Assessment of the Toxicity of Quantum Dots through Biliometric Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Source of the Data and Search Strategy
2.2. Data Collection
2.3. Statistical Methods
3. Results
3.1. Publication Outputs and Growth Prediction
3.2. Distribution by Journals
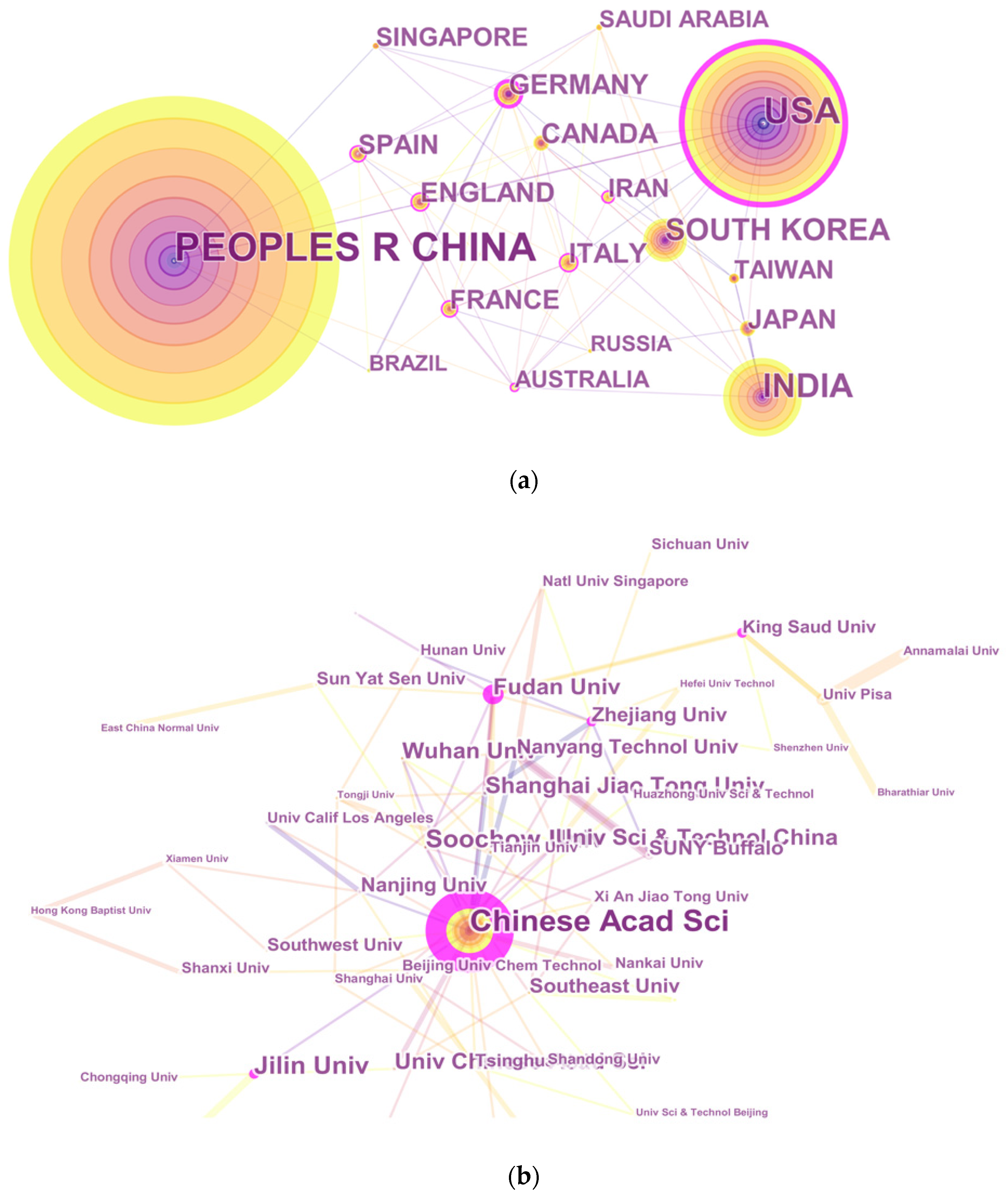
3.3. Distribution by Country and Institution
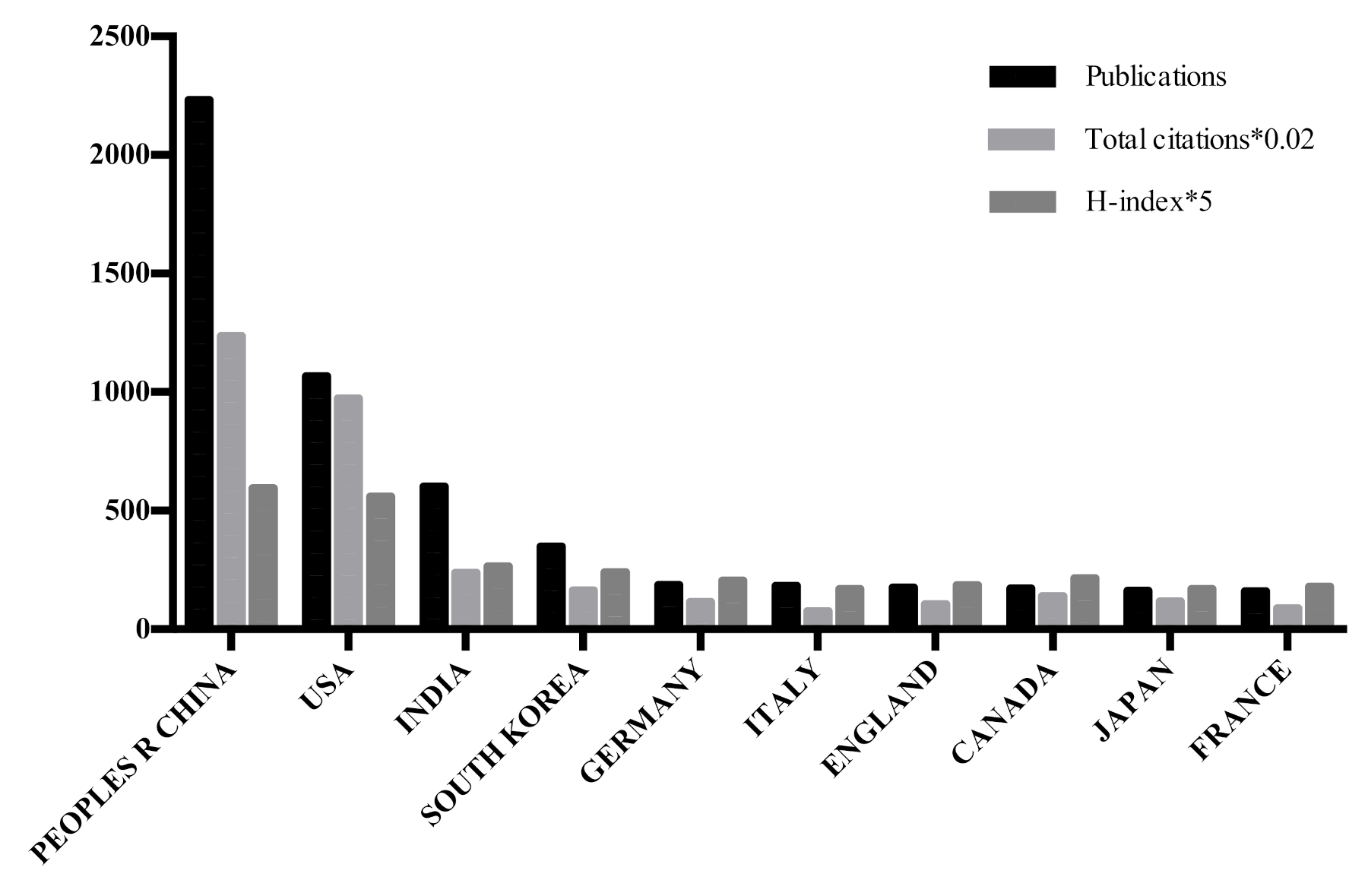
3.4. Analysis of Publications, Total Citations, and H-Index
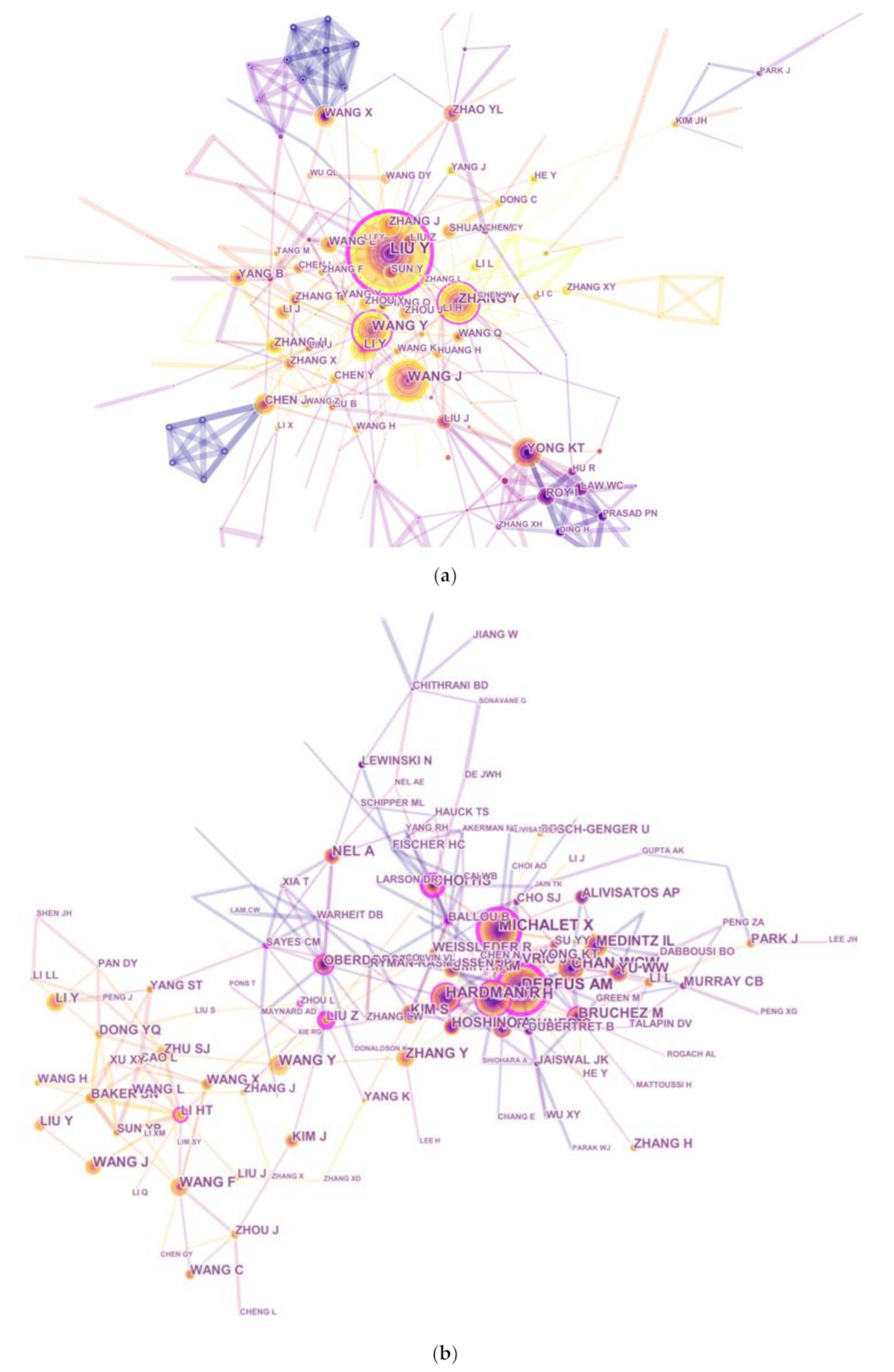
3.5. Contribution by Authors
3.6. Analysis of References
3.7. Analysis of Keywords
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rossetti, R.; Brus, L. Electron-hole recombination emission as a probe of surface-chemistry in aqueous cds colloids. J. Phys. Chem. 1982, 86, 4470–4472. [Google Scholar] [CrossRef]
- Alivisatos, A.P. Semiconductor clusters, nanocrystals, and quantum dots. Science 1996, 271, 933–937. [Google Scholar] [CrossRef] [Green Version]
- Wu, T.S.; Zhang, T.; Chen, Y.L.; Tang, M. Research advances on potential neurotoxicity of quantum dots. J. Appl. Toxicol. 2016, 36, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.S.; Tang, M. Review of the effects of manufactured nanoparticles on mammalian target organs. J. Appl. Toxicol. 2018, 38, 25–40. [Google Scholar] [CrossRef]
- Wang, Y.W.; Yang, K.; Tang, H.O.; Chen, D.; Bai, Y.L. Toxicity assessment of repeated intravenous injections of arginine-glycine-aspartic acid peptide conjugated CdSeTe/ZnS quantum dots in mice. Int. J. Nanomed. 2014, 9, 4809–4817. [Google Scholar]
- Mattoussi, H.; Palui, G.; Na, H.B. Luminescent quantum dots as platforms for probing in vitro and in vivo biological processes. Adv. Drug Deliv. Rev. 2012, 64, 138–166. [Google Scholar] [CrossRef]
- Kairdolf, B.A.; Smith, A.M.; Stokes, T.H.; Wang, M.D.; Young, A.N.; Nie, S.M. Semiconductor Quantum Dots for Bioimaging and Biodiagnostic Applications. In Annual Review of Analytical Chemistry; Cooks, R.G., Pemberton, J.E., Eds.; Annual Reviews: Palo Alto, CA, USA, 2013; Volume 6, pp. 143–162. [Google Scholar]
- Matea, C.T.; Mocan, T.; Tabaran, F.; Pop, T.; Mosteanu, O.; Puia, C.; Iancu, C.; Mocan, L. Quantum dots in imaging, drug delivery and sensor applications. Int. J. Nanomed. 2017, 12, 5421–5431. [Google Scholar] [CrossRef] [Green Version]
- Pohanka, M. Quantum Dots in the Therapy: Current Trends and Perspectives. Mini-Rev. Med. Chem. 2017, 17, 650–656. [Google Scholar] [CrossRef]
- Wang, Y.; Tang, M. Review of in vitro toxicological research of quantum dot and potentially involved mechanisms. Sci. Total Environ. 2018, 625, 940–962. [Google Scholar] [CrossRef]
- Wu, T.S.; Tang, M. Toxicity of quantum dots on respiratory system. Inhal. Toxicol. 2014, 26, 128–139. [Google Scholar] [CrossRef]
- Zhang, T.; Wang, Y.; Lu, K.; Xue, Y.; Tang, M. Threshold Dose of Three Types of Quantum Dots (QDs) Induces Oxidative Stress Triggers DNA Damage and Apoptosis in Mouse Fibroblast L929 Cells. Int. J. Environ. Res. Public Health 2015, 12, 13435–13454. [Google Scholar] [CrossRef]
- Wang, Y.; Tang, M. Dysfunction of various organelles provokes multiple cell death after quantum dot exposure. Int. J. Nanomed. 2018, 13, 2729–2742. [Google Scholar] [CrossRef] [Green Version]
- Liang, X.; Tang, M. Research Advances on Cytotoxicity of Cadmium-Containing Quantum Dots. J. Nanosci. Nanotechnol. 2019, 19, 5375–5387. [Google Scholar] [CrossRef]
- Wang, Q.; Yang, Z.; Yang, Y.; Long, C.; Li, H. A bibliometric analysis of research on the risk of engineering nanomaterials during 1999–2012. Sci. Total Environ. 2014, 473–474, 483–489. [Google Scholar] [CrossRef]
- Teles, R.H.G.; Moralles, H.F.; Cominetti, M.R. Global trends in nanomedicine research on triple negative breast cancer: A bibliometric analysis. Int. J. Nanomed. 2018, 13, 2321–2336. [Google Scholar] [CrossRef] [Green Version]
- Chen, X. Mapping the Decadal (2009–2018) Research Landscape of Nanotoxicity: Insights from a Bibliometric Study. Nanosci. Nanotechnol. Lett. 2019, 11, 1327–1337. [Google Scholar] [CrossRef]
- Yi, F.; Yang, P.; Sheng, H. Tracing the scientific outputs in the field of Ebola research based on publications in the Web of Science. BMC Res. Notes 2016, 9, 221. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.M.; Hu, Z.G.; Liu, S.B.; Tseng, H. Emerging trends in regenerative medicine: A scientometric analysis in CiteSpace. Expert Opin. Biol. 2012, 12, 593–608. [Google Scholar] [CrossRef]
- Xu, J.; He, H.; Wang, Y.Y.; Yan, R.; Zhou, L.J.; Liu, Y.Z.; Jiang, F.L.; Maskow, T.; Liu, Y. New aspects of the environmental risks of quantum dots: Prophage activation. Environ. Sci. Nano 2018, 5, 1556–1566. [Google Scholar] [CrossRef]
- Jin, J.C.; Wang, B.B.; Xu, Z.Q.; He, X.H.; Zou, H.F.; Yang, Q.Q.; Jiang, F.L.; Liu, Y. A novel method for the detection of silver ions with carbon dots: Excellent selectivity, fast response, low detection limit and good applicability. Sens. Actuators B Chem. 2018, 267, 627–635. [Google Scholar] [CrossRef]
- Xiang, X.; Gao, T.; Zhang, B.R.; Jiang, F.L.; Liu, Y. Surface functional groups affect CdTe QDs behavior at mitochondrial level. Toxicol. Res. 2018, 7, 1071–1080. [Google Scholar] [CrossRef] [Green Version]
- Shen, C.; Wang, J.; Cao, Y.; Lu, Y. Facile access to B-doped solid-state fluorescent carbon dots toward light emitting devices and cell imaging agents. J. Mater. Chem. C 2015, 3, 6668–6675. [Google Scholar] [CrossRef]
- Sun, Y.; Shen, C.; Wang, J.; Lu, Y. Facile synthesis of biocompatible N,S-doped carbon dots for cell imaging and ion detecting. RSC Adv. 2015, 5, 16368–16375. [Google Scholar] [CrossRef]
- Xia, B.; Li, J.C.; Shi, J.S.; Zhang, Y.; Zhang, Q.; Chen, Z.Y.; Wang, B. Biodegradable and Magnetic-Fluorescent Porous Silicon@lron Oxide Nanocomposites for Fluorescence/Magnetic Resonance Bimodal Imaging of Tumor In Vivo. ACS Biomater. Sci. Eng. 2017, 3, 2579–2587. [Google Scholar] [CrossRef]
- Guo, D.; Zhao, Y.; Zhang, Y.U.; Wang, Q.; Huang, Z.; Ding, Q.; Guo, Z.; Zhou, X.; Zhu, L.; Gu, N. The Cellular Uptake and Cytotoxic Effect of Silver Nanoparticles on Chronic Myeloid Leukemia Cells. J. Biomed. Nanotechnol. 2014, 10, 669–678. [Google Scholar] [CrossRef]
- Derfus, A.M.; Chan, W.C.W.; Bhatia, S.N. Probing the cytotoxicity of semiconductor quantum dots. Nano Lett. 2004, 4, 11–18. [Google Scholar] [CrossRef] [Green Version]
- Derfus, A.M.; Chan, W.C.W.; Bhatia, S.N. Intracellular delivery of quantum dots for live cell labeling and organelle tracking. Adv. Mater. 2004, 16, 961–966. [Google Scholar] [CrossRef]
- Antelman, J.; Wilking-Chang, C.; Weiss, S.; Michalet, X. Nanometer Distance Measurements between Multicolor Quantum Dots. Nano Lett. 2009, 9, 2199–2205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, X.H.; Cui, Y.Y.; Levenson, R.M.; Chung, L.W.K.; Nie, S.M. In vivo cancer targeting and imaging with semiconductor quantum dots. Nat. Biotechnol. 2004, 22, 969–976. [Google Scholar] [CrossRef] [PubMed]
- Hardman, R. A toxicologic review of quantum dots: Toxicity depends on physicochemical and environmental factors. Environ. Health Perspect. 2006, 114, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Kurakula, M.; Naveen, N.R. Prospection of recent chitosan biomedical trends: Evidence from patent analysis (2009–2020). Int. J. Biol. Macromol. 2020, 165 Pt B, 1924–1938. [Google Scholar] [CrossRef]
- Chen, C.M. CiteSpace II: Detecting and visualizing emerging trends and transient patterns in scientific literature. J. Am. Soc. Inf. Sci. Technol. 2006, 57, 359–377. [Google Scholar] [CrossRef] [Green Version]
- Miao, Y.; Xu, S.Y.; Chen, L.S.; Liang, G.Y.; Pu, Y.P.G.; Yin, L.H. Trends of long noncoding RNA research from 2007 to 2016: A bibliometric analysis. Oncotarget 2017, 8, 83114–83127. [Google Scholar] [CrossRef]
- Zhou, X.D.; Zhao, G.H. Global liposome research in the period of 1995–2014: A bibliometric analysis. Scientometrics 2015, 105, 231–248. [Google Scholar] [CrossRef]
- Su, Y.Y.; Hu, M.; Fan, C.H.; He, Y.; Li, Q.N.; Li, W.X.; Wang, L.H.; Shen, P.P.; Huang, Q. The cytotoxicity of CdTe quantum dots and the relative contributions from released cadmium ions and nanoparticle properties. Biomaterials 2010, 31, 4829–4834. [Google Scholar] [CrossRef]
- Wu, T.S.; He, K.Y.; Zhan, Q.L.; Ang, S.J.; Ying, J.L.; Zhang, S.H.; Zhang, T.; Xue, Y.Y.; Tang, M. MPA-capped CdTe quantum dots exposure causes neurotoxic effects in nematode Caenorhabditis elegans by affecting the transporters and receptors of glutamate, serotonin and dopamine at the genetic level, or by increasing ROS, or both. Nanoscale 2015, 7, 20460–20473. [Google Scholar] [CrossRef]
- Peng, L.; He, M.; Chen, B.; Qiao, Y.; Hu, B. Metallomics Study of CdSe/ZnS Quantum Dots in HepG2 Cells. ACS Nano 2015, 9, 10324–10334. [Google Scholar] [CrossRef]
- Su, Y.Y.; He, Y.; Lu, H.T.; Sai, L.M.; Li, Q.N.; Li, W.X.; Wang, L.H.; Shen, P.P.; Huang, Q.; Fan, C.H. The cytotoxicity of cadmium based, aqueous phase—Synthesized, quantum dots and its modulation by surface coating. Biomaterials 2009, 30, 19–25. [Google Scholar] [CrossRef]
- Dunpall, R.; Nejo, A.A.; Pullabhotla, V.S.R.; Opoku, A.R.; Revaprasadu, N.; Shonhai, A. An in vitro assessment of the interaction of cadmium selenide quantum dots with DNA, iron, and blood platelets. IUBMB Life 2012, 64, 995–1002. [Google Scholar] [CrossRef]
- Pandey, V.; Pandey, G.; Tripathi, V.K.; Yadav, S.; Mudiam, M.K.R. Nucleation temperature-controlled synthesis and in vitro toxicity evaluation of L-cysteine-capped Mn:ZnS quantum dots for intracellular imaging. Luminescence 2016, 31, 341–347. [Google Scholar] [CrossRef]
- Wang, Y.B.; Liang, X.H.; Ma, X.; Hu, Y.H.; Hu, X.Y.; Li, X.H.; Fan, J. Simple and greener synthesis of highly photoluminescence Mn2+-doped ZnS quantum dots and its surface passivation mechanism. Appl. Surf. Sci. 2014, 316, 54–61. [Google Scholar] [CrossRef]
- Wu, T.S.; Liang, X.; He, K.Y.; Wei, T.T.; Wang, Y.; Zou, L.Y.; Lu, J.; Yao, Y.; Liu, N.; Zhang, T.; et al. Transcriptome analysis of different sizes of 3-mercaptopropionic acid-modified cadmium telluride quantum dot-induced toxic effects reveals immune response in rat hippocampus. J. Appl. Toxicol. 2018, 38, 1177–1194. [Google Scholar] [CrossRef] [PubMed]
- Brunetti, V.; Chibli, H.; Fiammengo, R.; Galeone, A.; Malvindi, M.A.; Vecchio, G.; Cingolani, R.; Nadeau, J.L.; Pompa, P.P. InP/ZnS as a safer alternative to CdSe/ZnS core/shell quantum dots: In vitro and in vivo toxicity assessment. Nanoscale 2013, 5, 307–317. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.S.; Liang, X.; He, K.Y.; Wei, T.T.; Wang, Y.; Lu, J.; Yao, Y.; Zhang, T.; Xue, Y.Y.; Tang, M. MPA-modified CdTe quantum dots increased interleukin-1beta secretion through MyD88-dependent Toll-like receptor pathway and NLRP3 inflammasome activation in microglia. Toxicol. Vitr. 2018, 52, 41–51. [Google Scholar] [CrossRef]
- Xiong, Z.; Zhang, X.; Zhang, S.; Lei, L.; Ma, W.; Li, D.; Wang, W.; Zhao, Q.; Xing, B. Bacterial toxicity of exfoliated black phosphorus nanosheets. Ecotoxicol. Environ. Saf. 2018, 161, 507–514. [Google Scholar] [CrossRef]
- Qi, X.; Shan, H.; Wei, S.; Li, P.; Yi, L. Evaluate the potential toxicity of quantum dots on bacterial metabolism by microcalorimetry. Thermochim. Acta 2013, 552, 98–105. [Google Scholar]
- Hossain, S.T.; Mukherjee, S.K. Toxicity of cadmium sulfide (CdS) nanoparticles against Escherichia coli and HeLa cells. J. Hazard. Mater. 2013, 260, 1073–1082. [Google Scholar] [CrossRef]
- Baslak, C.; Kars, M.D.; Karaman, M.; Kus, M.; Cengeloglu, Y.; Ersoz, M. Biocompatible multi-walled carbon nanotube–CdTe quantum dot–polymer hybrids for medical applications. J. Lumin. 2015, 160, 9–15. [Google Scholar] [CrossRef]
- Huiquan, T.; Kai, Y.; Zhen, M.; Jianmei, W.; Youjiu, Z.; Zhenhui, K.; Zhuang, L. In vivo NIR fluorescence imaging, biodistribution, and toxicology of photoluminescent carbon dots produced from carbon nanotubes and graphite. Small 2012, 8, 281–290. [Google Scholar]
- Li, H.; Huang, J.; Song, Y.; Zhang, M.; Wang, H.; Lu, F.; Huang, H.; Liu, Y.; Dai, X.; Gu, Z.; et al. Degradable Carbon Dots with Broad-Spectrum Antibacterial Activity. ACS Appl. Mater. Interfaces 2018, 10, 26936–26946. [Google Scholar] [CrossRef]




| Rank | Journal Title | Country | Count | Percent | 5-Year IF |
|---|---|---|---|---|---|
| 1 | RSC Advances | England | 237 | 4.37% | 3.096 |
| 2 | Nanoscale | England | 178 | 3.29% | 7.713 |
| 3 | ACS Applied Materials Interfaces | United States | 129 | 2.38% | 8.284 |
| 4 | Biomaterials | England | 126 | 2.33% | 9.315 |
| 5 | Journal of Materials Chemistry B | England | 107 | 1.98% | 4.959 |
| 6 | ACS Nano | United States | 96 | 1.77% | 14.82 |
| 7 | Scientific Reports | England | 84 | 0.016 | 4.609 |
| 8 | Nanotechnology | England | 83 | 1.53% | 3.467 |
| 9 | Journal of Nanoscience and Nanotechnology | United States | 79 | 1.46% | 1.103 |
| 10 | Sensors and Actuators B Chemical | Switzerland | 73 | 1.35% | 5.118 |
| Rank | Country | Count | Institution | Count |
|---|---|---|---|---|
| 1 | Peoples Republic of China | 2233 | Chinese Acad Sci | 368 |
| 2 | USA | 1067 | Jilin Univ | 125 |
| 3 | India | 603 | Soochow Univ | 103 |
| 4 | South Korea | 349 | Univ Chinese Acad Sci | 90 |
| 5 | Germany | 188 | Wuhan Univ | 89 |
| 6 | Italy | 183 | Fudan Univ | 83 |
| 7 | England | 177 | Shanghai Jiao Tong Univ | 83 |
| 8 | Canada | 172 | Nanyang Technol Univ | 75 |
| 9 | Japan | 162 | Southeast Univ | 68 |
| 10 | France | 161 | Univ Sci Technol China | 63 |
| Rank | Author | Count | Co-Cited Author | Count | Co-Cited Reference | Count |
|---|---|---|---|---|---|---|
| 1 | Liu Y | 106 | Derfus AM | 593 | Hardman R, 2006, Environ Health Persp, V114, P165 | 304 |
| 2 | Wang J | 66 | Michalet X | 492 | Michalet X, 2005, Science, V307, P538 | 292 |
| 3 | Zhang Y | 61 | Gao XH | 432 | Derfus AM, 2004, Nano Lett, V4, P11 | 266 |
| 4 | Wang Y | 56 | Chan WCW | 410 | Baker SN, 2010, Angew Chem Int Edit, V49, P6726 | 222 |
| 5 | Li Y | 48 | Medintz IL | 388 | Choi HS, 2007, Nat Biotechnol, V25, P1165 | 206 |
| 6 | Yong KT | 46 | Hardman R | 379 | Kirchner C, 2005, Nano Lett, V5, P331 | 199 |
| 7 | Zhang J | 43 | Zhang Y | 294 | Gao XH, 2004, Nat Biotechnol, V22, P969 | 190 |
| 8 | Wang X | 38 | Choi HS | 286 | Medintz IL, 2005, Nat Mater, V4, P435 | 177 |
| 9 | Yang B | 38 | Wang Y | 277 | NEL A, 2006, Science, V311, P622 | 173 |
| 10 | Zhang H | 36 | Bruchez M | 274 | Cho SJ, 2007, Langmuir, V23, P1974 | 170 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhong, L.; Zhang, L.; Li, Y.; Liang, X.; Kong, L.; Shen, X.; Wu, T. Assessment of the Toxicity of Quantum Dots through Biliometric Analysis. Int. J. Environ. Res. Public Health 2021, 18, 5768. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph18115768
Zhong L, Zhang L, Li Y, Liang X, Kong L, Shen X, Wu T. Assessment of the Toxicity of Quantum Dots through Biliometric Analysis. International Journal of Environmental Research and Public Health. 2021; 18(11):5768. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph18115768
Chicago/Turabian StyleZhong, Lishi, Lili Zhang, Yimeng Li, Xue Liang, Lu Kong, Xiaobing Shen, and Tianshu Wu. 2021. "Assessment of the Toxicity of Quantum Dots through Biliometric Analysis" International Journal of Environmental Research and Public Health 18, no. 11: 5768. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph18115768






