Thallium Contamination of Drinking Water: Health Implications in a Residential Cohort Study in Tuscany (Italy)
Abstract
:1. Introduction
2. Materials and Methods
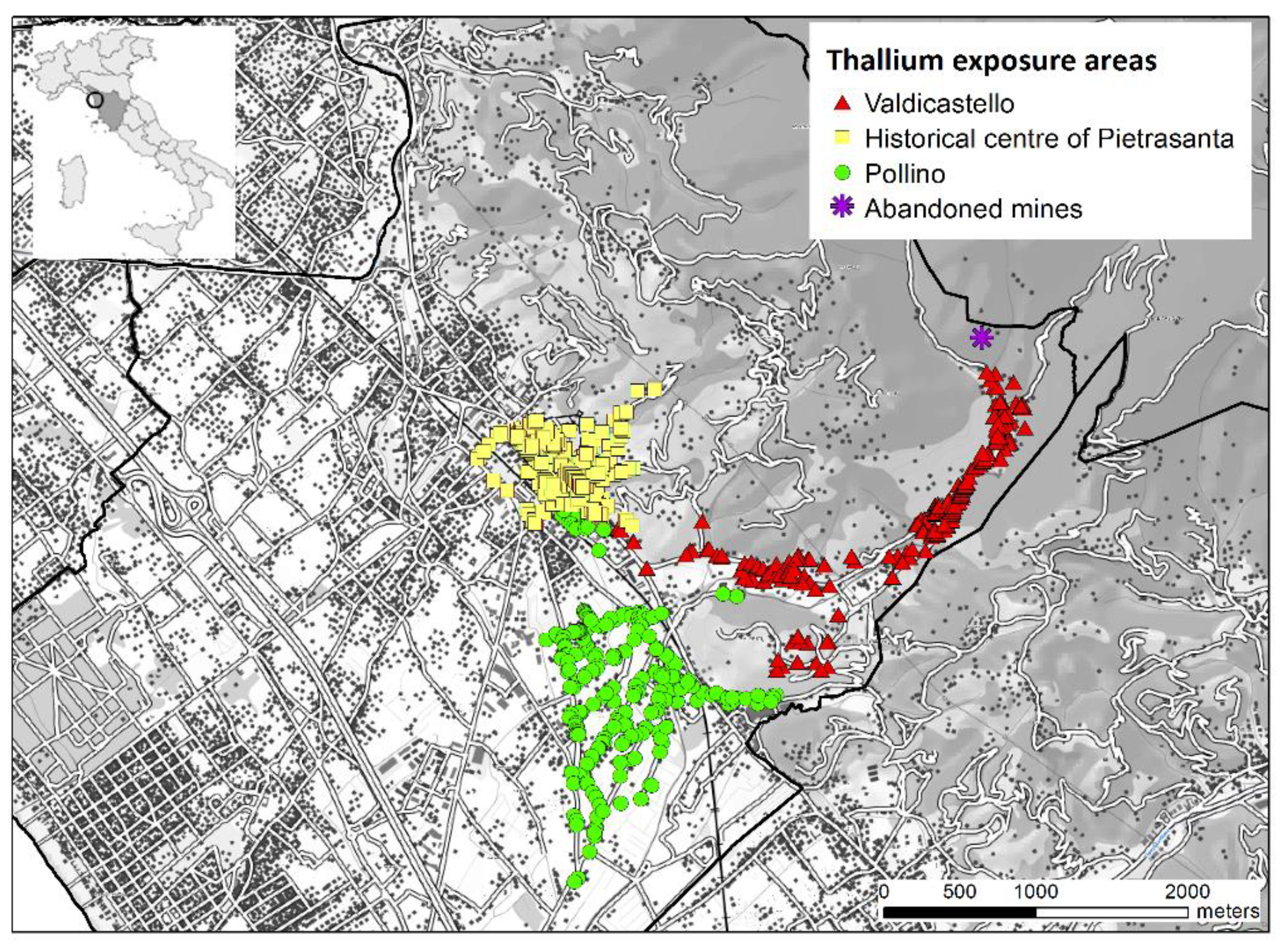
2.1. Evaluation of Thallium Exposure
2.2. The Definition of the Cohort
2.3. Follow-Up Procedures and Health Data
2.4. Statistical Analysis
3. Results
3.1. Characteristics of the Study Cohort
3.2. Association between Residence in Thallium Exposure Areas and Mortality
3.3. Association between Residence in Exposure Areas and Hospitalizations
3.4. Association between Residence in Exposure Areas and Adverse Pregnancy Outcomes
4. Discussion
4.1. Mortality and Morbidity Outcomes
4.2. Adverse Pregnancy Outcomes
4.3. Strenghts and Limitations of the Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization WHO. Thallium. International Programme on Chemical Safety; Environmental Health Criteria 182; WHO: Geneva, Switzerland, 1996. [Google Scholar]
- EPA U.S. Environmental Protection Agency. Toxicological Review of Thallium and Compounds; EPA: Washington, DC, USA, 2009.
- ATSDR Agency for Toxic Substances and Disease Registry U.S. Public Health Service. Toxicological Profile for Thallium; ATSDR: Atlanta, GA, USA, 1992.
- Kemper, F.H.; Bertram, H.P. Thallium. In Metals in the Environment; Merian, E., Ed.; Verlag Chemie: Weinheim, Germany, 1984; pp. 571–583. [Google Scholar]
- Manzo, L.; Sabbioni, E. Thallium toxicity and the nervous system. In Metal Neurotoxicity; Bondy, S.C., Prasad, K.N., Eds.; CRC Press: Boca Raton, FL, USA, 1988; pp. 35–54. [Google Scholar]
- Pearson, A.J.; Ashmore, E. Risk assessment of antimony, barium, beryllium, boron, bromine, lithium, nickel, strontium, thallium and uranium concentrations in the New Zealand diet. Food Addit. Contam. Part A 2019, 37, 451–464. [Google Scholar] [CrossRef] [PubMed]
- Sherlock, J.C.; Smart, G.A. Thallium in foods and the diet. Food Addit. Contam. 1986, 3, 363–370. [Google Scholar] [CrossRef]
- Yu, H.-Y.; Chang, C.; Li, F.; Wang, Q.; Chen, M.; Zhang, J. Thallium in flowering cabbage and lettuce: Potential health risks for local residents of the Pearl River Delta, South China. Environ. Pollut. 2018, 241, 626–635. [Google Scholar] [CrossRef] [PubMed]
- Nisse, C.; Tagne-Fotso, R.; Howsam, M.; Richeval, C.; Labat, L.; Leroyer, A. Blood and urinary levels of metals and metalloids in the general adult population of Northern France: The IMEPOGE study, 2008–2010. Int. J. Hyg. Environ. Health 2017, 220, 341–363. [Google Scholar] [CrossRef] [PubMed]
- CDC NHANES. Fourth National Report on Human Exposure to Environmental Chemicals Updated Tables, January 2019 Volume Two. US Department of Health an Human Services Centers for Disease Control and Prevention. Available online: https://www.cdc.gov/exposurereport/pdf/FourthReport_UpdatedTables_Volume2_Jan2019-508.pdf (accessed on 15 December 2019).
- Osorio-Rico, L.; Santamaria, A.; Galván-Arzate, S. Thallium Toxicity: General Issues, Neurological Symptoms, and Neurotoxic Mechanisms. Adv. Neurobiol. 2017, 18, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Karbowska, B. Presence of thallium in the environment: Sources of contaminations, distribution and monitoring methods. Environ. Monit. Assess. 2016, 188, 1–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galván-Arzate, S.; Santamaría, A. Thallium toxicity. Toxicol. Lett. 1998, 99, 1–13. [Google Scholar] [CrossRef]
- Kemper, F.H. Thallium poisonings. Münch. Med. Wochenschr. 1979, 121, 1357–1358. [Google Scholar]
- Potes-Gutierrez, J.; Del Real, E. Acute thallium intoxication. Ind. Med. Surg. 1966, 35, 618–619. [Google Scholar]
- Sharma, J.; Sharma, R.; Singh, H.; Satake, M. Hazards and analysis of thallium—A review. Toxicol. Environ. Chem. 1986, 11, 93–116. [Google Scholar] [CrossRef]
- Saddique, A.; Peterson, C.D. Thallium poisoning: A review. Vet. Hum. Toxicol. 1983, 25, 16–22. [Google Scholar] [PubMed]
- Malcolm, D. Toxicology of metals other than lead. In Current Approaches to Occupational Medicine; Gardner, A.W., Ed.; John Wright and Sons Ltd.: Bristol, UK, 1979; pp. 18–43. [Google Scholar]
- Triebig, G.; Büttner, J. Occupational relevant neurotoxins: I. Metals and their compounds—A literature review of the years 1970 to 1982. Zent. Bakteriol. Mikrobiol. Hyg. 1983, 177, 11–36. [Google Scholar]
- Schoer, J. Thallium. In The Handbook of Environmental Chemistry; Anthropogenic Compounds, Part C; Hutzinger, O., Ed.; Springer: Berlin, Germany, 1984; Volume 3, pp. 143–214. [Google Scholar]
- Moeschlin, S. Poisoning: Diagnosis and Treatment; Georg Thieme: Stuttgart, Germany, 1965. [Google Scholar]
- Buschke, A.; Langer, E. The forensic and the industrial-hygienic importance of thallium. Münch. Med. Wochenschr. 1927, 74, 1494–1497. [Google Scholar]
- Gefel, A.; Liron, M.; Hirsch, W. Chronic thallium poisoning. Isr. J. Med. Sci. 1970, 6, 380–382. [Google Scholar] [PubMed]
- Goldblatt, D. Pollutants and industrial hazards. In Merritt’s Textbook of Neurology, 8th ed.; Rowland, L.P., Ed.; Lea and Febinger: Philadelphia, PA, USA, 1989; pp. 919–928. [Google Scholar]
- Aprea, M.C.; Apostoli, P.; Bettinelli, M.; Lovreglio, P.; Negri, S.; Perbellini, L.; Perico, A.; Ricossa, M.C.; Salamon, F.; Scapellato, M.L.; et al. Urinary levels of metal elements in the non-smoking general population in Italy: SIVR study 2012–2015. Toxicol. Lett. 2018, 298, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Minoia, C.; Sabbioni, E.; Apostoli, P.I.E.T.R.A.; Pietra, R.; Pozzoli, L.; Gallorini, M.; Nicolaou, G.; Alessio, L.; Capodaglio, E. Trace element reference values in tissues from inhabitants of the European Community I. A study of 46 elements in urine, blood and serum of Italian subjects. Sci. Total Environ. 1990, 95, 89–105. [Google Scholar] [CrossRef]
- Sabbioni, E.; Minoia, C.; Ronchi, A.; Hansen, B.G.; Pietra, R.; Balducci, C. Trace element reference values in tissues from inhabitants of the European Union. VIII. Thallium in the Italian population. Sci. Total Environ. 1994, 158, 227–236. [Google Scholar] [CrossRef]
- Alimonti, A.; Bocca, B.; Mattei, D.; Pino, A. Biomonitoraggio della popolazione italiana per l’esposizione ai metalli: Valori di riferimento 1990–2009. Rapp. Istisan 2010, 10, 22. [Google Scholar]
- National Research Council (NRC). Monitoring Human Tissues for Toxic Substances. Washington; National Academy Press: Washington, DC, USA, 1991. [Google Scholar]
- Becker, K.; Conrad, A.; Kirsch, N.; Kolossa-Gehring, M.; Schulz, C.; Seiwert, M.; Seifert, B. German Environmental Survey (GerES): Human biomonitoring as a tool to identify exposure pathways. Int. J. Hyg. Environ. Health 2007, 210, 267–269. [Google Scholar] [CrossRef]
- Schulz, C.; Angerer, J.; Ewers, U.; Heudorf, U.; Wilhelm, M. Human Biomonitoring Commission of the German Federal Environment Agency. Revised and new reference values for environmental pollutants in urine or blood of children in Germany derived from the German environmental survey on children 2003–2006 (GerES IV). Int. J. Hyg. Environ. Health 2009, 212, 637–647. [Google Scholar] [CrossRef]
- Schulz, C.; Wilhelm, M.; Heudorf, U.; Kolossa-Gehring, M. Human Biomonitoring Commission of the German Federal Environment Agency. Update of the reference and HBM values derived by the German Human Biomonitoring Commission. Int. J. Hyg. Environ. Health 2011, 215, 26–35. [Google Scholar] [CrossRef]
- Dolgner, R.; Brockhaus, A.; Ewers, U.; Wiegand, H.; Majewski, F.; Soddemann, H. Repeated surveillance of exposure to thallium in a population living in the vicinity of a cement plant emitting dust containing thallium. Int. Arch. Occup. Environ. Health 1983, 52, 79–94. [Google Scholar] [CrossRef]
- Brockhaus, A.; Dolgner, R.; Ewers, U.; Krämer, U.; Soddemann, H.; Wiegand, H. Intake and health effects of thallium among a population living in the vicinity of a cement plant emitting thallium containing dust. Int. Arch. Occup. Environ. Health 1981, 48, 375–389. [Google Scholar] [CrossRef]
- Xiao, T.; Guha, J.; Liu, C.-Q.; Zheng, B.; Wilson, G.; Ning, Z.; He, L. Potential health risk in areas of high natural concentrations of thallium and importance of urine screening. Appl. Geochem. 2007, 22, 919–929. [Google Scholar] [CrossRef]
- Zhou, D.X.; Liu, D.N. Chronic thallium poisoning in a rural area of Guizhou Province, China. J. Environ. Health 1985, 48, 14–18. [Google Scholar]
- Xia, W.; Du, X.; Zheng, T.; Zhang, B.; Li, Y.; Bassig, B.A.; Zhou, A.; Wang, Y.; Xiong, C.; Li, Z.; et al. A Case-Control Study of Prenatal Thallium Exposure and Low Birth Weight in China. Environ. Health Perspect. 2016, 124, 164–169. [Google Scholar] [CrossRef]
- Hu, X.; Zheng, T.; Cheng, Y.; Holford, T.; Lin, S.; Leaderer, B.; Qiu, J.; Bassig, B.A.; Shi, K.; Zhang, Y.; et al. Distributions of heavy metals in maternal and cord blood and the association with infant birth weight in China. J. Reprod. Med. 2015, 60, 21–29. [Google Scholar] [PubMed]
- Curković, M.; Sipos, L.; Puntarić, D.; Dodig-Ćurković, K.; Pivac, N.; Kralik, K. Detection of thallium and uranium in well water and biological specimens of an eastern Croatian population. Arh. Hig. Rada Toksikol. 2013, 64, 385–394. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aprea, M.C.; Nuvolone, D.; Petri, D.; Voller, F.; Bertelloni, S.; Aragona, I. Human biomonitoring to assess exposure to thallium following the contamination of drinking water. PLoS ONE 2020, 15, e0241223. [Google Scholar] [CrossRef]
- Campanella, B.; Onor, M.; D’Ulivo, A.; Giannecchini, R.; D’Orazio, M.; Petrini, R.; Bramanti, E. Human exposure to thallium through tap water: A study from Valdicastello Carducci and Pietrasanta (northern Tuscany, Italy). Sci. Total Environ. 2016, 548–549, 33–42. [Google Scholar] [CrossRef]
- Hoet, P.; Jacquerye, C.; Deumer, G.; Lison, D.; Haufroid, V. Reference values and upper reference limits for 26 trace elements in the urine of adults living in Belgium. Clin. Chem. Lab. Med. 2013, 51, 839–849. [Google Scholar] [CrossRef] [PubMed]
- Morton, J.; Tan, E.; Leese, E.; Cocker, J. Determination of 61 elements in urine samples collected from a non-occupationally exposed UK adult population. Toxicol. Lett. 2014, 231, 179–193. [Google Scholar] [CrossRef] [Green Version]
- Saravanabhavan, G.; Werry, K.; Walker, M.; Haines, D.; Malowany, M.; Khoury, C. Human biomonitoring reference values for metals and trace elements in blood and urine derived from the Canadian Health Measures Survey 2007–2013. Int. J. Hyg. Environ. Health 2017, 220, 189–200. [Google Scholar] [CrossRef] [Green Version]
- Stillerman, K.P.; Mattison, D.R.; Giudice, L.C.; Woodruff, T.J. Environmental Exposures and Adverse Pregnancy Outcomes: A Review of the Science. Reprod. Sci. 2008, 15, 631–650. [Google Scholar] [CrossRef] [PubMed]
- Wigle, D.T.; Arbuckle, T.E.; Turner, M.C.; Bérubé, A.; Yang, Q.; Liu, S.; Krewski, D. Epidemiologic Evidence of Relationships Between Reproductive and Child Health Outcomes and Environmental Chemical Contaminants. J. Toxicol. Environ. Health Part B 2008, 11, 373–517. [Google Scholar] [CrossRef] [PubMed]
- Wigle, N.T.; Arbuckle, T.E.; Walker, M.; Wade, M.G.; Liu, S.; Krewski, D. Environmental Hazards: Evidence for Effects on Child Health. J. Toxicol. Environ. Health Part B 2007, 10, 3–39. [Google Scholar] [CrossRef] [PubMed]
| MEN | WOMEN | TOTAL | |||||
|---|---|---|---|---|---|---|---|
| N. | % | N. | % | N. | % | ||
| Total | 15,940 | 100 | 17,768 | 100 | 33,708 | 100 | |
| Age Group (Years) at Baseline | |||||||
| <1 | 1377 | 8.6 | 1380 | 7.8 | 2757 | 8.2 | |
| 1–9 | 1263 | 7.9 | 1256 | 7.1 | 2519 | 7.5 | |
| 10–19 | 1244 | 7.8 | 1233 | 6.9 | 2477 | 7.3 | |
| 20–29 | 2104 | 13.2 | 2289 | 12.9 | 4393 | 13.0 | |
| 30–39 | 2915 | 18.3 | 2980 | 16.8 | 5895 | 17.5 | |
| 40–49 | 2328 | 14.6 | 2342 | 13.2 | 4670 | 13.9 | |
| 50–59 | 1886 | 11.8 | 2134 | 12.0 | 4020 | 11.9 | |
| 60–69 | 1618 | 10.2 | 1997 | 11.2 | 3615 | 10.7 | |
| 70–79 | 966 | 6.1 | 1523 | 8.6 | 2489 | 7.4 | |
| ≥80 | 239 | 1.5 | 634 | 3.6 | 873 | 2,6 | |
| Socioeconomic Deprivation Index (SES) | |||||||
| Low | 2595 | 16.3 | 3053 | 17.2 | 5648 | 16.8 | |
| Medium | 4910 | 30.8 | 5507 | 31.0 | 10,417 | 30.9 | |
| High | 8431 | 52.9 | 9205 | 51.8 | 17,636 | 52.3 | |
| Missing | 3 | 0.02 | 4 | 0.02 | 7 | 0.02 | |
| Calendar Period | |||||||
| Present at 1 January 2000 | 10,894 | 68.3 | 12037 | 67.7 | 22,931 | 68.0 | |
| Entered between 2000 and 2004 | 1461 | 9.2 | 1594 | 9.0 | 3055 | 9.1 | |
| Entered between 2005 and 2009 | 1890 | 11.9 | 2162 | 12.2 | 4052 | 12.0 | |
| Entered between 2010 and 2015 | 1695 | 10.6 | 1975 | 11.1 | 3670 | 10.9 | |
| Birth Place | |||||||
| Pietrasanta | 7628 | 47.9 | 8096 | 45.6 | 15,724 | 46.6 | |
| Tuscany | 5579 | 35.0 | 6072 | 34.2 | 11,651 | 34.6 | |
| Italy | 1568 | 9.8 | 1844 | 10.4 | 3412 | 10.1 | |
| Foreign | 1165 | 7.3 | 1756 | 9.9 | 2921 | 8.7 | |
| Vital Status at 15 March 2015 | |||||||
| Alive | 10,837 | 68.0 | 12,297 | 69.2 | 23,134 | 68.6 | |
| Emigrant | 3059 | 19.2 | 3399 | 19.1 | 6458 | 19.2 | |
| Dead | 1669 | 10.5 | 1773 | 10.0 | 3442 | 10.2 | |
| Other | 375 | 2.4 | 299 | 1.7 | 674 | 2.0 | |
| Model 1: Residence in Valdicastello | Model 2: Overall Exposure | Not Contaminated Area | ||||
|---|---|---|---|---|---|---|
| N. | % | N. | % | N. | % | |
| 1682 | 100 | 6854 | 100 | 26,854 | 100 | |
| Gender | ||||||
| Men | 772 | 45.9 | 3232 | 47.1 | 12,708 | 47.3 |
| Women | 910 | 54.1 | 3622 | 52.9 | 14,146 | 52.7 |
| Age at Baseline | ||||||
| Mean (SD) | 48.5 (23.4) | 49.7 (23.5) | 50.9 (23.9) | |||
| Socioeconomic Deprivation Index (SES) | ||||||
| Low | 654 | 38.9 | 705 | 10.9 | 4897 | 18.2 |
| Medium | 704 | 41.9 | 2110 | 30.8 | 8308 | 30.9 |
| High | 324 | 19.3 | 3987 | 58.2 | 13,649 | 50.8 |
| Calendar Period | ||||||
| Present at 1 January 2000 | 1135 | 67.5 | 4288 | 62.6 | 18,643 | 69.4 |
| Entered between 2000 and 2004 | 169 | 10.0 | 801 | 11.7 | 2254 | 8.4 |
| Entered between 2005 and 2009 | 187 | 11.1 | 912 | 13.3 | 3140 | 11.7 |
| Entered between 2010 and 2015 | 191 | 11.4 | 853 | 12.4 | 2817 | 10.5 |
| Birth Place | ||||||
| Pietrasanta | 954 | 56.7 | 3580 | 52.2 | 12,144 | 45.2 |
| Tuscany | 467 | 27.8 | 1878 | 27.4 | 9773 | 36.4 |
| Italy | 118 | 7.0 | 667 | 9.7 | 2745 | 10.2 |
| Foreign | 143 | 8.5 | 729 | 10.6 | 2192 | 8.2 |
| Model 1: Residence in Valdicastello | Model 2: Overall Exposure * | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Cause of Death (ICD-9) | Reference N. | N. | HR ** | 95% CI | N. | HR | 95% CI | ||
| All Causes (001–899) | 2797 | 138 | 0.76 | 0.64 | 0.91 | 645 | 0.88 | 0.81 | 0.96 |
| Malignant Neoplasms (140–209) | 764 | 31 | 0.60 | 0.42 | 0.86 | 157 | 0.81 | 0.68 | 0.96 |
| Stomach (151) | 47 | 2 | 0.67 | 0.16 | 2.77 | 9 | 0.77 | 0.38 | 1.58 |
| Colon Rectum (153,154,159) | 97 | 4 | 0.62 | 0.23 | 1.69 | 21 | 0.85 | 0.53 | 1.37 |
| Liver (155,156) | 89 | 6 | 0.92 | 0.40 | 2.11 | 27 | 1.21 | 0.79 | 1.87 |
| Pancreas (157) | 49 | 1 | 0.31 | 0.04 | 2.25 | 8 | 0.63 | 0.30 | 1.34 |
| Lung (162) | 141 | 2 | 0.23 | 0.06 | 0.92 | 21 | 0.58 | 0.36 | 0.92 |
| Breast (174,175) | 43 | 1 | 0.29 | 0.04 | 2.11 | 11 | 0.99 | 0.51 | 1.93 |
| Ovary (183) | 18 | 1 | 0.70 | 0.09 | 5.26 | 4 | 0.89 | 0.30 | 2.65 |
| Bladder (188) | 18 | 3 | 1.81 | 0.53 | 6.16 | 9 | 2.07 | 0.92 | 4.63 |
| Lymphatic and Hematopoietic Tissue (200–208) | 66 | 4 | 0.96 | 0.35 | 2.66 | 19 | 1.09 | 0.65 | 1.82 |
| Diabetes Mellitus (250) | 75 | 1 | 0.25 | 0.03 | 1.78 | 7 | 0.34 | 0.16 | 0.74 |
| Nervous System and Sense Organs (320–289) | 96 | 3 | 0.53 | 0.17 | 1.67 | 20 | 0.79 | 0.49 | 1.29 |
| Circulatory System (390–459) | 836 | 48 | 0.89 | 0.66 | 1.19 | 217 | 0.97 | 0.84 | 1.13 |
| Ischemic Heart Disease (410–414) | 228 | 19 | 1.18 | 0.73 | 1.89 | 66 | 1.13 | 0.86 | 1.48 |
| Respiratory System (460–519) | 137 | 8 | 0.96 | 0.47 | 1.97 | 38 | 1.03 | 0.72 | 1.48 |
| COPD (490–492,494–496) | 71 | 3 | 0.73 | 0.23 | 2.35 | 19 | 1.02 | 0.61 | 1.69 |
| Digestive System (520–579) | 113 | 3 | 0.43 | 0.14 | 1.37 | 18 | 0.60 | 0.37 | 0.99 |
| Chronic Liver Disease and Cirrhosis (571) | 46 | 0 | n.d. | n.d. | n.d. | 6 | 0.49 | 0.21 | 1.15 |
| Genitourinary System (580–629) | 35 | 2 | 0.94 | 0.22 | 4,00 | 11 | 1.15 | 0.59 | 2.28 |
| Renal Failure (584–586) | 21 | 0 | n.d. | n.d. | n.d. | 4 | 0.71 | 0.24 | 2.08 |
| Model 1: Residence in Valdicastello | Model 2: Overall Exposure * | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Diagnoses (ICD-9) | Reference N. | N. | HR ** | 95% CI | N. | HR ** | 95% CI | ||
| Malignant Neoplasms (140–209) | 1926 | 96 | 0.70 | 0.57 | 0.86 | 425 | 0.85 | 0.77 | 0.95 |
| Stomach (151) | 75 | 1 | 0.21 | 0.03 | 1.51 | 9 | 0.47 | 0.24 | 0.94 |
| Colon Rectum (153,154,159) | 229 | 13 | 0.78 | 0.44 | 1.36 | 58 | 1.00 | 0.75 | 1.34 |
| Liver (155,156) | 123 | 8 | 0.93 | 0.45 | 1.90 | 28 | 0.91 | 0.60 | 1.37 |
| Pancreas (157) | 43 | 2 | 0.67 | 0.16 | 2.76 | 9 | 0.83 | 0.40 | 1.70 |
| Larynx (161) | 51 | 3 | 0.94 | 0.29 | 3.04 | 15 | 1.19 | 0.67 | 2.12 |
| Lung (162) | 165 | 8 | 0.76 | 0.37 | 1.55 | 36 | 0.85 | 0.59 | 1.23 |
| Connective and Other Soft Tissue (171) | 8 | 1 | 1.58 | 0.20 | 12.75 | 4 | 1.96 | 0.59 | 6.53 |
| Breast (174,175) | 270 | 14 | 0.63 | 0.37 | 1.07 | 69 | 0.97 | 0.75 | 1.27 |
| Ovary (183) | 31 | 3 | 1.13 | 0.35 | 3.71 | 9 | 1.10 | 0.52 | 2.33 |
| Bladder (188) | 187 | 13 | 1.08 | 0.61 | 1.90 | 40 | 0.87 | 0.62 | 1.22 |
| Kidney (189) | 73 | 5 | 1,00 | 0.40 | 2.48 | 13 | 0.75 | 0.41 | 1.35 |
| Central Nervous System (191,192, 225, 2396) | 81 | 7 | 1.24 | 0.57 | 2.71 | 14 | 0.68 | 0.39 | 1.20 |
| Thyroid (193) | 70 | 6 | 1.28 | 0.55 | 2.96 | 13 | 0.73 | 0.40 | 1.32 |
| Lymphatic and Hematopoietic Tissue (200–208) | 168 | 10 | 0.87 | 0.46 | 1.65 | 39 | 0.90 | 0.63 | 1.27 |
| Diabetes Mellitus (250) | 136 | 8 | 0.89 | 0.43 | 1.82 | 21 | 0.60 | 0.38 | 0.94 |
| Nervous System and Sense Organs (320–289) | 1632 | 87 | 0.74 | 0.60 | 0.92 | 374 | 0.88 | 0.78 | 0.98 |
| Circulatory System (390–459) | 3636 | 192 | 0.75 | 0.65 | 0.87 | 798 | 0.85 | 0.79 | 0.92 |
| Ischemic Heart Disease (410–414) | 1070 | 56 | 0.77 | 0.59 | 1.01 | 252 | 0.92 | 0.80 | 1.06 |
| Respiratory System (460–519) | 2550 | 163 | 0.88 | 0.75 | 1.03 | 590 | 0.90 | 0.82 | 0.98 |
| COPD (490–492,494–496) | 882 | 64 | 0.99 | 0.77 | 1.28 | 200 | 0.87 | 0.75 | 1.02 |
| Acute Respiratory Infections (460–466) | 135 | 6 | 0.72 | 0.32 | 1.63 | 27 | 0.75 | 0.50 | 1.14 |
| Digestive System (520–579) | 3239 | 197 | 0.85 | 0.73 | 0.98 | 772 | 0.93 | 0.86 | 1.00 |
| Chronic Liver Disease and Cirrhosis (571) | 238 | 13 | 0.74 | 0.42 | 1.29 | 66 | 1.08 | 0.82 | 1.42 |
| Genitourinary System (580–629) | 2364 | 143 | 0.79 | 0.67 | 0.94 | 554 | 0.90 | 0.82 | 0.99 |
| Renal Failure (584–586) | 288 | 11 | 0.57 | 0.31 | 1.04 | 59 | 0.79 | 0.60 | 1.05 |
| Congenital Anomalies (740–759) | 374 | 20 | 0.74 | 0.47 | 1.16 | 67 | 0.72 | 0.55 | 0.93 |
| . | Reference | Model 1: Residence in Valdicastello | Model 2: Overall Exposure * | ||||||
|---|---|---|---|---|---|---|---|---|---|
| N. | N. | OR ** | 95% CI | N. | OR | 95% CI | |||
| Low birth weight | 102 | 6 | 0.90 | 0.38 | 2.12 | 30 | 1.43 | 0.91 | 2.25 |
| Preterm birth | 75 | 3 | 0.55 | 0.17 | 1.78 | 22 | 1.40 | 0.82 | 2.37 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nuvolone, D.; Petri, D.; Aprea, M.C.; Bertelloni, S.; Voller, F.; Aragona, I. Thallium Contamination of Drinking Water: Health Implications in a Residential Cohort Study in Tuscany (Italy). Int. J. Environ. Res. Public Health 2021, 18, 4058. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph18084058
Nuvolone D, Petri D, Aprea MC, Bertelloni S, Voller F, Aragona I. Thallium Contamination of Drinking Water: Health Implications in a Residential Cohort Study in Tuscany (Italy). International Journal of Environmental Research and Public Health. 2021; 18(8):4058. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph18084058
Chicago/Turabian StyleNuvolone, Daniela, Davide Petri, Maria Cristina Aprea, Silvano Bertelloni, Fabio Voller, and Ida Aragona. 2021. "Thallium Contamination of Drinking Water: Health Implications in a Residential Cohort Study in Tuscany (Italy)" International Journal of Environmental Research and Public Health 18, no. 8: 4058. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph18084058






