of chemical fractionation results obtained by the use of the B.C.R. procedure. Anal. Chim. Acta 2004, 523, 125–132. [Google Scholar] [CrossRef]
- Frentiu, T.; Ponta, M.; Levei, E.; Gheorghiu, E.; Kasler, I.; Cordos, E.A. Validation of the Tessier scheme for speciation of metals in soil using the Bland and Altman test. Chem. Pap. 2008, 62, 114–122. [Google Scholar] [CrossRef]
- Nagajyoti, P.C.; Lee, K.D.; Sreekanth, T.V.M. Heavy metals, occurrence and toxicity for plants: A review. Environ. Chem. Lett. 2010, 8, 199–216. [Google Scholar] [CrossRef]
- Menzies, N.W.; Donn, M.J.; Kopittke, P.M. Evaluation of extractants for estimation of the phytoavailable traces metals in soils. Environ. Poll. 2007, 145, 121–130. [Google Scholar] [CrossRef] [Green Version]
- Reis, A.T.; Davidson, C.M.; Vale, C.; Pereira, E. Overview and challenges of mercury fractionation and speciation in soils. Trends Anal. Chem. 2016, 82, 109–117. [Google Scholar] [CrossRef] [Green Version]
- Hageman, P.L. US Geological Survey field leach test for assessing water reactivity and leaching potential of mine wastes, soils and other geologic and environmental materials. In U.S. Geological Survey Techniques and Methods; U.S. Geological Survey: Reston, VA, USA, 2007; Volume 5, p. 14. [Google Scholar]
- Sahuquillo, A.; López-Sánchez, J.F.; Rubio, R.; Rauret, G.; Hatje, V. Sequential extraction of trace-metals from sediments. Validation of CR determination in the extract by AAS. Fresenius J. Anal. Chem. 1995, 351, 197–203. [Google Scholar] [CrossRef]
- Martínez-Fernández, M.; Barciela-Alonso, M.C.; Moreda-Pineiro, A.; Bermejo-Barrera, P. Matrix solid phase dispersion-assisted BCR sequential extraction method for metal partitioning in surface estuarine sediments. Talanta 2011, 83, 840–849. [Google Scholar] [CrossRef]
- Teng, Y.G.; Yang, J.; Wang, J.S.; Song, L.T. Bioavailability of vanadium extracted by EDTA, HCl, HOAC and NaNO3 in Topsoil in the Panzhihua Urban Park, located in Southwest China. Biol. Trace Elem. Res. 2011, 144, 1394–1404. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.H.; Huang, D.Y.; Liu, S.L.; Luo, Z.C.; Zhu, H.H.; Zhou, B.; Lei, M.; Rao, Z.X.; Cao, X.L. Assessment of single extraction methods for evaluating the immobilization effect of amendments on cadmium in contaminated acidic paddy soil. Plant Soil Environ. 2012, 58, 98–103. [Google Scholar] [CrossRef] [Green Version]
- Li, M.S.; Yang, S.X. Heavy Metal Contamination in Soils and Phytoaccumulation in a Manganese Mine Wasteland, South China. Air Soil Water Res. 2020, 1, 31–41. [Google Scholar] [CrossRef] [Green Version]
- Kabata-Pendias, A. Behavioral properties of trace metal in soils. Appl. Geochem. 1993, 8, 3–9. [Google Scholar] [CrossRef]
- Remon, E.; Bouchardon, J.-L.; Cornier, B.; Guy, B.; Leclerc, J.-C.; Faure, O. Soil characteristics, heavy metal availability and vegetation recovery at a former metallurgical landfill: Implications in risk assessment and site restoration. Environ. Poll. 2005, 137, 316–323. [Google Scholar] [CrossRef]
- Wang, W.S.; Shan, X.Q.; Wen, B.; Zhang, S.Z. Relationship between the extractable metals from soils and metals taken up by maize roots and shoots. Chemosphere 2003, 53, 523–530. [Google Scholar] [CrossRef]
- Azzoni, R.G.; Giordani, M.; Bartoli, D.T.W.; Viaroli, P. Iron, sulphur and phosphorus cycling in the rhizosphere sediments of an eutrophic Ruppia cirrhosa meadow (Valle Smarlacca, Italy). J. Sea Res. 2001, 45, 15–26. [Google Scholar] [CrossRef]
- Válega, M.; Lillebø, A.I.; Pereira, M.E.; Duarte, A.C.; Pardal, M.A. Long-term effects of mercury in a salt marsh: Hysteresis in the distribution of vegetation following recovery from contamination. Chemosphere 2008, 71, 765–772. [Google Scholar] [CrossRef] [Green Version]
- Bouhlel, S.; Fortuné, J.P. Contribution à l’étude minéralogique et géochimique des minéraux de Pb-Zn-Cu de la province fluorée tunisienne. Notes Serv. Géol. Tunis. 1985, 51, 155–176. [Google Scholar]
- Mejri, H. Caractérisation hydrogéologique, hydro-chimique et isotopique des nappes Haffouz et Bou Hafna (Tunisie centrale). MD Thesis, Université de Sfax, Sfax, Tunisie, 13 July 2010. [Google Scholar]
- Hamed, Y.; Redhaounia, B.; Sâad, A.; Hadji, R.; Zahri, F.; Zighmi, K. Hydrothermal waters from karst aquifer: Case study of the Trozza basin (Central Tunisia). J. Tethys 2017, 5, 33–44. [Google Scholar]
- US EPA. Test Methods for Evaluating Solid Waste, Physical/Chemical Methods, SW 846. In Method 1312, Synthetic Precipitation Leaching Procedure, 3rd ed.; United States Environmental Protection Agency: Washington, DC, USA, 1994. [Google Scholar]
- Sladek, C.; Gustin, M.S. Evaluation of sequential and selective extraction methods for determination of mercury speciation and mobility in mine waste. Appl. Geochem. 2003, 18, 567–576. [Google Scholar] [CrossRef]
- Higueras, P.; Esbrí, J.M.; García-Ordiales, E.; González-Corrochano, B.; López-Berdonces, M.A.; García-Noguero, E.M.; Alonso-Azcárate, J.; Martínez-Coronado, A. Potentially harmful elements in soils and holm-oak trees (Quercus ilex L.) growing in mining sites at the Valle de Alcudia Pb-Zn district (Spain)–Some clues on plant metal uptake. J. Geochem. Explor. 2016, 182, 166–179. [Google Scholar] [CrossRef]
- Sahuquillo, A.; López-Sánchez, J.F.; Rubio, R.; Rauret, G.; Thomas, R.P.; Davidson, C.M.; Ure, A.M. Use of a certified reference material for extractable trace metals to assess sources of uncertainty in the BCR three-stage sequential procedure. Anal. Chim. Acta 1999, 382, 317–327. [Google Scholar] [CrossRef]
- Lindsay, W.L. Chemical Equilibria in Soils; John Wiley and Sons: New York, NY, USA, 1979. [Google Scholar]
- Gee, C.; Ramsey, M.H.; Maskall, J.; Thornton, I. Buffering from secondary minerals as a migration limiting factor in lead polluted soils at historical smelting sites. Appl. Geochem. 2001, 16, 1193–1199. [Google Scholar] [CrossRef]
- Chlopecka, A.; Bacon, J.R.; Wilson, M.J.; Kay, J. Forms of cadmium lead and zinc in contaminated soils from southwest Poland. J. Environ. Qual. 1996, 25, 69–79. [Google Scholar] [CrossRef]
- Rodriguez, L.; Ruiz, E.; Alonso-Azcarate, J.; Rincón, J. Heavy metals distribution and chemical speciation in tailings and soils around a Pb-Zn mine in Spain. J. Environ. Manag. 2009, 90, 1106–1116. [Google Scholar] [CrossRef]
- Hall, G.E.M. Analytical perspective on trace element species of interest in exploration. J. Geochem. Explor. 1998, 61, 1–19. [Google Scholar] [CrossRef]
- Boussen, S. Evolution de haldes plombo-zincifères dans le nord de la Tunisie: L’exemple d’un contexte carbonaté. Ph.D. Thesis, Faculté des Sciences de Tunis, Université de Tunis El Manar, Tunis, Tunisia, 2 July 2010. [Google Scholar]
- Lillo, J.; Oyarzun, R.; Esbrí, J.M.; García-Lorenzo, M.-L.; Higueras, P. Pb–Zn–Cd–As Pollution in Soils Affected by Mining Activities in Central and Southern Spain: A Scattered Legacy Posing Potential Environmental and Health Concerns. In Environment, Energy and Climate Change I; Springer: Cham, Switzerland, 2014; Volume 32. [Google Scholar]
- Callender, E. Heavy Metals in the Evvironnement; US Geological Servey: Westerly, RI, USA, 2004.
- Manceau, A.; Boisset, M.C.; Sarret, G.; Hazemann, J.L.; Mench, M.; Cambier, P.; Prost, R. Direct determination of lead speciation in contaminated soils by EXAFS spectroscopy. Environ. Sci. Technol. 1996, 30, 1540–1552. [Google Scholar] [CrossRef]
- Roberts, D.R.; Scheinost, A.C.; Sparks, D.L. Zinc speciation in a smelter-contaminated soil profile using bulk and microspectroscopic techniques. Environ. Sci. Technol. 2002, 36, 1742–1750. [Google Scholar] [CrossRef] [PubMed]
- Fan, T.; Wang, Y.; Li, C.; He, J.; Gao, J.; Zhou, D.; Friedman, S.P.; Sparks, D.L. Effect of Organic Matter on Sorption of Zn on Soil: Elucidation by Wien Effect Measurements and EXAFS Spectroscopy. Environ. Sci. Technol. 2016, 50, 2931–2937. [Google Scholar] [CrossRef]
- Lin, Y.; Tiegeng, L. Sphalerite chemistry, Niujiaotang Cd-rich zinc deposit, Guizhou, Southwest China. Chin. J. Geochem. 1999, 18, 62–68. [Google Scholar] [CrossRef]
- Soon, Y.K.; Bates, T.E. Chemical pools of Cd, Ni and Zn in polluted soils and some preliminary indications of their availability to plants. J. Soil Sci. 1982, 33, 477–488. [Google Scholar] [CrossRef]
- Antoniadis, V.; Alloway, B.J. The role of dissolved organic carbon in the mobility of Cd, Ni and Zn in sewage sludge-amended soils. Environ. Pollut. 2002, 117, 515–521. [Google Scholar] [CrossRef]
| Materials | Samples | Zn (mg·kg−1) | Pb (mg·kg−1) | Cd (mg·kg−1) |
|---|---|---|---|---|
| Tailings | DF1 | 3795 | 1865 | 25 |
| DF2 | 3815 | 1675 | 35 | |
| DF3 | 4235 | 3740 | 40 | |
| DG1 | 3720 | 2110 | 50 | |
| DG2 | 4855 | 3425 | 15 | |
| DG3 | 5135 | 2395 | 70 | |
| Sediments | SED4 | 1445 | 1770 | 20 |
| SED6 | 730 | 1870 | 40 | |
| SED12 | 1645 | 1530 | 15 | |
| Soils | S2 | 955 | 860 | 10 |
| S3 | 1005 | 1395 | 9 | |
| S5 | 1020 | 960 | 10 | |
| S8 | 130 | 280 | 5 | |
| S9 | 325 | 495 | 7 | |
| S10 | 305 | 260 | 5 | |
| S11 | 215 | 220 | 5 | |
| S13 | 1080 | 1825 | 12 | |
| S14 | 855 | 1075 | 10 | |
| S15 | 245 | 370 | 4 |
| Samples | Concentrations (mg·kg−1) | ||
|---|---|---|---|
| Zn | Cd | Pb | |
| DG1 | 1516 (1.36%) | 0.4 (0.39%) | 508 (1.77%) |
| DG2 | 1800 (1.51%) | 0.4 (0.35%) | 442 (1.47%) |
| DG3 | 1854 (1.48%) | 0.4 (0.35%) | 610 (2.04%) |
| DG4 | 1356 (1.29%) | 0.4 (0.37%) | 340 (1.62%) |
| DG5 | 1200 (1.22%) | 0.3 (0.27%) | 466 (2.54%) |
| DF1 | 1136 (1.33%) | 0.4 (0.46%) | 778 (1.94%) |
| DF2 | 858 (1.13%) | 0.5 (0.5%) | 1162 (2.24%) |
| DF3 | 1306 (1.71%) | 0.5 (0.43%) | 526 (0.88%) |
| DF4 | 1252 (1.005) | 0.2 (0.16%) | 590 (1.81%) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elmayel, I.; Esbrí, J.M.; Efrén, G.-O.; García-Noguero, E.-M.; Elouear, Z.; Jalel, B.; Farieri, A.; Roqueñí, N.; Cienfuegos, P.; Higueras, P. Evolution of the Speciation and Mobility of Pb, Zn and Cd in Relation to Transport Processes in a Mining Environment. Int. J. Environ. Res. Public Health 2020, 17, 4912. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph17144912
Elmayel I, Esbrí JM, Efrén G-O, García-Noguero E-M, Elouear Z, Jalel B, Farieri A, Roqueñí N, Cienfuegos P, Higueras P. Evolution of the Speciation and Mobility of Pb, Zn and Cd in Relation to Transport Processes in a Mining Environment. International Journal of Environmental Research and Public Health. 2020; 17(14):4912. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph17144912
Chicago/Turabian StyleElmayel, Intissar, José María Esbrí, García-Ordiales Efrén, Eva-María García-Noguero, Zouhair Elouear, Bouzid Jalel, Alessandro Farieri, Nieves Roqueñí, Pablo Cienfuegos, and Pablo Higueras. 2020. "Evolution of the Speciation and Mobility of Pb, Zn and Cd in Relation to Transport Processes in a Mining Environment" International Journal of Environmental Research and Public Health 17, no. 14: 4912. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph17144912





