The Preventive Effect of Lactoferrin-Containing Yogurt on Gastroenteritis in Nursery School Children—Intervention Study for 15 Weeks
Abstract
:1. Introduction
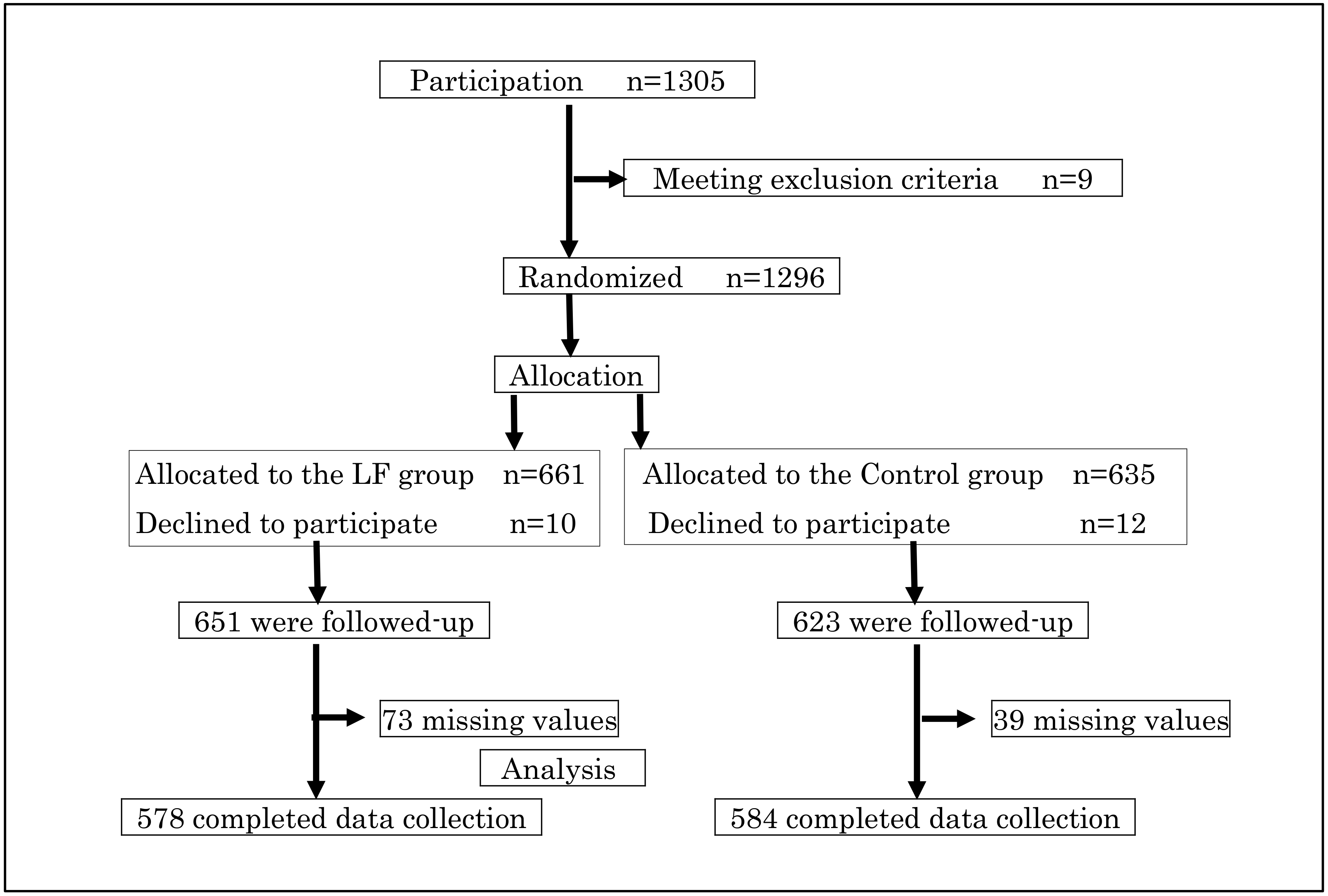
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Thongprachum, A.; Takanashi, S.; Kalesaran, A.F.; Okitsu, S.; Mizuguchi, M.; Hayakawa, S.; Ushijima, H. Four-year study of viruses that cause diarrhea in Japanese pediatric outpatients. J. Med. Virol. 2015, 87, 1141–1148. [Google Scholar] [CrossRef]
- Bartlett, A.V.; Moore, M.; Gary, G.W.; Starko, K.M.; Erben, J.J.; Meredith, B.A. Diarrheal illness among infants and toddlers in day care centers. II. Comparison with day care homes and households. J. Pediatr. 1985, 107, 503–509. [Google Scholar] [CrossRef]
- Bradley, R.H.; National Institute of Child Health and Human Development (NICHD) Early Child Care Research Network. Child care and common communicable illnesses in children aged 37 to 54 months. Arch. Pediatr. Adolesc. Med. 2003, 157, 196–200. [Google Scholar] [PubMed]
- Glass, R.I.; Parashar, U.D.; Estes, M.K. Norovirus gastroenteritis. N. Engl. J. Med. 2009, 361, 1776–1785. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rockx, B.; De Wit, M.; Vennema, H.; Vinjé, J.; De Bruin, E.; Van Duynhoven, Y.; Koopmans, M. Natural history of human calicivirus infection: A prospective cohort study. Clin. Infect. Dis. 2002, 35, 246–253. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akihara, S.; Phan, T.G.; Nguyen, T.A.; Hansman, G.; Okitsu, S.; Ushijima, H. Existence of multiple outbreaks of viral gastroenteritis among infants in a day care center in Japan. Arch. Virol. 2005, 150, 2061–2075. [Google Scholar] [CrossRef]
- Iizuka, S.; Tsunomori, Y.; Tabara, K.; Tsuda, K.; Fukuma, T. An outbreak of mixed infection of enterohemorrhagic Escherichia coli O26:H11 and norovirus genogroup II at a kindergarten in Shimane, Japan. Jpn. J. Infect. Dis. 2005, 58, 329–330. [Google Scholar]
- Johansson, B. Isolation of an iron-containing red protein from human milk. Acta Chem. Scand. 1960, 14, 510–512. [Google Scholar] [CrossRef]
- Groves, M.L. The isolation of a red protein from milk. J. Am. Chem. Soc. 1960, 82, 3345–3350. [Google Scholar] [CrossRef]
- Masson, P.L.; Heremans, J.F. Lactoferrin in milk from different species. Comp. Biochem. Physiol. 1971, 39, 119–129. [Google Scholar] [CrossRef]
- Berlutti, F.; Pantanella, F.; Natalizi, T.; Frioni, A.; Paesano, R.; Polimeni, A.; Valenti, P. Antiviral properties of lactoferrin—A natural immunity molecule. Molecules 2011, 16, 6992–7018. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McCann, K.B.; Lee, A.; Wan, J.; Roginski, H.; Coventry, M.J. The effect of bovine lactoferrin and lactoferricin B on the ability of feline calicivirus (a norovirus surrogate) and poliovirus to infect cell cultures. J. Appl. Microbiol. 2003, 95, 1026–1033. [Google Scholar] [CrossRef] [Green Version]
- Masson, P.L.; Heremans, J.F.; Schonne, E. Lactoferrin, an iron-binding protein in neutrophilic leukocytes. J. Exp. Med. 1969, 130, 643–658. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ellison, R.T., III; Giehl, T.J. Killing of gram-negative bacteria by lactoferrin and lysozyme. J. Clin. Investig. 1991, 88, 1080–1091. [Google Scholar] [CrossRef] [PubMed]
- Sekine, K.; Ushida, Y.; Kuhara, T.; Iigo, M.; Baba-Toriyama, H.; Moore, M.A.; Murakoshi, M.; Satomi, Y.; Nishino, H.; Kakizoe, T.; et al. Inhibition of initiation and early stage development of aberrant crypt foci and enhanced natural killer activity in male rats administered bovine lactoferrin concomitantly with azoxymethane. Cancer Lett. 1997, 121, 211–216. [Google Scholar] [CrossRef]
- Gomez, H.F.; Ochoa, T.J.; Herrera-Insua, I.; Carlin, L.G.; Cleary, T.G. Lactoferrin protects rabbits from Shigella flexneri-induced inflammatory enteritis. Infect. Immun. 2002, 70, 7050–7053. [Google Scholar] [CrossRef] [Green Version]
- Wakabayashi, H.; Takakura, N.; Teraguchi, S.; Tamura, Y. Lactoferrin feeding augments peritoneal macrophage activities in mice intraperitoneally injected with inactivated Candida albicans. Microbiol. Immunol. 2003, 47, 37–43. [Google Scholar] [CrossRef]
- Wakabayashi, H.; Yamauchi, K.; Takase, M. Lactoferrin research, technology and applications. Int. Dairy J. 2006, 16, 1241–1251. [Google Scholar] [CrossRef]
- Takakura, N.; Wakabayashi, H.; Ishibashi, H.; Yamauchi, K.; Teraguchi, S.; Tamura, Y.; Yamaguchi, H.; Abe, S. Effect of orally administered bovine lactoferrin on the immune response in the oral candidiasis murine model. J. Med. Microbiol. 2004, 54, 495–500. [Google Scholar] [CrossRef] [Green Version]
- Oda, H.; Wakabayashi, H.; Yamauchi, K.; Abe, F. Lactoferrin and bifidobacteria. Biometals 2014, 27, 915–922. [Google Scholar] [CrossRef]
- Egashira, M.; Takayanagi, T.; Moriuchi, M.; Moriuchi, H. Does daily intake of bovine lactoferrin-containing products ameliorate rotaviral gastroenteritis? Acta Paediatr. 2007, 96, 1242–1244. [Google Scholar] [CrossRef]
- Oda, H. The researches on the body defense effect of lctoferrin. Milk Sci. 2013, 62, 105–109. (In Japanese) [Google Scholar]
- Ochoa, T.J.; Chea-Woo, E.; Baiocchi, N.; Pecho, I.; Campos, M.; Prada, A.; Valdiviezo, G.; Lluque, A.; Lai, D.; Cleary, T.G. Randomized double-blind controlled trial of bovine lactoferrin for prevention of diarrhea in children. J. Pediatr. 2013, 162, 349–356. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, K.; Chai, L.; Li, H.; Zhang, Y.; Xie, H.M.; Shang, J.; Tian, W.; Yang, P.; Jiang, A.C. Effect of bovine lactoferrin from iron-fortified formulas on morbidity of diarrhea and respiratory tract infections of weaned infants in a randomized controlled trial. Nutrition 2016, 32, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Mori, K.; Hayashi, Y.; Akiba, T.; Noguchi, Y.; Yoshida, Y.; Kai, A.; Yamada, S.; Sakai, S.; Hara, M. Effects of handwashing on Feline Calicivirus removal as Norovirus surrogate. Kansenshogaku Zasshi 2006, 80, 496–500. (In Japanese) [Google Scholar] [CrossRef] [Green Version]
- Belliot, G.; Lopman, B.A.; Ambert-Balay, K.; Pothier, P. The burden of norovirus gastroenteritis: An important foodborne and healthcare-related infection. Clin. Microbiol. Infect. 2014, 20, 724–730. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Hao, L.; Pan, L.; Xue, C.; Liu, Q.; Zhao, X.; Zhu, W. Age, primary symptoms, and genotype characteristics of norovirus outbreaks in Shanghai schools in 2017. Sci. Rep. 2018, 8, 15238. [Google Scholar] [CrossRef]
- Embleton, N.D.; Berrington, J.E.; McGuire, W.; Stewart, C.J.; Cummings, S.P. Lactoferrin: Antimicrobial activity and therapeutic potential. Semin. Fetal Neonatal Med. 2013, 18, 143–149. [Google Scholar] [CrossRef]
- Ishikawa, H.; Awano, N.; Fukui, T.; Sasaki, H.; Kyuwa, S. The protective effects of lactoferrin against murine norovirus infection through inhibition of both viral attachment and replication. Biochem. Biophys. Res. Commun. 2013, 434, 791–796. [Google Scholar] [CrossRef]
- Binns, C.W.; Lee, A.H.; Harding, H.; Gracey, M.; Barclay, D.V. The CUPDAY study: Prebiotic-probiotic milk product in 1-3-yearold children attending childcare centres. Acta Paediatr. 2007, 96, 1646–1650. [Google Scholar] [CrossRef]
- Fox, M.J.; Ahuja, K.D.; Robertson, I.K.; Ball, M.J.; Eri, R.D. Can probiotic yogurt prevent diarrhoea in children on antibiotics? A double-blind, randomised, placebo-controlled study. BMJ Open 2015, 14, e006474. [Google Scholar] [CrossRef]
- Nakamura, M.; Hamazaki, K.; Matsumura, K.; Kasamatsu, H.; Tsuchida, A.; Inadera, H.; Japan Environment and Children’s Study Group. Infant dietary intake of yogurt and cheese and gastroenteritis at 1 year of age: The Japan Environment and Children’s Study. PLoS ONE 2019, 14, e0223495. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Szajewska, H.; Kotowska, M.; Mrukowicz, J.Z.; Armanska, M.; Mikolajczyk, W. Efficacy of Lactobacillus GG in prevention of nosocomial diarrhea in infants. J. Pediatr. 2001, 138, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Sur, D.; Manna, B.; Niyogi, S.K.; Ramamurthy, T.; Palit, A.; Nomoto, K.; Takahashi, T.; Shima, T.; Tsuji, H.; Kurakawa, T.; et al. Role of probiotic in preventing acute diarrhoea in children: A community-based, randomized, double-blind placebo-controlled field trial in an urban slum. Epidemiol. Infect. 2011, 139, 919–926. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hong Chau, T.T.; Minh Chau, N.N.; Hoang Le, N.T.; Chung The, H.; Voong Vinh, P.; Nguyen To, N.T.; Ngoc, N.M.; Tuan, H.M.; Chau Ngoc, T.L.; Kolader, M.E.; et al. A double-blind, randomized, placebo-controlled trial of lactobacillus acidophilus for the treatment of acute watery diarrhea in vietnamese children. Pediatr. Infect. Dis. J. 2018, 37, 35–42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El Manouni El Hassani, S.; de Boer, N.K.H.; Jansen, F.M.; Benninga, M.A.; Budding, A.E.; de Meij, T.G.J. Effect of daily intake of lactobacillus casei on microbial diversity and dynamics in a healthy pediatric population. Curr. Microbiol. 2019, 76, 1020–1027. [Google Scholar] [CrossRef] [Green Version]
- Nagata, S.; Asahara, T.; Ohta, T.; Yamada, T.; Kondo, S.; Bian, L.; Wang, C.; Yamashiro, Y.; Nomoto, K. Effect of the continuous intake of probiotic-fermented milk containing Lactobacillus casei strain Shirota on fever in a mass outbreak of norovirus gastroenteritis and the faecal microflora in a health service facility for the aged. Br. J. Nutr. 2011, 106, 549–556. [Google Scholar] [CrossRef] [Green Version]
- Allen, S.J.; Wareham, K.; Wang, D.; Bradley, C.; Hutchings, H.; Harris, W.; Dhar, A.; Brown, H.; Foden, A.; Gravenor, M.B.; et al. Lactobacilli and bifidobacteria in the prevention of antibiotic-associated diarrhoea and Clostridium difficile diarrhoea in older inpatients (PLACIDE): A randomised, double-blind, placebo-controlled, multicentre trial. Lancet 2013, 382, 1249–1257. [Google Scholar] [CrossRef] [Green Version]
- Orisaka, S.; Iwatsuki, K. Haloperidol-like effects of α-lactalbumin, lactoferrin and lysozyme in laboratory animals. Milk Sci. 2012, 61, 1–9. (In Japanese) [Google Scholar]
- Ammendoria, M.G.; Agamennone, K.; Pietrantoni, A.; Lannutti, F.; Siciliano, R.A.; De Giulio, B.; Amici, C.; Superti, F. Bovine lactoferrin-derived peptides as novel broad-spectrum inhibitors of influenza virus. Pathog. Glob. Health 2012, 106, 12–19. [Google Scholar] [CrossRef] [Green Version]
- Pietrantoni, A.; Ammendolia, M.G.; Superti, F. Bovine lactoferrin: Involvement of metal saturation and carbohydrates in the inhibition of influenza virus infection. Biochem. Cell Biol. 2012, 90, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Shin, K.; Wakabayashi, H.; Yamauchi, K.; Teraguchi, S.; Tamura, Y.; Kurokawa, M.; Shiraki, K. Effects of orally administered bovine lactoferrin and lactoperoxidase on influenza virus infection in mice. J. Med. Microbiol. 2005, 54, 717–723. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oda, H.; Miyakawa, M.; Mizuki, M.; Misawa, Y.; Tsukahara, T.; Tanaka, M.; Yamauchi, K.; Abe, F.; Nomiyama, T. Effects of lactoferrin on subjective skin conditions in winter: A Preliminary, randomized, double-blinded, placebo-controlled trial. Clin. Cosmet. Investig. Dermatol. 2019, 12, 875–880. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Characteristics | All | LF Group | Control Group | p Value |
|---|---|---|---|---|
| Number of participants | 1162 | 578 | 584 | |
| Sex | ||||
| Boys n (%) | 632 (54.4) | 318 (55.0) | 314 (53.8) | |
| Girls n (%) | 530 (45.6) | 260 (45.0) | 270 (46.2) | 0.67 a |
| Age (years) Mean ± SD | 4.7 ± 1.0 | 4.7 ± 0.9 | 4.7 ± 1.0 | 0.86 b |
| 3 years n (%) | 132 (11.4) | 63 (10.9) | 69 (11.8) | 0.90 a |
| 4 years n (%) | 350 (30.1) | 178 (30.8) | 172 (29.5) | |
| 5 years n (%) | 408 (35.1) | 205 (35.5) | 203 (34.8) | |
| 6 years n (%) | 272 (23.4) | 132 (22.8) | 140 (24.0) | |
| Height (cm) Mean ± SD | 107.4 ± 7.1 | 107.2 ± 7.3 | 107.6 ± 6.9 | 0.37 c |
| Weight (kg) Mean ± SD | 17.8 ± 3.0 | 17.7 ± 3.1 | 17.9 ± 3.0 | 0.23 c |
| Hand-washing | ||||
| Habitually | 1042 (89.7) | 514 (88.9) | 528 (90.4) | 0.44 a |
| Not habitually | 120 (10.3) | 64 (11.1) | 56 (9.6) | |
| Rotavirus vaccination n (%) | 13 (1.1) | 4 (0.7) | 9 (1.5) | 0.17 a |
| Reasons for Absence | All | LF Group | Control Group | p Value |
|---|---|---|---|---|
| Number of participants | 1162 | 578 | 584 | |
| Norovirus gastroenteritis | ||||
| the total number of absentees n (%) | 15 (1.3) | 7 (1.2) | 8 (1.4) | 0.81 |
| absent days (days) Mean ± SD | 2.2 ± 1.1 | 2.0 ± 0.8 | 2.4 ± 1.4 | 0.81 |
| Gastroenteritis | ||||
| the total number of absentees n (%) | 43 (3.7) | 27 (4.7) | 16 (2.7) | 0.08 |
| absent days (days) Mean ± SD | 2.1 ± 1.1 | 1.9 ± 1.1 | 2.5 ± 1.0 | 0.08 |
| Infectios gastroenteritis | ||||
| the total number of absentees n (%) | 29 (2.5) | 14 (2.4) | 15 (2.6) | 0.87 |
| absent days (days) Mean ± SD | 2.0 ± 1.1 | 2.1 ± 0.9 | 1.8 ± 1.3 | 0.18 |
| Diarrhea | ||||
| the total number of absentees n (%) | 31 (2.7) | 17 (2.9) | 14 (2.4) | 0.57 |
| absent days (days) Mean ± SD | 1.7 ± 1.1 | 1.5 ± 0.7 | 1.9 ± 1.4 | 0.67 |
| Vomiting | ||||
| the total number of absentees n (%) | 93 (8.0) | 44 (7.6) | 49 (8.4) | 0.63 |
| absent days (days) Mean ± SD | 1.5 ± 0.8 | 1.6 ± 0.8 | 1.5 ± 0.8 | 0.96 |
| All absence due to illnesses | ||||
| the total number of absentees n (%) | 892 (76.8) | 447 (77.3) | 445 (76.2) | 0.65 |
| absent days (days) Mean ± SD | 4.1 ± 3.0 | 4.0 ± 2.8 | 4.1 ± 3.2 | 0.77 |
| Reasons for Absence | 3 Days or More Than 3 Days Per Week | p Value a | 2 Days or Less Than 2 Days Per Week | p Value b | Control Group | p for Trend |
|---|---|---|---|---|---|---|
| Number of participants | 234 | 344 | 584 | |||
| Norovirus gastroenteritis | ||||||
| the total number of absentees n (%) | 3 (1.3) | 1 | 4 (1.2) | 1 | 8 (1.4) | 0.92 |
| absent days (days) Mean ± SD | 1.7 ± 0.6 | 0.5 | 2.3 ± 1.0 | 0.86 | 2.4 ± 1.4 | 0.6 |
| Gastroenteritis | ||||||
| the total number of absentees n (%) | 11 (4.7) | 0.16 | 16 (4.7) | 0.12 | 16 (2.7) | 0.09 |
| absent days (days) Mean ± SD | 1.7 ± 0.9 | 0.06 | 2.1 ± 1.2 | 0.23 | 2.5 ± 1.0 | 0.06 |
| Infectios gastroenteritis | ||||||
| the total number of absentees n (%) | 2 (0.9) | 0.17 | 12 (3.5) | 0.42 | 15 (2.6) | 0.5 |
| absent days (days) Mean ± SD | 2.0 ± 1.4 | 0.74 | 2.2 ± 0.9 | 0.16 | 1.8 ± 1.3 | 0.21 |
| Diarrhea | ||||||
| the total number of absentees n (%) | 9 (3.8) | 0.26 | 8 (2.3) | 0.95 | 14 (2.4) | 0.39 |
| absent days (days) Mean ± SD | 1.3 ± 0.5 | 0.45 | 1.8 ± 0.9 | 0.94 | 1.9 ± 1.4 | 0.44 |
| Vomiting | ||||||
| the total number of absentees n (%) | 10 (4.3) | 0.04 | 34 (9.9) | 0.43 | 49 (8.4) | 0.25 |
| absent days (days) Mean ± SD | 1.3 ± 0.5 | 0.57 | 1.6 ± 0.9 | 0.75 | 1.5 ± 0.8 | 0.88 |
| All absence due to illnesses | ||||||
| the total number of absentees n (%) | 175 (74.8) | 0.67 | 272 (79.1) | 0.31 | 445 (76.2) | 0.91 |
| absent days (days) Mean ± SD | 3.6 ± 2.8 | 0.02 | 4.3 ± 2.9 | 0.19 | 4.1 ± 3.2 | 0.17 |
| Variables | Crude Odds Ratio | Adjusted Odds Ratio | |||||
|---|---|---|---|---|---|---|---|
| n | Odds ratio | 95 % CI | p Value | Odds ratio | 95 % CI | p Value | |
| Sex | |||||||
| Boys | 632 | 1.30 | 0.84–2.00 | 0.24 | 1.34 | 0.87–2.07 | 0.19 |
| Girls | 530 | reference | reference | ||||
| Age | |||||||
| 3 years | 132 | 1.14 | 0.58–2.21 | 0.71 | 1.06 | 0.55–2.08 | 0.86 |
| 4 years | 350 | 0.50 | 0.27–0.91 | 0.02 | 0.49 | 0.27–0.89 | 0.02 |
| 5 years | 408 | 0.72 | 0.42–1.22 | 0.22 | 0.71 | 0.41–1.22 | 0.21 |
| 6 years | 272 | reference | reference | ||||
| The habit of hand-washing | |||||||
| Habitually | 1042 | reference | reference | ||||
| Not habitually | 120 | 0.80 | 0.38 –1.70 | 0.57 | 0.76 | 0.36–1.62 | 0.49 |
| LF-containing yogurt intake | |||||||
| Control Group | 584 | 2.05 | 1.02–4.12 | 0.044 | 2.03 | 1.01–4.09 | 0.047 |
| LF group < 3 days | 344 | 2.46 | 1.19–5.08 | 0.02 | 2.48 | 1.19–5.14 | 0.02 |
| LF group ≥ 3 days | 234 | reference | reference | ||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsukahara, T.; Fujimori, A.; Misawa, Y.; Oda, H.; Yamauchi, K.; Abe, F.; Nomiyama, T. The Preventive Effect of Lactoferrin-Containing Yogurt on Gastroenteritis in Nursery School Children—Intervention Study for 15 Weeks. Int. J. Environ. Res. Public Health 2020, 17, 2534. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph17072534
Tsukahara T, Fujimori A, Misawa Y, Oda H, Yamauchi K, Abe F, Nomiyama T. The Preventive Effect of Lactoferrin-Containing Yogurt on Gastroenteritis in Nursery School Children—Intervention Study for 15 Weeks. International Journal of Environmental Research and Public Health. 2020; 17(7):2534. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph17072534
Chicago/Turabian StyleTsukahara, Teruomi, Anri Fujimori, Yuka Misawa, Hirotsugu Oda, Koji Yamauchi, Fumiaki Abe, and Tetsuo Nomiyama. 2020. "The Preventive Effect of Lactoferrin-Containing Yogurt on Gastroenteritis in Nursery School Children—Intervention Study for 15 Weeks" International Journal of Environmental Research and Public Health 17, no. 7: 2534. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph17072534






