Exposure Assessment of Environmental Tobacco Aerosol from Heated Tobacco Products: Nicotine and PM Exposures under Two Limited Conditions
Abstract
:1. Introduction
2. Methods
2.1. No-Observable-Adverse-Effect Level for Nicotine
2.2. Toxicological Risk Assessment of Nicotine
2.3. Using HTPs and Smoking Cigarette
2.4. Concentration of Nicotine
2.5. Concentration of Particulate Matter 2.5 (PM2.5)
2.6. Test Shower Cubicle
2.7. Test Room
3. Results
3.1. Indoor Air Quality in the Small Room
3.2. Indoor Air Quality under the Usual Condition
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Disclaimer
References
- World Health Organization (WHO). Heated Tobacco Products (HTPs) Information Sheet. Available online: https://apps.who.int/iris/bitstream/handle/10665/272875/WHO-NMH-PND-17.6-eng.pdf?ua=1 (accessed on 24 September 2020).
- Mallock, N.; Pieper, E.; Hutzler, C.; Henkler-Stephani, F.; Luch, A. Heated tobacco products: A review of current knowledge and initial assessments. Front. Public Health 2019, 7, 287. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sutanto, E.; Miller, C.; Smith, D.M.; O’Connor, R.J.; Quah, A.C.K.; Cummings, K.M.; Xu, S.; Fong, G.T.; Hyland, A.; Ouimet, J.; et al. Prevalence, use behaviors, and preferences among users of heated tobacco products: Findings from the 2018 ITC Japan survey. Int. J. Environ. Res. Public Health 2019, 16, 4630. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bialous, S.A.; Glantz, S.A. Heated tobacco products: Another tobacco industry global strategy to show progress in tobacco control. Tob. Control 2018, 27, s111–s117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tabuchi, T.; Gallus, S.; Shinozaki, T.; Nakaya, T.; Kunugita, N.; Colwell, B. Heat-not-burn tobacco product use in Japan: Its prevalence, predictors and perceived symptoms from exposure to secondhand heat-not-burn tobacco aerosol. Tob. Control 2018, 27, e25–e33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakamura, M. Judou-Kitsuen Boushi-Tou No Tabako-Taisaku No Suishin-Ni Kansuru Kenkyu; Heisei 29 Nendo Soukatsu Buntan Kenkyu Houkokusyo (MHLW Granted Research Group’s Report; Research for Promotion to Prevent Environmental Tobacco Smoke and Others. Fiscal year 2018). Available online: https://mhlw-grants.niph.go.jp/niph/search/NIDD02.do?resrchNum=201809001A (accessed on 24 September 2020). (In Japanese).
- Simonavicius, E.; McNeill, A.; Shahab, L.; Brose, L.S. Heat-not-burn tobacco products: A systematic literature review. Tob. Control 2019, 28, 582–594. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). WHO Report on Global Tobacco Epidemic. 2019. Available online: https://www.who.int/teams/health-promotion/tobacco-control/who-report-on-the-global-tobacco-epidemic-2019 (accessed on 24 September 2020).
- Ratajczak, A.; Jankowski, P.; Strus, P.; Feleszko, W. Heat not burn tobacco product—A new global trend: Impact of heat-not-burn tobacco products on public health, a systematic review. Int. J. Environ. Res. Public Health 2020, 17, 409. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, P.N.; Djurdjevic, S.; Weitkunat, R.; Baker, G. Estimating the population health impact of introducing a reduced-risk tobacco product into Japan. The effect of differing assumptions, and some comparisons with U.S. Regul. Toxocol. Pharmacol. 2018, 100, 92–104. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health, Labour and Welfare. Partial Revision of Health Promotion Act (No. 78 of 2108). Available online: https://www.mhlw.go.jp/english/policy/health-medical/health/dl/201904kenko.pdf (accessed on 24 September 2020).
- International Union Against Tuberculosis and Lung Disease. The Union’s Position on Heat-Not-Burn (HNB) Tobacco Products. Available online: https://theunion.org/news/union-position-on-heat-not-burn-tobacco-products-published-precautionary-principle-advised (accessed on 24 September 2020).
- Mitova, M.I.; Campelos, P.B.; Goujon-Ginglinger, C.G.; Maeder, S.; Mottier, N.; Rouget, E.G.R.; Tharin, M.; Tricker, A.R. Comparison of the impact of the Tobacco Heating System 2.2 and a cigarette on indoor air quality. Regul. Toxicol. Pharmacol. 2016, 80, 91–101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rijksinstituut voor Volksgezondheid en Milieu (National Institute for Public Health and the Environment, RIVM). De Gezondheidsrisico’s Van E-sigaretten Voor Omstanders. Briefrapport 2016-0036. Available online: https://www.rivm.nl/bibliotheek/rapporten/2016-0036.pdf (accessed on 24 September 2020).
- Environmental Protection Agency (US EPA). Reregistration Eligibility Decision for Nicotine. Available online: https://archive.epa.gov/pesticides/reregistration/web/pdf/nicotine_red.pdf (accessed on 24 September 2020).
- National Institute of Technology and Evaluation (NITE) and Chemicals Evaluation and Research Institute Japan (CERI) Kagaku Busshitsu no Shoki-hyouka Shisin (Guidelines for Initial Risk Assessment of Chemical Substances) Version 2.0. Available online: https://www.nite.go.jp/chem/chrip/chrip_search/dt/pdf/CI_02_001/guidance_ver2_20070115.pdf (accessed on 24 September 2020). (In Japanese).
- Tobacco Institute of Japan. Joui 20 Meigara Suii (Changes in Top 20 Cigarette Brand Names by Sales Volume). Data Related to Tobacco. Available online: https://www.tioj.or.jp/data/pdf/200424_03.pdf (accessed on 24 September 2020). (In Japanese).
- International Standard Organization (ISO). Environmental Tobacco Smoke—Determination of Aerosol Phase Nicotine and 3-Ethenylpyridine in Air—Gas-Chromatographic Method; International Standard Organization: Geneva, Switzerland, 2003; ISO 18145:2003. [Google Scholar]
- Moyer, T.P.; Charlson, J.R.; Enger, R.J. Simultaneous analysis of nicotine, nicotine metabolites, and tobacco alkaloids in serum or urine by tandem mass spectrometry, with clinically relevant metabolic profiles. Clin. Chem. 2002, 48, 1460–1471. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ministry of Health, Labour and Welfare. Tobacco no K-chi Sokutei no Gaiyou (Outline of Measurement of K Value for Tobacco Smoke). Available online: https://www.mhlw.go.jp/file/05-Shingikai-11201000-Roudoukijunkyoku-Soumuka/0000070587.pdf (accessed on 24 September 2020). (In Japanese)
- Lee, K.; Hahn, E.J.; Pieper, N.; Okoli, C.T.C.; Repace, J.; Troutman, A. Differential impacts of smoke-free laws on indoor air quality. J. Environ. Health 2008, 70, 24–30. [Google Scholar] [PubMed]
- Ministry of Health, Labour and Welfare. Shokuba NI Okeru Judou-Kitsuen-Boushi-Taisaku-Kijun Kentou-Iinkai Houkokusho (The Report on Committee on Standards for Measures to Prevent Second-Hand Smoking in the Workplace). Available online: https://www.mhlw.go.jp/file/05-Shingikai-11201000-Roudoukijunkyoku-Soumuka/0000066649.pdf (accessed on 24 September 2020). (In Japanese)
- Bekki, K.; Inaba, Y.; Uchiyama, S.; Kunugita, N. Comparison of chemicals in mainstream smoke in heat-not-burn tobacco and combustion cigarettes. J. Univ. Occup. Environ. Health Jpn. 2017, 39, 201–207. [Google Scholar]
- Uchiyama, S.; Noguchi, M.; Takagi, M.; Hayashida, H.; Inaba, Y.; Ogura, H.; Kunugita, N. Simple determination of gaseous and particulate compounds generated from heated tobacco products. Chem. Res. Toxicol. 2018, 31, 585–593. [Google Scholar] [CrossRef] [PubMed]
- Khachatoorian, C.; Jacob, P., III; Benowitz, N.L.; Talbot, P. Electronic cigarette chemicals transfer from a vape shop to a nearby business in a multiple-tenant retail building. Tob. Control 2019, 28, 519–525. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, R.; Aherrera, A.; Isichei, C.; Olmedo, P.; Jarmul, S.; Cohen, J.E.; Navas-Acien, A.; Rule, A.M. Assessment of indoor air quality at an electronic cigarette (Vaping) convention. J. Expo. Sci. Environ. Epidemiol. 2018, 28, 522–529. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kawai, H. Ganbarazu-ni Suppari Yamerareru Kin-en (Smoking can be Quitted without Trying Hard); Sunmark Publishing: Shinjuku-ku, Tokyo, Japan, 2017. (In Japanese) [Google Scholar]
- Goujon-Ginglinger, C.G. Indoor Air Chemistry—Assessment of Environmental Aerosols Generated by the Tobacco Heating System 2.2; Society of Indoor Environment: Okinawa, Japan, 2015; Available online: https://www.pmiscience.com/resources/docs/default-source/library-documents/c-_goujon_jpn.pdf?sfvrsn=8cb0f606_2 (accessed on 24 September 2020).
- US Environmental Protection Agency (USEPA). The National Ambient Air Quality Standards for Particle Pollution, Revised Air Quality Standards for Particle Pollution and Update to the Air Quality Index (AQI). Available online: https://www.epa.gov/sites/production/files/2016-04/documents/2012_aqi_factsheet.pdf (accessed on 24 September 2020).
- Ministry of the Environment. Chu-I Kanki No Tameno Zanteiteki-Na Shishin (Interim Guidelines for Alerting—Response to Air Pollution Caused by Particulate Matter (PM2.5)). Available online: http://www.env.go.jp/air/osen/pm/info.html#GUIDELINE (accessed on 24 September 2020). (In Japanese)
- International Agency for Research on Cancer (IARC). Tobacco Smoke and Involuntary Smoking; IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; 83 Series; IARC: Lyon, France, 2004; Volume 83. [Google Scholar]
- Stephens, W.E. Comparing the cancer potencies of emissions from vapourised nicotine products including e-cigarettes with those of tobacco smoke. Tob. Control 2018, 27, 10–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- National Institute of Technology and Evaluation. Risk Assessment on Chemicals—For Better Understanding. Version 8. Available online: https://www.nite.go.jp/data/000009058.pdf. (accessed on 24 September 2020).
- Endo, O.; Matsumoto, M.; Inaba, Y.; Sugita, K.; Nakajima, D.; Goto, S.; Ogata, H.; Suzuki, G. Nicotine, tar, and mutagenicity of mainstream smoke generated by machine smoking with International Organization for Standardization and Health Canada Intense regimes of major Japanese cigarette brands. J. Health Sci. 2009, 55, 421–427. [Google Scholar] [CrossRef] [Green Version]
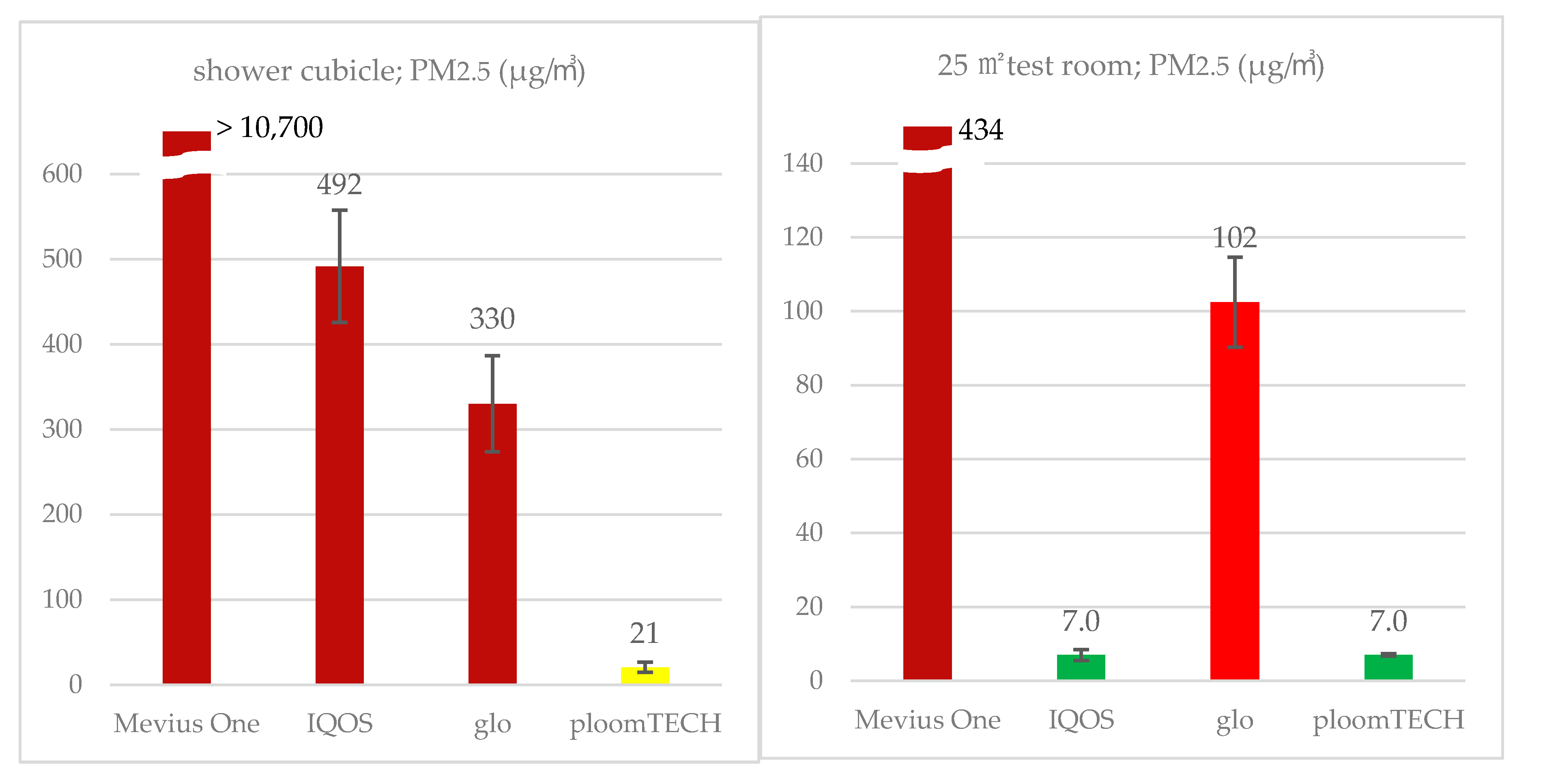
| HTP | Product | Nicotine (μg/m3) | PM2.5 (μg/m3) | ||||
|---|---|---|---|---|---|---|---|
| 1.0 m | 1.8 m | 1.0 m | 1.8 m | ||||
| Mean | SD | Mean | SD | ||||
| ploomTECH | Mevius regular | 29.3 | 25.9 | 21 | 55 | 10 | 6.6 |
| Glo | Kent | 160 | 111 | 330 | 564 | 99 | 119 |
| IQOS | Marlboro regular | 257 | 212 | 492 | 667 | 413 | 466 |
| (cigarette) | Mevius One | 1040 | 2420 | >10,700 | - | >5800 | - |
| HTP | Product | Nicotine (μg/m3) | PM2.5 (μg/m3) | ||||
|---|---|---|---|---|---|---|---|
| 1.5 m | 2.5 m | 1.5 m | 2.5 m | ||||
| Mean | SD | Mean | SD | ||||
| ploomTECH | Mevius Regular | 0.48 | 0.41 | 6.5 | 5.8 | 7.0 | 2.7 |
| Glo | Kent | 2.3 | 3.0 | 102 | 95 | 56 | 56 |
| IQOS | Marlboro Regular | 2.6 | 2.7 | 7.0 | 11.6 | 6.9 | 4.0 |
| (cigarette) | Mevius One | 130 | 160 | 378 | 215 | 434 | 243 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hirano, T.; Shobayashi, T.; Takei, T.; Wakao, F. Exposure Assessment of Environmental Tobacco Aerosol from Heated Tobacco Products: Nicotine and PM Exposures under Two Limited Conditions. Int. J. Environ. Res. Public Health 2020, 17, 8536. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph17228536
Hirano T, Shobayashi T, Takei T, Wakao F. Exposure Assessment of Environmental Tobacco Aerosol from Heated Tobacco Products: Nicotine and PM Exposures under Two Limited Conditions. International Journal of Environmental Research and Public Health. 2020; 17(22):8536. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph17228536
Chicago/Turabian StyleHirano, Tomoyasu, Tokuaki Shobayashi, Teiji Takei, and Fumihiko Wakao. 2020. "Exposure Assessment of Environmental Tobacco Aerosol from Heated Tobacco Products: Nicotine and PM Exposures under Two Limited Conditions" International Journal of Environmental Research and Public Health 17, no. 22: 8536. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph17228536





