A Comparative Evaluation of Nanohydroxyapatite-Enriched Hydrogen Peroxide Home Bleaching System on Color, Hardness and Microstructure of Dental Enamel
Abstract
:1. Introduction
2. Materials and Methods
2.1. Color Evaluation
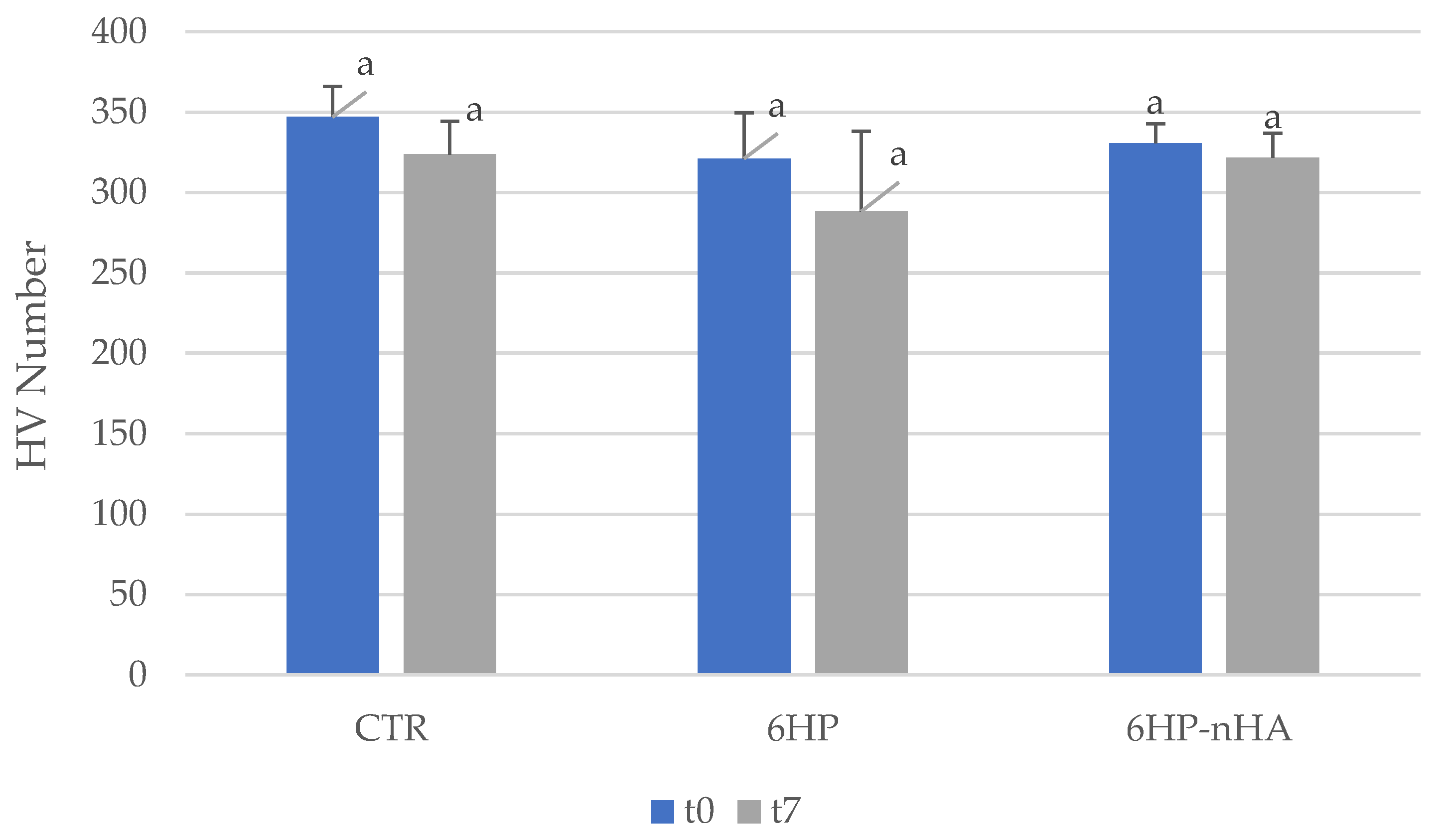
2.2. Microhardness Evaluation
2.3. Scanning Electron Microscopy Observations
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Goettems, M.L.; Fernandez, M.D.S.; Donassollo, T.A.; Donassollo, S.H.; Demarco, F.F. Impact of tooth bleaching on oral health-related quality of life in adults: A triple-blind randomised clinical trial. J. Dent. 2021, 105, 103564. [Google Scholar] [CrossRef]
- Jin, J.; Xu, X.; Lai, G.; Kunzelmann, K.-H. Efficacy of tooth whitening with different calcium phosphate-based formulations. Eur. J. Oral Sci. 2013, 121, 382–388. [Google Scholar] [CrossRef] [PubMed]
- Pontes, M.; Gomes, J.; Lemos, C.; Leão, R.S.; Moraes, S.; Vasconcelos, B.; Pellizzer, E.P. Effect of Bleaching Gel Concentration on Tooth Color and Sensitivity: A Systematic Review and Meta-analysis. Oper. Dent. 2020, 45, 265–275. [Google Scholar] [CrossRef] [PubMed]
- De Geus, J.L.; Wambier, L.M.; Kossatz, S.; Loguercio, A.D.; Reis, A. At-home vs In-office Bleaching: A Systematic Review and Meta-analysis. Oper. Dent. 2016, 41, 341–356. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luque-Martinez, I.; Reis, A.; Schroeder, M.; Muñoz, M.A.; Loguercio, A.D.; Masterson, D.; Maia, L. Comparison of efficacy of tray-delivered carbamide and hydrogen peroxide for at-home bleaching: A systematic review and meta-analysis. Clin. Oral Investig. 2016, 20, 1419–1433. [Google Scholar] [CrossRef]
- De la Peña, V.A.; Ratón, M.L. Randomized Clinical Trial on the Efficacy and Safety of Four Professional At-Home Tooth Whitening Gels. Oper. Dent. 2014, 39, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Sutil, E.; da Silva, K.L.; Terra, R.M.O.; Burey, A.; Rezende, M.; Reis, A.; Loguercio, A.D. Effectiveness and adverse effects of at-home dental bleaching with37% versus 10%carbamide peroxide: Arandomized, blind clinical trial. J. Esthet. Restor. Dent. 2020. [Google Scholar] [CrossRef] [PubMed]
- Alkahtani, R.; Stone, S.; German, M.; Waterhouse, P. A review on dental whitening. J. Dent. 2020, 100, 103423. [Google Scholar] [CrossRef]
- Cordeiro, D.; Toda, C.; Hanan, S.; Arnhold, L.P.; Reis, A.; Loguercio, A.D.; Bandeira, M.C.L. Clinical Evaluation of Different Delivery Methods of At-Home Bleaching Gels Composed of 10% Hydrogen Peroxide. Oper. Dent. 2019, 44, 13–23. [Google Scholar] [CrossRef]
- Monteiro, M.J.F.; Lindoso, J.B.C.; Conde, N.C.D.O.; Da Silva, L.M.; Loguercio, A.D.; Pereira, J.V. Evaluation of the genotoxic potential of different delivery methods of at-home bleaching gels: A single-blind, randomized clinical trial. Clin. Oral Investig. 2019, 23, 2199–2206. [Google Scholar] [CrossRef]
- Rodríguez-Martínez, J.; Valiente, M.; Sánchez-Martín, M. Tooth whitening: From the established treatments to novel approaches to prevent side effects. J. Esthet. Restor. Dent. 2019, 31, 431–440. [Google Scholar] [CrossRef]
- Maran, B.M.; Matos, T.D.P.; Castro, A.D.S.D.; Vochikovski, L.; Amadori, A.L.; Loguercio, A.D.; Reis, A.; Berger, S.B. In-office bleaching with low/medium vs. high concentrate hydrogen peroxide: A systematic review and meta-analysis. J. Dent. 2020, 103, 103499. [Google Scholar] [CrossRef]
- Orilisi, G.; Tosco, V.; Monterubbianesi, R.; Notarstefano, V.; Özcan, M.; Putignano, A.; Orsini, G. ATR-FTIR, EDS and SEM evaluations of enamel structure after treatment with hydrogen peroxide bleaching agents loaded with nano-hydroxyapatite particles. PeerJ 2021, 9, e10606. [Google Scholar] [CrossRef] [PubMed]
- Orlovskii, V.P.; Komlev, V.; Barinov, S.M. Hydroxyapatite and Hydroxyapatite-Based Ceramics. Inorg. Mater. 2002, 38, 973–984. [Google Scholar] [CrossRef]
- Szcześ, A.; Hołysz, L.; Chibowski, E. Synthesis of hydroxyapatite for biomedical applications. Adv. Colloid Interface Sci. 2017, 249, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Kuśnieruk, S.; Wojnarowicz, J.; Chodara, A.; Chudoba, T.; Gierlotka, S.; Lojkowski, W. Influence of hydrothermal synthesis parameters on the properties of hydroxyapatite nanoparticles. Beilstein J. Nanotechnol. 2016, 7, 1586–1601. [Google Scholar] [CrossRef] [Green Version]
- Rao, A.; Swarup, J.S. Enamel surface remineralization: Using synthetic nanohydroxyapatite. Contemp. Clin. Dent. 2012, 3, 433–436. [Google Scholar] [CrossRef] [PubMed]
- Vano, M.; Derchi, G.; Barone, A.; Genovesi, A.; Covani, U. Tooth bleaching with hydrogen peroxide and nano-hydroxyapatite: A 9-month follow-up randomized clinical trial. Int. J. Dent. Hyg. 2015, 13, 301–307. [Google Scholar] [CrossRef]
- Joiner, A.; Luo, W. Tooth colour and whiteness: A review. J. Dent. 2017, 67, S3–S10. [Google Scholar] [CrossRef]
- Tao, D.; Smith, R.N.; Zhang, Q.; Sun, J.N.; Philpotts, C.J.; Ricketts, S.R.; Naeeni, M.; Joiner, A. Tooth whitening evaluation of blue covarine containing toothpastes. J. Dent. 2017, 67, S20–S24. [Google Scholar] [CrossRef]
- Jiang, N.; Zhang, C.; Agingu, C.; Attin, T.; Cheng, H.; Yu, H. Comparison of Whitening Dentifrices on the Effectiveness of In-office Tooth Bleaching: A Double-blind Randomized Controlled Clinical Trial. Oper. Dent. 2019, 44, 138–145. [Google Scholar] [CrossRef]
- Epple, M.; Meyer, F.; Enax, J. A critical Review of Modern Concepts for Teeth Whitening. Dent. J. 2019, 7, 79. [Google Scholar] [CrossRef] [Green Version]
- Coceska, E.; Gjorgievska, E.; Coleman, N.J.; Gabric, D.; Slipper, I.J.; Stevanovic, M.; Nicholson, J.W. Enamel alteration following tooth bleaching and remineralization. J. Microsc. 2015, 262, 232–244. [Google Scholar] [CrossRef] [PubMed]
- Vaez, S.C.; Faria-E-Silva, A.L.; Loguércio, A.D.; Fernandes, M.T.G.; Nahsan, F.P.S. Preemptive use of etodolac on tooth sensitivity after in-office bleaching: A randomized clinical trial. J. Appl. Oral Sci. 2018, 26, e20160473. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lima, S.N.L.; Ribeiro, I.S.; Grisotto, M.A.; Fernandes, E.S.; Hass, V.; Tavarez, R.R.D.J.; Pinto, S.C.S.; Lima, D.M.; Loguercio, A.D.; Bandeca, M.C. Evaluation of several clinical parameters after bleaching with hydrogen peroxide at different concentrations: A randomized clinical trial. J. Dent. 2018, 68, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Santos, L.F.T.F.; Torres, C.R.G.; Caneppele, T.M.F.; Magalhães, A.C.; Borges, A.B. Effect of home-bleaching gels modified by calcium and/or fluoride and the application of nano-hydroxyapatite paste on in vitro enamel erosion susceptibility. Acta Odontol. Scand. 2015, 74, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, R.T.; Catelan, A.; Bertoldo, E.D.S.; Venâncio, P.C.; Groppo, F.C.; Ambrosano, G.M.B.; Marchi, G.M.; Lima, D.A.N.L.; Aguiar, F.H.B. Effect of 7.5% hydrogen peroxide containing remineralizing agents on hardness, color change, roughness and micromorphology of human enamel. Am. J. Dent. 2015, 28, 261–267. [Google Scholar] [PubMed]
- Paravina, R.D.; Pérez, M.M.; Ghinea, R. Acceptability and perceptibility thresholds in dentistry: A comprehensive review of clinical and research applications. J. Esthet. Restor. Dent. 2018, 31, 103–112. [Google Scholar] [CrossRef]
- Lilaj, B.; Dauti, R.; Agis, H.; Schmid-Schwap, M.; Franz, A.; Kanz, F.; Moritz, A.; Schedle, A.; Cvikl, B. Comparison of Bleaching Products With Up to 6% and With More Than 6% Hydrogen Peroxide: Whitening Efficacy Using BI and WID and Side Effects—An in vitro Study. Front. Physiol. 2019, 10, 919. [Google Scholar] [CrossRef]
- Pinto, A.V.D.; Carlos, N.R.; Amaral, F.L.B.D.; França, F.M.G.; Turssi, C.P.; Basting, R.T. At-Home, in-Office and Combined Dental Bleaching Techniques Using Hydrogen Peroxide: Randomized Clinical Trial Evaluation of Effectiveness, Clinical Parameters and Enamel Mineral Content. Am. J. Dent. 2019, 32, 124–132. [Google Scholar]
- Kutuk, Z.B.; Ergin, E.; Cakir, F.Y.; Gurgan, S. Effects of in-office bleaching agent combined with different desensitizing agents on enamel. J. Appl. Oral Sci. 2018, 27, e20180233. [Google Scholar] [CrossRef] [PubMed]
- Vaz, V.T.P.; Jubilato, D.P.; De Oliveira, M.R.M.; Bortolatto, J.F.; Floros, M.C.; Dantas, A.; Junior, O.B.D.O. Whitening toothpaste containing activated charcoal, blue covarine, hydrogen peroxide or microbeads: Which one is the most effective? J. Appl. Oral Sci. 2019, 27, e20180051. [Google Scholar] [CrossRef] [PubMed]
- Shamel, M.; Al-Ankily, M.M.; Bakr, M.M. Influence of different types of whitening tooth pastes on the tooth color, enamel surface roughness and enamel morphology of human teeth. F1000Research 2019, 8, 1764. [Google Scholar] [CrossRef] [PubMed]
- Bergesch, V.; Aguiar, F.H.B.; Turssi, C.P.; França, F.M.G.; Basting, R.T.; Amaral, F.L.B. Shade changing effectiveness of plasdone and blue covarine-based whitening toothpaste on teeth stained with chlorhexidine and black tea. Eur. J. Dent. 2017, 11, 432–437. [Google Scholar] [CrossRef]
- Orsini, G.; Tosco, V.; Monterubbianesi, R.; Orilisi, G.; Putignano, A. A New Era in Restorative Dentistry. In The First Out-Standing 50 Years of “Università Politecnica delle Marche”: Research Achievements in Life Sciences; Longhi, S., Monteriù, A., Freddi, A., Aquilanti, L., Ceravolo, M.G., Carnevali, O., Giordano, M., Moroncini, G., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 319–334. ISBN 978-3-030-33832-9. [Google Scholar]
- Vilhena, K.F.B.; Nogueira, B.C.L.; Fagundes, N.C.F.; Loretto, S.C.; Angelica, R.S.; Lima, R.R.; E Souza, M.H.S. Dental enamel bleached for a prolonged and excessive time: Morphological changes. PLoS ONE 2019, 14, e0214948. [Google Scholar] [CrossRef] [Green Version]
- De Carvalho, A.-C.-G.; De Souza, T.-F.; Liporoni, P.-C.-S.; Pizi, E.-C.-G.; Matuda, L.-S.-D.A.; Catelan, A. Effect of bleaching agents on hardness, surface roughness and color parameters of dental enamel. J. Clin. Exp. Dent. 2020, 12, e670–e675. [Google Scholar] [CrossRef]
- Orilisi, G.; Monterubbianesi, R.; Notarstefano, V.; Tosco, V.; Vitiello, F.; Giuliani, G.; Putignano, A.; Orsini, G. New Insights from Raman MicroSpectroscopy and Scanning Electron Microscopy on the Microstructure and Chemical Composition of Vestibular and Lingual Surfaces in Permanent and Deciduous Human Teeth. Spectrochim Acta A Mol Biomol Spectrosc 2021, 260, 119966. [Google Scholar] [CrossRef]
- Oltu, U.; Gurgan, S. Effects of three concentrations of carbamide peroxide on the structure of enamel. J. Oral Rehabil. 2000, 27, 332–340. [Google Scholar] [CrossRef]
- McGuckin, R.S.; Babin, J.; Meyer, B. Alterations in human enamel surface morphology following vital bleaching. J. Prosthet. Dent. 1992, 68, 754–760. [Google Scholar] [CrossRef]
- Jiang, T.; Ma, X.; Wang, Y.; Tong, H.; Shen, X.; Hu, Y.; Hu, J. Investigation of the effects of 30% hydrogen peroxide on human tooth enamel by Raman scattering and laser-induced fluorescence. J. Biomed. Opt. 2008, 13, 014019. [Google Scholar] [CrossRef] [PubMed]
- Cavalli, V.; Rodrigues, L.K.A.; Paes-Leme, A.F.; Brancalion, M.L.; Arruda, M.A.Z.; Berger, S.B.; Giannini, M. Effects of bleaching agents containing fluoride and calcium on human enamel. Quintessence Int. 2010, 41, 157–165. [Google Scholar]
- Perdigão, J.; Baratieri, L.N.; Arcari, G.M. Contemporary trends and techniques in tooth whitening: A review. Pract. Proced. Aesthet. Dent. 2004, 16, 185–192. [Google Scholar] [PubMed]
- Levrini, L.; Di Benedetto, G.; Raspanti, M. Dental Wear: A Scanning Electron Microscope Study. BioMed Res. Int. 2014, 2014, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Bistey, T.; Nagy, I.P.; Simó, A.; Hegedűs, C. In vitro FT-IR study of the effects of hydrogen peroxide on superficial tooth enamel. J. Dent. 2007, 35, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Bordea, I.R.; Candrea, S.; Alexescu, G.T.; Bran, S.; Băciuț, M.; Băciuț, G.; Lucaciu, O.; Dinu, C.M.; Todea, D.A. Nano-hydroxyapatite use in dentistry: A systematic review. Drug Metab. Rev. 2020, 52, 319–332. [Google Scholar] [CrossRef]
- Onuma, K.; Yamagishi, K.; Oyane, A. Nucleation and growth of hydroxyapatite nanocrystals for nondestructive repair of early caries lesions. J. Cryst. Growth 2005, 282, 199–207. [Google Scholar] [CrossRef]
- Juntavee, N.; Juntavee, A.; Plongniras, P. Remineralization potential of nano-hydroxyapatite on enamel and cementum surrounding margin of computer-aided design and computer-aided manufacturing ceramic restoration. Int. J. Nanomed. 2018, 13, 2755–2765. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Xiao, Z.; Yang, J.; Lu, D.; Kishen, A.; Li, Y.; Chen, Z.; Que, K.; Zhang, Q.; Deng, X.; et al. Oriented and Ordered Biomimetic Remineralization of the Surface of Demineralized Dental Enamel Using HAP@ACP Nanoparticles Guided by Glycine. Sci. Rep. 2017, 7, 40701. [Google Scholar] [CrossRef] [Green Version]
- Kwon, S.R.; Wertz, P.W. Review of the Mechanism of Tooth Whitening. J. Esthet. Restor. Dent. 2015, 27, 240–257. [Google Scholar] [CrossRef]
- Pérez, M.M.; Herrera, L.J.; Carrillo, F.; Pecho, O.E.; Dudea, D.; Gasparik, C.; Ghinea, R.; Della Bona, A. Whiteness difference thresholds in dentistry. Dent. Mater. 2019, 35, 292–297. [Google Scholar] [CrossRef]

| Experimental Group | Manufacturer | Chemical Composition | Exposure Time |
|---|---|---|---|
| 6HP | White Dental Beauty, Novon, Optident Ilkley, West Yorkshire, UK | 6% hydrogen peroxide | 50 min/day for 7 days |
| 6HP-nHA | BioWhiten, Biodent Ltd., Istanbul, Turkey | water, glycerin, alcohol, sodium bicarbonate, sodium hydroxide, 6% hydrogen peroxide and nano hydroxyapatite * (ratio 1:3) | 50 min/day for 7 days |
| CTR | 6HP | 6HP-nHA | ||||
|---|---|---|---|---|---|---|
| t0 | t7 | t0 | t7 | t0 | t7 | |
| L* | 65.60 ± 6.59 | 67.59 ± 6.03 | 64.07 ± 2.88 | 74.44 ± 3.31 | 64.91 ± 2.85 | 75.88 ± 3.26 |
| a* | 3.56 ± 2.26 | 3.76 ± 2.02 | 3.62 ± 0.39 | 1.74 ± 0.96 | 2.73 ± 1.17 | 1.28 ± 0.65 |
| b* | 20.83 ± 3.00 | 17.57 ± 3.48 | 22.69 ± 2.10 | 14.29 ± 2.35 | 19.02 ± 3.49 | 11.25 ± 2.73 |
| CTR | 6HP | 6HP-nHA | |
|---|---|---|---|
| ΔE*ab | 5.80 ± 2.34 a | 14.09 ± 2.52 b | 14.06 ± 2.90 b |
| ΔL* | 2.21 ± 3.69 a | 10.37 ± 2.52 b | 10.97± 3.48 b |
| Δa* | 0.20 ± 1.03 a | −1.88 ± 1.05 a | −1.45 ± 0.82 a |
| Δb* | −2.78 ± 3.70 a | −8.4 ± 2.47 b | −7.63 ± 2.76 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Monterubbianesi, R.; Tosco, V.; Bellezze, T.; Giuliani, G.; Özcan, M.; Putignano, A.; Orsini, G. A Comparative Evaluation of Nanohydroxyapatite-Enriched Hydrogen Peroxide Home Bleaching System on Color, Hardness and Microstructure of Dental Enamel. Materials 2021, 14, 3072. https://0-doi-org.brum.beds.ac.uk/10.3390/ma14113072
Monterubbianesi R, Tosco V, Bellezze T, Giuliani G, Özcan M, Putignano A, Orsini G. A Comparative Evaluation of Nanohydroxyapatite-Enriched Hydrogen Peroxide Home Bleaching System on Color, Hardness and Microstructure of Dental Enamel. Materials. 2021; 14(11):3072. https://0-doi-org.brum.beds.ac.uk/10.3390/ma14113072
Chicago/Turabian StyleMonterubbianesi, Riccardo, Vincenzo Tosco, Tiziano Bellezze, Giampaolo Giuliani, Mutlu Özcan, Angelo Putignano, and Giovanna Orsini. 2021. "A Comparative Evaluation of Nanohydroxyapatite-Enriched Hydrogen Peroxide Home Bleaching System on Color, Hardness and Microstructure of Dental Enamel" Materials 14, no. 11: 3072. https://0-doi-org.brum.beds.ac.uk/10.3390/ma14113072









