The Antimicrobial Properties of Cedar Leaf (Thuja plicata) Oil; A Safe and Efficient Decontamination Agent for Buildings
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. Methods
3. Results
3.1. Antibacterial Tests with Liquid CLO
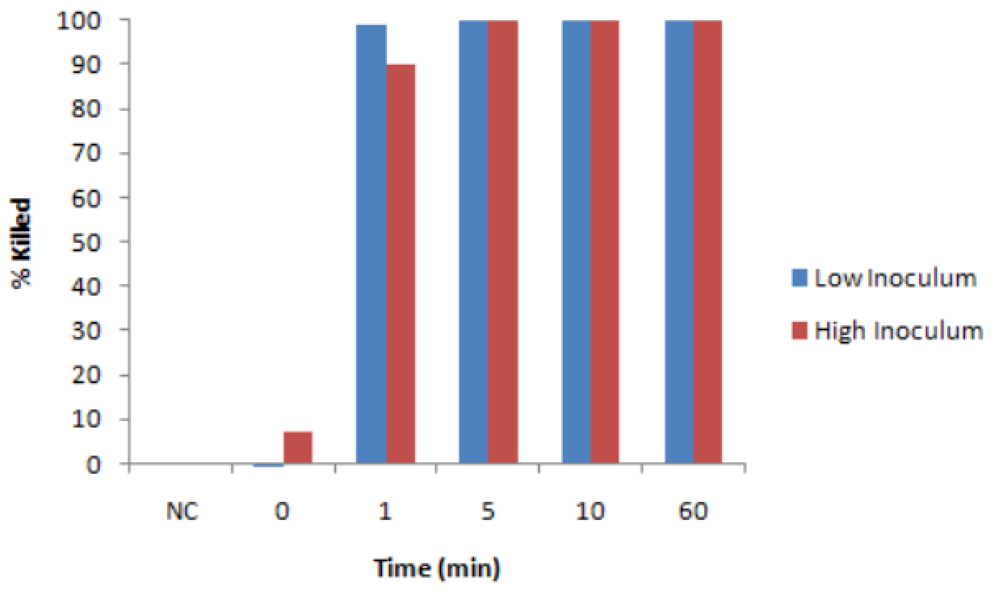
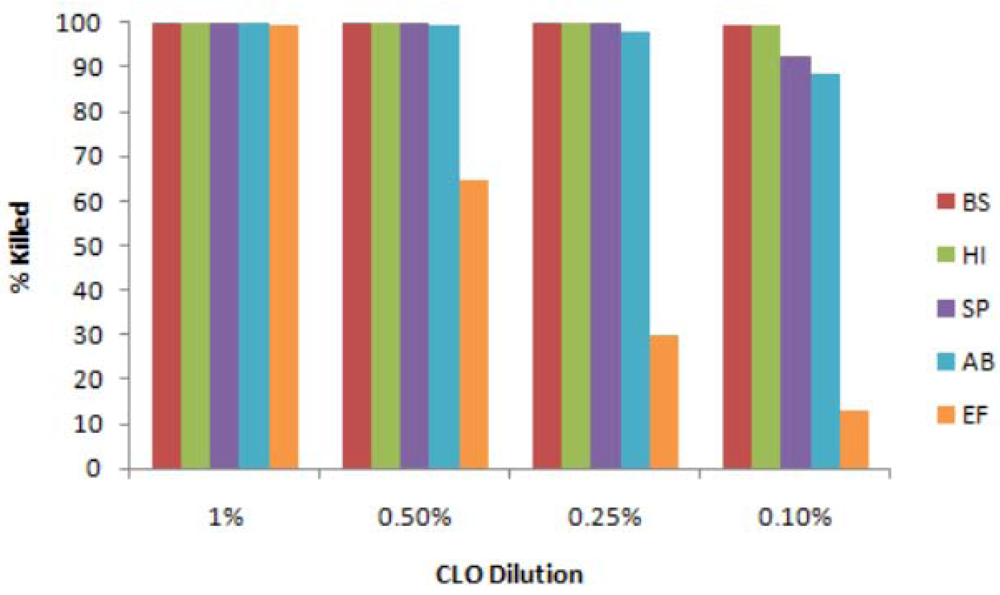
3.2. Possible Effect of Light

3.3. Sporicidal Activity of CLO
| Exposure time hours | 1% CLO | 5% CLO | 10% CLO |
|---|---|---|---|
| 24 | 30.5 | 48.0 | 63.6 |
| 48 | 41.2 | 50.0 | 88.0 |
| 72 | 64.5 | 70.2 | 93.4 |
3.4. Antibacterial Effects of CLO Vapor on Dried Films

3.5. Effect of CLO on Candida albicans
| Exposure time hours | C. albicans (cfu) | % kill |
|---|---|---|
| 0 | 1,270 ± 58 | |
| 4 | 67 ± 3.4 | 95 |
| 24 | 0 | >99.9 |
3.6. Effect of CLO-Vapor on Aspergillus niger
3.7. Cytotoxicity of CLO Vapor
| Time of exposure to CLO vapor (min) | Cell survival% of control |
|---|---|
| 10 | 97.5 ± 13.7 |
| 30 | 96.7 ± 7.6 |
| 60 | 97.0 ± 8.3 |
4. Discussion
Acknowledgments
Conflict of Interest
References
- Norback, D. An update on sick building syndrome. Curr. Opin. Allergy Clin. Immunol. 2009, 9, 55–59. [Google Scholar]
- Terr, A.I. Sick building syndrome: Is mould the cause? Med. Mycol. 2009, 47 (Suppl 1), S217–S222. [Google Scholar]
- Straus, D.C. Molds, mycotoxins, and sick building syndrome. Toxicol. Ind. Health 2009, 25, 617–635. [Google Scholar]
- Eduard, W. Fungal spores: A critical review of the toxicological and epidemiological evidence as a basis for occupational exposure limit setting. Crit. Rev. Toxicol. 2009, 39, 799–864. [Google Scholar]
- Wood, J.P.; Choi, Y.W.; Rogers, J.V.; Kelly, T.J.; Riggs, K.B.; Willenberg, Z.J. Efficacy of liquid spray decontaminants for inactivation of Bacillus anthracis spores on building and outdoor materials. J. Appl. Microbiol. 2011, 110, 1262–1273. [Google Scholar]
- Sabbah, S.; Springthorpe, S.; Sattar, S.A. Use of a mixture of surrogates for infectious bioagents in a standard approach to assessing disinfection of environmental surfaces. Appl. Environ. Microbiol. 2010, 76, 6020–6022. [Google Scholar]
- Hudson, J.; Vimalanathan, S. Echinacea—A source of potent antivirals for respiratory virus infections. Pharmaceuticals 2011, 4, 1019–1031. [Google Scholar]
- Small, B.M. Creating healthier buildings. Toxicol. Ind. Health 2009, 25, 731–735. [Google Scholar]
- McDonnell, G.; Russell, A.D. Antiseptics and disinfectants: Activity, action and resistance. Clin. Microbiol. Rev. 1999, 12, 147–179. [Google Scholar]
- Setlow, P. Spores of Bacillus subtilis: Their resistance to and killing by radiation, heat and chemicals. J. Appl. Microbiol. 2005, 101, 514–525. [Google Scholar]
- Hudson, J.B.; Sharma, M. The practical application of ozone gas as an anti-fungal (anti-mold) agent. Ozone Sci. Eng. 2009, 31, 326–332. [Google Scholar]
- Moat, J.; Cargill, J.; Shone, J.; Upton, M. Application of a novel decontamination process using gaseous ozone. Can. J. Microbiol. 2009, 55, 928–933. [Google Scholar]
- Carson, C.F.; Hammer, K.A.; Riley, T.V. Melaleuca alternifolia (Tea Tree) oil: A review of antimicrobial and other medicinal properties. Clin. Microbiol. Rev. 2006, 19, 50–62. [Google Scholar]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar]
- Sadlon, A.E.; Lamson, D.W. Immune-modifying and antimicrobial effects of eucalyptus oil and simple inhalation devices. Altern. Med. Rev. 2010, 15, 33–47. [Google Scholar]
- Lawrence, H.A.; Palombo, E.A. Activity of essential oils against Bacillus subtilis spores. J. Microbiol. Biotechnol. 2009, 19, 1590–1595. [Google Scholar]
- Tsiri, D.; Graikou, K.; Poblocka-Olech, L.; Krauze-Baranowska, M.; Spyropoulos, C.; Chnou, I. Chemosystematic value of essential oil compostion of Thuja species cultivated in Poland—Antimicrobial activity. Molecules 2009, 14, 4707–4715. [Google Scholar]
- Sharma, M.; Vohra, S.; Arnason, J.T.; Hudson, J.B. Echinacea extracts contain significant and selective activities against human pathogenic bacteria. Pharm. Biol. 2008, 46, 111–116. [Google Scholar]
- Sorg, J.A.; Dineen, S.S. Laboratory maintenance of Clostridium difficile. Curr. Protoc. Microbiol. 2009, 12, 9A.1:1–9A.1:10. [Google Scholar]
- Hammer, K.A.; Carson, C.F.; Dunstan, J.A.; Hale, J.; Lehmann, H.; Robinson, C.J.; Prescott, S.L.; Riley, T.V. Antimicrobial and anti-inflammatory activity of five Taxandria fragrans oils in vitro. Microbiol. Immunol. 2008, 52, 522–530. [Google Scholar]
- Stark, H.; Roponen, M.; Purokivi, M.; Randell, J.; Tukiainen, H.; Hirvonen, M.R. Aspergillus fumigatus challenge increases cytokine levels in nasal lavage fluid. Inhal. Toxicol. 2006, 18, 1033–1039. [Google Scholar]
- Hope, A.P.; Simon, R.A. Excess dampness and mold growth in homes: An evidence-based review of the aeroirritant effect and its potential causes. Allergy Asthma Proc. 2007, 28, 262–270. [Google Scholar]
- Naser, B.; Bodinet, C.; Tegtmeier, M.; Lindequist, U. Thuja occidentalis (Arbor vitae): A review of its pharmaceutical, pharmacological and clinical properties. Evid. Based Complement. Alternat. Med. 2005, 2, 69–78. [Google Scholar]
- Tyagi, A.K.; Malik, A. Antimicrobial potential and chemical composition of Eucalyptus globulus oil in liquid and vapour phase against food spoilage microorganisms. Food Chem. 2011, 126, 228–235. [Google Scholar]
- Wullt, M.; Odenholt, I.; Walder, M. Activity of three disinfectants and acidified nitrite against Clostridium difficile spores. Infect. Control Hosp. Epidemiol. 2003, 24, 765–768. [Google Scholar]
- Gertsch, J. Botanical drugs, synergy, and network pharmacology: Forth and back to intelligent mixtures. Planta Med. 2011, 77, 1086–1098. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hudson, J.; Kuo, M.; Vimalanathan, S. The Antimicrobial Properties of Cedar Leaf (Thuja plicata) Oil; A Safe and Efficient Decontamination Agent for Buildings. Int. J. Environ. Res. Public Health 2011, 8, 4477-4487. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph8124477
Hudson J, Kuo M, Vimalanathan S. The Antimicrobial Properties of Cedar Leaf (Thuja plicata) Oil; A Safe and Efficient Decontamination Agent for Buildings. International Journal of Environmental Research and Public Health. 2011; 8(12):4477-4487. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph8124477
Chicago/Turabian StyleHudson, James, Michael Kuo, and Selvarani Vimalanathan. 2011. "The Antimicrobial Properties of Cedar Leaf (Thuja plicata) Oil; A Safe and Efficient Decontamination Agent for Buildings" International Journal of Environmental Research and Public Health 8, no. 12: 4477-4487. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph8124477




