The Association between Body Mass Index and Leisure-Time Physical Activity in Adults with Multiple Sclerosis
Abstract
:1. Introduction
2. Method
2.1. Study Design and Participants
2.2. Godin Leisure-Time Exercise Questionnaire (GLTEQ)
2.3. Weight Status
2.4. Expanded Disability Status Scale (EDSS)
2.5. Statistical Analysis
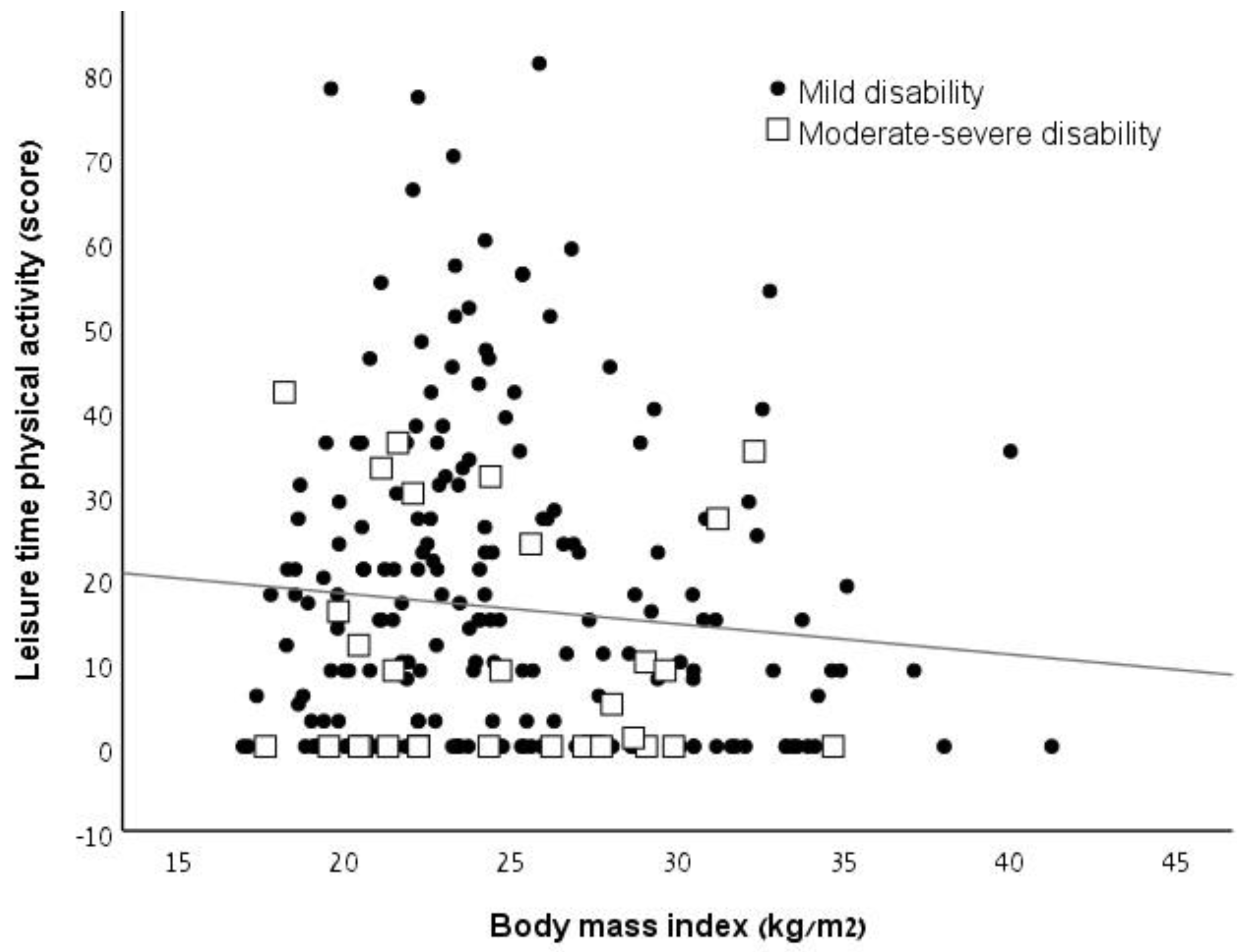
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Correale, J.; Gaitán, M.I.; Ysrraelit, M.C.; Fiol, M.P. Progressive multiple sclerosis: From pathogenic mechanisms to treatment. Brain 2017, 140, 527–546. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marck, C.H.; Neate, S.L.; Taylor, K.L.; Weiland, T.J.; Jelinek, G.A. Prevalence of comorbidities, overweight and obesity in an international sample of people with multiple sclerosis and associations with modifiable lifestyle factors. PLoS ONE 2016, 11, e0148573. [Google Scholar] [CrossRef] [PubMed]
- Swift, D.L.; McGee, J.E.; Earnest, C.P.; Carlisle, E.; Nygard, M.; Johannsen, N.M. The effects of exercise and physical activity on weight loss and maintenance. Prog. Cardiovasc. Dis. 2018, 61, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Pilutti, L.A.; Dlugonski, D.; Sandroff, B.M.; Klaren, R.E.; Motl, R.W. Internet delivered lifestyle activity interventions improves body composition in multiple sclerosis: Preliminary evidence from a randomized controlled trial. Arch. Phys. Med. Rehabil. 2014, 957, 1283–1288. [Google Scholar] [CrossRef] [PubMed]
- Keytsman, C.; Hansen, D.; Wens, I.; OEijnde, B. Impact of high intensity concurrent training on cardiovascular risk factors in persons with multiple sclerosis: Pilot study. Disabil. Rehabil. 2019, 41, 430–435. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, S.; Wonneberger, M. Long-term endurance exercise improves aerobic capacity in patients with relapsing-remitting multiple sclerosis: Impact of baseline fatigue. J. Neurol. Sci. 2014, 336, 29–35. [Google Scholar] [CrossRef]
- Wens, I.; Keytsman, C.; Deckx, N.; Cools, N.; Dalgas, U.; OEijnde, B. Brain derived neurotrophic factor in multiple sclerosis: Effect of 24 weeks endurance and resistance training. Eur. J. Neurol. 2016, 23, 1028–1035. [Google Scholar] [CrossRef]
- Polman, C.H.; Reingold, S.C.; Banwell, B.; Clanet, M.; Cohen, J.A.; Filippi, M.; Fujihara, K.; Havrdova, E.; Hutchinson, M.; Kappos, L.; et al. Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann. Neurol. 2011, 69, 292–302. [Google Scholar] [CrossRef] [Green Version]
- Kurtzke, J.F. Rating neurologic impairment in multiple sclerosis: An expanded disability status scale (EDSS). Neurology 1983, 33, 1444–1452. [Google Scholar] [CrossRef] [Green Version]
- Godin, G.; Shephard, R.J. A simple method to assess exercise behavior in the community. Can. J. Appl. Sport Sci. 1985, 10, 141–146. [Google Scholar]
- Sikes, E.M.; Richardson, E.V.; Cederberg, K.J.; Sasaki, J.E.; Sandroff, B.M.; Motl, R.W. Use of the Godin leisure-time exercise questionnaire in multiple sclerosis research: A comprehensive narrative review. Disabil. Rehabil. 2019, 41, 1243–1267. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Physical Status: The Use and Interpretation of Anthropometry; Report of a WHO Committee, World Health Organ Technical Report Series; World Health Organization: Geneva, Switzerland, 1995; Volume 854, pp. 1–452. [Google Scholar]
- Pilutti, L.A.; Motl, R.W. Body mass index underestimates adiposity in persons with multiple sclerosis. Arch. Phys. Med. Rehabil. 2016, 97, 405–412. [Google Scholar] [CrossRef]
- Pilluti, L.A.; McAuley, E.; Motl, R.W. Weight status and disability in multiple sclerosis: An examination of bi-directional associations over a 24-month period. Mult. Scler. Relat. Disord. 2012, 1, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Ward, C.L.; Suh, Y.; Lane, A.D.; Yan, H.; Ranadive, S.M.; Fernhall, B.; Motl, R.W.; Evans, E.M. Body composition and physical function in women with multiple sclerosis. J. Rehabil. Res. Dev. 2013, 50, 1139–1147. [Google Scholar] [CrossRef] [PubMed]
- Khurana, S.R.; Bamer, A.M.; Turner, A.P.; Wadhwani, R.V.; Bowen, J.D.; Leipertz, S.L.; Haselkorn, J.K. The prevalence of overweight and obesity in veterans with multiple sclerosis. Am. J. Phys. Med. Rehabil. 2009, 88, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Ewanchuk, B.W.; Gharagozloo, M.; Peelen, E.; Pilluti, L.A. Exploring the role of physical activity and exercise for managing vascular comorbidities in people with multiple sclerosis: A scoping review. Mult. Scler. Relat. Disord. 2018, 26, 19–32. [Google Scholar] [CrossRef]
- Mokhtarzade, M.; Agha-Alinejad, H.; Motl, R.W.; Negaresh, R.; Baker, J.S.; Zimmer, P. Weight control and physical activity in people with multiple sclerosis: Current knowledge and future perspectives. Complement Ther. Med. 2019, 43, 240–246. [Google Scholar] [CrossRef] [Green Version]
- Marrie, R.; Horwitz, R.; Cutter, G.; Tyry, T.; Campagnolo, D.; Vollmer, T. High frequency of adverse health behaviors in multiple sclerosis. Mult. Scler. 2009, 15, 105–113. [Google Scholar] [CrossRef]
- Slawta, J.N.; Wilcox, A.R.; McCubbin, J.A.; Nalle, D.J.; Fox, S.D.; Anderson, G. Health behaviors, body composition, and coronary heart disease risk in women with multiple sclerosis. Arch. Phys. Med. Rehabil. 2003, 84, 1823–1830. [Google Scholar] [CrossRef]
- Lambert, C.P.; Archer, R.L.; Evans, W.J. Body composition in ambulatory women with multiple sclerosis. Arch. Phys. Med. Rehabil. 2002, 83, 1559–1561. [Google Scholar] [CrossRef] [Green Version]
- Wens, I.; Dalgas, U.; Stenager, E.; OEijnde, B. Risk factors related to cardiovascular disease and the metabolic syndrome in multiple sclerosis-A systematic review. Mult. Scler. J. 2013, 19, 1556–1564. [Google Scholar] [CrossRef] [Green Version]
- Dalgas, U.; Langeskov-Christensen, M.; Stenager, E.; Riemenschneider, M.; Hvid, L.G. Exercise as medicine in multiple sclerosis-time for a paradigm shift: Preventive, symptomatic, and disease-modifying aspects and perspectives. Curr. Neurol. Neurosci. Rep. 2019, 19, 88. [Google Scholar] [CrossRef] [PubMed]
- Coulter, E.H.; Bond, S.; Dalgas, U.; Paul, L. The effectiveness of interventions targeting physical activity and/or sedentary behaviour in people with multiple sclerosis: A systematic review. Disabil. Rehabil. 2018, 5, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Motl, R.W.; Bollaert, R.E.; Sandroff, B.M. Validation of the Godin Leisure-Time Exercise Questionnaire classification coding system using accelerometry in multiple sclerosis. Rehabil. Psychol. 2018, 63, 77–82. [Google Scholar] [CrossRef] [PubMed]
| Variables | World Health Organization Standard of Obesity | Obesity as Defined for People with MS | |||||
|---|---|---|---|---|---|---|---|
| Normal (n = 144) | Overweight (n = 57) | Obese (n = 37) | p-Value | Normal (n = 142) | Obese (n = 96) | p-Value | |
| Age (years) | 38.07 ± 12.15 | 46.18 ± 13.04 | 40.93 ± 13.09 | <0.001 * | 38.06 ± 12.23 | 43.99 ± 13.14 | <0.001 * |
| Sex (female/male) | 87/57 | 31/26 | 20/17 | 86/56 | 52/44 | ||
| Height (cm) | 169.14 ± 9.08 | 168.32 ± 9.15 | 167.43 ± 10.29 | 0.576 | 169.11 ± 8.99 | 168.03 ± 9.70 | 0.379 |
| Weight (kg) | 62.04 ± 9.35 | 77.31 ± 10.13 | 92.86 ± 12.39 | <0.001 * | 61.88 ± 9.21 | 83.21 ± 13.46 | <0.001 * |
| BMI (kg/m2) | 21.60 ± 1.99 | 27.18 ± 1.38 | 33.06 ± 2.64 | <0.001 * | 21.55 ± 1.97 | 29.39 ± 3.52 | <0.001 * |
| Disease duration (years) | 5.52 ± 8.03 | 9.31 ± 9.06 | 5.38 ± 6.86 | 0.020 * | 5.52 ± 8.03 | 7.69 ± 8.41 | 0.068 |
| EDSS (score) | 2.53 ± 1.73 | 2.82 ± 1.65 | 2.70 ± 1.58 | 0.573 | 2.54 ± 1.74 | 2.76 ± 1.60 | 0.341 |
| Pyramidal | 1.37 ± 1.23 | 1.56 ± 1.29 | 1.58 ± 1.20 | 0.518 | 1.37 ± 1.23 | 1.56 ± 1.24 | 0.274 |
| Cerebellar | 0.74 ± 0.97 | 0.67 ± 0.92 | 0.73 ± 0.91 | 0.917 | 0.74 ± 0.97 | 0.69 ± 0.91 | 0.663 |
| Sensory | 0.79 ± 1.05 | 0.87 ± 0.97 | 0.85 ± 1.00 | 0.895 | 0.78 ± 1.05 | 0.87 ± 0.98 | 0.531 |
| GLTEQ (score) | 18.18 ± 18.16 | 15.26 ± 19.27 | 11.43 ± 13.96 | 0.106 | 18.16 ± 18.14 | 13.88 ± 17.47 | 0.071 |
| BMI | Active vs. Insufficiently Active | |||||
|---|---|---|---|---|---|---|
| n | ß | SE | OR | 95% CI | p-Value | |
| Model 1 (without adjustment for age, sex, and disability status) | ||||||
| Normal | 144 | Reference | 1.000 | |||
| Overweight | 57 | 0.067 | 0.341 | 1.070 | 0.548–2.086 | 0.844 |
| Obesity | 37 | 0.499 | 0.438 | 1.648 | 0.698–3.888 | 0.254 |
| Model 2 (with adjustment for age, sex, and disability status) | ||||||
| Normal | 144 | Reference | 1.000 | |||
| Overweight | 57 | 0.119 | 0.394 | 1.126 | 0.521–2.436 | 0.763 |
| Obesity | 37 | 0.089 | 0.462 | 1.093 | 0.442–2.702 | 0.847 |
| BMI | Active | Insufficiently Active | Total | x2 | p-Value |
|---|---|---|---|---|---|
| Normal | 44 (31.0%) | 98 (69.0%) | 142 (100.0%) | 0.420 | 0.564 |
| Obesity | 26 (27.1%) | 70 (72.9%) | 96 (100.0%) | ||
| Total | 70 (29.4%) | 168 (70.6%) | 238 (100.0%) |
| BMI | Mild vs. Moderate-Severe | |||||
|---|---|---|---|---|---|---|
| n | ß | SE | OR | 95% CI | p-Value | |
| Model 1 (without adjustment for age, sex, and physical activity) | ||||||
| Normal | 144 | Reference | 1.000 | |||
| Overweight | 57 | 0.515 | 0.420 | 1.674 | 0.735–3.810 | 0.220 |
| Obesity | 37 | −0.482 | 0.653 | 0.618 | 0.172–2.221 | 0.460 |
| Model 2 (with adjustment for age, sex, and physical activity) | ||||||
| Normal | 144 | Reference | 1.000 | |||
| Overweight | 57 | 0.122 | 0.452 | 1.130 | 0.466–2.738 | 0.787 |
| Obesity | 37 | −0.806 | 0.676 | 0.447 | 0.119–1.682 | 0.234 |
| BMI | Mild Disability | Moderate-Severe Disability | Total | x2 | p-Value |
|---|---|---|---|---|---|
| Normal | 124 (87.3%) | 18 (12.7%) | 142 (100.0%) | 0.179 | 0.701 |
| Obesity | 82 (85.4%) | 14 (14.6%) | 96 (100.0%) | ||
| Total | 206 (86.6%) | 32 (13.4%) | 238 (100.0%) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
So, W.-Y.; Kalron, A. The Association between Body Mass Index and Leisure-Time Physical Activity in Adults with Multiple Sclerosis. Int. J. Environ. Res. Public Health 2020, 17, 920. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph17030920
So W-Y, Kalron A. The Association between Body Mass Index and Leisure-Time Physical Activity in Adults with Multiple Sclerosis. International Journal of Environmental Research and Public Health. 2020; 17(3):920. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph17030920
Chicago/Turabian StyleSo, Wi-Young, and Alon Kalron. 2020. "The Association between Body Mass Index and Leisure-Time Physical Activity in Adults with Multiple Sclerosis" International Journal of Environmental Research and Public Health 17, no. 3: 920. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph17030920





