Chimeric Capsid Proteins Impact Transduction Efficiency of Haploid Adeno-Associated Virus Vectors
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plasmids and Site-Directed Mutagenesis
2.2. Virus Production
2.3. In Vitro Transduction Assay
2.4. Animal Study
2.5. Quantitation of Luciferase Expression in Mouse Tissue
2.6. Detection of AAV Genome Copy Number in Mouse Tissue
2.7. Immune-Blot of Heating or pH-Treated AAV Particles
2.8. Statistical Analysis
3. Results
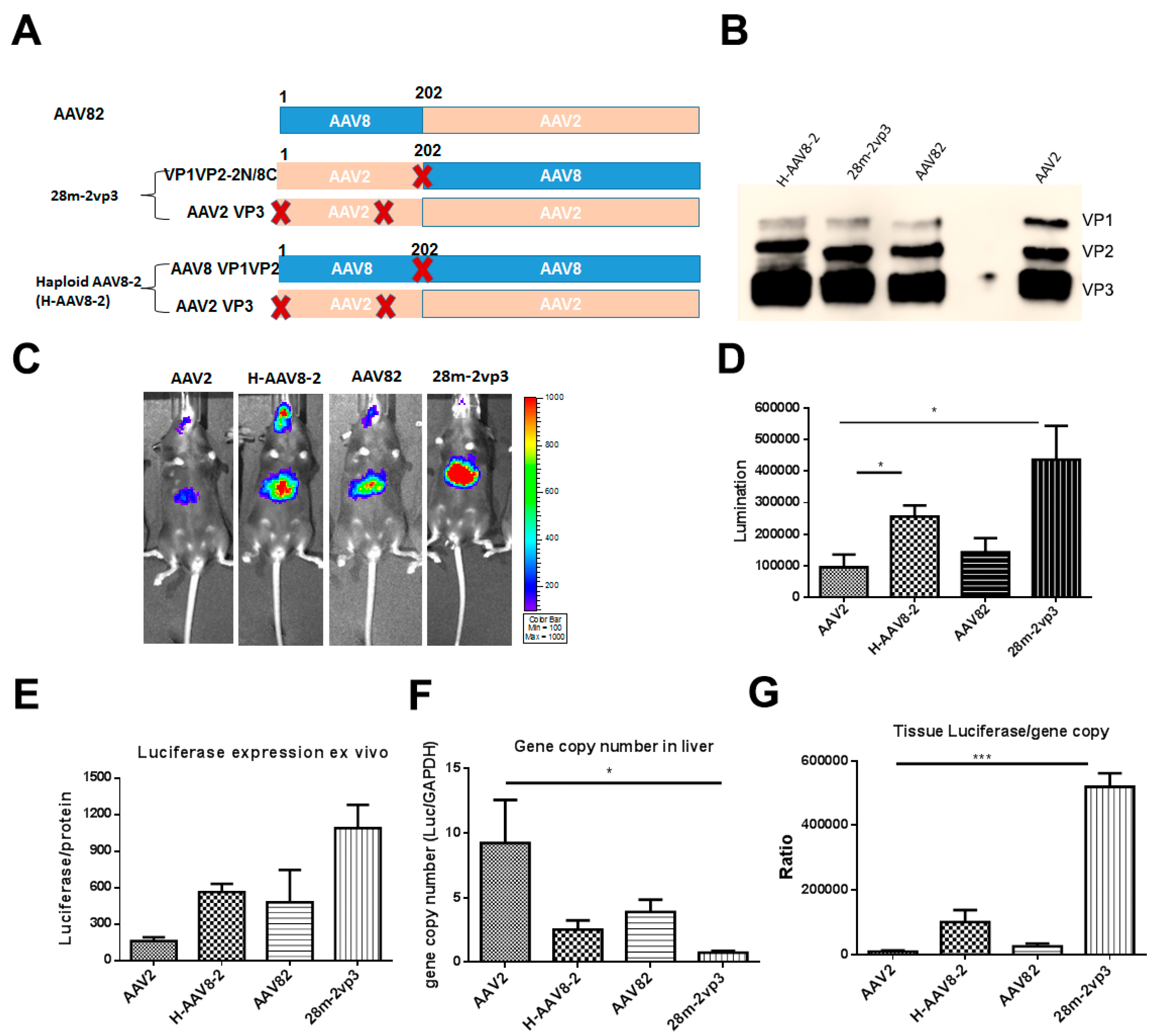
3.1. Haploid Vectors with VP3 of AAV2 and VP1/VP2 of Other Serotypes Enhanced the Liver Transduction of AAV2
3.2. Haploid Vector with Chimeric VP1/VP2 further Enhanced AAV2 Liver Transduction
3.3. Haploid Vectors with the VP3 of AAV3 and VP1/VP2 of AAV8 or 9 Enhanced the Liver Transduction
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Srivastava, A.; Lusby, E.W.; Berns, K.I. Nucleotide sequence and organization of the adeno-associated virus 2 genome. J. Virol. 1983, 45, 555–564. [Google Scholar]
- Srivastava, A. In vivo tissue-tropism of adeno-associated viral vectors. Curr. Opin. Virol. 2016, 21, 75–80. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manno, C.S.; Chew, A.J.; Hutchison, S.; Larson, P.J.; Herzog, R.W.; Arruda, V.R.; Tai, S.J.; Ragni, M.V.; Thompson, A.; Ozelo, M.; et al. AAV-mediated factor IX gene transfer to skeletal muscle in patients with severe hemophilia B. Blood 2003, 101, 2963–2972. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lisowski, L.; Tay, S.S.; Alexander, I.E. Adeno-associated virus serotypes for gene therapeutics. Curr. Opin. Pharmacol. 2015, 24, 59–67. [Google Scholar] [CrossRef]
- Boye, S.E.; Boye, S.L.; Lewin, A.S.; Hauswirth, W.W. A Comprehensive Review of Retinal Gene Therapy. Mol. Ther. 2013, 21, 509–519. [Google Scholar] [CrossRef] [Green Version]
- Nathwani, A.C.; Reiss, U.M.; Tuddenham, E.G.D.; Rosales, C.; Chowdary, P.; McIntosh, J.; Della Peruta, M.; Lheriteau, E.; Patel, N.; Raj, D.; et al. Long-Term Safety and Efficacy of Factor IX Gene Therapy in Hemophilia B. N. Engl. J. Med. 2014, 371, 1994–2004. [Google Scholar] [CrossRef] [Green Version]
- Nathwani, A.C.; Tuddenham, E.G.D.; Rangarajan, S.; Rosales, C.; McIntosh, J.; Linch, D.C.; Chowdary, P.; Riddell, A.; Pie, A.J.; Harrington, C.; et al. Adenovirus-Associated Virus Vector–Mediated Gene Transfer in Hemophilia B. N. Engl. J. Med. 2011, 365, 2357–2365. [Google Scholar] [CrossRef]
- Simioni, P.; Tormene, D.; Tognin, G.; Gavasso, S.; Bulato, C.; Iacobelli, N.P.; Finn, J.D.; Spiezia, L.; Radu, C.; Arruda, V.R. X-Linked Thrombophilia with a Mutant Factor IX (Factor IX Padua). N. Engl. J. Med. 2009, 361, 1671–1675. [Google Scholar] [CrossRef]
- Saraiva, J.; Nobre, R.J.; Pereira de Almeida, L. Gene therapy for the CNS using AAVs: The impact of systemic delivery by AAV9. J. Controll. Release 2016, 241, 94–109. [Google Scholar] [CrossRef]
- Smalley, E. First AAV gene therapy poised for landmark approval. Nat. Biotechnol. 2017, 35, 998. [Google Scholar] [CrossRef]
- Chai, Z.; Sun, J.; Rigsbee, K.M.; Wang, M.; Samulski, R.J.; Li, C. Application of polyploid adeno-associated virus vectors for transduction enhancement and neutralizing antibody evasion. J. Controll. Release 2017, 262, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Jin, D.Y.; Zhang, T.P.; Gui, T.; Stafford, D.W.; Monahan, P.E. Creation of a mouse expressing defective human factor IX. Blood 2004, 104, 1733–1739. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shen, S.; Bryant, K.D.; Brown, S.M.; Randell, S.H.; Asokan, A. Terminal N-Linked Galactose Is the Primary Receptor for Adeno-associated Virus 9. J. Biol. Chem. 2011, 286, 13532–13540. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shen, S.; Horowitz, E.D.; Troupes, A.N.; Brown, S.M.; Pulicherla, N.; Samulski, R.J.; Agbandje-McKenna, M.; Asokan, A. Engraftment of a galactose receptor footprint onto adeno-associated viral capsids improves transduction efficiency. J. Biol. Chem. 2013, 288, 28814–28823. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rabinowitz, J.E.; Bowles, D.E.; Faust, S.M.; Ledford, J.G.; Cunningham, S.E.; Samulski, R.J. Cross-dressing the virion: The transcapsidation of adeno-associated virus serotypes functionally defines subgroups. J. Virol. 2004, 78, 4421–4432. [Google Scholar] [CrossRef] [Green Version]
- Grieger, J.C.; Johnson, J.S.; Gurda-Whitaker, B.; Agbandje-McKenna, M.; Samulski, R.J. Surface-exposed adeno-associated virus Vp1-NLS capsid fusion protein rescues infectivity of noninfectious wild-type Vp2/Vp3 and Vp3-only capsids but not that of fivefold pore mutant virions. J. Virol. 2007, 81, 7833–7843. [Google Scholar] [CrossRef] [Green Version]
- Girod, A.; Wobus, C.E.; Zádori, Z.; Ried, M.; Leike, K.; Tijssen, P.; Kleinschmidt, J.A.; Hallek, M. The VP1 capsid protein of adeno-associated virus type 2 is carrying a phospholipase A2 domain required for virus infectivity. J. Gen. Virol. 2002, 83, 973–978. [Google Scholar] [CrossRef] [Green Version]
- Muralidhar, S.; Becerra, S.P.; Rose, J.A. Site-directed mutagenesis of adeno-associated virus type 2 structural protein initiation codons: Effects on regulation of synthesis and biological activity. J. Virol. 1994, 68, 170–176. [Google Scholar]
- Ruffing, M.; Zentgraf, H.; Kleinschmidt, J.A. Assembly of viruslike particles by recombinant structural proteins of adeno-associated virus type 2 in insect cells. J. Virol. 1992, 66, 6922–6930. [Google Scholar]
- Warrington, K.H.; Gorbatyuk, O.S.; Harrison, J.K.; Opie, S.R.; Zolotukhin, S.; Muzyczka, N. Adeno-Associated Virus Type 2 VP2 Capsid Protein Is Nonessential and Can Tolerate Large Peptide Insertions at Its N Terminus. J. Virol. 2004, 78, 6595–6609. [Google Scholar] [CrossRef] [Green Version]
- Kronenberg, S.; Kleinschmidt, J.A.; Böttcher, B. Electron cryo-microscopy and image reconstruction of adeno-associated virus type 2 empty capsids. EMBO Rep. 2001, 2, 997–1002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xie, Q.; Bu, W.; Bhatia, S.; Hare, J.; Somasundaram, T.; Azzi, A.; Chapman, M.S. The atomic structure of adeno-associated virus (AAV-2), a vector for human gene therapy. Proc. Natl. Acad. Sci. USA 2002, 99, 10405–10410. [Google Scholar] [CrossRef] [Green Version]
- Rayaprolu, V.; Kruse, S.; Kant, R.; Venkatakrishnan, B.; Movahed, N.; Brooke, D.; Lins, B.; Bennett, A.; Potter, T.; McKenna, R.; et al. Comparative Analysis of Adeno-Associated Virus Capsid Stability and Dynamics. J. Virol. 2013, 87, 13150–13160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gruntman, A.M.; Su, L.; Su, Q.; Gao, G.; Mueller, C.; Flotte, T.R. Stability and Compatibility of Recombinant Adeno-Associated Virus Under Conditions Commonly Encountered in Human Gene Therapy Trials. Hum. Gene Ther. Methods 2015, 26, 71–76. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, G.-P.; Alvira, M.R.; Wang, L.; Calcedo, R.; Johnston, J.; Wilson, J.M. Novel adeno-associated viruses from rhesus monkeys as vectors for human gene therapy. Proc. Natl. Acad. Sci. USA 2002, 99, 11854–11859. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davidoff, A.M.; Gray, J.T.; Ng, C.Y.C.; Zhang, Y.; Zhou, J.; Spence, Y.; Bakar, Y.; Nathwani, A.C. Comparison of the ability of adeno-associated viral vectors pseudotyped with serotype 2, 5, and 8 capsid proteins to mediate efficient transduction of the liver in murine and nonhuman primate models. Mol. Ther. 2005, 11, 875–888. [Google Scholar] [CrossRef]
- Zincarelli, C.; Soltys, S.; Rengo, G.; Rabinowitz, J.E. Analysis of AAV Serotypes 1–9 Mediated Gene Expression and Tropism in Mice after Systemic Injection. Mol. Ther. 2008, 16, 1073–1080. [Google Scholar] [CrossRef]
- Foust, K.D.; Nurre, E.; Montgomery, C.L.; Hernandez, A.; Chan, C.M.; Kaspar, B.K. Intravascular AAV9 preferentially targets neonatal neurons and adult astrocytes. Nat. Biotechnol. 2009, 27, 59–65. [Google Scholar] [CrossRef] [Green Version]
- Venkatakrishnan, B.; Yarbrough, J.; Domsic, J.; Bennett, A.; Bothner, B.; Kozyreva, O.G.; Samulski, R.J.; Muzyczka, N.; McKenna, R.; Agbandje-McKenna, M. Structure and Dynamics of Adeno-Associated Virus Serotype 1 VP1-Unique N-Terminal Domain and Its Role in Capsid Trafficking. J. Virol. 2013, 87, 4974–4984. [Google Scholar] [CrossRef] [Green Version]
- Ling, C.; Yin, Z.; Li, J.; Zhang, D.; Aslanidi, G.; Srivastava, A. Strategies to generate high-titer, high-potency recombinant AAV3 serotype vectors. Mol. Ther. Methods Clin. Dev. 2016, 3, 16029. [Google Scholar] [CrossRef]
- Chen, L.; Yuan, W.; Yuanhui, Z.; Anila, E.; Zifei, Y.; Yuan, L.; Lina, W.; Meng, W.; Jun, L.; Zhongbo, H.; et al. Selective In Vivo Targeting of Human Liver Tumors by Optimized AAV3 Vectors in a Murine Xenograft Model. Hum. Gene Ther. 2014, 25, 1023–1034. [Google Scholar]
- Ding, W.; Zhang, L.; Yan, Z.; Engelhardt, J.F. Intracellular trafficking of adeno-associated viral vectors. Gene Ther. 2005, 12, 873. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Joo, K.I.; Wang, P. Endocytic processing of adeno-associated virus type 8 vectors for transduction of target cells. Gene Ther. 2013, 20, 308–317. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Keiser, N.W.; Yan, Z.; Zhang, Y.; Lei-Butters, D.C.M.; Engelhardt, J.F. Unique Characteristics of AAV1, 2, and 5 Viral Entry, Intracellular Trafficking, and Nuclear Import Define Transduction Efficiency in HeLa Cells. Hum. Gene Ther. 2011, 22, 1433–1444. [Google Scholar] [CrossRef] [Green Version]
- Thomas, C.E.; Storm, T.A.; Huang, Z.; Kay, M.A. Rapid Uncoating of Vector Genomes Is the Key to Efficient Liver Transduction with Pseudotyped Adeno-Associated Virus Vectors. J. Virol. 2004, 78, 3110–3122. [Google Scholar] [CrossRef] [Green Version]





© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chai, Z.; Zhang, X.; Dobbins, A.L.; Frost, E.A.; Samulski, R.J.; Li, C. Chimeric Capsid Proteins Impact Transduction Efficiency of Haploid Adeno-Associated Virus Vectors. Viruses 2019, 11, 1138. https://0-doi-org.brum.beds.ac.uk/10.3390/v11121138
Chai Z, Zhang X, Dobbins AL, Frost EA, Samulski RJ, Li C. Chimeric Capsid Proteins Impact Transduction Efficiency of Haploid Adeno-Associated Virus Vectors. Viruses. 2019; 11(12):1138. https://0-doi-org.brum.beds.ac.uk/10.3390/v11121138
Chicago/Turabian StyleChai, Zheng, Xintao Zhang, Amanda Lee Dobbins, Ellie Azure Frost, R. Jude Samulski, and Chengwen Li. 2019. "Chimeric Capsid Proteins Impact Transduction Efficiency of Haploid Adeno-Associated Virus Vectors" Viruses 11, no. 12: 1138. https://0-doi-org.brum.beds.ac.uk/10.3390/v11121138




