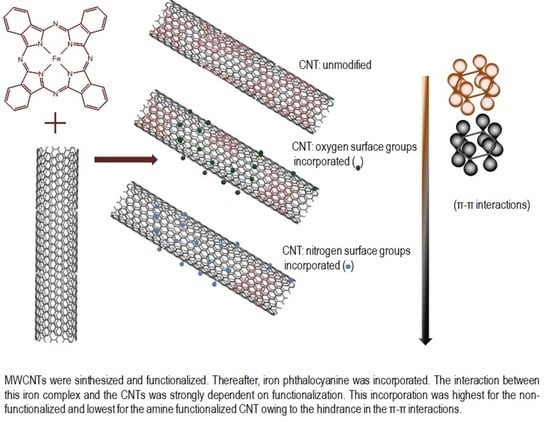
Study of the Interaction of an Iron Phthalocyanine Complex over Surface Modified Carbon Nanotubes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthesis of Carbon Materials
2.2. Synthesis of Composities
2.3. Characterisation
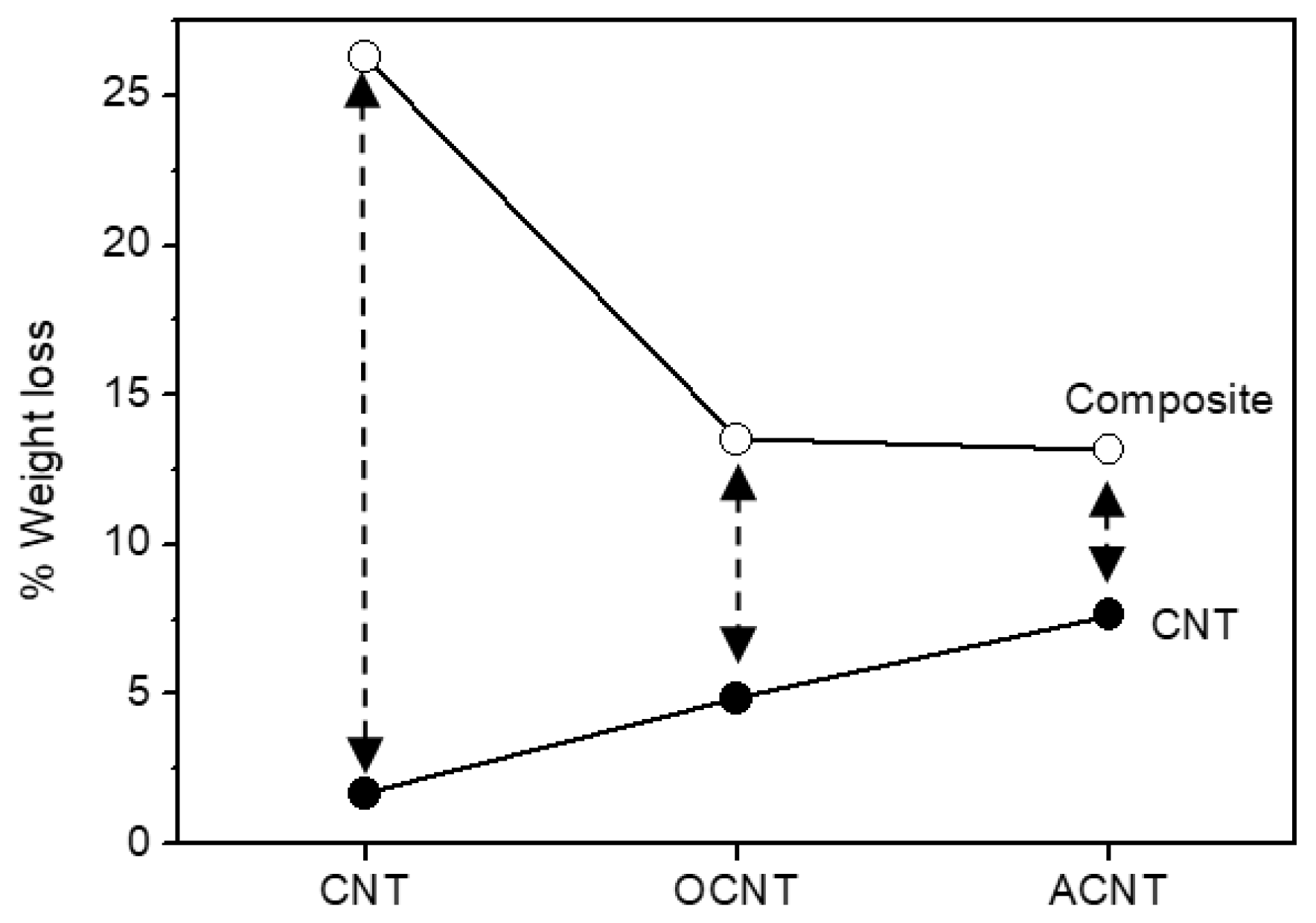
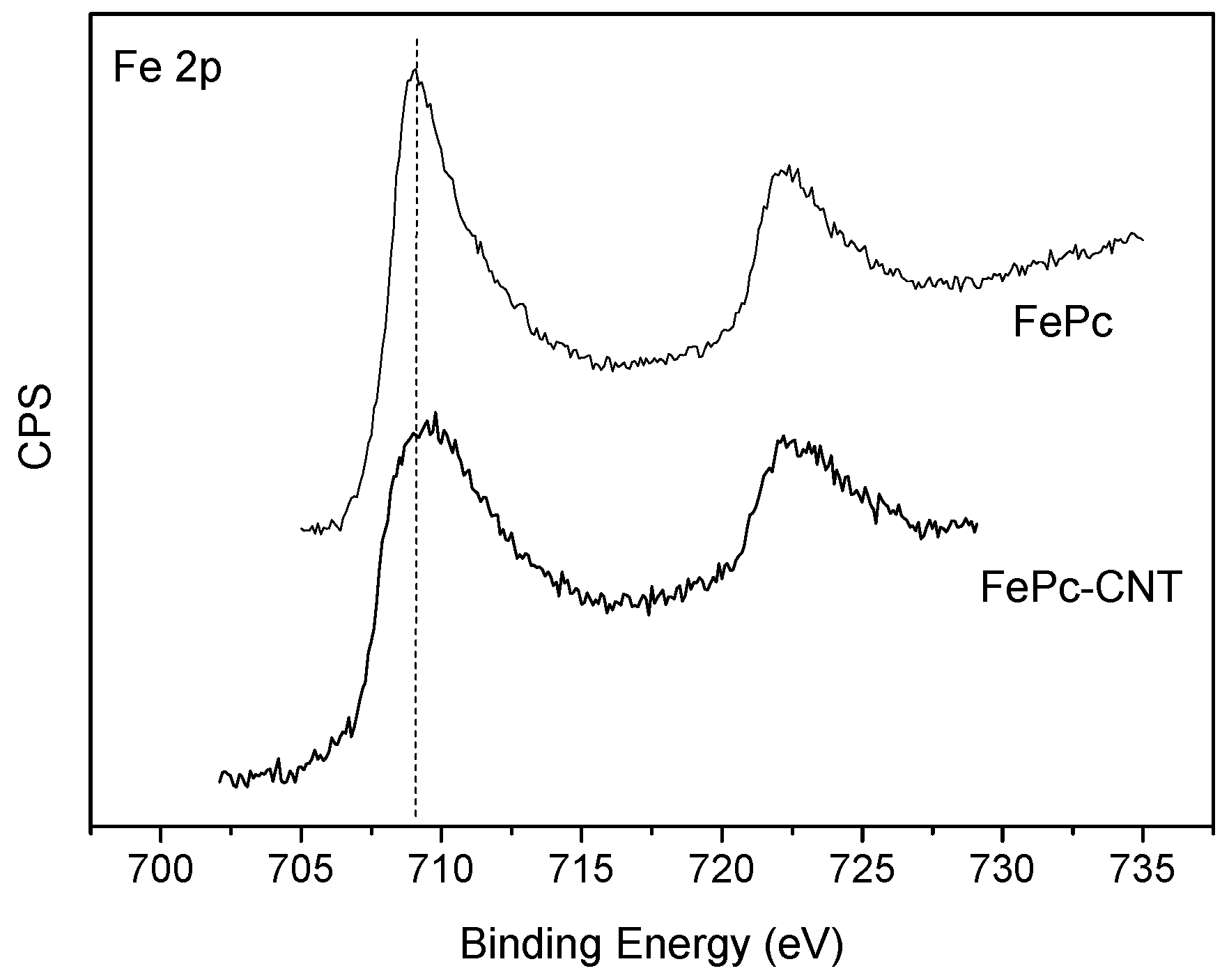
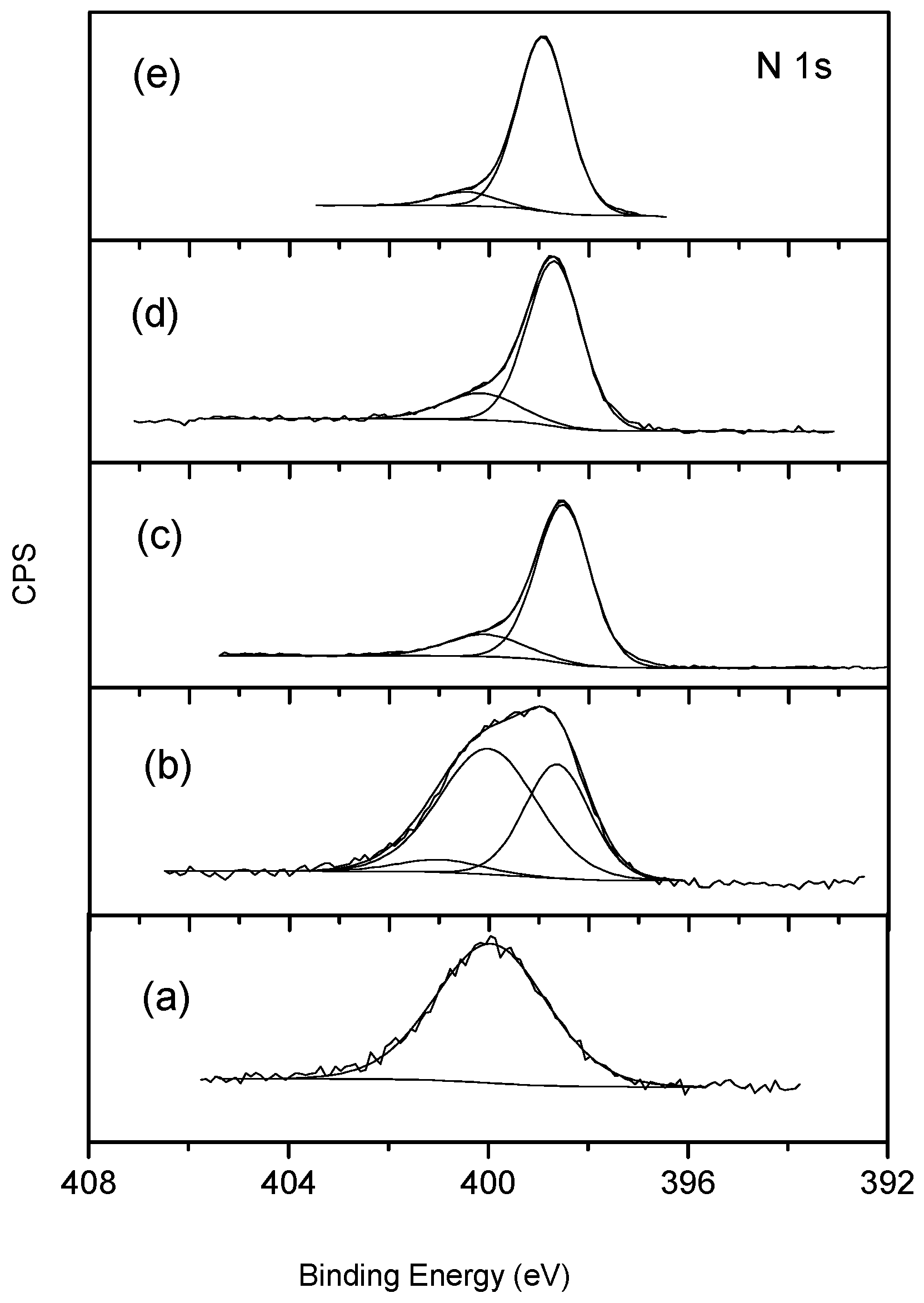
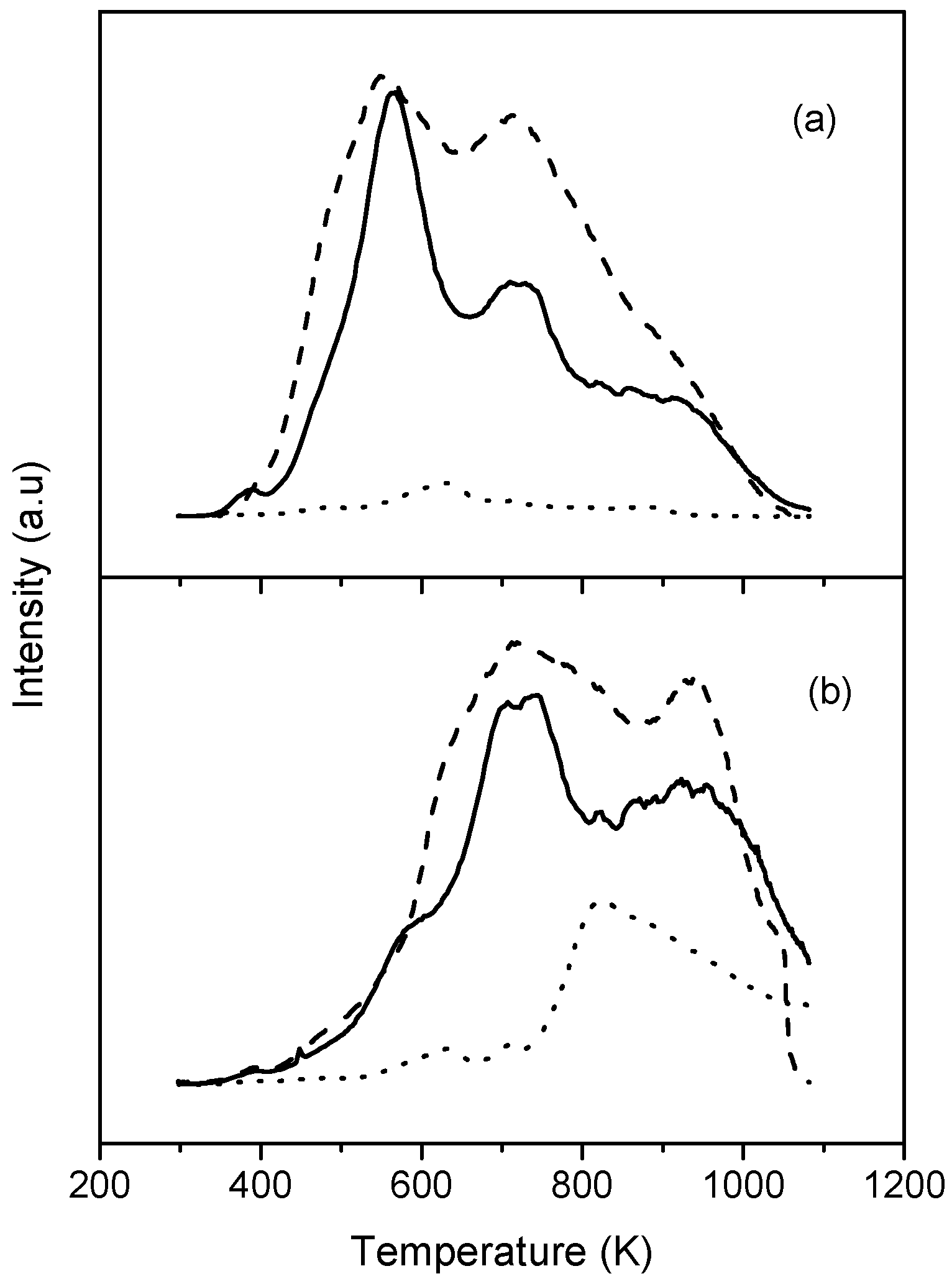
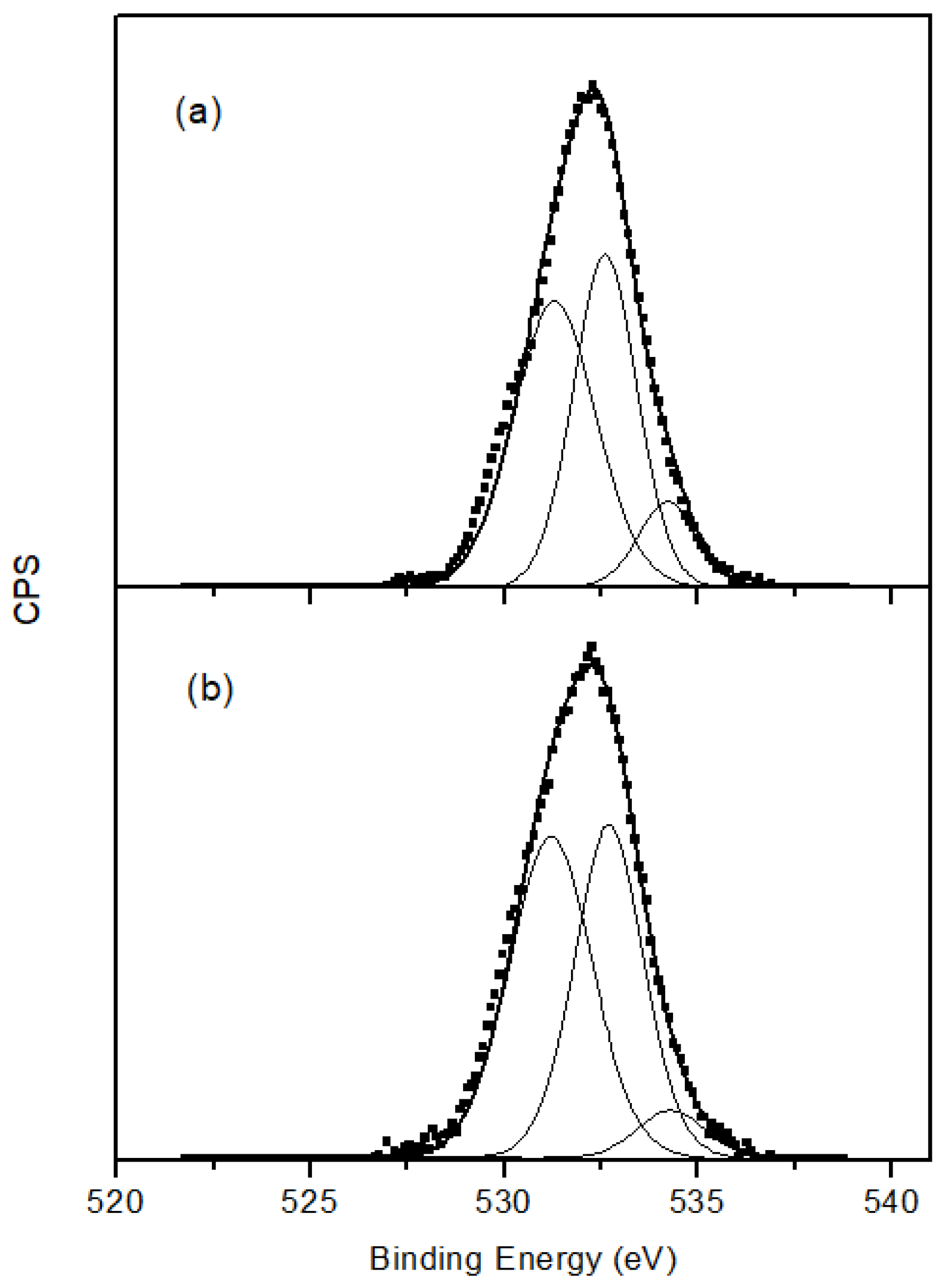
3. Results and Discussion
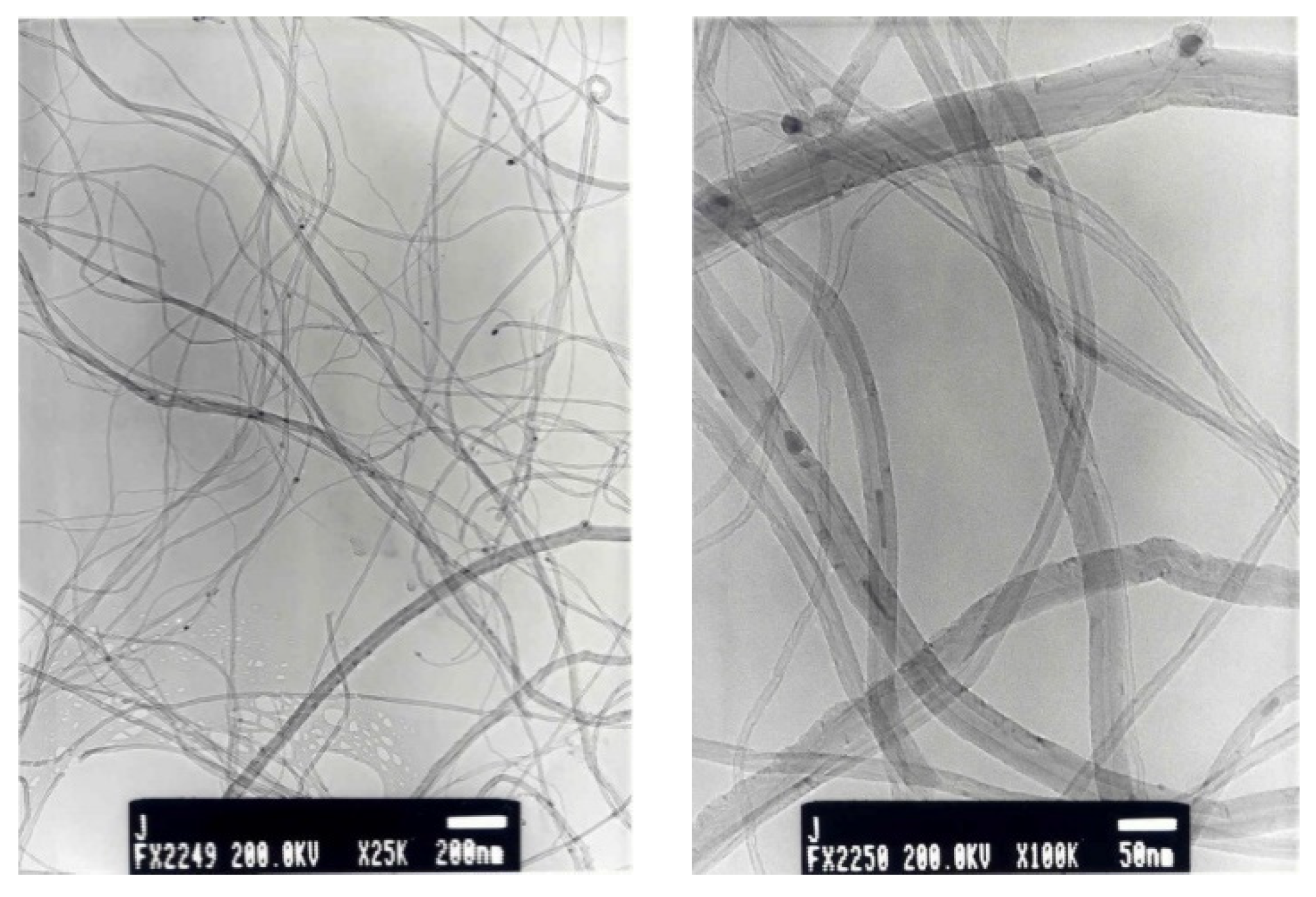
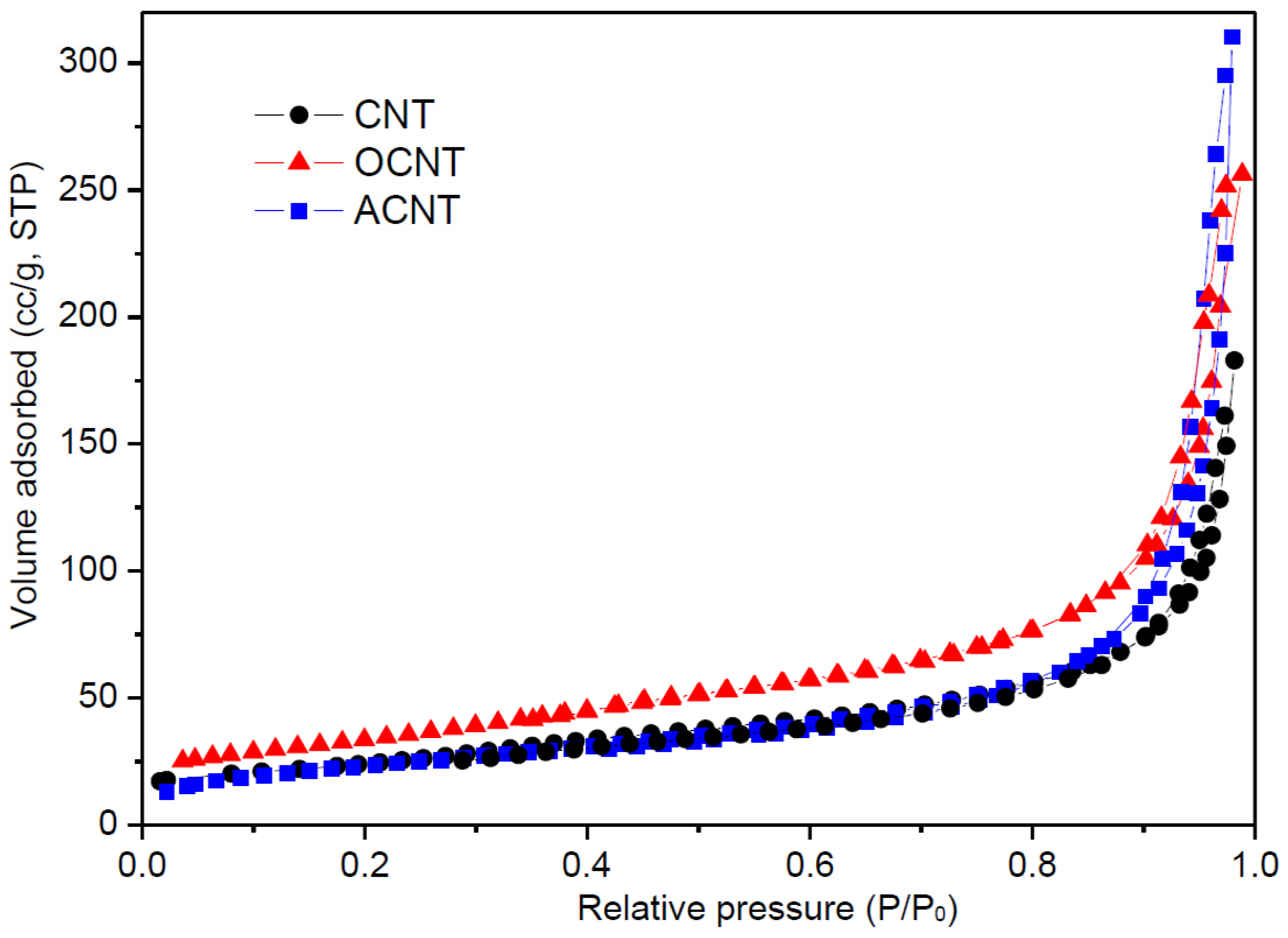
3.1. Carbon Nanotubes
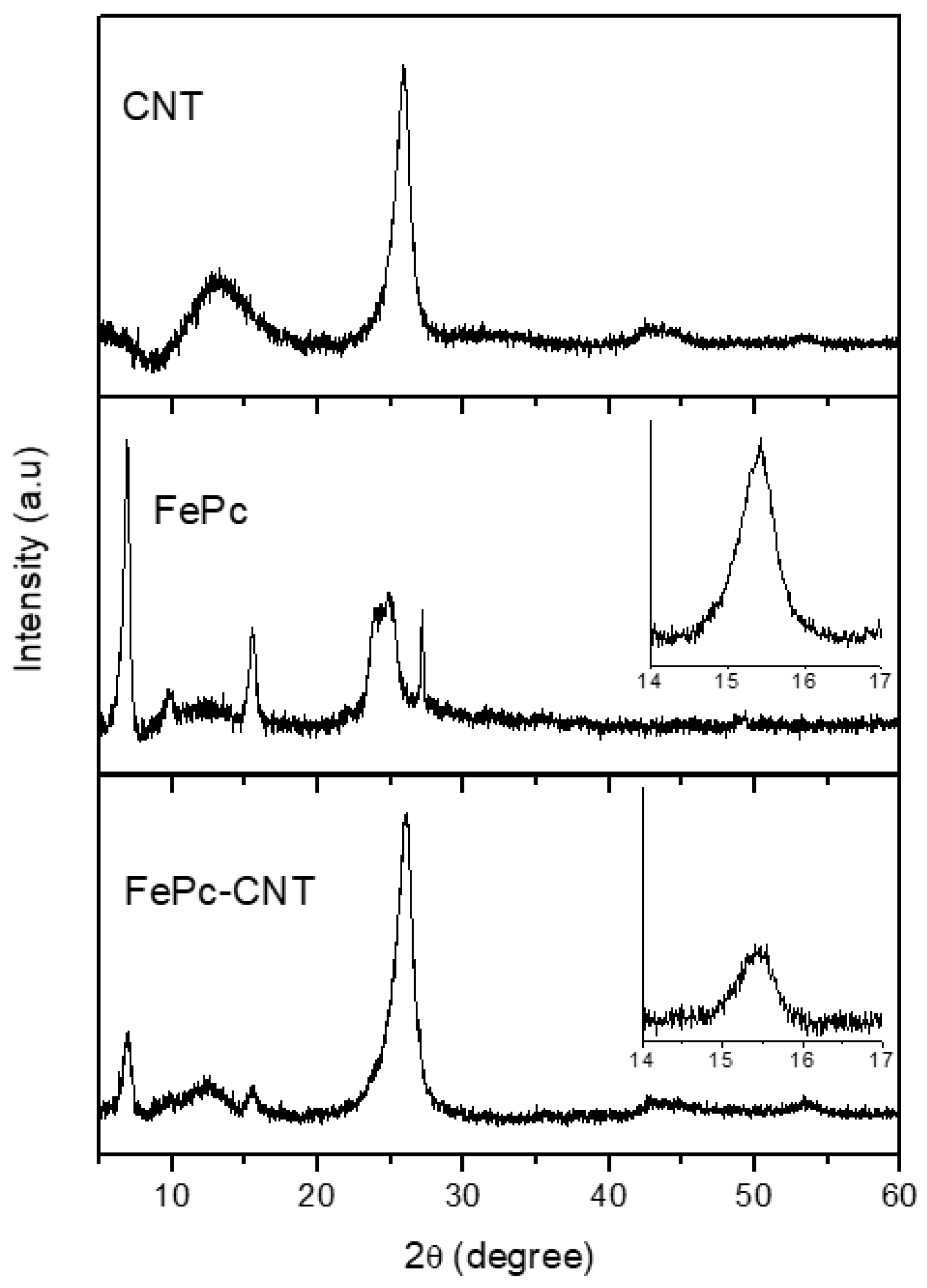
3.2. FePc-CNT Composites
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- De Oliveira, G.A.R.; Leme, D.M.; de Lapuente, J.; Brito, L.B.; Porredón, C.; Rodrigues, L.B.; Brull, N.; Serret, J.T.; Borràs, M.; Disner, G.R.; et al. A test battery for assessing the ecotoxic effects of textile dyes. Chem. Biol. Interact. 2018, 291, 171–179. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, X.; Zhang, M.; Chen, Z.; Bu, Y.; Gao, X.; Sui, Y.; Yu, Y. Sodium Alginate Micelle-Encapsulating Zinc Phthalocyanine Dye-Sensitized Photoelectrochemical Biosensor with CdS as the Photoelectric Material for Hg2+ Detection. ACS Appl. Mater. Interfaces 2021, 13, 16828–16836. [Google Scholar] [CrossRef]
- Günay, I.; Orman, E.B.; Altındal, A.; Salih, B.; Özer, M.; Özkaya, A.R. Novel tetrakis 4-(hydroxymethyl)-2,6-dimethoxyphenoxyl substituted metallophthalocyanines: Synthesis, electrochemical redox, electrocatalytic oxygen reducing, and volatile organic compounds sensing and adsorption properties. Dyes Pigm. 2018, 154, 72–187. [Google Scholar] [CrossRef]
- Rydosz, A.; Maciak, E.; Wincza, K.; Gruszczynski, S. Microwave-based sensors with phthalocyanine films for acetone, ethanol and methanol detection. Sens. Actuators B Chem. 2016, 237, 439. [Google Scholar] [CrossRef]
- Brown, M.D.; Schoenfisch, M.H. Catalytic selectivity of metallophthalocyanines for electrochemical nitric oxide sensing. Electrochim. Acta 2018, 273, 98–104. [Google Scholar] [CrossRef]
- Dao, Q.D.; Fujii, A.; Tsuji, R.; Kateoka, Y. Efficiency enhancement in perovskite solar cell utilizing solution-processable phthalocyanine hole transport layer with thermal annealing. Org. Electron. 2017, 43, 156–161. [Google Scholar] [CrossRef]
- Azzouzi, S.; Ben Ali, M.; Abbas, M.N.; Bausells, J.; Zine, N.; Errachid, A. Novel iron (III) phthalocyanine derivative functionalized semiconductor based transducers for the detection of citrate. Org. Electron. 2016, 34, 200–207. [Google Scholar] [CrossRef]
- Paraboschi, I.; Turnock, S.; Kramer-Marek, G.; Musleh, L.; Barisa, M.; Anderson, J.; Giuliani, S. Near-InfraRed PhotoImmunoTherapy (NIR-PIT) for the local control of solid cancers: Challenges and potentials for human applications. Crit. Rev. Oncol. Hematol. 2021, 161, 103325. [Google Scholar] [CrossRef]
- Medina, W.S.G.; Santos, N.A.G.; Curti, C.; Tedesco, A.C.; Santos, A.C. Effects of zinc phthalocyanine tetrasulfonate-based photodynamic therapy on rat brain isolated mitochondria. Chem. Biol. Interact. 2009, 179, 402–406. [Google Scholar] [CrossRef]
- Zorlu, Y.; Dumoulin, F.; Bouchu, D.; Ahsen, V.; Lafont, D. Monoglycoconjugated water-soluble phthalocyanines. Design and synthesis of potential selectively targeting PDT photosensitisers. Tetrahedron Lett. 2010, 51, 6615–6618. [Google Scholar] [CrossRef]
- Leznoff, C.C.; Lever, A.B.P. Advanced Materials, Phthalocyanines: Properties and Applications; VCH: Weinheim, Germany, 1993; Volume 3, p. 303. [Google Scholar] [CrossRef]
- Bilgiçli, A.T.; Yaraşir, M.N.; Kandaz, M.; Özkaya, A.R. H-type aggregation of functional metal ion sensing phthalocyanines: Syn-thesis, characterization and electrochemistry. Polyhedron 2010, 29, 2498–2512. [Google Scholar] [CrossRef]
- Günsel, A.; Bilgiçli, A.T.; Pişkin, H.; Delibaş, N.C.; Yarasir, M.N.; Gündüz, B. Synthesis, characterization, and optical and surface properties of (4-(trifluoromethylthio)phenoxy) copper(II) phthalocyanine. New J. Chem. 2018, 42, 6013–6022. [Google Scholar] [CrossRef]
- Hashimoto, K.; Toukai, N. Removal of NOx with cobalt phthalocyanine supported on Y-type faujasite and alumina. J. Mol. Catal. 2003, 195, 275–282. [Google Scholar] [CrossRef]
- Valente, A.; Palma, C.; Fonseca, I.M.; Ramos, A.M.; Vital, J. Oxidation of pinane over phthalocyanine complexes supported on activated carbon: Effect of the support surface treatment. Carbon 2003, 41, 2793–2803. [Google Scholar] [CrossRef]
- Fedoruk, G.G.; Sagaidak, D.I.; Misevich, A.V.; Pochtenny, A.E. Electrical and gas sensing properties of copper phthalocyanine-polymer composites. Sens. Actuators B Chem. 1998, 48, 351–355. [Google Scholar] [CrossRef]
- Jerkovic, A.; Abou-Ahmed, S.; Ertl, P.; StoeSSl, B.; Lengauer, V.; Samphao, A.; Kalcher, K.; Leitinger, G.; Wernitznig, S.; Ortner, A. Development of a cobalt(II) phthalocyanine- MWCNT modified carbon paste electrode for the detection of polyunsaturated fatty acids. Anal. Chim. Acta 2018, 1038, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Moraes, F.C.; Golinelli, D.L.C.; Mascaro, L.H.; Machado, S.A.S. Determination of epinephrine in urine using multi-walled carbon nanotube modified with cobalt phthalocyanine in a paraffin composite electrode. Sens. Actuators B Chem. 2010, 148, 492–497. [Google Scholar] [CrossRef]
- Yin, H.; Zhou, Y.; Xu, J.; Ai, S.; Cui, L.; Zhu, L. Amperometric biosensor based on tyrosinase immobilized onto multiwalled carbon nanotubes-cobalt phthalocyanine-silk fibroin film and its application to determine bisphenol A. Anal. Chim. Acta 2010, 659, 144–150. [Google Scholar] [CrossRef]
- Baba, A.; Kanetsuna, Y.; Sriwichai, S.; Ohdaira, Y.; Shinbo, K.; Kato, K.; Phanichphant, S.; Kaneko, F. Nanostructured carbon nano-tubes/copper phthalocyanine hybrid multilayers prepared using layer-by-layer self-assembly approach. Thin Solid Films 2010, 518, 2200–2205. [Google Scholar] [CrossRef]
- Alzate-Carvajal, N.; Bolivar-Pineda, L.M.; Meza-Laguna, V.; Basiuk, V.A.; Basiuk, E.V.; Baranova, E.A. Oxygen Evolution Reaction on Single-Walled Carbon Nanotubes Noncovalently Functionalized with Metal Phthalocyanines. ChemElectroChem 2020, 7, 428–436. [Google Scholar] [CrossRef]
- Morozan, A.; Campidelli, S.; Filoramo, A.; Jousselme, B.; Palacin, S. Catalytic activity of cobalt and iron phthalocyanines or porphyrins supported on different carbon nanotubes towards oxygen reduction reaction. Carbon 2011, 49, 4839–4847. [Google Scholar] [CrossRef]
- Sherrington, D.C.; Kybett, A.P.; McKeown, N.B.; Li, H.; Makhseed, S. Supported catalysts and their applications. Towards phthalocyanine network polymers for heterogeneous catalysis. R. Soc. Chem. 2001, 214–218. [Google Scholar] [CrossRef]
- Hu, J.; Liu, H.; Wang, L.; Li, N.; Xu, T.; Lu, W.; Zhu, Z.; Chen, W. Electronic properties of carbon nanotubes linked covalently with iron phthalocyanine to determine the formation of high-valent iron intermediates or hydroxyl radicals. Carbon 2016, 100, 408–416. [Google Scholar] [CrossRef]
- González-Gaitán, C.; Ruiz-Rosas, R.; Morallon, E.; Cazorla-Amoros, D. The relevance of the interaction between the M-phthalocyanines and Carbon Nanotubes in the electroactivity towards ORR. Langmuir 2017, 33, 11945–11955. [Google Scholar] [CrossRef]
- Silva, N.; Castro-Castillo, C.; Oyarzún, M.P.; Ramírez, S.; Gutierrez-Ceron, C.; Marco, J.F.; Silva, J.F.; Zagal, J.H. Modulation of the electrocatalytic activity of Fe phthalocyanine to carbon nanotubes: Electrochemistry of l-cysteine and l-cystine. Electrochim. Acta 2019, 308, 295–306. [Google Scholar] [CrossRef]
- Brunauer, S.; Deming, L.S.; Deming, W.E.; Teller, E. On a Theory of the van der Waals Adsorption of Gases. J. Am. Chem. Soc. 1940, 62, 1723–1732. [Google Scholar] [CrossRef]
- Rouquerol, F.; Rouquerol, J.; Sing, K. Adsorption by Powders and Porous Solids; Academic Press: Cambridge, MA, USA, 1999. [Google Scholar]
- Porro, S.; Musso, S.; Vinante, M.; Vanzetti, L.; Anderle, M.; Trotta, F.; Tagliaferro, A. Purification of carbon nanotubes grown by thermal CVD. Physica E Low Dimens. Syst. Nanostruct. 2007, 37, 58–61. [Google Scholar] [CrossRef]
- Shah, K.A.; Tali, B.A. Synthesis of carbon nanotubes by catalytic chemical vapour deposition: A review on carbon sources, catalysts and substrates. Mater. Sci. Semicond. Process 2016, 41, 67–82. [Google Scholar] [CrossRef]
- Yan, X.; Xu, X.; Zhong, Z.; Liu, J.; Tian, X.; Kang, L.; Yao, J. The effect of oxygen content of carbon nanotubes on the catalytic activity of carbon-based iron phthalocyanine for oxygen reduction reaction. Electrochim. Acta 2018, 281, 562–570. [Google Scholar] [CrossRef]
- Wang, B.; Gao, F.; Ma, H. Preparation and XPS studies of macromolecule mixed-valent Cu(I, II) and Fe(II, III) complexes. J. Hazard. Mater. 2007, 144, 363–368. [Google Scholar] [CrossRef]
- Jansen, R.J.J.; van Bekkum, H. XPS of nitrogen-containing functional groups on activated carbon. Carbon 1995, 33, 1021–1027. [Google Scholar] [CrossRef]
- Pantea, D.; Darmstadt, H.; Kaliaguine, S.; Sümmchen, L.; Roy, C. Electrical conductivity of thermal carbon blacks—Influence of surface chemistry. Carbon 2001, 39, 1147–1158. [Google Scholar] [CrossRef]
- Lee, D.Y.; Lee, M.H.; Kim, K.J.; Heo, S.; Kim, B.Y.; Lee, S.J. Effect of multiwalled carbon nanotube (M-CNT) loading on M-CNT distribution behavior and the related electromechanical properties of the M-CNT dispersed ionomeric nanocomposites. Surf. Coat. Technol. 2005, 200, 1920–1925. [Google Scholar] [CrossRef]
- Hsieh, C.T.; Wei, J.L.; Lin, J.Y.; Chen, W.Y. Hydrogenation and dehydrogenation of Mg2Co nanoparticles and carbon nanotube composites. J. Power Sources 2008, 183, 92–97. [Google Scholar] [CrossRef]
- Kumar, A.; Yasin, G.; Koraib, R.M.; Slimani, Y.; Ali, M.F.; Tabish, M.; Nazir, M.T.; Nguyen, T.A. Boosting oxygen reduction reaction activity by incorporating the iron phthalocyanine nanoparticles on carbon nanotubes network. Inorg. Chem. Commun. 2020, 120, 108160. [Google Scholar] [CrossRef]
- Harnisch, F.; Savastenko, N.A.; Zhao, F.; Steffen, H.; Brüser, V.; Schröder, U. Comparative study on the performance of pyrolyzed and plasma-treated iron(II) phthalocyanine-based catalysts for oxygen reduction in pH neutral electrolyte solutions. J. Power Sources 2009, 193, 86–92. [Google Scholar] [CrossRef]
- Wang, S.; Song, X.L. Reduced graphene oxide-carbon black composite supported iron phthalocyanine as a high-efficiency and stable catalyst for oxygen. Dyes Pigm. 2021, 189, 109254. [Google Scholar] [CrossRef]
- Basiuk, E.V.; Huerta, L.; Basiuk, V.A. Noncovalent bonding of 3d metal(II) phthalocyanines with single-walled carbon nanotubes: A combined DFT and XPS study. Appl. Surf. Sci. 2019, 470, 622–630. [Google Scholar] [CrossRef]
- Chávez-Colorado, E.; Basiuk, A. Noncovalent interactions of free-base phthalocyanine with elongated fullerenes as carbon nanotube models. Struct. Chem. 2017, 28, 1765–1773. [Google Scholar] [CrossRef]
- Jiang, Y.; Lu, Y.; Lv, X.; Han, D.; Zhang, Q.; Niu, L.; Chen, W. Enhanced Catalytic Performance of Pt-Free Iron Phthalocyanine by Graphene Support for Efficient Oxygen Reduction Reaction. ACS Catal. 2013, 3, 1263–1271. [Google Scholar] [CrossRef]
| Sample | C 1s | O 1s | N 1s |
|---|---|---|---|
| CNT | 97.50 | 2.36 | 0.00 |
| OCNT | 92.16 | 7.69 | 0.00 |
| ACNT | 93.74 | 4.08 | 2.12 |
| FePc-CNT | 90.28 | 2.98 | 6.32 |
| FePc-OCNT | 89.94 | 5.66 | 4.03 |
| FePc-ACNT | 92.40 | 4.62 | 2.91 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pérez-Cadenas, M.; Asedegbega-Nieto, E.; Carter, J.; Anderson, J.A.; Rodríguez-Ramos, I.; Guerrero-Ruiz, A. Study of the Interaction of an Iron Phthalocyanine Complex over Surface Modified Carbon Nanotubes. Materials 2021, 14, 4067. https://0-doi-org.brum.beds.ac.uk/10.3390/ma14154067
Pérez-Cadenas M, Asedegbega-Nieto E, Carter J, Anderson JA, Rodríguez-Ramos I, Guerrero-Ruiz A. Study of the Interaction of an Iron Phthalocyanine Complex over Surface Modified Carbon Nanotubes. Materials. 2021; 14(15):4067. https://0-doi-org.brum.beds.ac.uk/10.3390/ma14154067
Chicago/Turabian StylePérez-Cadenas, María, Esther Asedegbega-Nieto, Jonathan Carter, James A. Anderson, Inmaculada Rodríguez-Ramos, and Antonio Guerrero-Ruiz. 2021. "Study of the Interaction of an Iron Phthalocyanine Complex over Surface Modified Carbon Nanotubes" Materials 14, no. 15: 4067. https://0-doi-org.brum.beds.ac.uk/10.3390/ma14154067