Outcome Expectations for Exercise and Decisional Balance Questionnaires Predict Adherence and Efficacy of Exercise Programs in Dialysis Patients
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Criteria And Ethical Considerations
2.2. Study Design
2.3. The Main Study Outcomes
2.3.1. Decisional Balance Scale
2.3.2. Outcome Expectations for Exercise Scale
2.3.3. Patient Adherence
2.4. Statistical Analysis
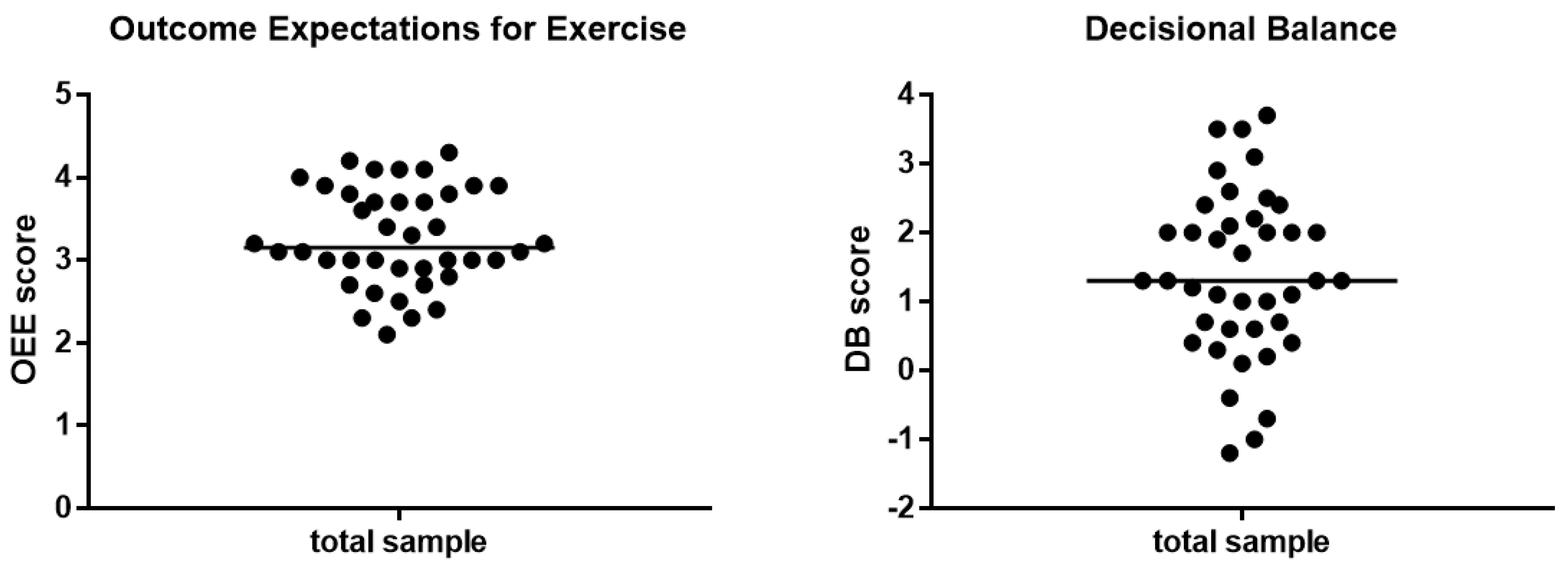
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Jha, V.; Garcia-Garcia, G.; Iseki, K.; Li, Z.; Naicker, S.; Plattner, B.; Saran, R.; Wang, A.Y.M.; Yang, C.W. Chronic kidney disease: Global dimension and perspectives. Lancet 2013, 382, 260–272. [Google Scholar] [CrossRef]
- Clarke, A.L.; Young, H.M.L.; Hull, K.L.; Hudson, N.; Burton, J.O.; Smith, A.C. Motivations and barriers to exercise in chronic kidney disease: A qualitative study. Nephrol. Dial. Transplant. 2015, 30, 1885–1892. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shlipak, M.G.; Fried, L.F.; Cushman, M.; Manolio, T.A.; Peterson, D.; Stehman-Breen, C.; Bleyer, A.; Newman, A.; Siscovick, D.; Psaty, B. Cardiovascular mortality risk in chronic kidney disease: Comparison of traditional and novel risk factors. J. Am. Med. Assoc. 2005, 293, 1737–1745. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hiraki, K.; Yasuda, T.; Hotta, C.; Izawa, K.P.; Morio, Y.; Watanabe, S.; Sakurada, T.; Shibagaki, Y.; Kimura, K. Decreased physical function in pre-dialysis patients with chronic kidney disease. Clin. Exp. Nephrol. 2013, 17, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Bučar Pajek, M.; Čuk, I.; Leskošek, B.; Mlinšek, G.; Buturović Ponikvar, J.; Pajek, J. Six-Minute Walk Test in Renal Failure Patients: Representative Results, Performance Analysis and Perceived Dyspnea Predictors. PLoS ONE 2016, 11, e0150414. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bučar Pajek, M.; Svilan, K.; Vivoda, T.; Škoberne, A.; Pajek, J. Isolated Effects of Renal Failure, Anthropometric Indices, and Serum Total Iron-Binding Capacity as Determinants of Muscle Performance in Hemodialysis Patients. J. Ren. Nutr. 2016, 26, 245–252. [Google Scholar] [CrossRef]
- Workeneh, B.T.; Mitch, W.E. Review of muscle wasting associated with chronic kidney disease. Am. J. Clin. Nutr. 2010, 91, 1128S–1132S. [Google Scholar] [CrossRef] [Green Version]
- Abdel-Kader, K.; Unruh, M.L.; Weisbord, S.D. Symptom burden, depression, and quality of life in chronic and end-stage kidney disease. Clin. J. Am. Soc. Nephrol. 2009, 4, 1057–1064. [Google Scholar] [CrossRef]
- Dashtidehkordi, A.; Shahgholian, N.; Attari, F. Exercise during hemodialysis and health promoting behaviors: a clinical trial. BMC Nephrol. 2019, 20, 96. [Google Scholar] [CrossRef] [Green Version]
- Bogataj, Š.; Pajek, M.; Pajek, J.; Buturović Ponikvar, J.; Paravlic, A. Exercise-Based Interventions in Hemodialysis Patients: A Systematic Review with a Meta-Analysis of Randomized Controlled Trials. J. Clin. Med. 2020, 9, 43. [Google Scholar] [CrossRef] [Green Version]
- Goodman, E.D.; Ballou, M.B. Perceived barriers and motivators to exercise in hemodialysis patients. Nephrol. Nurs. J. 2004, 31, 23–29. [Google Scholar] [PubMed]
- Pinto, B.M.; Clark, M.M.; Cruess, D.G.; Szymanski, L.; Pera, V. Changes in self-efficacy and decisional balance for exercise among obese women in a weight management program. Obes. Res. 1999, 7, 288–292. [Google Scholar] [CrossRef] [PubMed]
- Plotnikoff, R.C.; Blanchard, C.; Hotz, S.B.; Rhodes, R. Validation of the decisional balance scales in the exercise domain from the transtheoretical model: A longitudinal test. Meas. Phys. Educ. Exerc. Sci. 2001, 5, 191–206. [Google Scholar] [CrossRef]
- Jette, A.M.; Rooks, D.; Lachman, M.; Lin, T.H.; Levenson, C.; Heislein, D.; Giorgetti, M.M.; Harris, B.A. Home-based resistance training: Predictors of participation and adherence. Gerontologist 1998, 38, 412–421. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Resnick, B.; Zimmerman, S.I.; Orwig, D.; Furstenberg, A.-L.; Magaziner, J. Outcome Expectations for Exercise Scale: Utility and Psychometrics. Journals Gerontol. Ser. B Psychol. Sci. Soc. Sci. 2000, 55, S352–S356. [Google Scholar] [CrossRef] [Green Version]
- Gough, A.; Kee, F. Physical activity and behaviour change: the role of distributed motivation. Crit. Public Health 2018. [Google Scholar] [CrossRef] [Green Version]
- Samdal, G.B.; Eide, G.E.; Barth, T.; Williams, G.; Meland, E. Effective behaviour change techniques for physical activity and healthy eating in overweight and obese adults; systematic review and meta-regression analyses. Int. J. Behav. Nutr. Phys. Act. 2017, 14, 42. [Google Scholar] [CrossRef] [Green Version]
- Michie, S.; Ashford, S.; Sniehotta, F.F.; Dombrowski, S.U.; Bishop, A.; French, D.P. A refined taxonomy of behaviour change techniques to help people change their physical activity and healthy eating behaviours: The CALO-RE taxonomy. Psychol. Heal. 2011, 26, 1479–1498. [Google Scholar] [CrossRef]
- Keller, C.; Fleury, J.; Gregor-Holt, N.; Thompson, T. Predictive ability of social cognitive theory in exercise research: an integrated literature review. Online J. Knowl. Synth. Nurs. 1999, 6. [Google Scholar] [CrossRef]
- Anding, K.; Bär, T.; Trojniak-Hennig, J.; Kuchinke, S.; Krause, R.; Rost, J.M.; Halle, M. A structured exercise programme during haemodialysis for patients with chronic kidney disease: clinical benefit and long-term adherence. BMJ Open 2015, 5, e008709. [Google Scholar] [CrossRef]
- Heiwe, S.; Jacobson, S.H. Exercise training for adults with chronic kidney disease. In Cochrane Database of Systematic Reviews; Heiwe, S., Ed.; John Wiley & Sons, Ltd: Chichester, UK, 2011; p. CD003236. [Google Scholar]
- Smart, N.; Steele, M. Exercise Training in Hemodialysis Patients: A Systematic Review and Meta-Analysis. Nephrology 2011, 16, 626–632. [Google Scholar] [CrossRef] [PubMed]
- Koufaki, P.; Greenwood, S.A.; Macdougall, I.C.; Mercer, T.H. Exercise therapy in individuals with chronic kidney disease: a systematic review and synthesis of the research evidence. Annu. Rev. Nurs. Res. 2013, 31, 235–275. [Google Scholar] [CrossRef] [PubMed]
- Cheema, B.S.; Chan, D.; Fahey, P.; Atlantis, E. Effect of progressive resistance training on measures of skeletal muscle hypertrophy, muscular strength and health-related quality of life in patients with chronic kidney disease: A systematic review and meta-analysis. Sport. Med. 2014, 44, 1125–1138. [Google Scholar] [CrossRef] [PubMed]
- Sheng, K.; Zhang, P.; Chen, L.; Cheng, J.; Wu, C.; Chen, J. Intradialytic exercise in hemodialysis patients: A systematic review and meta-analysis. Am. J. Nephrol. 2014, 40, 478–490. [Google Scholar] [CrossRef]
- Heiwe, S.; Jacobson, S.H. Exercise training in adults with CKD: a systematic review and meta-analysis. Am. J. Kidney Dis. 2014, 64, 383–393. [Google Scholar] [CrossRef]
- Phan, K.; Jia, F.; Kamper, S.J. Effects of regular physical exercise training in adults with chronic kidney disease (PEDro synthesis). Br. J. Sports Med. 2016, 50, 317–318. [Google Scholar] [CrossRef]
- Young, H.M.L.; March, D.S.; Graham-Brown, M.P.M.; Jones, A.W.; Curtis, F.; Grantham, C.S.; Churchward, D.R.; Highton, P.; Smith, A.C.; Singh, S.J.; et al. Effects of intradialytic cycling exercise on exercise capacity, quality of life, physical function and cardiovascular measures in adult haemodialysis patients: a systematic review and meta-analysis. Nephrol. Dial. Transplant. 2018. [Google Scholar] [CrossRef] [Green Version]
- Van Lummel, R.C.; Ainsworth, E.; Lindemann, U.; Zijlstra, W.; Chiari, L.; Van Campen, P.; Hausdorff, J.M. Automated approach for quantifying the repeated sit-to-stand using one body fixed sensor in young and older adults. Gait Posture 2013, 38, 153–156. [Google Scholar] [CrossRef] [Green Version]
- Norman, K.; Stobäus, N.; Gonzalez, M.C.; Schulzke, J.-D.; Pirlich, M. Hand grip strength: Outcome predictor and marker of nutritional status. Clin. Nutr. 2011, 30, 135–142. [Google Scholar] [CrossRef]
- Bogataj, Š.; Pajek, J.; Buturović Ponikvar, J.; Hadžić, V.; Pajek, M. Kinesiologist-guided functional exercise in addition to intradialytic cycling program in end-stage kidney disease patients: a randomised controlled trial. Sci. Rep. 2020, 10. [Google Scholar] [CrossRef]
- Bohannon, R.W.; Bubela, D.J.; Magasi, S.R.; Wang, Y.-C.; Gershon, R.C. Sit-to-stand test: Performance and determinants across the age-span. Isokinet. Exerc. Sci. 2010, 18, 235–240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ross, R.M.; Murthy, J.N.; Wollak, I.D.; Jackson, A.S. The six minute walk test accurately estimates mean peak oxygen uptake. BMC Pulm. Med. 2010, 10, 31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bellace, J.V.; Healy, D.; Besser, M.P.; Byron, T.; Hohman, L. Validity of the Dexter Evaluation System’s Jamar dynamometer attachment for assessment of hand grip strength in a normal population. J. Hand Ther. 2000, 13, 46–51. [Google Scholar] [CrossRef]
- Beckham, S.G.; Harper, M. Functional training. ACSMs. Health Fit. J. 2010, 14, 24–30. [Google Scholar] [CrossRef]
- Boyle, M. Functional Training for Sports; Human Kinetics: Champaign, IL, USA, 2004; ISBN 9780736046817. [Google Scholar]
- Brill, P.A. Functional Fitness for Older Adults; Human Kinetics: Champaign, IL, USA, 2004; ISBN 9780736046565. [Google Scholar]
- Marcus, B.H.; Rakowski, W.; Rossi, J.S. Assessing motivational readiness and decision making for exercise. Health Psychol. 1992, 11, 257–261. [Google Scholar] [CrossRef]
- Hopkins, W. How to Interpret Changes in an Athletic Performance Test. Sportscience 2004, 8, 1–7. [Google Scholar]
- Kontos, P.C.; Miller, K.L.; Brooks, D.; Jassal, S.V.; Spanjevic, L.; Devins, G.M.; Souza, M.J.; Heck, C.; Laprade, J.; Naglie, G. Factors influencing exercise participation by older adults requiring chronic hemodialysis: A qualitative study. Int. Urol. Nephrol. 2007, 39, 1303–1311. [Google Scholar] [CrossRef]
- Patterson, M.S.; Meyer, M.R.U.; Beaujean, A.A.; Bowden, R.G. Using the social cognitive theory to understand physical activity among dialysis patients. Rehabil. Psychol. 2014, 59, 278–288. [Google Scholar] [CrossRef]
- Bandura, A. Self-efficacy: The Exercise of Control; W H Freeman/Times Books/Henry Holt & Co: New York, NY, USA, 1997; ISBN 0-7167-2626-2 (Hardcover); 0-7167-2850-8 (Paperback). [Google Scholar]
- Brown, S.A. Measuring perceived benefits and perceived barriers for physical activity. Am. J. Health Behav. 2005, 29, 107–116. [Google Scholar] [CrossRef] [Green Version]
- Taylor, R.S.; Brown, A.; Ebrahim, S.; Jolliffe, J.; Noorani, H.; Rees, K.; Skidmore, B.; Stone, J.A.; Thompson, D.R.; Oldridge, N. Exercise-based rehabilitation for patients with coronary heart disease: Systematic review and meta-analysis of randomized controlled trials. Am. J. Med. 2004, 116, 682–692. [Google Scholar] [CrossRef]
- Felson, D.T.; Lawrence, R.C.; Hochberg, M.C.; McAlindon, T.; Dieppe, P.A.; Minor, M.A.; Blair, S.N.; Berman, B.M.; Fries, J.F.; Weinberger, M.; et al. Osteoarthritis: New insights. Part 2: Treatment approaches. Ann. Intern. Med. 2000, 133, 726–737. [Google Scholar]
- Boulé, N.G.; Haddad, E.; Kenny, G.P.; Wells, G.A.; Sigal, R.J. Effects of exercise on glycemic control and body mass in type 2 diabetes mellitus: A meta-analysis of controlled clinical trials. J. Am. Med. Assoc. 2001, 286, 1218–1227. [Google Scholar] [CrossRef] [PubMed]
- Vickers, K.S.; Nies, M.A.; Patten, C.A.; Dierkhising, R.; Smith, S.A. Patients with diabetes and depression may need additional support for exercise. Am. J. Health Behav. 2006, 30, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Pajek, M.; Jerman, A.; Osredkar, J.; Ponikvar, J.B.; Pajek, J. Association of uremic toxins and inflammatory markers with physical performance in dialysis patients. Toxins (Basel) 2018, 10, 403. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Inclusion Criteria | Exclusion Criteria | Withdrawal Criteria |
|---|---|---|
| patients on HD renal replacement therapy > 3 months | chronic malignant or infectious disease | any intercurrent illness or trauma |
| 18−90 years old | uncontrolled arterial hypertension | the occurrence of an acute illness |
| stable medical condition | angina pectoris of Canadian Cardiovascular Society grade 2−4 | diagnosis of malignant disease during the research |
| capable of independent walking and feeding | New York Heart Association heart failure grade 3 or 4 | withdrawal of the consent to participate in the research |
| the presence of psychotic illness or a mental disability | ||
| a history of limb amputation (more than two fingers on the lower limb and/or upper limb) |
| Variable | All Participants (n = 40) | Experimental Group (n = 20) | Control Group (n = 20) |
|---|---|---|---|
| Age (years) | 63.6 ± 12.5 | 65.2 ± 12.1 | 61.9 ± 13.0 |
| Male sex (%) | 55% | 60% | 50% |
| Height (cm) | 167.9 ± 9.8 | 168.4 ± 9.6 | 167.5 ± 10.2 |
| Weight (kg) | 72.1 ± 15.8 | 72.6 ± 16.1 | 71.7 ± 15.9 |
| Dialysis vintage (years) | 7.4 ± 7.7 | 7.4 ± 8.1 | 7.5 ± 7.3 |
| Weekly dialysis duration (h) | 12.9 ± 2.3 | 12.5 ± 2.7 | 13.3 ± 1.9 |
| Lean tissue index (kg/m2) | 13.3 ± 2.6 | 13.6 ± 3.2 | 12.9 ± 2.0 |
| Fat tissue index (kg/m2) | 11.5 ± 5.4 | 11.4 ± 4.8 | 11.6 ± 6.1 |
| Phase angle (°) | 5.0 ± 0.9 | 5.2 ± 0.9 | 4.7 ± 0.9 |
| BIA assessed overhydration (L) | 1.4 ± 1.9 | 0.9 ± 1.1 | 1.9 ± 2.4 |
| Variable | OEE | DB | ||||||
|---|---|---|---|---|---|---|---|---|
| B | 95% CI | p | Partial Eta Squared | B | 95% CI | p | Partial Eta Squared | |
| 6MWT | 0.006 | 2.97 ± 0.16 (2.64 to 3.29) | 0.001 | 0.26 | 0.002 | 1.39 ± 0.17 (1.04 to 1.73) | 0.257 | 0.03 |
| STS10 | −0.043 | 2.97 ± 0.18 (2.6 to 3.33) | 0.079 | 0.08 | −0.028 | 1.39 ± 0.17 (1.04 to 1.73) | 0.224 | 0.04 |
| HG | 0.059 | 2.97 ± 0.18 (2.61 to 3.32) | 0.024 | 0.13 | −0.006 | 1.39 ± 0.18 (1.03 to 1.74) | 0.799 | 0.002 |
| BMI | 0.034 | 2.97 ± 0.19 (2.59 to 3.34) | 0.376 | 0.02 | 0.024 | 1.39 ± 0.18 (1.03 to 1.74) | 0.517 | 0.01 |
| LTI | 0.150 | 2.97 ± 0.18 (2.61 to 3.32) | 0.033 | 0.11 | 0.012 | 1.39 ± 0.18 (1.03 to 1.74) | 0.864 | 0.001 |
| FTI | −0.005 | 2.97 ± 0.19 (2.59 to 3.35) | 0.891 | 0.001 | 0.014 | 1.39 ± 0.18 (1.03 to 1.74) | 0.678 | 0.01 |
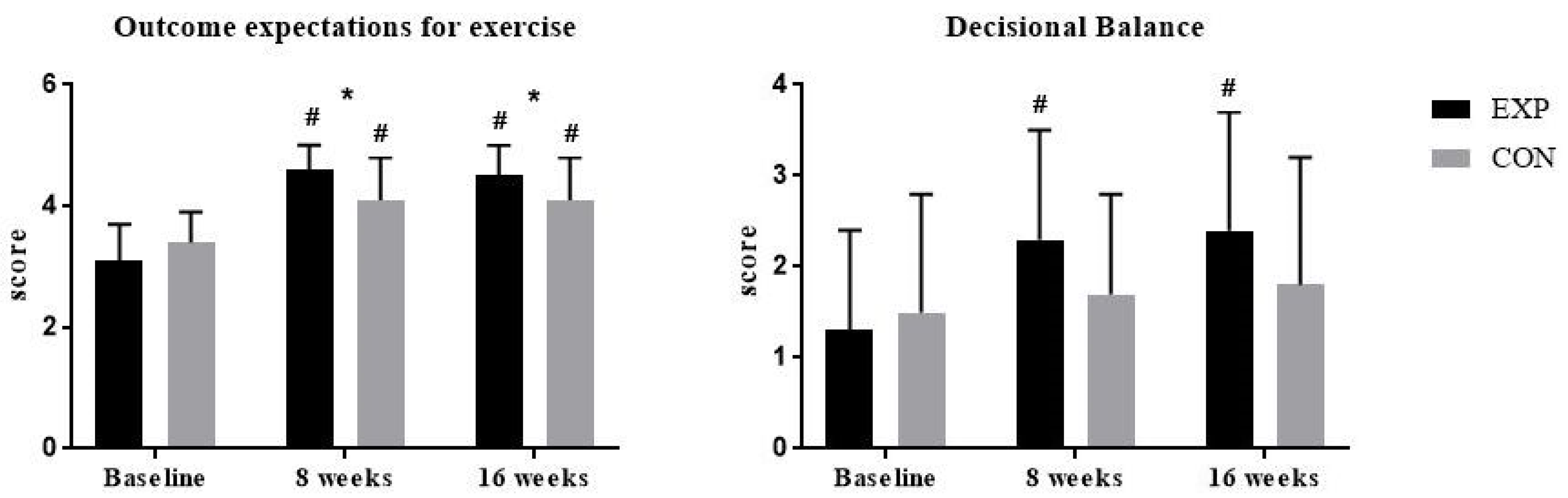
| Group | Outcome Expectations for Exercise | Decisional Balance | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Baseline mean ± SD | 8 weeks mean ± SD | ES | 16 weeks mean ± SD | ES | Baseline mean ± SD | 8 weeks mean ± SD | ES | 16 weeks mean ± SD | ES | |
| EXP | 3.1 ± 0.6 | 4.6 ± 0.4 | 2.9 | 4.5 ± 0.5 | 2.5 | 1.3 ± 1.1 | 2.3 ± 1.2 | 0.9 | 2.4 ± 1.3 | 0.9 |
| CON | 3.4 ± 0.5 | 4.1 ± 0.7 | 1.2 | 4.1 ± 0.7 | 1.2 | 1.5 ± 1.3 | 1.7 ± 1.1 | 0.2 | 1.8 ± 1.4 | 0.2 |
| Adherence | OEE High Group | OEE Low Group | p | Mean Difference (95% CI) |
| average adherence at cycling exercie (both study phases, n = 34) | 89% ± 11% | 76% ± 18% | 0.012 | 13.0 ± 4.9 (3.1 to 22.9) |
| average adherence to supervised predialysis functional exercise (phase 1, n = 17) * | 93% ± 7% | 81% ± 13% | 0.001 | 12.4 ± 3.4 (5.5 to 19.3) |
| average adherence to home-based functional exercise (phase 2, n = 16) * | 85% ± 8% | 60% ± 22% | <0.001 | 24.2 ± 5.3 (13.4 to 34.9) |
| DB High Group | DB Low Group | p | Mean Difference (95% CI) | |
| average adherence at cycling exercise (both study phases, n = 34) | 89% ± 11% | 77% ± 17% | 0.018 | 11.6 ± 4.7 (2.1 to 21.1) |
| average adherence to supervised predialysis functional exercise (phase 1, n = 17) * | 86% ± 15% | 87% ± 9% | 0.731 | 1.4 ± 4.0 (−6.8 to 9.6) |
| average adherence to home-based functional exercise (phase 2, n = 16) * | 75% ± 20% | 71% ± 22% | 0.631 | 3.4 ± 7.0 (−10.9 to 17.7) |
| Change after 16 weeks | OEE | DB | ||||||
|---|---|---|---|---|---|---|---|---|
| B | 95% CI | P | Partial Eta Squared | B | 95% CI | p | Partial Eta Squared | |
| 6MWT | 17.71 | −18.7 to 54.1 | 0.329 | 0.03 | 22.864 | 6.1 to 39.6 | 0.009 | 0.21 |
| STS10 | −0.53 | −4.6 to 3.5 | 0.790 | 0.002 | −1.84 | −3.8 to 0.1 | 0.062 | 0.11 |
| HG | 0.122 | −1.9 to 2.1 | 0.900 | 0.001 | 1.22 | 0.3 to 2.2 | 0.013 | 0.18 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bogataj, Š.; Pajek, M.; Buturović Ponikvar, J.; Pajek, J. Outcome Expectations for Exercise and Decisional Balance Questionnaires Predict Adherence and Efficacy of Exercise Programs in Dialysis Patients. Int. J. Environ. Res. Public Health 2020, 17, 3175. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph17093175
Bogataj Š, Pajek M, Buturović Ponikvar J, Pajek J. Outcome Expectations for Exercise and Decisional Balance Questionnaires Predict Adherence and Efficacy of Exercise Programs in Dialysis Patients. International Journal of Environmental Research and Public Health. 2020; 17(9):3175. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph17093175
Chicago/Turabian StyleBogataj, Špela, Maja Pajek, Jadranka Buturović Ponikvar, and Jernej Pajek. 2020. "Outcome Expectations for Exercise and Decisional Balance Questionnaires Predict Adherence and Efficacy of Exercise Programs in Dialysis Patients" International Journal of Environmental Research and Public Health 17, no. 9: 3175. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph17093175







