Adherence Is More Than Just Being Present: Example of a Lay-Led Home-Based Programme with Physical Exercise, Nutritional Improvement and Social Support, in Prefrail and Frail Community-Dwelling Older Adults
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.1.1. Characteristics of the Participants
2.1.2. Intervention: Week 1–12
2.1.3. Intervention: Week 13–24
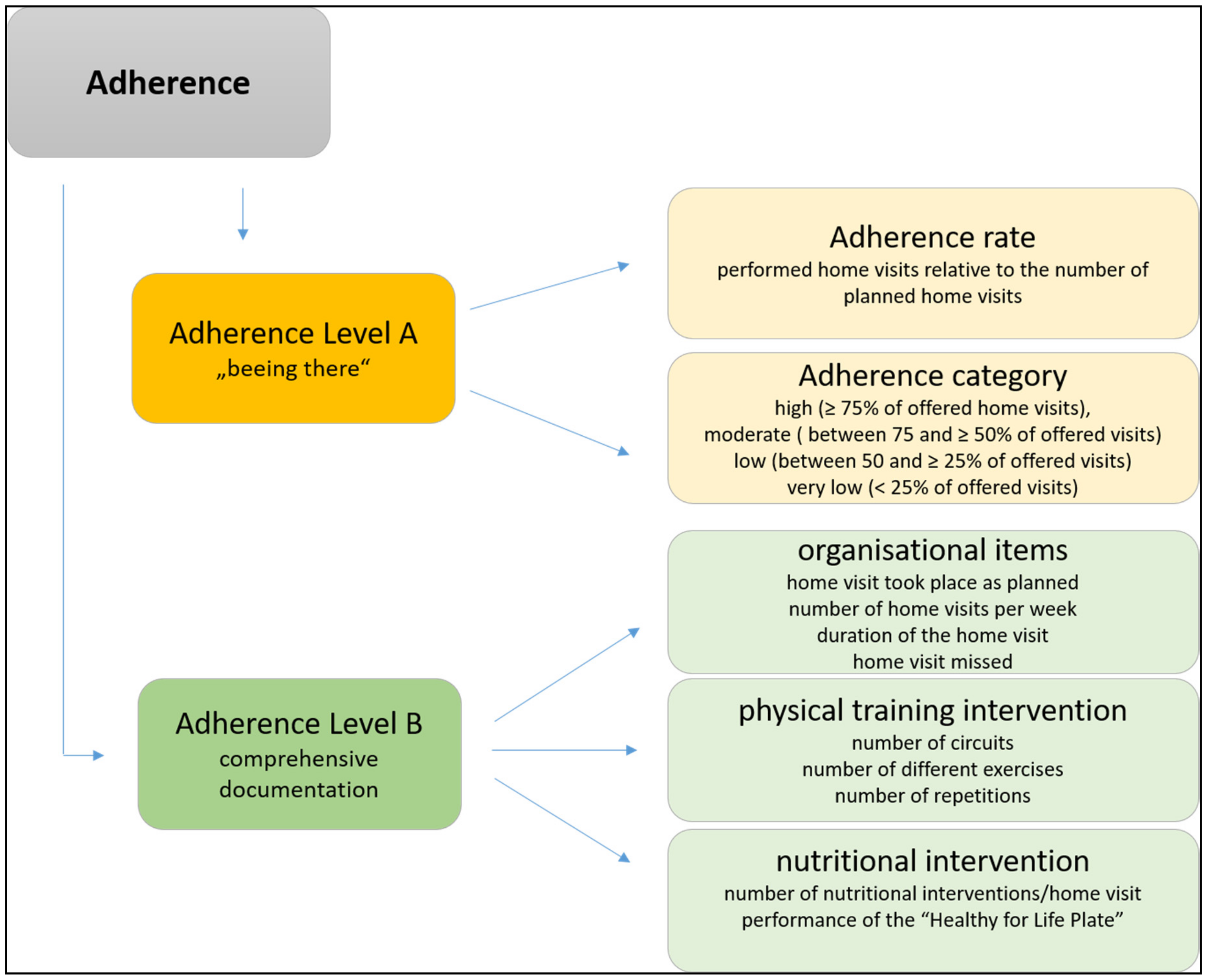
2.2. Measurements of Different Aspects of Adherence
2.2.1. Log Book Determined Adherence Rate and Category
- high (carried out ≥75% of offered home visits),
- moderate (carried out between 75 and ≥50% of offered home visits),
- low (carried out between 50 and ≥25% of offered home visits), or
- very low (drop-outs or less than 25% of offered home visits) [21].
2.2.2. Comprehensive Documentation of the Lifestyle Intervention
- organizational items: home visit took place as planned (yes/no); number of home visits per week; duration of the home visit; home visit missed due to the prefrail/frail participant or the buddy;
- physical training intervention: number of circuits; number of different exercises; number of repetitions;
- nutritional intervention: number of nutritional interventions per home visit (≥1 nutritional message discussed); performance of the “Healthy for Life Plate” during a visit (yes/no).
2.2.3. Self-Perceived Adherence Rate
- “How many home visits were carried out?”
- “How many times were nutritional aspects discussed?”
- “How many times was the predetermined physical activity program realized?”
2.2.4. Continuation
- “Did you regularly continue the study intervention (home visits with physical training and nutritional intervention) in the last 6 months?” (yes/no)
- “Did you sporadically continue the study intervention in the last 6 months?” (yes/no)
- “Did you stop the home visits in the last 6 months?” (yes/no)
- “How many home visits were performed in the last 6 months?” (number)
- “How many physical training interventions were performed in the last 6 months?” (number)
- “How many nutritional interventions were performed in the last 6 months?” (number)
2.2.5. Association with Adherence Rate
2.3. Ethical Considerations
2.4. Statistical Analyses
3. Results
3.1. Baseline Characteristics of the Participants
3.2. Adherence Rate & Category
3.3. Quality of the Intervention (Based on the Comprehensive Documentation)
3.4. Variables Associated with Adherence Category
3.5. Self-Perceived Adherence Parameters
3.6. Continuation after 12 Months
4. Discussion
- –
- How much of the planned intervention has to be done to gain effects?
- –
- How should the overall adherence to an intervention respectively the performed quality be described?
- –
- Is a psycho-social context even more important than a physiological context?
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Locher, J.L.; Roth, D.L.; Ritchie, C.S.; Cox, K.; Sawyer, P.; Bodner, E.V.; Allman, R.M. Body mass index, weight loss, and mortality in community-dwelling older adults. J. Gerontol. A Biol. Sci. Med. Sci. 2007, 62, 1389–1392. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hubbard, R.E.; O’Mahony, M.S.; Calver, B.L.; Woodhouse, K.W. Plasma esterases and inflammation in ageing and frailty. Eur. J. Clin. Pharmacol. 2008, 64, 895–900. [Google Scholar] [CrossRef] [PubMed]
- Dorner, T.; Kranz, A.; Zettl-Wiedner, K.; Ludwig, C.; Rieder, A.; Gisinger, C. The effect of structured strength and balance training on cognitive function in frail, cognitive impaired elderly long-term care residents. Aging Clin. Exp. Res. 2007, 19, 400–405. [Google Scholar] [CrossRef] [PubMed]
- Mello Ade, C.; Engstrom, E.M.; Alves, L.C. Health-related and socio-demographic factors associated with frailty in the elderly: A systematic literature review. Cad. Saude Publica 2014, 30, 1143–1168. [Google Scholar] [CrossRef]
- Haider, S.; Grabovac, I.; Drgac, D.; Mogg, C.; Oberndorfer, M.; Dorner, T.E. Impact of physical activity, protein intake and social network and their combination on the development of frailty. Eur. J. Public Health 2020, 30, 340–346. [Google Scholar] [CrossRef] [PubMed]
- Grabovac, I.; Haider, S.; Mogg, C.; Majewska, B.; Drgac, D.; Oberndorfer, M.; Dorner, T.E. Frailty Status Predicts All-Cause and Cause-Specific Mortality in Community Dwelling Older Adults. J. Am. Med. Dir. Assoc. 2019, 20, 1230–1235.e2. [Google Scholar] [CrossRef]
- Bachner, F.; Bobek, J.; Habimana, K.; Ladurner, J.; Lepuschütz, L.; Ostermann, H.; Rainer, L.; Schmidt, A.; Zuba, M.; Quentin, W.; et al. Austria: Health System Review 2018. Health Systems in Transition; WHO Regional Office for Europe: Copenhagen, Denmark, 2018. [Google Scholar]
- Loh, D.A.; Hairi, N.N.; Choo, W.Y.; Mohd Hairi, F.; Peramalah, D.; Kandiben, S.; Lee, P.L.; Gani, N.; Madzlan, M.F.; Abd Hamid, M.A.; et al. MultiComponent Exercise and theRApeutic lifeStyle (CERgAS) intervention to improve physical performance and maintain independent living among urban poor older people—A cluster randomised controlled trial. BMC Geriatr. 2015, 15, 8. [Google Scholar] [CrossRef] [Green Version]
- Denison, H.J.; Cooper, C.; Sayer, A.A.; Robinson, S.M. Prevention and optimal management of sarcopenia: A review of combined exercise and nutrition interventions to improve muscle outcomes in older people. Clin. Interv. Aging 2015, 10, 859–869. [Google Scholar]
- Cesari, M.; Vellas, B.; Hsu, F.C.; Newman, A.B.; Doss, H.; King, A.C.; Manini, T.M.; Church, T.; Gill, T.M.; Miller, M.E.; et al. A physical activity intervention to treat the frailty syndrome in older persons-results from the LIFE-P study. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2015, 70, 216–222. [Google Scholar] [CrossRef]
- Woo, J.; Goggins, W.; Sham, A.; Ho, S.C. Social determinants of frailty. Gerontology 2005, 51, 402–408. [Google Scholar] [CrossRef]
- Arrebola-Moreno, M.; Petrova, D.; Sánchez, M.J.; Rivera-López, R.; Ramírez-Hernández, J.A. Who does what the cardiologist recommends? Psychosocial markers of unhealthy behavior in coronary disease patients. PLoS ONE 2020, 15, e0228262. [Google Scholar] [CrossRef]
- Karfopoulou, E.; Anastasiou, C.A.; Avgeraki, E.; Kosmidis, M.H.; Yannakoulia, M. The role of social support in weight loss maintenance: Results from the MedWeight study. J. Behav. Med. 2016, 39, 511–518. [Google Scholar] [CrossRef]
- Brown, A.; Wilson, D.K.; Sweeney, A.M.; Van Horn, M.L. The Moderating Effects of Social Support and Stress on Physical Activity in African American Women. Ann. Behav. Med. 2020, 55, 376–382. [Google Scholar] [CrossRef]
- Krukowski, R.A.; Pope, R.A.; Love, S.; Lensing, S.; Felix, H.C.; Prewitt, T.E.; West, D. Examination of costs for a lay health educator-delivered translation of the Diabetes Prevention Program in senior centers. Prev. Med. 2013, 57, 400–402. [Google Scholar] [CrossRef]
- Adair, R.; Wholey, D.R.; Christianson, J.; White, K.M.; Britt, H.; Lee, S. Improving chronic disease care by adding laypersons to the primary care team: A parallel randomized trial. Ann. Intern. Med. 2013, 159, 176–184. [Google Scholar] [CrossRef]
- Geraedts, H.A.; Zijlstra, W.; Zhang, W.; Bulstra, S.; Stevens, M. Adherence to and effectiveness of an individually tailored home-based exercise program for frail older adults, driven by mobility monitoring: Design of a prospective cohort study. BMC Public Health 2014, 14, 570. [Google Scholar] [CrossRef] [Green Version]
- Aartolahti, E.; Tolppanen, A.M.; Lonnroos, E.; Hartikainen, S.; Hakkinen, A. Health condition and physical function as predictors of adherence in long-term strength and balance training among community-dwelling older adults. Arch. Gerontol. Geriatr. 2015, 61, 452–457. [Google Scholar] [CrossRef]
- Niemela, K.; Vaananen, I.; Leinonen, R.; Laukkanen, P. Benefits of home-based rocking-chair exercise for physical performance in community-dwelling elderly women: A randomized controlled trial. Aging Clin. Exp. Res. 2011, 23, 279–287. [Google Scholar] [CrossRef]
- Sjosten, N.M.; Salonoja, M.; Piirtola, M.; Vahlberg, T.J.; Isoaho, R.; Hyttinen, H.K.; Aarnio, P.T.; Kivela, S.L. A multifactorial fall prevention programme in the community-dwelling aged: Predictors of adherence. Eur. J. Public Health 2007, 17, 464–470. [Google Scholar] [CrossRef] [Green Version]
- Van Het Reve, E.; Silveira, P.; Daniel, F.; Casati, F.; de Bruin, E.D. Tablet-based strength-balance training to motivate and improve adherence to exercise in independently living older people: Part 2 of a phase II preclinical exploratory trial. J. Med. Internet Res. 2014, 16, e159. [Google Scholar] [CrossRef] [Green Version]
- Furlan, A.D.; Pennick, V.; Bombardier, C.; van Tulder, M. 2009 updated method guidelines for systematic reviews in the Cochrane Back Review Group. Spine 2009, 34, 1929–1941. [Google Scholar] [CrossRef]
- Spink, M.J.; Menz, H.B.; Fotoohabadi, M.R.; Wee, E.; Landorf, K.B.; Hill, K.D.; Lord, S.R. Effectiveness of a multifaceted podiatry intervention to prevent falls in community dwelling older people with disabling foot pain: Randomised controlled trial. BMJ 2011, 342, d3411. [Google Scholar] [CrossRef] [Green Version]
- Barker, A.L.; Talevski, J.; Bohensky, M.A.; Brand, C.A.; Cameron, P.A.; Morello, R.T. Feasibility of Pilates exercise to decrease falls risk: A pilot randomized controlled trial in community-dwelling older people. Clin. Rehabil. 2015, 30, 984–996. [Google Scholar] [CrossRef]
- Gill, T.M.; Baker, D.I.; Gottschalk, M.; Gahbauer, E.A.; Charpentier, P.A.; de Regt, P.T.; Wallace, S.J. A prehabilitation program for physically frail community-living older persons. Arch. Phys. Med. Rehabil. 2003, 84, 394–404. [Google Scholar] [CrossRef]
- Craig, P.; Dieppe, P.; Macintyre, S.; Michie, S.; Nazareth, I.; Petticrew, M. Developing and evaluating complex interventions: The new Medical Research Council guidance. Int. J. Nurs. Stud. 2013, 50, 587–592. [Google Scholar] [CrossRef] [Green Version]
- Rychetnik, L.; Frommer, M.; Hawe, P.; Shiell, A. Criteria for evaluating evidence on public health interventions. J. Epidemiol. Community Health 2002, 56, 119–127. [Google Scholar] [CrossRef]
- Oakley, A.; Strange, V.; Bonell, C.; Allen, E.; Stephenson, J. Process evaluation in randomised controlled trials of complex interventions. BMJ 2006, 332, 413–416. [Google Scholar] [CrossRef] [Green Version]
- Moore, G.F.; Audrey, S.; Barker, M.; Bond, L.; Bonell, C.; Hardeman, W.; Moore, L.; O’Cathain, A.; Tinati, T.; Wight, D.; et al. Process evaluation of complex interventions: Medical Research Council guidance. BMJ 2015, 350, h1258. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lackinger, C.; Wilfinger, J.; Mayerhofer, J.; Strehn, A.; Dick, D.; Dorner, T.E. Adherence to and effects on physical function parameters of a community-based standardised exercise programme for overweight or obese patients carried out by local sports clubs. Public Health 2017, 147, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Dorner, T.E.; Lackinger, C.; Haider, S.; Luger, E.; Kapan, A.; Luger, M.; Schindler, K.E. Nutritional intervention and physical training in malnourished frail community-dwelling elderly persons carried out by trained lay “buddies”: Study protocol of a randomized controlled trial. BMC Public Health 2013, 13, 1232. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haider, S.; Luger, E.; Kapan, A.; Schindler, K.; Lackinger, C.; Dorner, T.E. A home-based strength training and nutritional program effectively improves handgrip strength and physical performance in prefrail and frail older adults, even when carried out by lay volunteers. In Proceedings of the International Conference on Frailty Sarcopenia Research, Philadelphia, PA, USA, 28 April 2016. [Google Scholar]
- Kapan, A.; Winzer, E.; Haider, S.; Titze, S.; Schindler, K.; Lackinger, C.; Dorner, T.E. Impact of a lay-led home-based intervention programme on quality of life in community-dwelling pre-frail and frail older adults: A randomized controlled trial. BMC Geriatr. 2017, 17, 154. [Google Scholar] [CrossRef] [Green Version]
- Luger, E.; Dorner, T.E.; Haider, S.; Kapan, A.; Lackinger, C.; Schindler, K. Effects of a Home-Based and Volunteer-Administered Physical Training, Nutritional, and Social Support Program on Malnutrition and Frailty in Older Persons: A Randomized Controlled Trial. J. Am. Med. Dir. Assoc. 2016, 17, 671.e9–671.e16. [Google Scholar] [CrossRef]
- Romero-Ortuno, R.; Walsh, C.D.; Lawlor, B.A.; Kenny, R.A. A frailty instrument for primary care: Findings from the Survey of Health, Ageing and Retirement in Europe (SHARE). BMC Geriatr. 2010, 10, 57. [Google Scholar] [CrossRef] [Green Version]
- Kondrup, J.; Allison, S.P.; Elia, M.; Vellas, B.; Plauth, M. ESPEN guidelines for nutrition screening 2002. Clin. Nutr. 2003, 22, 415–421. [Google Scholar] [CrossRef]
- Grabovac, I.; Haider, S.; Winzer, E.; Kapan, A.; Schindler, K.E.; Lackinger, C.; Dorner, T.E. Changes in health parameters in older lay volunteers who delivered a lifestyle-based program to frail older people at home. Wien. Klin. Wochenschr. 2018, 130, 637–644. [Google Scholar] [CrossRef] [Green Version]
- University of Harvard Healthy Eating Plate. Available online: www.hsph.harvard.edu/nutritionsource/healthy-eating-plate/ (accessed on 10 September 2020).
- Cyarto, E.V.; Brown, W.J.; Marshall, A.L. Retention, adherence and compliance: Important considerations for home- and group-based resistance training programs for older adults. J. Sci. Med. Sport 2006, 9, 402–412. [Google Scholar] [CrossRef]
- Dale, O.; Salo, M. The Helsinki Declaration, research guidelines and regulations: Present and future editorial aspects. Acta Anaesthesiol. Scand. 1996, 40, 771–772. [Google Scholar] [CrossRef]
- Titze, S.; Lackinger, C.; Fessl, C.; Dorner, T.E.; Zeuschner, V. Austrian Physical Activity Guidelines for Adults and Older Adults with and without Physical, Sensory, or Mental Disabilities, as well as for Adults with Chronic Diseases. Gesundh. Bundesverb. Arzte Offentl. Gesundh. Ger. 2020, 82, S170–S176. [Google Scholar]
- Pate, R.R.; Pratt, M.; Blair, S.N.; Haskell, W.L.; Macera, C.A.; Bouchard, C.; Buchner, D.; Ettinger, W.; Heath, G.W.; King, A.C.; et al. Physical activity and public health. A recommendation from the Centers for Disease Control and Prevention and the American College of Sports Medicine. JAMA 1995, 273, 402–407. [Google Scholar] [CrossRef]
- De Vreede, P.L.; Samson, M.M.; van Meeteren, N.L.; van der Bom, J.G.; Duursma, S.A.; Verhaar, H.J. Functional tasks exercise versus resistance exercise to improve daily function in older women: A feasibility study. Arch. Phys. Med. Rehabil. 2004, 85, 1952–1961. [Google Scholar] [CrossRef]
- Stineman, M.G.; Strumpf, N.; Kurichi, J.E.; Charles, J.; Grisso, J.A.; Jayadevappa, R. Attempts to reach the oldest and frailest: Recruitment, adherence, and retention of urban elderly persons to a falls reduction exercise program. Gerontologist 2011, 51 (Suppl. 1), S59–S72. [Google Scholar] [CrossRef]
- Vestergaard, S.K.C.; Puggaard, L. Home-based video exercise intervention for community-dwelling frail older women: A randomized controlled trial. Aging Clin. Exp. Res. 2008, 20, 479–486. [Google Scholar] [CrossRef]
- Faber, M.; Andersen, M.H.; Sevel, C.; Thorborg, K.; Bandholm, T.; Rathleff, M. The majority are not performing home-exercises correctly two weeks after their initial instruction-an assessor-blinded study. PeerJ 2015, 3, e1102. [Google Scholar] [CrossRef]
- Vincent, K.R.; Braith, R.W.; Feldman, R.A.; Magyari, P.M.; Cutler, R.B.; Persin, S.A.; Lennon, S.L.; Gabr, A.H.; Lowenthal, D.T. Resistance exercise and physical performance in adults aged 60 to 83. J. Am. Geriatr. Soc. 2002, 50, 1100–1107. [Google Scholar] [CrossRef] [Green Version]
- Bauer, R.; Dorner, T.E.; Felder-Puig, R.; Fessl, C.; Gollner, E.; Halbwachs, C.; Kayer, B.; Lackinger, C.; Lechner, N.; Lercher, P.; et al. Österreichische Bewegungsempfehlungen; Fonds Gesundes Österreich: Vienna, Austria, 2020; Volume 17. [Google Scholar]
- Haider, S.; Dorner, T.E.; Luger, E.; Kapan, A.; Titze, S.; Lackinger, C.; Schindler, K.E. Impact of a Home-Based Physical and Nutritional Intervention Program Conducted by Lay-Volunteers on Handgrip Strength in Prefrail and Frail Older Adults: A Randomized Control Trial. PLoS ONE 2017, 12, e0169613. [Google Scholar] [CrossRef]
- Haider, S.; Grabovac, I.; Winzer, E.; Kapan, A.; Schindler, K.E.; Lackinger, C.; Titze, S.; Dorner, T.E. Change in inflammatory parameters in prefrail and frail persons obtaining physical training and nutritional support provided by lay volunteers: A randomized controlled trial. PLoS ONE 2017, 12, e0185879. [Google Scholar] [CrossRef] [Green Version]
- Titze, S.; Lackinger, C.; Grossschaedl, L.; Strehn, A.; Dorner, T.E.; Niebauer, J.; Schebesch-Ruf, W. How Does Counselling in a Stationary Health Care Setting Affect the Attendance in a Standardised Sports Club Programme? Process Evaluation of a Quasi-Experimental Study. Int. J. Environ. Res. Public Health 2018, 15, 134. [Google Scholar] [CrossRef] [Green Version]
- Titze, S.; Schebesch-Ruf, W.; Lackinger, C.; Großschädl, L.; Strehn, A.; Dorner, T.E.; Niebauer, J. Short-and Long-Term Effectiveness of a Physical Activity Intervention with Coordinated Action between the Health Care Sector and Local Sports Clubs. A Pragmatic Trial in Austrian Adults. Int. J. Environ. Res. Public Health 2019, 16, 2362. [Google Scholar] [CrossRef] [Green Version]
- Haider, S.; Luger, E.; Kapan, A.; Titze, S.; Lackinger, C.; Schindler, K.E.; Dorner, T.E. Associations between daily physical activity, handgrip strength, muscle mass, physical performance and quality of life in prefrail and frail community-dwelling older adults. Qual. Life Res. 2016, 25, 3129–3138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Prefrail/Frail Participants (n = 80) | Buddies (n = 70) | |
|---|---|---|
| Age (years) | 82.6 ± 8.1 | 60.1 ± 6.9 |
| Sex (Female, %) | 83.8 | 89.1 |
| Body height (cm) | 162.1 ± 8.6 | 167.1 ± 6.9 |
| Body weight (kg) | 71.4 ± 12.7 | 71.3 ± 14.9 |
| Body mass index (kg/m2) | 27.4 ± 4.4 | 25.6 ± 5.4 |
| Waist circumference (cm) | 103.5 ± 11.5 | 93.3 ± 14.8 |
| Hand grip strength (kg) | 16.9 ± 7.3 | 32.1 ± 7.9 |
| Share-FI categories (%) * | ||
| Robust | 1.3 | |
| Prefrail | 35.0 | |
| Frail | 63.7 | |
| Educational level (%) | ||
| Primary | 52.5 | 21.4 |
| Secondary | 35.0 | 54.3 |
| Tertiary | 12.5 | 18.6 |
| Missing | 05.7 | |
| Living with a partner (%) | 17.5 | 55.9 |
| PTN | SoSu | |||
|---|---|---|---|---|
| Weeks 1–12 | Weeks 13–24 | Weeks 1–12 | Weeks 13–24 | |
| Number of weeks between follow-ups | 10.5 ± 2.8 | 9.9 ± 2.3 1 | 9.4 ± 2.6 | 10.8 ± 2.1 3 |
| Number of visits per week | 1.6 ± 0.4 | 1.6 ± 0.7 2 | 1.5 ± 0.5 | 1.5 ± 0.5 4 |
| Adherence rate | 84.0% | 80.5% | 75.5% | 72.5% |
| Adherence category 5,6 | ||||
| High adherent (%) | 61.9 | 59.5 | 47.4 | 36.8 |
| Moderately adherent (%) | 26.2 | 14.3 | 21.1 | 15.8 |
| Low adherent (%) | 2.4 | 7.1 | 5.3 | 7.9 |
| Very low adherent (%) | 0 | 2.4 | 5.3 | 2.6 |
| Drop outs (% of the whole sample) | 11.9 | 16.6 | 23.7 | 36.8 |
| Planned Intervention | Actual Intervention | Dose (%) | Actual Intervention | Dose (%) | p * | |
|---|---|---|---|---|---|---|
| after 12 weeks * | after 24 weeks ** | |||||
| Home visit per week (times) | 2 | 1.6 ± 0.5 | 80 | 1.6 ± 0.6 | 77 | 0.562 |
| Duration of home visit (hours) | 1 | 1.4 ± 0.5 | 140 | 1.5 ± 0.8 | 154 | 0.259 |
| Intervention missed due to prefrail/frail person | 0 | 4.4 ± 4.5 | 4.5 ± 4.9 | 0.880 | ||
| Intervention missed due to buddy | 0 | 2.2 ± 3.1 | 2.0 ± 2.3 | 0.628 | ||
| Number of physical training intervention per home visit | 1 | 0.4 ± 0.5 | 41 | 0.8 ± 0.6 | 81 | <0.001 |
| Number of circles per home visit | 2 | 2.1 ± 0.7 | 107 | 1.2 ± 0.6 | 60 | 0.006 |
| Number of conducted strength exercises per home visit | 6 | 6.4 ± 5.5 | 106 | 5.3 ± 1.3 | 89 | 0.016 |
| Number of repetitions per exercise | 15 | 7.3 ± 6.5 | 48 | 13.1 ± 4.6 | 87 | <0.001 |
| Number of nutritional interventions per home visit | 1 | 0.6 ± 0.7 | 56 | 0.5 ± 0.3 | 47 | 0.168 |
| Number of “Healthy for life plate” interventions per home visit | 1 | 5.2 ± 6.7 | 521 | 0.2 ± 0.3 | 22 | |
| Number of nutritional messages delivered per home visit | 1 | 5.1 ± 7.6 | 507 | 0.3 ± 0.4 | 26 | |
| High Adherent | Moderate, Low and Very Low Adherent | p | ||
|---|---|---|---|---|
| Age | Younger than 81 | 9 (23.1%) | 12 (60.0%) | 0.006 |
| Older than 81 | 30 (76.9%) | 8 (40.0%) | ||
| Sex | Female | 35 (89.7%) | 16 (80.0%) | 0.258 |
| Male | 4 (10.3%) | 4 (20.0%) | ||
| Education level | Primary | 24 (61.5%) | 8 (40.0%) | 0.124 |
| Secondary | 13 (33.3%) | 8 (40.0%) | ||
| Tertiary | 2 (5.1%) | 4 (20.0%) | ||
| SHARE-FI | Robust | 2 (5.1%) | 4 (20.0%) | 0.038 |
| Prefrail | 22 (56.4%) | 5 (25.0%) | ||
| Frail | 15 (38.5%) | 11 (55.0%) |
| High Adherent | Moderate, Low and Very Low Adherent | p | ||
|---|---|---|---|---|
| Age | Younger than 59 | 15 (60.0%) | 6 (40.0%) | 0.220 |
| Older than 59 | 10 (40.0%) | 9 (60.0%) | ||
| Sex | Female | 27 (93.1%) | 13 (86.7%) | 0.481 |
| Male | 2 (6.9%) | 2 (13.3%) | ||
| Educational level | Primary | 7 (25.0%) | 4 (28.6%) | 0.853 |
| Secondary | 15 (53.6%) | 8 (57.1%) | ||
| Tertiary | 6 (21.4%) | 2 (14.3%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lackinger, C.; Grabovac, I.; Haider, S.; Kapan, A.; Winzer, E.; Stein, K.V.; Dorner, T.E. Adherence Is More Than Just Being Present: Example of a Lay-Led Home-Based Programme with Physical Exercise, Nutritional Improvement and Social Support, in Prefrail and Frail Community-Dwelling Older Adults. Int. J. Environ. Res. Public Health 2021, 18, 4192. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph18084192
Lackinger C, Grabovac I, Haider S, Kapan A, Winzer E, Stein KV, Dorner TE. Adherence Is More Than Just Being Present: Example of a Lay-Led Home-Based Programme with Physical Exercise, Nutritional Improvement and Social Support, in Prefrail and Frail Community-Dwelling Older Adults. International Journal of Environmental Research and Public Health. 2021; 18(8):4192. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph18084192
Chicago/Turabian StyleLackinger, Christian, Igor Grabovac, Sandra Haider, Ali Kapan, Eva Winzer, K. Viktoria Stein, and Thomas E. Dorner. 2021. "Adherence Is More Than Just Being Present: Example of a Lay-Led Home-Based Programme with Physical Exercise, Nutritional Improvement and Social Support, in Prefrail and Frail Community-Dwelling Older Adults" International Journal of Environmental Research and Public Health 18, no. 8: 4192. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph18084192








