Overview of Dose Assessment Developments and the Health of Riverside Residents Close to the “Mayak” PA Facilities, Russia
Abstract
:1. Introduction
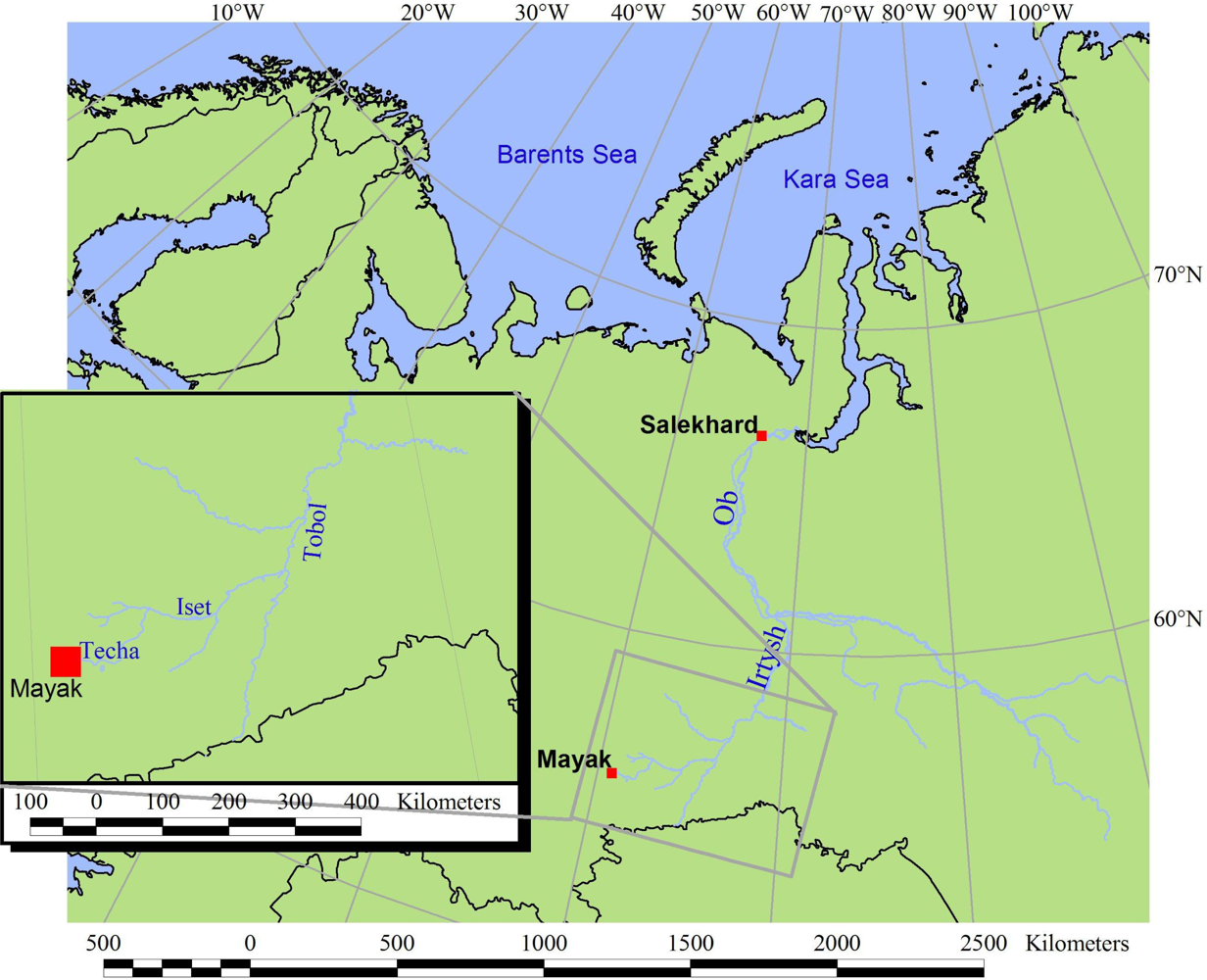
1.1. The Mayak Facilities
1.2. Contamination History
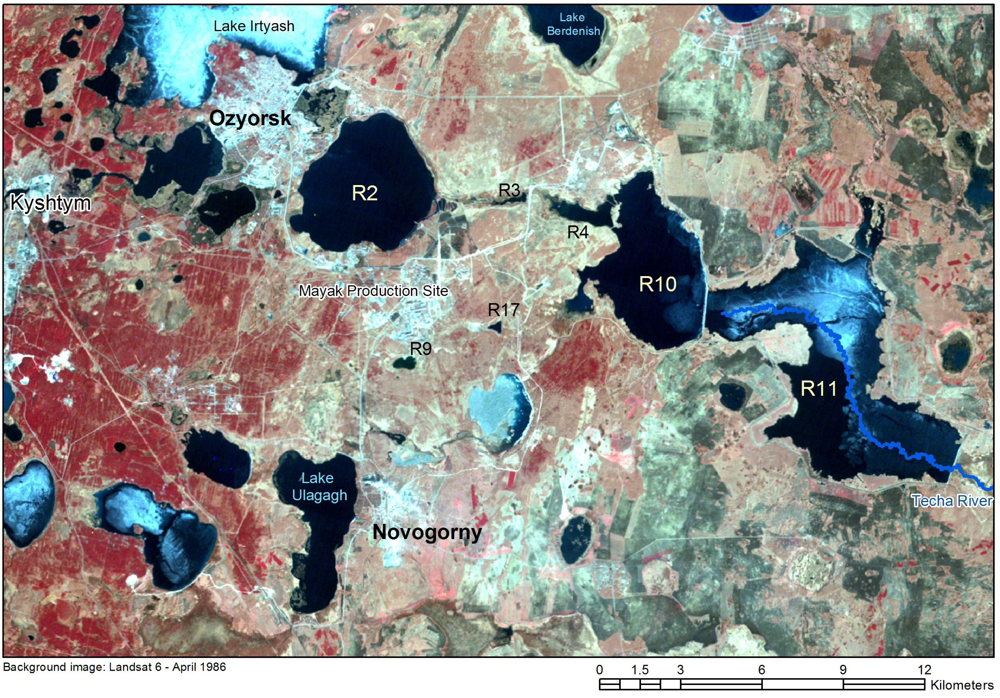
- Direct releases of radionuclides to the Techa (Reservoir 3) between 1949 and 1956.
- The Kyshtym accident – a thermal explosion in a high-level radioactive waste tank in 1957.
- Dispersal of radionuclides from the dried-out bed of Lake Karachay in 1967.
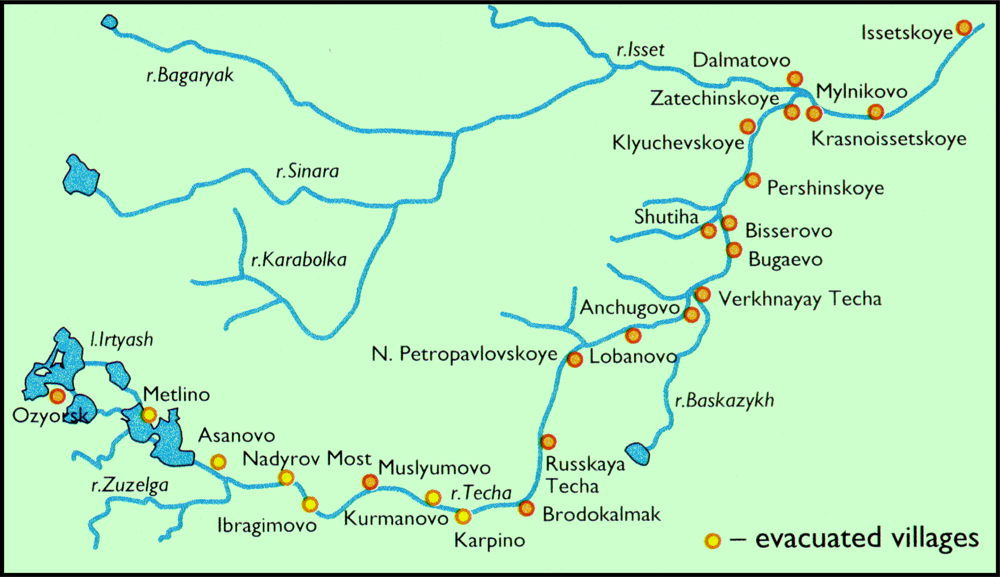
1.3. Radiation Affected Populations
- The workforce at the Mayak PA
- Participants in clean-up work carried out after the accidental releases in the Urals
- Local residents around the Mayak PA who were exposed due to environmental discharges of radioactivity and/or radiation accidents
1.4. Contamination Levels in Groundwater, Reservoir Water and River Water
1.5. Contamination Levels in Soils
1.6. Contamination Levels in Biota and Food Products
2. Dose Assessment
2.1. Dose Reconstruction
2.2. Internal and External Doses
2.3. Internal and External Doses Received from 1950 Onwards
2.4. Current Doses
- Group 1. People, who do not visit the river floodplain, do not consume milk from cows pastured on the floodplain or fish from the river Techa. This population group’s exposure doses are therefore not connected with contamination of the floodplain.
- Group 2. People, who visit the river floodplain in accordance with average occupancy factor values (0.03), consume 10% of their average annual milk consumption from cows pastured on the floodplain and 10% of their average annual consumption of fish from the river Techa. This population group receives doses that correspond to the average weighted dose in the settlement.
- Group 3. People, who visit the river floodplain with occupancy factor values for herdsmen (0.10), consume 100% of their milk from cows pastured on the river floodplain and 30% of their average annual consumption of fish from the river Techa. This can be defined as the critical population group.
3. Health and well-being of riverside residents
3.1. Potential Health Effects
3.2. Observed Health Effects in the Techa River Area
4. Conclusions
References
- Malyshev, SV; Vakulovsky, SM; Drozhko, EG; Romanov, GN; Glagolenko, YV; Mokrov, YuG; Westerlund, EA; Amundsen, I; Strand, P; Salbu, B; Oughton, DH; Christensen, GC; Bergan, TS. in Joint Norwegian-Russian Expert Group (JNREG) Sources contributing to radioactive contamination of the Techa River and the area surrounding the “Mayak” production association, Urals, RussiaProgramme of the Joint Norwegian-Russian Investigations of possible impacts of the Mayak PA activities on radioactive contamination of the Barents and Kara Seas; Norwegian Radiation Protection Authority: Østerås, Norway, 1997. [Google Scholar]
- Christensen, GC; Romanov, GN; Strand, P; Salbu, B; Malyshev, SV; Bergan, TD; Oughton, D; Drozkho, EG; Glagolenko, YV; Amundsen, I; Rudjord, AL; Bjerk, TO; Lind, B. Radioactive contamination in the environment of the nuclear enterprise ‘Mayak’ PA. Results from the joint Russian-Norwegian field work in 1994. Sci. Tot. Env 1997, 202, 237–248. [Google Scholar]
- Trapeznikov, AV; Pozolotina, VN; Chebotina, MYa; Chukanov, VN; Trapeznikova, VN; Kulikov, NV; Nielsen, SP; Aarkrog, A. Radioactive contamination of the Techa River, The Urals. Health Phys 1993, 65, 481–488. [Google Scholar]
- Cochran, TB; Standish Norris, R; Suokko, KL. Radioactive contamination at Chelyabinsk-65, Russia. Annu. Rev. Energy Environ 1993, 18, 507–528. [Google Scholar]
- Bradley, DJ; Jenquin, UP. Radioactive inventories and sources for contamination of the Kara Sea by fluvial transport. . Office of Naval Research Report, Contract N00014-94-1-0054. Pacific Northwest Laboratory: Richmond, Washington, U.S., 1995. [Google Scholar]
- Malyshev, SV; Westerlund, EA; Amundesen, I; Christensen, GC; Drozhko, EG; Salbu, B; Oughton, DH; Mokrov, YuG; Strand, P; Bergan, TDS; Romanov, GN. Waste handling, releases and accidents at “Mayak” PA - A description of sources contaminating the Techa River. In Third International Symposium on Environmental Radioactivity in the Arctic, Tromsø; Norwegian Radiation Protection Authority: Østerås, Norway, 1997; pp. 16–17. [Google Scholar]
- Karavaeva, YeN; Kulikov, NV; Molchanova, IV; Pozolotina, VN; Yushkov, PI. Accumulation and distribution of long-lived radionuclides in forest ecosystems of the Kyshtym accident zone. Sci. Tot. Env 1994, 157, 147–151. [Google Scholar]
- Kryshev, II; Romanov, GN; Isaeva, LN; Kholina, YuB. Radioecological state of lakes in the southern Ural impacted by radioactive release of the 1957 radiation accident. J. Envir. Rad 1997, 34, 223–235. [Google Scholar]
- Myasoedov, BF; Drozhko, EG. Up-to-date radioecological situation around the ‘Mayak’ nuclear facility. J. All. Com 1998. [Google Scholar]
- Aarkrog, A; Dahlgaard, H; Nielsen, SP; Trapeznikov, AV; Molchanova, IV; Pozolotina, VN; Karavaeva, EN; Yushkov, PI; Polikarpov, GG. Radioactive inventories from the Kyshtym and Karachay accidents: estimates based on soil samples collected in the South Urals (1990–1995). Sci. Tot. Environ 1997, 201, 137–154. [Google Scholar]
- Gerchikov, MYu; Mathers, D; North, M; Glagolenko, Y; Mokrov, YG; Rovny, S; Samsonova, LM. Current situation concerning radioactive waste management and restoration of contaminated territories in and around the Mayak PA site. In European Commission (EC) Nuclear Safety and the Environment Final Report: Serie EUR 18190EN; Office of Official Publications of the European Communities: Luxembourg, 2000. [Google Scholar]
- Mokrov, YuG; Glagolenko, YuV; Drozhko, G; Rovny, SI. The Techa Reservoir Cascade: Safety and Regulation Problems. In NATO Science for Peace and Security Series – C: Environmental Security. Challenges in Radiation Protection and Nuclear Safety Regulation of the Nuclear Legacy; Sneve, MK, Kiselev, MF, Eds.; Springer: Netherlands, 2008; pp. 163–174. [Google Scholar]
- Romanov, VV. Assessment of “Mayak” Environmental Impact, Presentation at Joint Norwegian-Russian Expert Group meeting, Oslo, Norway; November 2006.
- Bradley, DJ. Behind the nuclear curtain: Radioactive waste management in the Former Soviet Union; Payson, DR, Ed.; Battelle Memorial Institute: USA, 1997. [Google Scholar]
- Akleyev, AV; Lyubchansky, EE. Environmental and medical effects of nuclear weapon production in the southern Urals. Sci. Tot. Envir 1994, 142, 1–8. [Google Scholar]
- Akleyev, AV; Kostyuchenko, VA; Peremyslova, LM; Baturin, VA; Popova, IYa. Radioecological impacts of the Techa River contamination. Health Phys 2000, 79, 36–47. [Google Scholar]
- Shutov, VN; Travnikova, IG; Bruk, GYa; Golikov, VY; Balonov, MI; Howard, BJ; Brown, J; Strand, P; Kravtsova, EM; Gavrilov, AP; Kravtsova, OS; Mubasarov, AA. Current contamination by 137Cs and 90Sr of the inhabited part of the Techa river basin in the Urals. J. Environ. Rad 2002, 61, 91–109. [Google Scholar]
- Kryshev, II; Romanov, GN; Chumichev, VB; Sazykina, TG; Isaeva, LN; Ivanitskaya, MV. Radiological consequences of radioactive discharges into the Techa river on the southern Urals. J. Environ. Rad 1998, 38, 195–209. [Google Scholar]
- Cabianca, T; Bexon, AP; Pozolotina, V; Trapeznikov, A; Simmonds, J. Preliminary assessment of current radiation doses to the population of Brodokalmak from contamination of the Techa River. J. Environ. Rad 2000, 50, 193–206. [Google Scholar]
- Chesnokov, AV; Govorun, AP; Linnik, VG; Shcherbak, SB. 137Cs contamination of the Techa river flood plain near the village of Muslumovo. J. Environ. Rad 2000, 50, 179–191. [Google Scholar]
- Chesnokov, AV; Govorun, AP; Ivanitskaya, MV; Liksonov, VI; Shcherbak, SB. 137Cs contamination of Techa river flood plain in Brodokalmak settlement. App. Rad. Isotop 1999, 50, 1121–1129. [Google Scholar]
- Kryshev, II; Romanov, GN; Isaeva, LN; Kholina, YuB. Radioecological state of lakes in the southern Ural impacted by radioactive release of the 1957 radiation accident. J. Environ. Rad 1997, 34, 223–235. [Google Scholar]
- Radiation hygiene assessment of residing conditions of inhabitants in settlements by the river Techa within the borders of the Chelyabinsk region; Kravtsova, EM (Ed.) Centre for State Sanitary and Epidemiological Inspection in the Chelyabinsk region: Chelyabinsk, Russian, 1998.
- Strand, P; Howard, B; Balanov, M; Prister, B; Ratnikov, A; Shilov, A. RECLAIM Final report: Time dependent optimisation of strategies for counter measures use to reduce population radiation dose and reclaim abandoned land; Mehli, H, Ed.; EU contract ERBIC15CT960209Norwegian Radiation Protection Authority: Østerås, Norway, 2000. [Google Scholar]
- Romanov, GN. JNRI Final Report. The investigation of possible impact of the activity of PA “Mayak” on the radioactive contamination of the Barents and Kara Seas: Exposure population doses on the Techa river and possible consequences of the exposure for health of the population. Norwegian Radiation Protection Authority: Østerås, Norway, 1998. [Google Scholar]
- Kossenko, MM; Degteva, MO; Vyushkova, OV; Preston, DL; Mabuchi, K; Kozheurov, VP. Issues in the comparison of risk estimates for the population in the Techa River Region and atomic bomb survivors. Radiat. Res 1997, 148, 54–63. [Google Scholar]
- Kossenko, MM; Preston, DL; Krestinina, LY; Degteva, MO; Startsev, NV; Thomas, T; Vyushkova, OV; Anspaugh, LR; Napier, BA; Kozheurov, VP; Ron, E; Akleyev, AV. Studies on the extended Techa river cohort: cancer risk estimation. Radiat. Environ. Biopyhs 2002, 41, 45–48. [Google Scholar]
- Kossenko, MM; Degteva, MO. Cancer mortality and radiation risk evaluation for the Techa River population. Sci. Tot. Environ 1994, 142, 73–89. [Google Scholar]
- Degteva, MO; Kozheurov, VP; Vorobiova, MI. General approach to dose reconstruction in the population exposed as a result of the release of radioactive wastes into the Techa River. Sci. Tot. Environ 1994, 14, 49–61. [Google Scholar]
- Degteva, MO; Vorobiova, MI; Kozheurov, VP; Tolstykh, EI; Anspaugh, LR; Napier, BA. Dose reconstruction system for the exposed population living along the Techa River. Health Phys 2000, 78, 542–554. [Google Scholar]
- Vorobiova, MI; Degteva, MO; Kozyrev, AV; Anspaugh, LR; Napier, BA. Review of historical monitoring data on Techa river contamination. Health Phys 1999, 76, 605–618. [Google Scholar]
- Mokrov, YuG. Reconstruction of the radionuclide spectrum of liquid radioactive waste released into the Techa River in 1949–1951. Radiat. Environ. Biophys 2003, 42, 7–15. [Google Scholar]
- Mokrov, YuG. Radioactive contamination of bottom sediments in the upper reaches of the Techa River: analysis of the data obtained in 1950 and 1951. Radiat. Environ. Biophys 2003, 42, 155–168. [Google Scholar]
- Balonov, M; Alexakhin, R; Bouville, A; Liljinzin, J-O. Report from the Techa River Dosimetry Review Workshop held on 8–10 December 2003 at the State Research Centre Institute of Biophysics, Moscow, Russia. Health Phys 2006, 90, 97–113. [Google Scholar]
- Degteva, MO; Kozheurov, VP; Tolstykh, EI; Vorobiova, MI; Anspaugh, LR; Napier, BA; Kovtun, AN. The Techa River Dosimetry System: Methods for the reconstruction of internal dose. Health Phys 2000, 79, 24–35. [Google Scholar]
- International Commission on Radiological Protection (ICRP) Publication 67: Age-dependent doses to members of the public from intake of radionuclides: Part 2 Ingestion dose coefficients; ICRP: Stockholm, Sweden, 1994.
- Kozheurov, VP; Degteva, M. Dietary intake evaluation and dosimetric modelling for the Techa River residents based on in vivo measurements of strontium-90 in teeth and skeleton. Sci. Tot. Environ 1994, 142, 63–72. [Google Scholar]
- Kossenko, MM; Hoffman, DA; Thomas, TL. Stochastic effects of environmental radiation exposure in populations living near the Mayak Industrial Association: preliminary report on cancer morbidity. Health Phys 2000, 79, 55–62. [Google Scholar]
- Degteva, MO; Anspaugh, LR; Napier, BA; Vorobiova, MI; Tolstykh, EI; Kozheurov, VP; Kozyrev, AV; Bougrov, NG; Kovtun, AN; Shagina, NB; Shishkina, EA; Tokareva, EE; Taranenko, VA. Development of an improved dose reconstruction system for the general population affected by the operation of the Mayak Production association. In Final report: US-Russian Joint Coordinating Committee on Radiation Effects Research, Project 1.1.; Urals Research Center for Radiation Medicine: Chelyabinsk, Russia, 2000. [Google Scholar]
- Aarkrog, A; Simmonds, J; Strand, P; Christensen, G; Salbu, B. Radiological assessment of past, present and potential sources to environmental contamination in the Southern Urals and strategies for remedial measures (SUCON Final Report). Risø National Laboratory: Roskilde, Denmark, 2000. [Google Scholar]
- Balonov, MI; Bruk, GY; Golikov, VY; Barkovsky, AN; Kravtsova, EM; Kravtosova, OS; Mubasarov, AA; Shutov, VN; Travnikova, IG; Howard, BJ; Brown, JE; Strand, P. Assessment of current exposure of the population living in the Techa River basin from radioactive releases of the Mayak facility. Health Phys 2007, 92, 134–147. [Google Scholar]
- International Commission on Radiological Protection (ICRP) Publication 60: Recommendations of the International Commission on Radiological Protection. Annals of the ICRP 20/1, 1991.
- Hall, EJ. Radiobiology for the radiologist, Fourth Edition edJ.B. Lippincott Company: USA, 1994. [Google Scholar]
- Testa, A; Padovani, L; Mauro, F; Appolloni, M; Anzidei, P; Stronati, L. Cytogenic study on children living in southern Urals contaminated areas (nuclear incidents 1948 – 1967). Mutat. Res 1998, 401, 193–197. [Google Scholar]
- United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR), UNSCEAR 2006 Report Vol. 1 Effects of Ionizing Radiation: Report to the General Assembly, Scientific Annexes A and B. United Nations; UNSCEAR: New York, USA, 2008.
- Thompson, DE; Mabuchi, K; Ron, E; Soda, M; Tokunaga, M; Ochikubo, S; Sugimoto, S; Ikeda, T; Terasaki, M; Izumi, S. Cancer incidence in atomic bomb survivors. Part II: Solid tumors, 1958–1987. Rad. Res 1994, 137, 7–67. [Google Scholar]
- Kossenko, MM; Nikolayenko, LA; Yepidfanova, SB; Ostroumova, YV. Chronic radiation sickness among Techa riverside residents; Armed Forces Radiobiology Research Institute: Bethesda, Maryland, USA, 1998. [Google Scholar]
- Buldakov, LA. Medical consequences of radiation accidents. In: Proceedings of a Symposium, Environmental Impact of Radioactive Releases; IAEA: Vienna, Austria, 1995; pp. 467–478. [Google Scholar]
- Kossenko, MM; Degteva, MO; Petrushova, NA. Estimate of the risk of leukemia to residents exposed to radiation as a result of a nuclear accident in the Southern Urals. Physician. Soc. Responsibility 1992, 187–197. [Google Scholar]
- Kossenko, MM; Akleyev, AV; Startsev, NV; Degteva, MO. Epidemiological analysis of remote cancerogenesis effects on populations with chronic exposure to radiation in the Urals region. Int. J. Rad. Med 1999, 2, 34–41. [Google Scholar]
- Krestinina, LYu; Preston, DL; Ostroumova, EV; Degtava, MO; Ron, E; Vyushkova, OV; Startsev, NV; Kossenko, MM; Akleyev, AV. Protracted radiation exposure and cancer mortality in the Techa River Cohort. Radiat. Res 2005, 164, 602–611. [Google Scholar]
- Ilyinskikh, NN; Ilyinskikh, IN; Shakirov, NN; Smirnov, BV; Ilyinskikh, EN. Monitoring of radiation-exposed people close to Mayak nuclear facility in the Chelyabinsk region (Russia) using different biodosimetric methods. Environ. Monitor. Ass 2000, 61, 345–359. [Google Scholar]
- Kossenko, MM. Cancer mortality among Techa River residents and their offspring. Health Phys 1996, 71, 77–82. [Google Scholar]
| Reservoir | Area (km2) | Volume (m3 ×106) | Estimated inventory (TBq) | Percentage total activity in sediments (%) |
|---|---|---|---|---|
| 3 | 0.5–0.8 | 0.78 | 666 | 85 |
| 4 | 1.3 | 4.1–4.3 | 222 | 70 |
| 10 | 18–19 | 76.64 | 4070 (1200) | 5 (25) |
| 11 | 44 | 215.74 | 1443 (1300) | 40 (60) |
| Product | Cabianca et al., [19] | RECLAIM [24] | Romanov [25] | ||
|---|---|---|---|---|---|
| Brodokalmak | Brodokalmak | Muslyumovo | Brodokalmak | Muslyumovo | |
| Milk | 3.6 (1–5.6) | 0.8 (0.04–5.7)a | 1.7 (0.04–37)a | 0.82 (0.4–1.2)c | 0.60 (0.37–0.74)b |
| 0.80 (0.7–1.0)c | |||||
| Non-restricted pastures: 3.6 (1–4.9)
Flood plain pasture: 3.6 (1.6–5.6) | |||||
| Potatoes | 5.4 (3.2–9.4) | 0.63±0.37 | 0.65±0.34 | 0.38 (0.21–0.85)c | 0.30 (0.15–8.1)b |
| 0.55 (0.27–1.5)c | |||||
| Vegetables | 5.2–28.5 | <4 | <5 | ||
| Meat | 0.6 | <1 | <4 | ||
| Egg | 0.6 | 1.6±1.3 | 2.0 | ||
| Cereals | 1.4 (1.26–1.32) | ||||
| Fish | |||||
| Techa River | 340 | 150 | 2.7 | ||
| Lakes | 45 (41–48) | ||||
| Product | Cabianca et al.,[19] (Brodokalmak) | RECLAIM [24] | Romanov [25] | |||
|---|---|---|---|---|---|---|
| mean | range (n) | Brodokalmak | Muslyumovo | Brodokalmak | Muslyumovo | |
| Milk (all) | 55 | 1.8–230 (7) | 4.4 (0.04–292) (n=266)a | 15.3 (0.04–1890) (n=961)a | 0.71 (0.4–1.1)c | 1.3 (0.37–6.7)b |
| Non-restricted pastures: | 7.1 | 1.8–12 (5) | 2.8c | |||
| Floodplain pasture: | 175 | 120–230 (2) | ||||
| Potatoes | BDL | 0.16±0.10 | 0.36±0.20 | 0.74 (0.5–1.4)c | 0.63 (0.37–6.3)b | |
| 1.0 (0.8–1.3)c | ||||||
| Vegetables | BDL | <0.4 | <4.6 | |||
| Meat (poultry and beef) | 0.6 | <14 | ||||
| Egg | 2.8 | |||||
| Cereals | 1.3 | 1.26–1.32 | ||||
| Fish | ||||||
| Techa River | 580 | 220±48 | ||||
| Lakes | 58 | 38–92 (3) | ||||
| Food product | Cabianca et al., [19] | RECLAIM [24] | Romanov [25] | |||
|---|---|---|---|---|---|---|
| Average | Most exposed group | Average | Range | Range (age 1 to > 17 yrs) | ||
| Milk | 100 | 423 | 255 | 0 – 1280 | 164 – 197 | |
| Meat | 25 | 105 | 55 | 0 – 175 | 9 – 547 | |
| Potatoes | 91 | 250 | 146 | 26 – 365 | 46 – 120 | |
| Vegetables | 80 | 350 | 55 | 0 – 110 | 15 – 40 | |
| Fish | 24 | 100 | 37 | 0 – 183 | ||
| Bread | 130 | 270 | 77 – 237 | |||
| Water | 820 | 1200 | 24 – 547 | |||
| Organ | ≤ 1 mGy | 1–10 mGy | 10–100 mGy | 100 mGy–1 Gy | > 1 Gy |
|---|---|---|---|---|---|
| RBM | 7.9 | 12 | 23 | 55 | 1.7 |
| BS | 9.0 | 9.5 | 13 | 57 | 11 |
| LLI | 11 | 12 | 44 | 34 | – |
| ULI | 12 | 16 | 54 | 18 | – |
| SI | 14 | 56 | 22 | 7.7 | – |
| ST | 14 | 58 | 20 | 7.6 | – |
| Testes | 13 | 59 | 20 | 8.2 | – |
| Ovaries | 16 | 58 | 20 | 7.1 | – |
| Uterus | 15 | 58 | 20 | 7.1 | – |
| Age (y) | Muslyumovo | Brodokalmak | ||
|---|---|---|---|---|
| 90Sr | 137Cs | 90Sr | 137Cs | |
| <1 | 58 | 310 | 52 | 130 |
| 1 | 110 | 620 | 100 | 270 |
| 5 | 170 | 620 | 160 | 270 |
| 10 | 230 | 680 | 210 | 300 |
| 15 | 230 | 830 | 210 | 360 |
| >17 | 230 | 930 | 210 | 400 |
| Age (1999) | Muslyumovo | Brodokalmak | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Accumulated up to 1999 | Intake in 1999 | Accumulated up to 1999 | Intake in 1999 | |||||||||
| 90Sr | 137Cs | Total | 90Sr | 137Cs | Total | 90Sr | 137Cs | Total | 90Sr | 137Cs | Total | |
| <1 | 0.00022 | 0.0065 | 0.0067 | – | – | – | ||||||
| 1 | 0.00042 | 0.013 | 0.013 | 0.00048 | 0.015 | 0.016 | 0.0004 | 0.0057 | 0.0061 | 0.0002 | 0.0027 | 0.0029 |
| 5 | 0.0034 | 0.068 | 0.071 | 0.00056 | 0.012 | 0.013 | 0.0032 | 0.030 | 0.033 | 0.0004 | 0.0065 | 0.0070 |
| 10 | 0.0087 | 0.086 | 0.095 | 0.00060 | 0.015 | 0.016 | 0.0080 | 0.038 | 0.046 | 0.0005 | 0.0051 | 0.0056 |
| 15 | 0.012 | 0.17 | 0.18 | 0.00030 | 0.029 | 0.029 | 0.011 | 0.074 | 0.085 | 0.0004 | 0.013 | 0.013 |
| 17–18 | 0.014 | 0.24 | 0.25 | 0.00032 | 0.036 | 0.036 | 0.012 | 0.10 | 0.11 | 0.0002 | 0.016 | 0.016 |
| 18–48 | 0.042 | 1.0 | 1.1 | 0.00032 | 0.036 | 0.036 | 0.039 | 0.44 | 0.48 | 0.0002 | 0.016 | 0.016 |
| Year | Muslyumovo | Brodokalmak | |||||
|---|---|---|---|---|---|---|---|
| Children (> 2y) | Teenagers (12–17) | Adults (18–48) | Children (> 2y) | Teenagers (12–17) | Adults (18–48) | ||
| Minimum level | Up to 1995: | 0.51 (86)a 1.3 (93)b | 2.3 (92) | 11 (90) | 0.20 (84)a 0.52 (91)b | 0.92 (91) | 7.0 (93) |
| 1999: | 0.15 (91) | 0.17 (82) | 0.15 (75) | 0.0061 (90) | 0.068 (81) | 0.13 (87) | |
| Maximum level | Up to 1995: | 3.0 (93)a 7.8 (96)b | 14.5 (96) | 65 (95) | 1.2 (92)a 3.1 (96)b | 5.9 (96) | 28 (95) |
| 1999: | 0.78 (94) | 1.1 (92) | 0.67 (84) | 0.32 (94) | 0.42 (91) | 0.27 (82) | |
| Village | Annual effective dose (mSv) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| External | Internal | Total | ||||||||||
| mean | geom. Mean | 5% conf. | 95% conf. | mean | geom. mean | 5% conf. | 95% conf. | mean | geom. Mean | 5% conf. | 95% conf. | |
| Muslyumovo | ||||||||||||
| Group1 | 0.05 | 0.04 | 0.03 | 0.07 | 0.07 | 0.06 | 0.02 | 0.16 | 0.12 | 0.11 | 0.05 | 0.21 |
| Group2 | 0.28 | 0.23 | 0.08 | 0.70 | 0.09 | 0.07 | 0.03 | 0.19 | 0.39 | 0.34 | 0.15 | 0.78 |
| Group3 | 0.89 | 0.67 | 0.19 | 2.34 | 0.25 | 0.20 | 0.06 | 0.62 | 1.13 | 0.93 | 0.34 | 2.57 |
| Brodokalmak | ||||||||||||
| Group1 | 0.03 | 0.03 | 0.02 | 0.04 | 0.05 | 0.04 | 0.01 | 0.12 | 0.08 | 0.07 | 0.03 | 0.15 |
| Group2 | 0.09 | 0.08 | 0.03 | 0.21 | 0.05 | 0.04 | 0.02 | 0.12 | 0.15 | 0.13 | 0.06 | 0.29 |
| Group3 | 0.27 | 0.21 | 0.06 | 0.67 | 0.10 | 0.08 | 0.03 | 0.20 | 0.37 | 0.31 | 0.12 | 0.81 |
Share and Cite
Standring, W.J.F.; Dowdall, M.; Strand, P. Overview of Dose Assessment Developments and the Health of Riverside Residents Close to the “Mayak” PA Facilities, Russia. Int. J. Environ. Res. Public Health 2009, 6, 174-199. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph6010174
Standring WJF, Dowdall M, Strand P. Overview of Dose Assessment Developments and the Health of Riverside Residents Close to the “Mayak” PA Facilities, Russia. International Journal of Environmental Research and Public Health. 2009; 6(1):174-199. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph6010174
Chicago/Turabian StyleStandring, William J.F., Mark Dowdall, and Per Strand. 2009. "Overview of Dose Assessment Developments and the Health of Riverside Residents Close to the “Mayak” PA Facilities, Russia" International Journal of Environmental Research and Public Health 6, no. 1: 174-199. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph6010174




