12-Week Inspiratory Muscle Training Improves Respiratory Muscle Strength in Adult Patients with Stable Asthma: A Randomized Controlled Trial
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
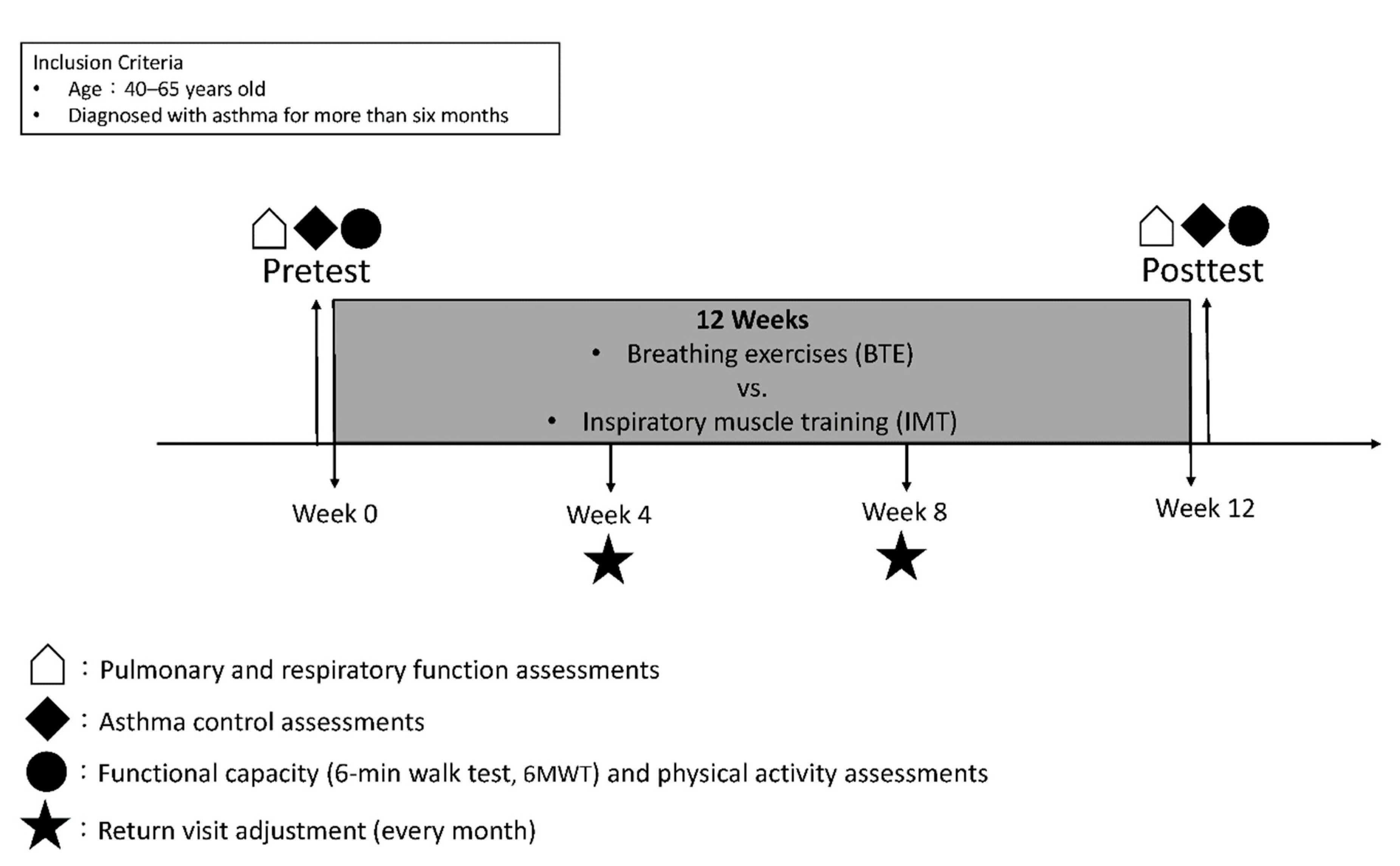
2.2. Experimental Design and Procedure
2.3. Measurements of Pulmonary Function
2.4. Evaluation of Respiratory Muscle Strength
2.5. Asthma Control Assessments—Asthma Control Test (ACT) and Asthma Control Questionnaire (ACQ)
2.6. Six-Minute Walk Test (6MWT), Heart Rate, and Blood Oxygen Saturation
2.7. Three-Day Physical Activity Log (3-D PAL)
2.8. Statistical Analysis
3. Results
3.1. Baseline Characteristics of the Participants
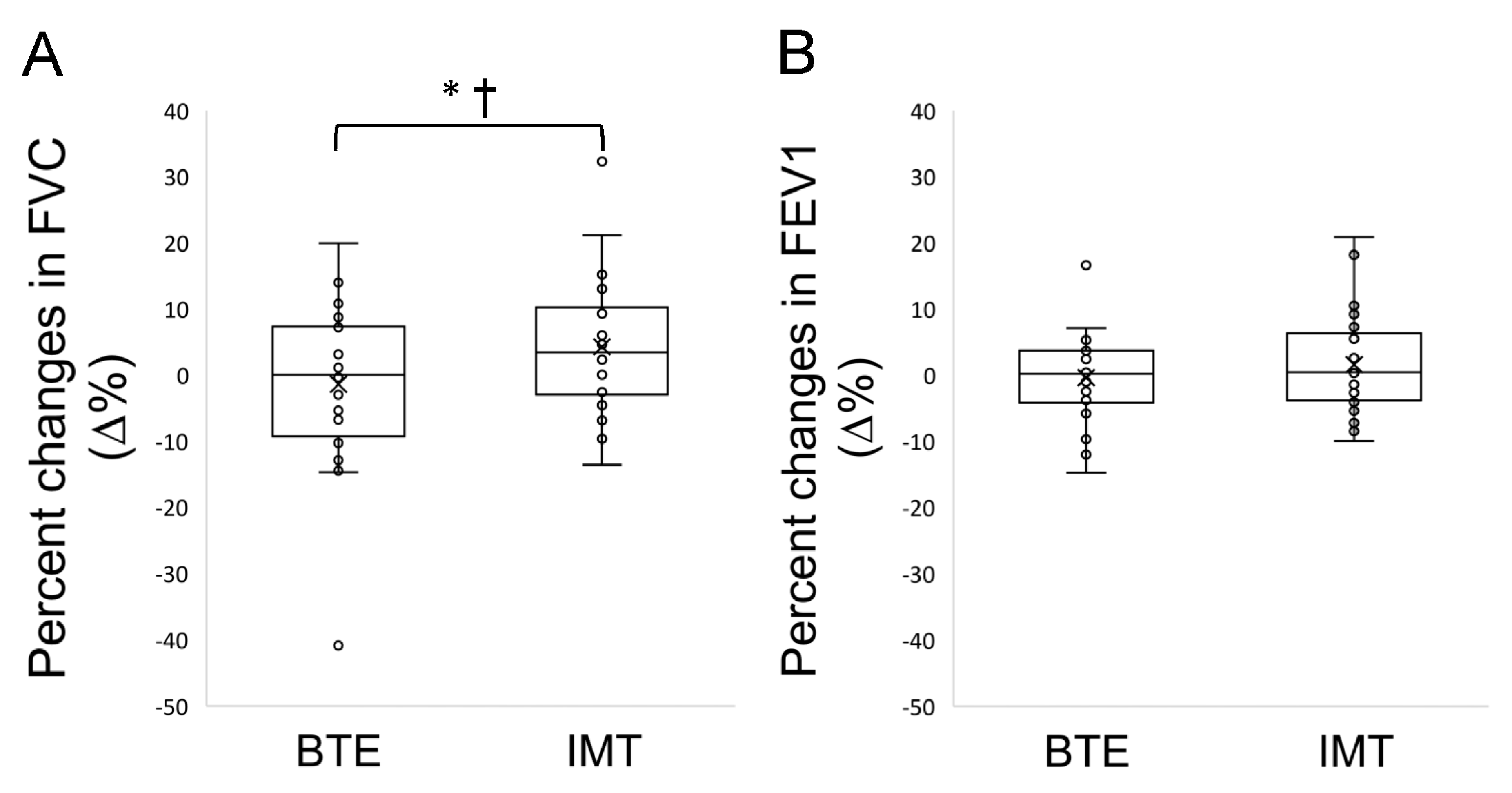
3.2. Pulmonary Function
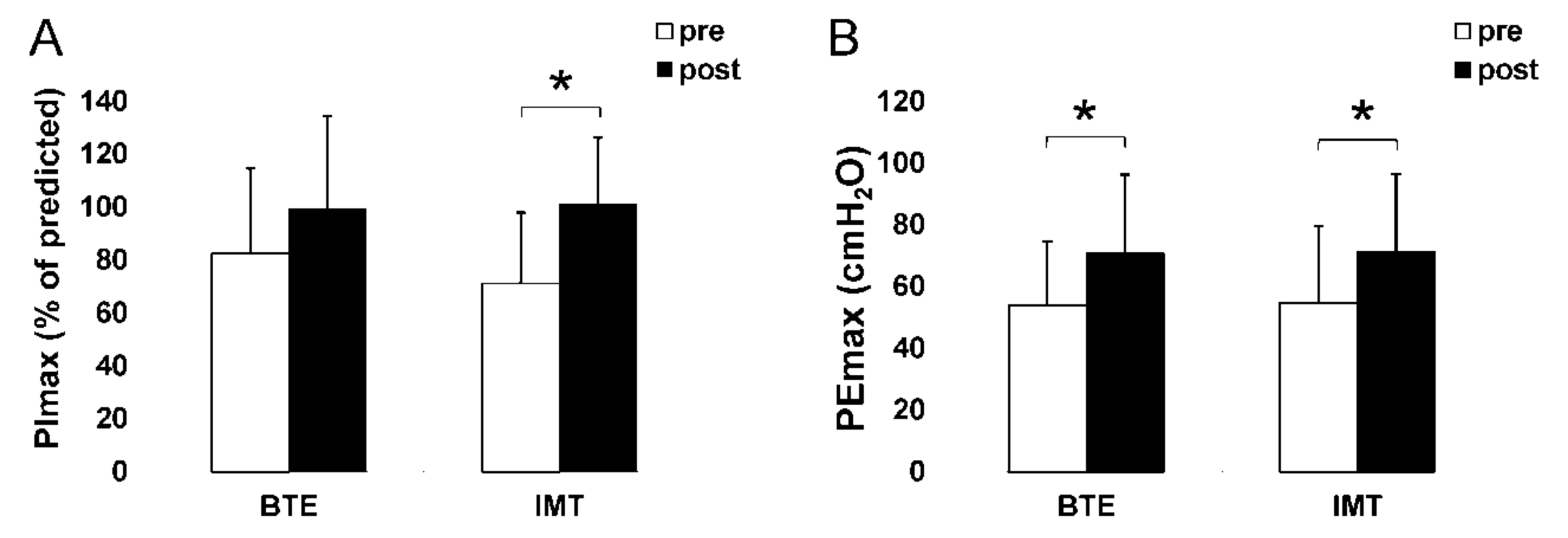
3.3. Respiratory Muscle Strength
3.4. Asthma Control
3.5. 6MWT
3.6. Physical Activity
4. Discussion
4.1. Primary Outcome
4.2. Secondary Outcomes
4.3. Limitation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- GBD 2015 Chronic Respiratory Disease Collaborators. Global, regional, and national deaths, prevalence, disability-adjusted life years, and years lived with disability for chronic obstructive pulmonary disease and asthma, 1990–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet Respir. Med. 2017, 5, 691–706. [Google Scholar] [CrossRef] [Green Version]
- Ying, Y.; Weng, Y.; Chang, K. The prevalence and patient’s quality of life for asthma in Taiwan. Int. J. Pul. Res. Sci. 2017, 1, 1–5. [Google Scholar] [CrossRef]
- Macedo, T.M.; Freitas, D.A.; Chaves, G.S.; Holloway, E.A.; Mendonca, K.M. Breathing exercises for children with asthma. Cochrane Database Syst. Rev. 2016, 4, CD011017. [Google Scholar] [CrossRef] [PubMed]
- Castilho, T.; Itaborahy, B.D.H.; Hoepers, A.; Brito, J.N.d.; Almeida, A.C.d.S.; Schivinski, C.I.S. Effects of inspiratory muscle training and breathing exercises in children with asthma: A systematic review. J. Hum. Growth Dev. 2020, 30, 291–300. [Google Scholar] [CrossRef]
- Bruton, A.; Thomas, M. The role of breathing training in asthma management. Curr. Opin. Allergy Clin. Immunol. 2011, 11, 53–57. [Google Scholar] [CrossRef] [PubMed]
- Freitas, P.D.; Ferreira, P.G.; da Silva, A.; Trecco, S.; Stelmach, R.; Cukier, A.; Carvalho-Pinto, R.; Salge, J.M.; Fernandes, F.L.A.; Mancini, M.C.; et al. The effects of exercise training in a weight loss lifestyle intervention on asthma control, quality of life and psychosocial symptoms in adult obese asthmatics: Protocol of a randomized controlled trial. BMC Pulm. Med. 2015, 15, 124. [Google Scholar] [CrossRef] [Green Version]
- Evaristo, K.B.; Saccomani, M.G.; Martins, M.A.; Cukier, A.; Stelmach, R.; Rodrigues, M.R.; Santaella, D.F.; Carvalho, C.R.F. Comparison between breathing and aerobic exercise on clinical control in patients with moderate-to-severe asthma: Protocol of a randomized trial. BMC Pulm. Med. 2014, 14, 160. [Google Scholar] [CrossRef] [Green Version]
- Shei, R.J.; Paris, H.L.; Wilhite, D.P.; Chapman, R.F.; Mickleborough, T.D. The role of inspiratory muscle training in the management of asthma and exercise-induced bronchoconstriction. Phys. Sportsmed. 2016, 44, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Silva, I.S.; Fregonezi, G.A.; Dias, F.A.; Ribeiro, C.T.; Guerra, R.O.; Ferreira, G.M. Inspiratory muscle training for asthma. Cochrane Database Syst. Rev. 2013, CD003792. [Google Scholar] [CrossRef]
- Hermes, B.M.; Cardoso, D.M.; Gomes, T.J.; Santos, T.D.; Vicente, M.S.; Pereira, S.N.; Barbosa, V.A.; Albuquerque, I.M. Short-term inspiratory muscle training potentiates the benefits of aerobic and resistance training in patients undergoing CABG in phase II cardiac rehabilitation program. Rev. Bras. Cir. Cardiovasc. 2015, 30, 474–481. [Google Scholar] [CrossRef] [Green Version]
- Prasanna, K.; Sowmiya, K.; Dhileeban, C. Effect of Buteyko breathing exercise in newly diagnosed asthmatic patients. Int. J. Med. Public Health 2015, 5. [Google Scholar] [CrossRef] [Green Version]
- Elnaggar, R.K. A randomized placebo-controlled study investigating the efficacy of inspiratory muscle training in the treatment of children with bronchial asthma. J. Asthma 2020, 1–9. [Google Scholar] [CrossRef]
- Santino, T.A.; Chaves, G.S.; Freitas, D.A.; Fregonezi, G.A.; Mendonca, K.M. Breathing exercises for adults with asthma. Cochrane Database Syst Rev 2020, 3, CD001277. [Google Scholar] [CrossRef] [PubMed]
- Bateman, E.D.; Hurd, S.S.; Barnes, P.J.; Bousquet, J.; Drazen, J.M.; FitzGerald, M.; Gibson, P.; Ohta, K.; O’Byrne, P.; Pedersen, S.E.; et al. Global strategy for asthma management and prevention: GINA executive summary. Eur. Respir. J. 2008, 31, 143–178. [Google Scholar] [CrossRef]
- Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention; Global Initiative for Asthma—GINA: Fontana, WI, USA, 2017. [Google Scholar]
- Gomieiro, L.T.; Nascimento, A.; Tanno, L.K.; Agondi, R.; Kalil, J.; Giavina-Bianchi, P. Respiratory exercise program for elderly individuals with asthma. Clinics 2011, 66, 1163–1169. [Google Scholar] [CrossRef] [Green Version]
- Bailey, S.J.; Romer, L.M.; Kelly, J.; Wilkerson, D.P.; DiMenna, F.J.; Jones, A.M. Inspiratory muscle training enhances pulmonary O(2) uptake kinetics and high-intensity exercise tolerance in humans. J. Appl. Physiol. 2010, 109, 457–468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dickinson, J.; Whyte, G.; McConnell, A. Inspiratory muscle training: A simple cost-effective treatment for inspiratory stridor. Br J. Sports Med. 2007, 41, 694–695. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, L.A.; McConnell, A.K. The influence of inspiratory and expiratory muscle training upon rowing performance. Eur. J. Appl. Physiol. 2007, 99, 457–466. [Google Scholar] [CrossRef]
- Schulz, K.F.; Altman, D.G.; Moher, D. CONSORT 2010 statement: Updated guidelines for reporting parallel group randomised trials. BMJ 2010, 340, c332. [Google Scholar] [CrossRef]
- Dressel, H.; Filser, L.; Fischer, R.; de la Motte, D.; Steinhaeusser, W.; Huber, R.M.; Nowak, D.; Jorres, R.A. Lung diffusing capacity for nitric oxide and carbon monoxide: Dependence on breath-hold time. Chest 2008, 133, 1149–1154. [Google Scholar] [CrossRef]
- Thomas, A.; Hanel, B.; Marott, J.L.; Buchvald, F.; Mortensen, J.; Nielsen, K.G. The Single-Breath Diffusing Capacity of CO and NO in Healthy Children of European Descent. PLoS ONE 2014, 9, e113177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- American Thoracic Society/European Respiratory, S. ATS/ERS Statement on respiratory muscle testing. Am. J. Respir. Crit. Care Med. 2002, 166, 518–624. [Google Scholar] [CrossRef]
- Thomas, M.; Kay, S.; Pike, J.; Williams, A.; Rosenzweig, J.R.C.; Hillyer, E.V.; Price, D. The Asthma Control Test (ACT) as a predictor of GINA guideline-defined asthma control: Analysis of a multinational cross-sectional survey. Prim. Care Respir. J. 2009, 18, 41–49. [Google Scholar] [CrossRef] [Green Version]
- Villiot-Danger, J.; Villiot-Danger, E.; Borel, J.; Pépin, J.; Wuyam, B.; Verges, S. Respiratory muscle endurance training in obese patients. Int. J. Obes. 2011, 35, 692–699. [Google Scholar] [CrossRef] [Green Version]
- Majewski, M.; Dąbrowska, G.; Pawik, M.; Rożek, K. Evaluation of a home-based pulmonary rehabilitation program for older females suffering from bronchial Asthma. Adv. Clin. Exp. Med. 2015, 24, 1079–1083. [Google Scholar] [CrossRef] [Green Version]
- Rondinel, T.Z.; Correa, I.F.; Hoscheidt, L.M.; Bueno, M.H.; Da Silva, L.M.; Reppold, C.T.; Dal Lago, P. Incentive spirometry combined with expiratory positive airway pressure improves asthma control and quality of life in asthma: A randomised controlled trial. J. Asthma 2015, 52, 220–226. [Google Scholar] [CrossRef]
- Nigro, C.A.; Dibur, E.; Rhodius, E. Pulse oximetry for the detection of obstructive sleep apnea syndrome: Can the memory capacity of oxygen saturation influence their diagnostic accuracy? Sleep Disord. 2011, 2011, 427028. [Google Scholar] [CrossRef]
- Bouchard, C.; Tremblay, A.; Leblanc, C.; Lortie, G.; Savard, R.; Thériault, G. A method to assess energy expenditure in children and adults. Am. J. Clin. Nutr. 1983, 37, 461–467. [Google Scholar] [CrossRef]
- Wu, W.-C.I.; Yang, Y.-T.C.; Hsieh, T.-Y. An application of cloud physical activity promotion system on high school female students’ physical activity. Int. J. e-Educ. e-Bus. e-Manag. e-Learn. 2013, 3, 155. [Google Scholar] [CrossRef]
- Pessoa, I.; Sclauser, M.; Parreira, V.F.; Fregonezi, G.A.; Sheel, A.W.; Chung, F.; Reid, W.D. Reference values for maximal inspiratory pressure: A systematic review. Can. Respir. J. 2014, 21, 43–50. [Google Scholar] [CrossRef] [Green Version]
- Bonini, M.; Di Paolo, M.; Bagnasco, D.; Baiardini, I.; Braido, F.; Caminati, M.; Carpagnano, E.; Contoli, M.; Corsico, A.; Del Giacco, S.; et al. Minimal clinically important difference for asthma endpoints: An expert consensus report. Eur. Respir. Rev. 2020, 29, 190137. [Google Scholar] [CrossRef] [PubMed]
- Weiner, P.; Azgad, Y.; Ganam, R.; Weiner, M. Inspiratory muscle training in patients with bronchial asthma. Chest 1992, 102, 1357–1361. [Google Scholar] [CrossRef]
- Shaw, B.S.; Shaw, I. Static standing posture and pulmonary function in moderate-persistent asthmatics following aerobic and diaphragmatic breathing training. Pak. J. Med. Sci. 2011, 27, 549–552. [Google Scholar]
- Shaw, B.S.; Shaw, I. Pulmonary function and abdominal and thoracic kinematic changes following aerobic and inspiratory resistive diaphragmatic breathing training in asthmatics. Lung 2011, 189, 131–139. [Google Scholar] [CrossRef]
- Turner, L.A.; Mickleborough, T.D.; McConnell, A.K.; Stager, J.M.; Tecklenburg-Lund, S.; Lindley, M.R. Effect of inspiratory muscle training on exercise tolerance in asthmatic individuals. Med. Sci. Sports Exerc. 2011, 43, 2031–2038. [Google Scholar] [CrossRef] [Green Version]
- McConnell, A.; Caine, M.; Donovan, K.; Toogood, A.; Miller, M. Inspiratory muscle training improves lung function and reduces exertional dyspnoea in mild/moderateasthmatics. Clin. Sci. 1998, 95, 4P. [Google Scholar] [CrossRef]
- Weiner, P.; Berar-Yanay, N.; Davidovich, A.; Magadle, R.; Weiner, M. Specific inspiratory muscle training in patients with mild asthma with high consumption of inhaled β2-agonists. Chest 2000, 117, 722–727. [Google Scholar] [CrossRef] [Green Version]
- Sampaio, L.M.M.; Jamami, M.; Pires, V.A.; e Silva, A.B.; Costa, D. Respiratory muscle strength in asthmatic patient submitted by respiratory muscle training and physical training. Physiother. Mag. Univ. Sao Paolo 2002, 9, 43–48. [Google Scholar]
- Weiner, P.; Magadle, R.; Massarwa, F.; Beckerman, M.; Berar-Yanay, N. Influence of gender and inspiratory muscle training on the perception of dyspnea in patients with asthma. Chest 2002, 122, 197–201. [Google Scholar] [CrossRef] [Green Version]
- Weiner, P.; Magadle, R.; Beckerman, M.; Berar-Yanay, N. The Relationship among the Inspiratory Muscle Strength, the Perception of Dyspnea and Inhaled Beta2-Agonists in Patients with Asthma. Can. Respir. J. 2002, 9, 307–312. [Google Scholar] [CrossRef] [Green Version]
- Lima, E.V.; Lima, W.L.; Nobre, A.; dos Santos, A.M.; Brito, L.M.; Costa Mdo, R. Inspiratory muscle training and respiratory exercises in children with asthma. J. Bras. Pneumol. 2008, 34, 552–558. [Google Scholar] [CrossRef] [Green Version]
- Gordon, A.; Glickman-Simon, R. Moxibustion and breech presentation, breathing exercises and asthma, coenzyme Q10 and heart failure, acupuncture and chronic low back pain, and cinnamon and diabetes. Explore (NY) 2014, 10, 198–202. [Google Scholar] [CrossRef]
- Andrade, L.B.; Britto, M.C.; Lucena-Silva, N.; Gomes, R.G.; Figueroa, J.N. The efficacy of aerobic training in improving the inflammatory component of asthmatic children. Randomized trial. Respir. Med. 2014, 108, 1438–1445. [Google Scholar] [CrossRef] [Green Version]
- Duruturk, N.; Acar, M.; Doğrul, M.I. Effect of inspiratory muscle training in the management of patients with asthma. J. Cardiopulm. Rehabil. Prev. 2018, 38, 198–203. [Google Scholar] [CrossRef]
- David, M.M.C.; Gomes, E.L.d.F.D.; Mello, M.C.; Costa, D. Noninvasive ventilation and respiratory physical therapy reduce exercise-induced bronchospasm and pulmonary inflammation in children with asthma: Randomized clinical trial. Ther. Adv. Respir. Dis. 2018, 12, 1753466618777723. [Google Scholar] [CrossRef]
- Wedri, N.; i Dewa Putu Gede, P.; Sulisnadewi, N.; Sipahutar, I.; Lestari, A.; Hendrajaya, K. Effect of Diaphragmatic Breathing Exercise on Peak Expiratory Flow (PEF) in Individual with Asthma. Indian J. Public Health Res. Dev. 2018, 9, 429. [Google Scholar] [CrossRef]
- Evaristo, K.B.; Mendes, F.A.R.; Saccomani, M.G.; Cukier, A.; Carvalho-Pinto, R.M.; Rodrigues, M.R.; Santaella, D.F.; Saraiva-Romanholo, B.M.; Martins, M.A.; Carvalho, C.R.F. Effects of aerobic training versus breathing exercises on asthma control: A randomized trial. J. Allergy Clin. Immunol. Pract. 2020, 8, 2989–2996. e4. [Google Scholar] [CrossRef]
- Chen, B.; Liu, G.; Shardonofsky, F.; Dowell, M.; Lakser, O.; Mitchell, R.W.; Fredberg, J.J.; Pinto, L.H.; Solway, J. Tidal breathing pattern differentially antagonizes bronchoconstriction in C57BL/6J vs. A/J mice. J. Appl. Physiol. (1985) 2006, 101, 249–255. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pereira, L.F.; Mancuzo, E.V.; Rezende, C.F.; Correa Rde, A. Six-minute walk test and respiratory muscle strength in patients with uncontrolled severe asthma: A pilot study. J. Bras. Pneumol. 2015, 41, 211–218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramos, E.; de Oliveira, L.V.; Silva, A.B.; Costa, I.P.; Correa, J.C.; Costa, D.; Alves, V.L.; Donner, C.F.; Stirbulov, R.; Arena, R.; et al. Peripheral muscle strength and functional capacity in patients with moderate to severe asthma. Multidiscip. Respir. Med. 2015, 10, 3. [Google Scholar] [CrossRef] [Green Version]
- Teng, Y.K.; Huang, J.L.; Yeh, K.W.; Fu, L.S.; Lin, C.H.; Ma, W.F.; Lee, S.D.; Chiang, L.C. Influential factors of insufficient physical activity among adolescents with asthma in Taiwan. PLoS ONE 2014, 9, e116417. [Google Scholar] [CrossRef]
- Verlaet, A.; Moreira, A.; Sá-Sousa, A.; Barros, R.; Santos, R.; Moreira, P.; Fonseca, J. Physical activity in adults with controlled and uncontrolled asthma as compared to healthy adults: A cross-sectional study. Clin. Transl. Allergy 2013, 3, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choudhari, A. Effects of Inspiratory Muscle Training on Heart Rate Variability. Master’s Thesis, Western Washington University, Bellingham, WA, USA, 2010. [Google Scholar]
- Washburn, R. Assessment of Physical Activity in Older Adults. Res. Q. Exerc. Sport 2000, 71, S79–S88. [Google Scholar] [CrossRef] [PubMed]
- Wickel, E.E.; Welk, G.J.; Eisenmann, J.C. Concurrent Validation of the Bouchard Diary with an Accelerometry-Based Monitor. Med. Sci. Sports Exerc. 2006, 38, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Chu, C.; Lin, K. Application of physical activity scales/questionnaires in Taiwan. J. Exerc. Sci. Fit. 2010, 11, 1–12. [Google Scholar]
- Prince, S.A.; Adamo, K.B.; Hamel, M.E.; Hardt, J.; Gorber, S.; Tremblay, M. A comparison of direct versus self-report measures for assessing physical activity in adults: A systematic review. Int. J. Behav. Nutr. Phys. Act. 2008, 5, 56. [Google Scholar] [CrossRef] [Green Version]
- Sylvia, L.G.; Bernstein, E.E.; Hubbard, J.L.; Keating, L.; Anderson, E.J. Practical guide to measuring physical activity. J. Acad. Nutr. Diet. 2014, 114, 199–208. [Google Scholar] [CrossRef] [Green Version]
- Soares, S.M.; Nucci, L.B.; da Silva, M.M.; Campacci, T.C. Pulmonary function and physical performance outcomes with preoperative physical therapy in upper abdominal surgery: A randomized controlled trial. Clin. Rehabil. 2013, 27, 616–627. [Google Scholar] [CrossRef]

| BTE | (n = 30) | IMT | (n = 30) | ||
|---|---|---|---|---|---|
| Mean | SD | Mean | SD | p Value | |
| Age (year) | 55.1 | 7.72 | 55.1 | 6.68 | 0.5 |
| Height (cm) | 159.57 | 8.69 | 160.87 | 7.55 | 0.26 |
| Weight (kg) | 64.31 | 11.62 | 64.47 | 14.5 | 0.17 |
| BMI (kg/m2) | 25.26 | 4.14 | 25.97 | 5.12 | 0.28 |
| Body fat (%) | 34.6 | 8.29 | 34.04 | 8.53 | 0.39 |
| Total | BTE | IMT | ||
|---|---|---|---|---|
| n (%) | n (%) | n (%) | p Value | |
| Participant | 60 (100%) | 30 (50%) | 30 (50%) | |
| Sex b | 0.38 | |||
| Male | 15 (25%) | 8 (26.7%) | 7 (23.3%) | |
| Female | 45 (75%) | 22 (73.3%) | 23 (76.7%) | |
| Age a | 0.5 | |||
| 40–49 years | 13 (21.6%) | 7 (23.3%) | 6 (20%) | |
| 50–59 years | 25 (41.7%) | 10 (33.3%) | 15 (50%) | |
| 60–65 years | 22 (36.7%) | 13 (43.3%) | 9 (30%) | |
| Asthma severity a | 0.5 | |||
| Moderate | 56 (93.3%) | 28 (93.3%) | 28 (93.3%) | |
| Severe | 4 (6.7%) | 2 (6.7%) | 2 (6.7%) |
| Outcome | Groups | Time Period | p Values | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Pre | 95% CI | Post | 95% CI | Δ | “Group” × “Time” | “Time” | “Group” | ||
| Pulmonary functions | |||||||||
| FVC (% predicted) | BTE | 87.05 ± 15.3 | 81.82–92.28 | 85.62 ± 14.75 | 80.78–90.46 | −1.43 ± 11.56 † | 0.045 * | 0.312 | 0.147 |
| IMT | 79.38 ± 13.26 | 74.15–84.62 | 83.63 ± 11.54 | 78.79–88.47 | 4.25 ± 9.82 † | ||||
| FEV1 (% predicted) | BTE | 82.77 ± 17.19 | 76.73–88.81 | 82.37 ± 16.56 | 77–87.74 | −0.4 ± 6.34 | 0.263 | 0.498 | 0.505 |
| IMT | 79.12 ± 15.84 | 73.08–85.17 | 80.73 ± 12.55 | 75.36–86.1 | 1.61 ± 7.36 | ||||
| Respiratory muscle strength | |||||||||
| PImax (% predicted) | BTE | 82.45 ± 32 | 71.69–93.22 | 99.38 ± 35.03 | 88.25–110.5 | 16.92 ± 12.94 † | 0.00 * | 0.00 * | 0.527 |
| IMT | 71.19 ± 26.66 | 60.42–81.95 | 101.03 ± 25.04 | 88.9–112.16 | 29.84 ± 13.16 † | ||||
| PEmax (cmH2O) | BTE | 53.87 ± 20.72 | 45.51–62.23 | 70.67 ± 25.5 | 61.39–79.94 | 16.8 ± 15.52 | 0.9 | 0.00 * | 0.918 |
| IMT | 54.7 ± 24.83 | 46.34–63.06 | 71.07 ± 25.26 | 61.79–80.34 | 16.37 ± 13.67 | ||||
| Asthma control | |||||||||
| ACT | BTE | 21.43 ± 2.06 | 20.5–22.36 | 22.06 ± 1.86 | 21.34–22.79 | 0.63 ± 1.49 | 0.437 | 0.001 * | 0.927 |
| IMT | 21.3 ± 2.95 | 20.37–22.23 | 22.3 ± 2.1 | 21.58–23.03 | 1 ± 2.03 | ||||
| ACQ | BTE | 1.15 ± 0.44 | 0.98–1.33 | 1.02 ± 0.43 | 0.87–1.17 | −0.13 ± 1.37 | 0.466 | 0.00 * | 0.883 |
| IMT | 1.2 ± 0.52 | 1.03–1.38 | 1 ± 0.41 | 0.85–1.16 | −0.2 ± 0.31 | ||||
| Functional capacity | |||||||||
| 6MWT | |||||||||
| Distance (meters) | BTE | 520.17 ± 74.8 | 493.35–546.98 | 545. ± 71.44 | 520.94–569.26 | 24.93 ± 42.03 | 0.237 | 0.00 * | 0.741 |
| IMT | 508.67 ± 71.92 | 481.85–535.48 | 545.06 ± 60.3 | 520.9–569.22 | 36.4 ± 31.45 | ||||
| Heart rate (beats/min) | BTE | 108.23 ± 13.8 | 103.02–113.45 | 109.7 ± 16.14 | 104.61–114.79 | 1.47 ± 16.39 | 0.318 | 0.076 | 0.524 |
| IMT | 104.36 ± 14.72 | 99.15–109.58 | 109.53 ± 11.29 | 104.44–114.62 | 5.17 ± 11.66 | ||||
| Blood oxygen saturation (%) | BTE | 94.86 ± 1.83 | 94.18–95.55 | 94.16 ± 1.93 | 93.48–94.85 | −0.7 ± 1.86 | 1 | 0.003 * | 0.440 |
| IMT | 94.53 ± 1.90 | 93.85–95.22 | 93.83 ± 1.82 | 93.15–94.52 | −0.7 ± 1.6 | ||||
| Physical activity | |||||||||
| 3-D PAL (Kcal/kg/day) | |||||||||
| Retired | BTE | 38.15 ± 5.67 | 35.33–40.98 | 39.51 ± 5.06 | 36.8–42.24 | 1.36 ± 2.29 | 0.587 | 0.001 * | 0.765 |
| IMT | 37.36 ± 3.95 | 34.43–40.31 | 39.15 ± 4.37 | 36.32–41.99 | 1.33 ± 2.53 | ||||
| Employed | BTE | 44.01 ± 4.57 | 40.76–47.28 | 45.34 ± 8.07 | 41.98–48.71 | 1.79 ± 1.5 | 0.462 | 0.001 * | 0.467 |
| IMT | 45.3 ± 8.06 | 42.14–48.48 | 47.33 ± 8.51 | 44.06–50.6 | 2.02 ± 2.65 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chung, Y.; Huang, T.-Y.; Liao, Y.-H.; Kuo, Y.-C. 12-Week Inspiratory Muscle Training Improves Respiratory Muscle Strength in Adult Patients with Stable Asthma: A Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2021, 18, 3267. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph18063267
Chung Y, Huang T-Y, Liao Y-H, Kuo Y-C. 12-Week Inspiratory Muscle Training Improves Respiratory Muscle Strength in Adult Patients with Stable Asthma: A Randomized Controlled Trial. International Journal of Environmental Research and Public Health. 2021; 18(6):3267. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph18063267
Chicago/Turabian StyleChung, Yi, Ting-Yu Huang, Yi-Hung Liao, and Yu-Chi Kuo. 2021. "12-Week Inspiratory Muscle Training Improves Respiratory Muscle Strength in Adult Patients with Stable Asthma: A Randomized Controlled Trial" International Journal of Environmental Research and Public Health 18, no. 6: 3267. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph18063267







