Using Geographically Weighted Regression (GWR) to Explore Spatial Varying Relationships of Immature Mosquitoes and Human Densities with the Incidence of Dengue
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Dengue Data
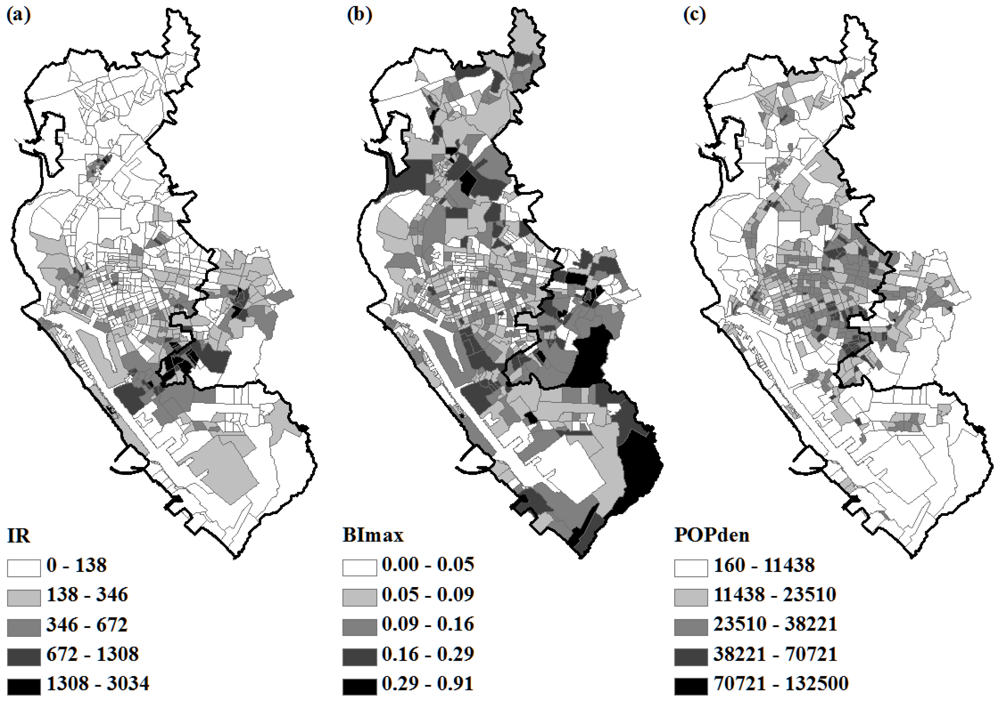
2.3. Immature Mosquito and Human Density
2.4. Statistical Analysis
3. Results
3.1. OLS Regression
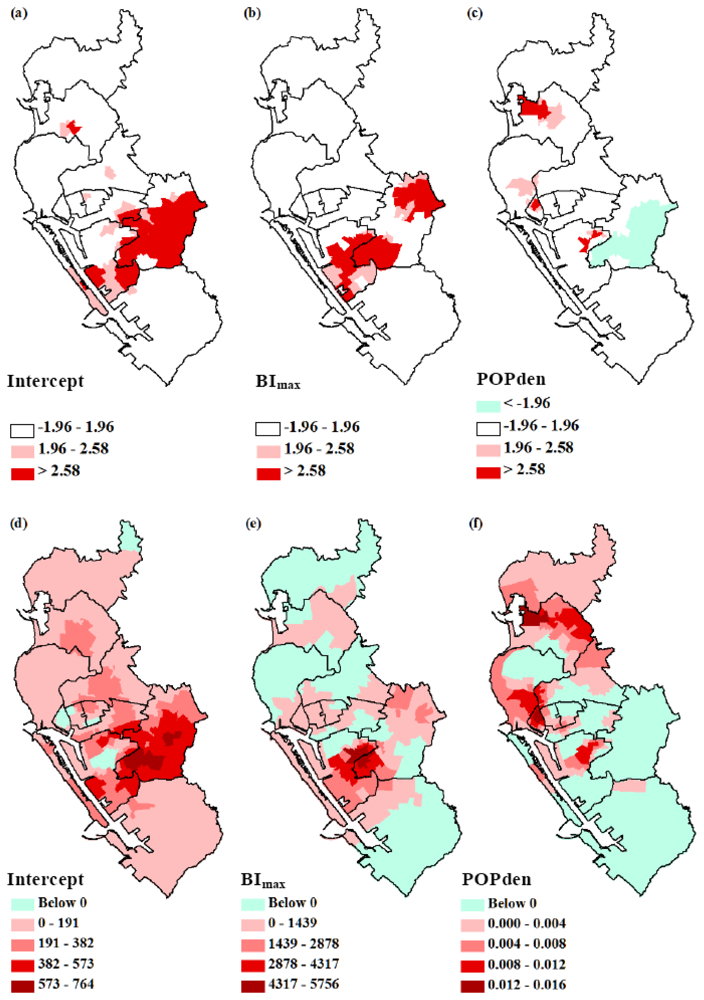
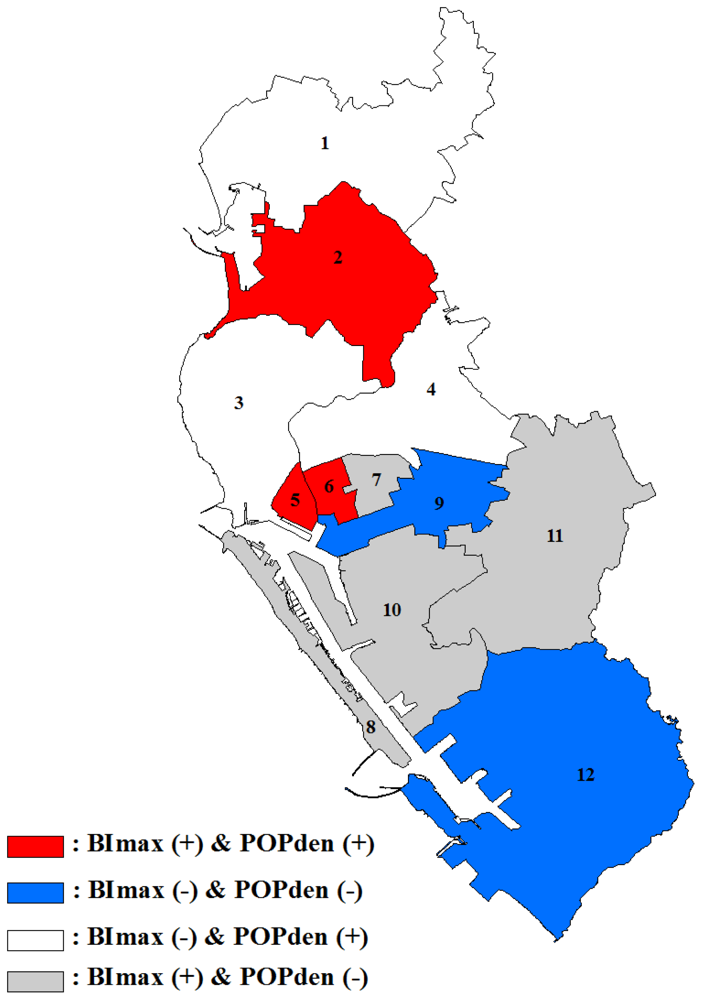
3.2. GWR Model and Spatial Variations
4. Discussion
5. Conclusions
Acknowledgements
References
- World Health Organization (WHO), Dengue: Guidelines for Diagnosis, Treatment, Prevention and Control-New Edition; WHO: Geneva, Switzerland, 2009.
- Akagi, K; Kojima, D. Experimental studies for the pathogen of dengue fever. J Formosan Med Assoc 1915, 158, 1049–1078. (in Japanese). [Google Scholar]
- Taiwan Centers For Disease Control (CDC), Guidelines for Dengue Control; CDC: Taipei city Taiwan, 2009.
- Gubler, DJ. Dengue and dengue hemorrhagic fever. Clin. Microbiol. Rev 1998, 11, 480–496. [Google Scholar]
- Guzman, A; Istúriz, RE. Update on the global spread of dengue. Int. J. Antimicrob. Agents 2010, 36, S40–S42. [Google Scholar]
- Wu, PC; Lay, JG; Guo, HR; Lin, CY; Lung, SC; Su, HJ. Higher temperature and urbanization affect the spatial patterns of dengue fever transmission in subtropical Taiwan. Sci. Total Environ 2009, 407, 2224–2233. [Google Scholar]
- Wu, PC; Guo, HR; Lung, SC; Lin, CY; Su, HJ. Weather as an effective predictor for occurrence of dengue fever in Taiwan. Acta Trop 2007, 103, 50–57. [Google Scholar]
- Focks, DA. A Review of Entomological Sampling Methods and Indicators for Dengue Vectors; World Health Organization: Geneva, Switzerland, 2003. [Google Scholar]
- Sulaiman, S; Pawanchee, ZA; Arifin, Z; Wahab, A. Relationship between Breteau and house indices and cases of dengue/dengue hemorrhagic fever in Kuala Lumpur, Malaysia. J. Am. Mosq. Control Assoc 1996, 12, 494–496. [Google Scholar]
- Teixeira, MG; Barreto, ML; Costa, MC; Ferreira, LD; Vasconcelos, PF; Cairncross, S. Dynamics of dengue virus circulation: a silent epidemic in a complex urban area. Trop. Med. Int. Health 2002, 7, 757–762. [Google Scholar]
- Chadee, DD; Shivnauth, B; Rawlins, SC; Chen, AA. Climate, mosquito indices and the epidemiology of dengue fever in Trinidad (2002–2004). Ann. Trop. Med. Parasitol 2007, 101, 69–77. [Google Scholar]
- Sanchez, L; Cortinas, J; Pelaez, O; Gutierrez, H; Concepcion, D; van der Stuyft, P. Breteau Index threshold levels indicating risk for dengue transmission in areas with low Aedes infestation. Trop. Med. Int. Health 2010, 15, 173–175. [Google Scholar]
- Thammapalo, S; Chongsuvivatwong, V; Geater, A; Dueravee, M. Environmental factors and incidence of dengue fever and dengue haemorrhagic fever in an urban area, Southern Thailand. Epidemiol. Infect 2008, 136, 135–143. [Google Scholar]
- Chadee, DD; Williams, FLR; Kitron, UD. Impact of vector control on a dengue fever outbreak in Trinidad, West Indies, in 1998. Trop. Med. Int. Health 2005, 10, 748–754. [Google Scholar]
- Siqueira, JB; Martelli, CMT; Maciel, IJ; Oliveira, RM; Ribeiro, MG; Amorim, FP; Moreira, BC; Cardoso, DDP; Souza, WV; Andrade, AL. Household survey of dengue infection in Central Brazil: Spatial point pattern analysis and risk factors assessment. Am. J. Trop. Med. Hyg 2004, 71, 646–651. [Google Scholar]
- Ko, YC. Epidemiology of dengue fever in Taiwan. Kaohsiung J. Med. Sci 1989, 5, 1–11. [Google Scholar]
- Morrison, AC; Getis, A; Santiago, M; Rigau-Perez, JG; Reiter, P. Exploratory space-time analysis of reported dengue cases during an outbreak in Florida, Puerto Rico, 1991–1992. Am. J. Trop. Med. Hyg 1998, 58, 287–298. [Google Scholar]
- Carbajo, AE; Schweigmann, N; Curto, SI; De Garin, A; Bejaran, R. Dengue transmission risk maps of Argentina. Trop. Med. Int. Health 2001, 6, 170–183. [Google Scholar]
- Kolivras, KN. Mosquito habitat and dengue risk potential in Hawaii: A conceptual framework and GIS application. Prof. Geographer 2006, 58, 139–154. [Google Scholar]
- Mammen, MP; Pimgate, C; Koenraadt, CJ; Rothman, AL; Aldstadt, J; Nisalak, A; Jarman, RG; Jones, JW; Srikiatkhachorn, A; Ypil-Butac, CA; et al. Spatial and temporal clustering of dengue virus transmission in Thai villages. PLos Med 2008, 5, e205. [Google Scholar]
- Menard, S. Applied Logistic Regression Analysis, 2nd ed; Sage: Newbury Park, CA, USA, 2002. [Google Scholar]
- Wong, DWS; Lee, J. Statistical Analysis of Geographic Information with ArcView GIS And ArcGIS; John Wiley and Sons: Hoboken, NJ, USA, 2005. [Google Scholar]
- Moran, PAP. Notes on Continuous Stochastic Phenomena. Biometrika 1950, 37, 17–23. [Google Scholar]
- Fotheringham, AS; Brunsdon, C; Charlton, M. Geographically Weighted Regression: The Analysis of Spatially Varying Relationships; Wiley: New York, NY, USA, 2002. [Google Scholar]
- Fotheringham, AS; Brunsdon, C; Charlton, M. Quantitative Geography: Perspectives on Spatial Data Analysis; Sage: Newbury Park, CA, USA, 2000. [Google Scholar]
- Wen, TH; Lin, NH; Chao, DY; Hwang, KP; Kan, CC; Lin, KCM; Wu, JTS; Huang, SYJ; Fan, IC; King, CC. Spatial-temporal patterns of dengue in areas at risk of dengue hemorrhagic fever in Kaohsiung, Taiwan, 2002. Int. J. Infect. Dis 2010, 14, e334–e343. [Google Scholar]
- Harrington, LC; Scott, TW; Lerdthusnee, KRIA; Coleman, RC; Costero, ADRI; Clark, GG; Jones, JJ; Kitthawee, SANG; Kittayapong, PATT; Sithiprasasna, RATA; et al. Dispersal of the dengue vector Aedes aegypti within and between rural communities. Am. J. Trop. Med. Hyg 2005, 72, 209–220. [Google Scholar]
- Favier, C; Schmit, D; Müller-Graf, CDM; Cazelles, B; Degallier, N; Mondet, B; Dubois, MA. Influence of spatial heterogeneity on an emerging infectious disease: the case of dengue epidemics. Proc. R. Soc B 2005, 272, 1171–1177. [Google Scholar]
- Vanwambeke, SO; van Benthem, BH; Khantikul, N; Burghoorn-Maas, C; Panart, K; Oskam, L; Lambin, E; Somboon, P. Multi-level analyses of spatial and temporal determinants for dengue infection. Int. J. Health Geogr. 2006, 5, 5. [Google Scholar]
- Koh, BK; Ng, LC; Kita, Y; Tang, CS; Li Ang, LW; Wong, KY; James, L; Goh, KT. The 2005 Dengue epidemic in Singapore-epidemiology, prevention and control. Ann. Acad. Med. Singapore 2008, 37, 538–545. [Google Scholar]
- Almeida, MCD; Caiaffa, WT; Assuncao, RM; Proietti, FA. Spatial vulnerability to dengue in a Brazilian urban area during a 7-year surveillance. J. Urban Health 2007, 84, 334–345. [Google Scholar]
- Thai, KTD; Nagelkerke, N; Phuong, HL; Nga, TTT; Giao, PT; Hung, LQ; Binh, TQ; Nam, NV; de Vries, PJ. Geographical heterogeneity of dengue transmission in two villages in southern Vietnam. Epidemiol. Infect 2010, 138, 585–591. [Google Scholar]
- Smith, DL; Dushoff, J; McKenzie, FE. The risk of a mosquito-borne infection in a heterogeneous environment. Plos Biol 2004, 2, 1957–1964. [Google Scholar]
- Cummings, DAT; Irizarry, RA; Huang, NE; Endy, TP; Nisalak, A; Ungchusak, K; Burke, DS. Travelling waves in the occurrence of dengue haemorrhagic fever in Thailand. Nature 2004, 427, 344–347. [Google Scholar]
- Shang, CS; Fang, CT; Liu, CM; Wen, TH; Tsai, KH; King, CC. The role of imported cases and favorable meteorological conditions in the onset of dengue epidemics. PLoS Neglected Trop. Dis 2010, 4, e775. [Google Scholar]
- Pai, HH; Lu, YL; Hong, YJ; Hsu, EL. The differences of dengue vectors and human behavior between families with and without members having dengue fever/dengue hemorrhagic fever. Int. J. Environ. Health Res 2005, 15, 263–269. [Google Scholar]
- Bohra, A; Andrianasolo, H. Application of GIS in modeling of dengue risk based on sociocultural data: Case of Jalore, Rajasthan, India. Dengue Bull 2001, 25, 92–102. [Google Scholar]
- Chowell, G; Torre, CA; Munayco-Escate, C; Suarez-Ognio, L; Lopez-Cruz, R; Hyman, JM; Castillo-Chavez, C. Spatial and temporal dynamics of dengue fever in Peru: 1994–2006. Epidemiol. Infect 2008, 136, 1667–1677. [Google Scholar]
- Reiter, P; Lathrop, S; Bunning, M; Biggerstaff, B; Singer, D; Tiwari, T; Baber, L; Amador, M; Thirion, J; Hayes, J; et al. Texas lifestyle limits transmission of dengue virus. Emerging Infect. Dis 2003, 9, 86–89. [Google Scholar]
- Connor, ME; Monroe, WM. Stegomyia Indices and Their Value in Yellow Fever Control. Am J Trop Med Hyg 1923. [Google Scholar]
- Sauthowo, TR; Tonn, RJ; Yasuno, M; Reader, PM; Murdie, G. Studies on Life Budget of Aedes aegypti in Wat Samphaya, Bangkok, Thailand. Bull. W. H. O 1972, 46, 211–226. [Google Scholar]
- Focks, DA; Brenner, RJ; Hayes, J; Daniels, E. Transmission thresholds for dengue in terms of Aedes aegypti pupae per person with discussion of their utility in source reduction efforts. Am. J. Trop. Med. Hyg 2000, 62, 11–18. [Google Scholar]
- Halstead, SB. Etiologies of the Experimental Dengues of Siler and Simmons. Am. J. Trop. Med. Hyg 1974, 23, 974–982. [Google Scholar]
- Isik, O; Pinarcioglu, MM. Geographies of a silent transition: A geographically weighted regression approach to regional fertility differences in Turkey. Eur. J. Popul 2006, 22, 399–421. [Google Scholar]
- Mennis, J. Mapping the results of geographically weighted regression. Cartogr. J 2006, 43, 171–179. [Google Scholar]
- Wen, TH; Chen, DR; Tsai, MJ. Identifying geographical variations in poverty-obesity relationships: empirical evidence from Taiwan. Geospat. Health 2010, 4, 257–265. [Google Scholar]
- Wheeler, D; Tiefelsdorf, M. Multicollinearity and correlation among local regression coefficients in geographically weighted regression. J. Geogr. Syst 2005, 7, 161–187. [Google Scholar]
- Qiu, X; Wu, SS. Global and Local Regression Analysis of Factors of American College Test (ACT) Score for Public High Schools in the State of Missouri. Ann. Assoc. Am. Geogr 2011, 101, 63–83. [Google Scholar]
- Waller, LA; Zhu, L; Gotway, CA; Gorman, DM; Gruenewald, PJ. Quantifying geographic variations in associations between alcohol distribution and violence: A comparison of geographically weighted regression and spatially varying coefficient models. Stoch. Environ. Res. Risk Assess 2007, 21, 573–588. [Google Scholar]





| Variable | Numerator | Denominator | |
|---|---|---|---|
| Dependent: | IR | 100,000 × number of cases | Populations |
| Independent: | BImax | 100 × number of positive containers | Number of premises inspected |
| POPden | Populations | The area of Li (km2) | |
| Parameter | Estimated Value | Standard Error | p-value | VIF |
|---|---|---|---|---|
| Intercept | 115.52 | 34.73 | 0.003 | |
| BImax | 947.93 | 202.48 | 0.013 | 1.02 |
| POPden | 0.00 | 0.00 | 0.111 | 1.02 |
| Adjusted R2 | 0.04 | |||
| AIC | 7,902.12 |
| Parameter | Minimum | 25% quartile | 50 % quartile | 75 % quartile | Maximum |
|---|---|---|---|---|---|
| Intercept | −272.60 | 78.46 | 166.09 | 320.92 | 1,088.31 |
| BImax | −2980.43 | −262.53 | 100.40 | 838.91 | 5,797.87 |
| POPden | −0.02 | −0.00 | 0.00 | 0.00 | 0.02 |
| Condition number | 2.96 | 4.67 | 5.83 | 7.32 10.39 | |
| Adjusted R2 | 0.59 | ||||
| AICc | 7,715.17 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lin, C.-H.; Wen, T.-H. Using Geographically Weighted Regression (GWR) to Explore Spatial Varying Relationships of Immature Mosquitoes and Human Densities with the Incidence of Dengue. Int. J. Environ. Res. Public Health 2011, 8, 2798-2815. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph8072798
Lin C-H, Wen T-H. Using Geographically Weighted Regression (GWR) to Explore Spatial Varying Relationships of Immature Mosquitoes and Human Densities with the Incidence of Dengue. International Journal of Environmental Research and Public Health. 2011; 8(7):2798-2815. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph8072798
Chicago/Turabian StyleLin, Chia-Hsien, and Tzai-Hung Wen. 2011. "Using Geographically Weighted Regression (GWR) to Explore Spatial Varying Relationships of Immature Mosquitoes and Human Densities with the Incidence of Dengue" International Journal of Environmental Research and Public Health 8, no. 7: 2798-2815. https://0-doi-org.brum.beds.ac.uk/10.3390/ijerph8072798




