Titers of Neutralizing Antibodies against SARS-CoV-2 Are Independent of Symptoms of Non-Severe COVID-19 in Young Adults
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cohort
2.2. Clinical Observations
2.3. Virus Propagation
2.4. Serological Assays
2.5. Serum Neutralization Test (SNT)
2.6. RT-qPCR
2.7. Graphical Representation and Statistical Analysis
3. Results
3.1. Neutralizing Antibody Titers Do Not Decline within One Month Post COVID-19 Diagnosis
3.2. Cumulative Neutralizing Capacity of Asymptomatic Cases Is Similar to Symptomatic Patients
3.3. The Live-Virus SNT Assay Comparison Reveals High Sensitivity for Live-Virus SNT Compared to Other Immunoassays
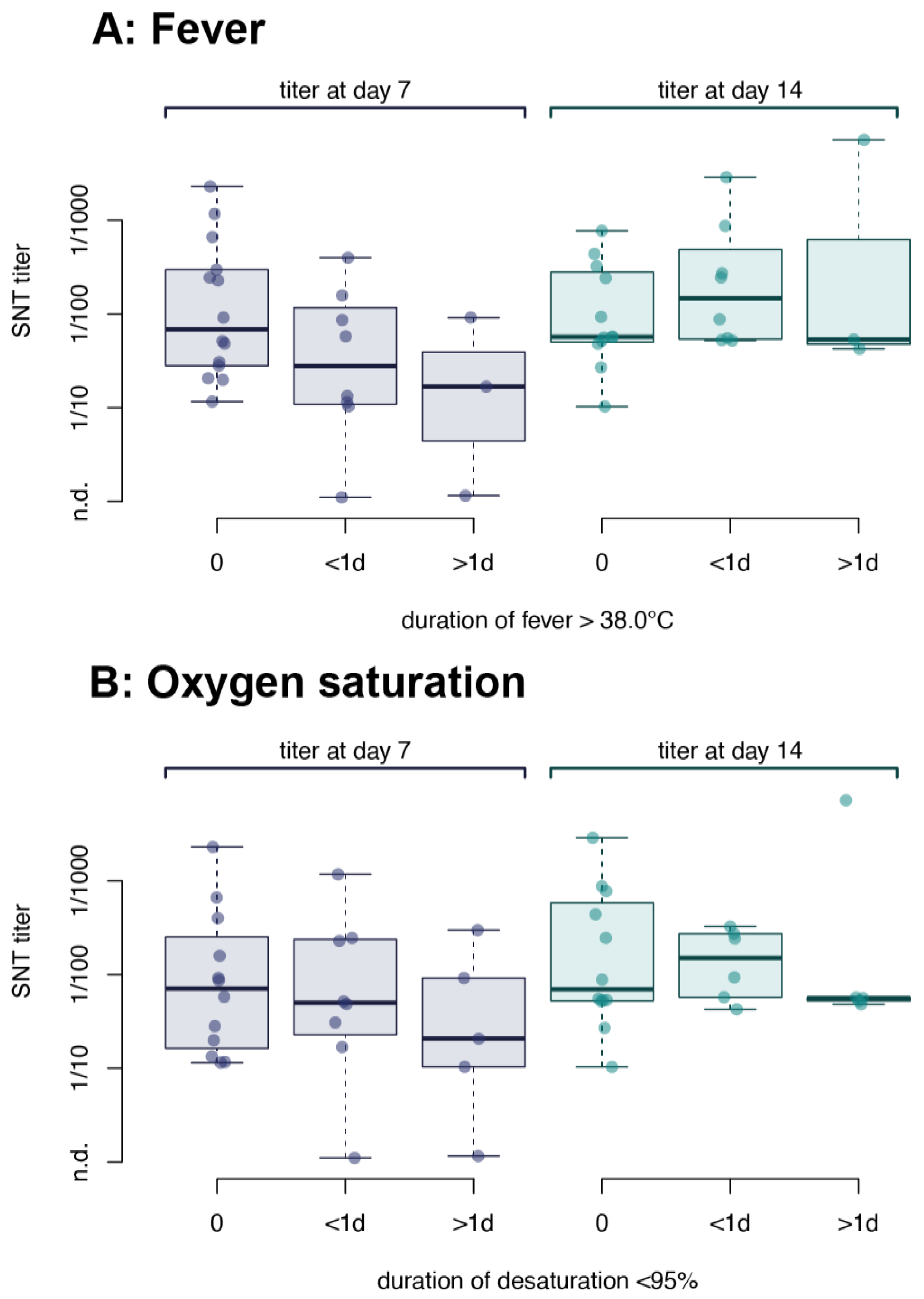
3.4. Clinical Presentation Does Not Correlate with Neutralizing Antibody Response in Young Adults with Non-Severe COVID-19
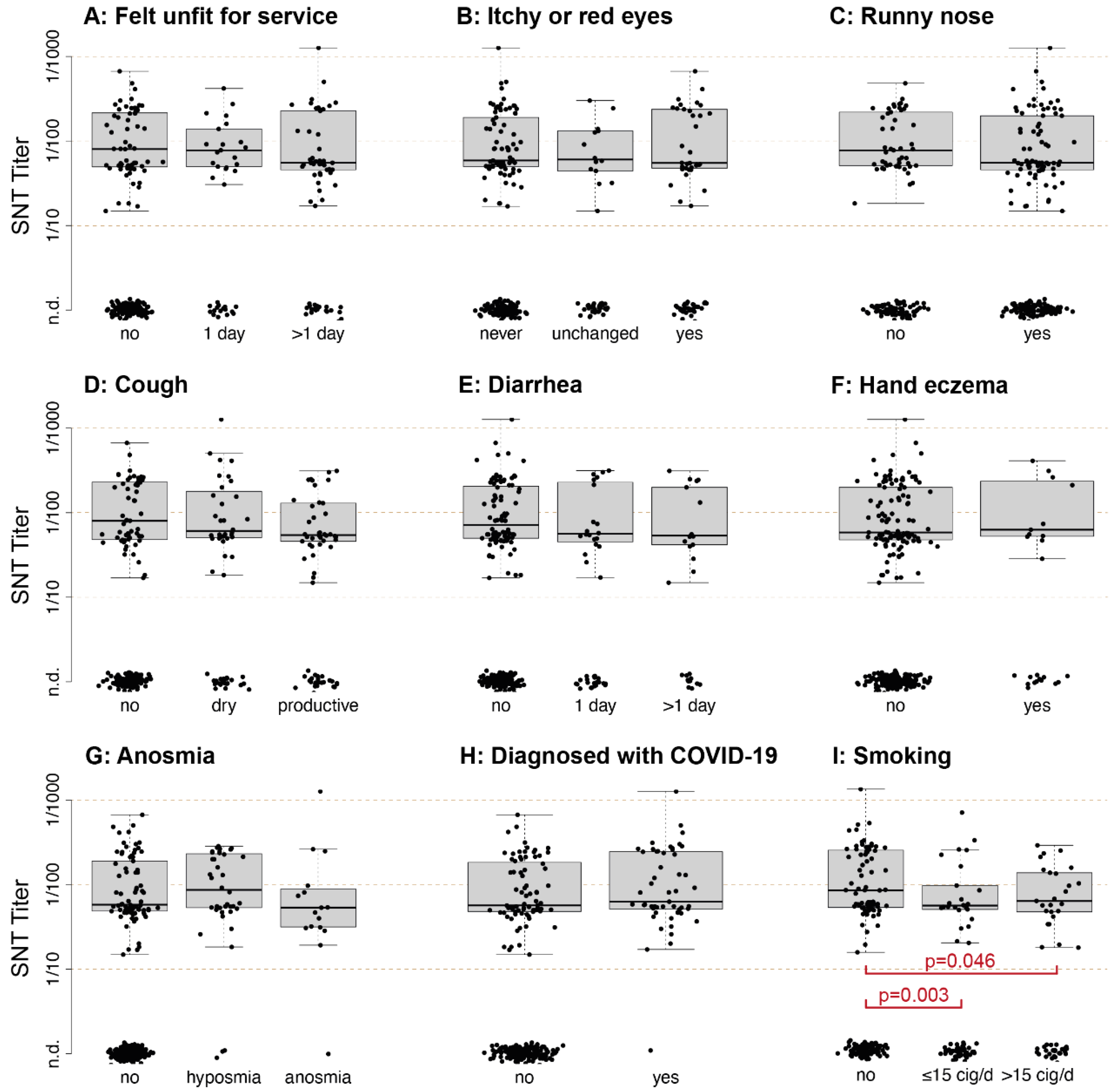
3.5. Common Symptoms of COVID-19 Are Not Related to the Quantitative Serological Response
3.6. Smokers Produce Significantly Lower Levels of nAbs Compared to Non-Smokers
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nikolich-Zugich, J.; Knox, K.S.; Rios, C.T.; Natt, B.; Bhattacharya, D.; Fain, M.J. SARS-CoV-2 and COVID-19 in older adults: What we may expect regarding pathogenesis, immune responses, and outcomes. Geroscience 2020, 42, 505–514. [Google Scholar] [CrossRef] [Green Version]
- Reilev, M.; Kristensen, K.B.; Pottegård, A.; Lund, L.C.; Hallas, J.; Ernst, M.T.; Christiansen, C.F.; Sørensen, H.T.; Johansen, N.B.; Brun, N.C.; et al. Characteristics and predictors of hospitalization and death in the first 11 122 cases with a positive RT-PCR test for SARS-CoV-2 in Denmark: A nationwide cohort. Int. J. Epidemiol. 2020, 49, 1468–1481. [Google Scholar] [CrossRef]
- Jamieson-Lane, A.; Cytrynbaum, E. Effects of age-targeted sequestration for COVID-19. J. Biol. Dyn. 2020, 14, 621–632. [Google Scholar] [CrossRef] [PubMed]
- Bielecki, M.; Züst, R.; Siegrist, D.; Meyerhofer, D.; Crameri, G.A.G.; Stanga, Z.G.; Stettbacher, A.; Buehrer, T.W.; Deuel, J.W. Social Distancing Alters the Clinical Course of COVID-19 in Young Adults: A Comparative Cohort Study. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.; Shah, S.T.H.; Imran, M.; Khan, A. The role of asymptomatic class, quarantine and isolation in the transmission of COVID-19. J. Biol. Dyn. 2020, 14, 389–408. [Google Scholar] [CrossRef]
- Yu, C.; Zhou, M.; Liu, Y.; Guo, T.; Ou, C.; Yang, L.; Li, Y.; Li, D.; Hu, X.; Shuai, L.; et al. Characteristics of asymptomatic COVID-19 infection and progression: A multicenter, retrospective study. Virulence 2020, 11, 1006–1014. [Google Scholar] [CrossRef] [PubMed]
- Ni, L.; Ye, F.; Cheng, M.L.; Feng, Y.; Deng, Y.Q.; Zhao, H.; Wei, P.; Ge, J.; Gou, M.; Li, X.; et al. Detection of SARS-CoV-2-Specific Humoral and Cellular Immunity in COVID-19 Convalescent Individuals. Immunity 2020, 52, 971–977.e3. [Google Scholar] [CrossRef]
- Lei, Q.; Li, Y.; Hou, H.Y.; Wang, F.; Ouyang, Z.Q.; Zhang, Y.; Lai, D.Y.; Banga Ndzouboukou, J.L.; Xu, Z.W.; Zhang, B.; et al. Antibody dynamics to SARS-CoV-2 in asymptomatic COVID-19 infections. Allergy 2020. Epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Pan, Z.; Yue, S.; Yu, F.; Zhang, J.; Yang, Y.; Li, R.; Liu, B.; Yang, X.; Gao, L.; et al. Disease severity dictates SARS-CoV-2-specific neutralizing antibody responses in COVID-19. Signal Transduct. Target. Ther. 2020, 5, 1–6. [Google Scholar] [CrossRef]
- Thao, T.T.; Labroussaa, F.; Ebert, N.; V’kovski, P.; Stalder, H.; Portmann, J.; Kelly, J.; Steiner, S.; Holwerda, M.; Kratzel, A.; et al. Rapid reconstruction of SARS-CoV-2 using a synthetic genomics platform. Nature 2020, 582, 561–565. [Google Scholar] [CrossRef] [PubMed]
- Corman, V.M.; Landt, O.; Kaiser, M.; Molenkamp, R.; Meijer, A.; Chu, D.K.; Bleicker, T.; Brünink, S.; Schneider, J.; Schmidt, M.L.; et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Eurosurveillance 2020, 25, 2000045. [Google Scholar] [CrossRef] [Green Version]
- Kissler, S.M.; Tedijanto, C.; Goldstein, E.; Grad, Y.H.; Lipsitch, M. Projecting the transmission dynamics of SARS-CoV-2 through the postpandemic period. Science 2020, 368, 860–868. [Google Scholar] [CrossRef] [PubMed]
- Callow, K.A.; Parry, H.F.; Sergeant, M.; Tyrrell, D.A.J. The time course of the immune response to experimental coronavirus infection of man. Epidemiol. Infect. 1990, 105, 435–446. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cao, W.-C.; Liu, W.; Zhang, P.-H.; Zhang, F.; Richardus, J. Disappearance of Antibodies to SARS-Associated Coronavirus after Recovery. N. Engl. J. Med. 2007, 357, 1162–1163. [Google Scholar] [CrossRef]
- Mo, H.; Zeng, G.; Ren, X.; Li, H.; Ke, C.; Tan, Y.; Cai, C.; Lai, K.; Chen, R.; Chan-Yeung, M.; et al. Longitudinal profile of antibodies against SARS-coronavirus in SARS patients and their clinical significance. Respirology 2006, 11, 49–53. [Google Scholar] [CrossRef] [Green Version]
- Wu, L.-P.; Wang, N.-C.; Chang, Y.-H.; Tian, X.-Y.; Na, D.-Y.; Zhang, L.-Y.; Zheng, L.; Lan, T.; Wang, L.-F.; Liang, G.-D. Duration of Antibody Responses after Severe Acute Respiratory Syndrome. Emerg. Infect. Dis. 2007, 13, 1562–1564. [Google Scholar] [CrossRef]
- Liu, W.; Fontanet, A.; Zhang, P.; Zhan, L.; Xin, Z.; Baril, L.; Tang, F.; Lv, H.; Cao, W. Two-Year Prospective Study of the Humoral Immune Response of Patients with Severe Acute Respiratory Syndrome. J. Infect. Dis. 2006, 193, 792–795. [Google Scholar] [CrossRef] [Green Version]
- Liu, L.; Xie, J.; Sun, J.; Han, Y.; Zhang, C.; Fan, H.; Liu, Z.; Qiu, Z.; He, Y.; Li, T. Longitudinal profiles of immunoglobulin G antibodies against severe acute respiratory syndrome coronavirus components and neutralizing activities in recovered patients. Scand. J. Infect. Dis. 2011, 43, 515–521. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choe, P.G.; Perera, R.A.; Park, W.B.; Song, K.H.; Bang, J.H.; Kim, E.S.; Kim, H.B.; Ko, L.W.; Park, S.W.; Kim, N.J.; et al. MERS-CoV Antibody Responses 1 Year after Symptom Onset, South Korea, 2015. Emerg. Infect. Dis. 2017, 23, 1079–1084. [Google Scholar] [CrossRef] [Green Version]
- Payne, D.C.; Biggs, H.M.; Al-Abdallat, M.M.; Alqasrawi, S.; Lü, X.; Abedi, G.R.; Haddadin, A.; Iblan, I.; Alsanouri, T.; Al Nsour, M.; et al. Multihospital Outbreak of a Middle East Respiratory Syndrome Coronavirus Deletion Variant, Jordan: A Molecular, Serologic, and Epidemiologic Investigation. Open Forum Infect. Dis. 2018, 5. [Google Scholar] [CrossRef] [Green Version]
- Okba, N.M.; Raj, V.S.; Widjaja, I.; GeurtsvanKessel, C.H.; De Bruin, E.; Chandler, F.D.; Park, W.B.; Kim, N.-J.; Farag, E.A.; Al-Hajri, M.; et al. Sensitive and Specific Detection of Low-Level Antibody Responses in Mild Middle East Respiratory Syndrome Coronavirus Infections. Emerg. Infect. Dis. 2019, 25, 1868–1877. [Google Scholar] [CrossRef]
- Long, Q.X.; Tang, X.J.; Shi, Q.L.; Li, Q.; Deng, H.J.; Yuan, J.; Hu, J.L.; Xu, W.; Zhang, Y.; Lv, F.J.; et al. Clinical and immunological assessment of asymptomatic SARS-CoV-2 infections. Nat. Med. 2020, 26, 1200–1204. [Google Scholar] [CrossRef]
- Liu, L.; To, K.K.-W.; Chan, K.-H.; Wong, Y.-C.; Zhou, R.; Kwan, K.-Y.; Fong, C.H.-Y.; Chen, L.-L.; Choi, C.Y.-K.; Lu, L.; et al. High neutralizing antibody titer in intensive care unit patients with COVID-19. Emerg. Microbes Infect. 2020, 9, 1664–1670. [Google Scholar] [CrossRef] [PubMed]
- Seow, J.; Graham, C.; Merrick, B.; Acors, S.; Steel, K.J.; Hemmings, O.; O’Bryne, A.; Kouphou, N.; Pickering, S.; Galao, R.; et al. Longitudinal observation and decline of neutralizing antibody responses in the three months following SARS-CoV-2 infection in humans. Nat. Microbiol. 2020, 5, 1598–1607. [Google Scholar] [CrossRef]
- Ho, M.-S.; Chen, W.-J.; Chen, H.-Y.; Lin, S.-F.; Wang, M.-C.; Di, J.; Lu, Y.-T.; Liu, C.-L.; Chang, S.-C.; Chao, C.-L.; et al. Neutralizing Antibody Response and SARS Severity. Emerg. Infect. Dis. 2005, 11, 1730–1737. [Google Scholar] [CrossRef] [PubMed]
- Verity, R.; Okell, L.C.; Dorigatti, I.; Winskill, P.; Whittaker, C.; Imai, N.; Cuomo-Dannenburg, G.; Thompson, H.; Walker, P.G.T.; Fu, H.; et al. Estimates of the severity of coronavirus disease 2019: A model-based analysis. Lancet Infect. Dis. 2020, 20, 669–677. [Google Scholar] [CrossRef]
- Gudbjartsson, D.F.; Norddahl, G.L.; Melsted, P.; Gunnarsdottir, K.; Holm, H.; Eythorsson, E.; Arnthorsson, A.O.; Helgason, D.; Bjarnadottir, K.; Ingvarsson, R.F.; et al. Humoral Immune Response to SARS-CoV-2 in Iceland. N. Engl. J. Med. 2020, 383, 1724–1734. [Google Scholar] [CrossRef]
- Arcavi, L.; Benowitz, N.L. Cigarette Smoking and Infection. Arch. Intern. Med. 2004, 164, 2206–2216. [Google Scholar] [CrossRef]
- Vardavas, C.I.; Nikitara, K. COVID-19 and smoking: A systematic review of the evidence. Tob. Induc. Dis. 2020, 18, 20. [Google Scholar] [CrossRef] [PubMed]
- Bielecki, M.; Crameri, G.A.G.; Schlagenhauf, P.; Buehrer, T.W.; Deuel, J.W. Body temperature screening to identify SARS-CoV-2 infected young adult travellers is ineffective. Travel Med. Infect. Dis. 2020, 37, 101832. [Google Scholar] [CrossRef]
- Wang, P.; Liu, L.; Nair, M.S.; Yin, M.T.; Luo, Y.; Wang, Q.; Yuan, T.; Mori, K.; Solis, A.G.; Yamashita, M.; et al. SARS-CoV-2 neutralizing antibody responses are more robust in patients with severe disease. Emerg. Microbes Infect. 2020, 9, 2091–2093. [Google Scholar] [CrossRef] [PubMed]



| Symptom | Without nAb | With nAb | Statistics | |
|---|---|---|---|---|
| Conjunctivitis | no/unchanged | 155 | 98 | p = 0.047 OR = 1.8 |
| yes | 95 | 34 | ||
| Cough | no | 138 | 59 | p < 10−6 OR = 3.3 |
| yes | 49 | 79 | ||
| Feeling unfit for service | no | 150 | 63 | p < 10−8 OR = 4.2 |
| yes | 37 | 66 | ||
| Diarrhea | no | 155 | 92 | p = 0.02 OR = 1.9 |
| yes | 32 | 37 | ||
| >1 of the above symptoms | no | 157 | 88 | p < 0.001 OR = 2.5 |
| yes | 29 | 41 |
| Symptom | Without nAb | With nAb | Statistics | |
|---|---|---|---|---|
| BMI | Median (25–75%) | 22.8 (20.8–25.2) | 23.0 (21.5–25.9) | n.s. |
| Smoker | non-smoker | 113 | 79 | n.s. |
| <15 cig/day | 46 | 25 | ||
| ≥15 cig/day | 28 | 25 | ||
| Influenza vaccination in the past season | no | 94 | 58 | n.s. |
| yes | 62 | 52 | ||
| Asthma | never | 159 | 112 | n.s. |
| prior/no therapy | 17 | 12 | ||
| yes/treated | 10 | 5 | ||
| Hay fever | no | 128 | 94 | n.s. |
| yes | 59 | 36 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jonsdottir, H.R.; Bielecki, M.; Siegrist, D.; Buehrer, T.W.; Züst, R.; Deuel, J.W. Titers of Neutralizing Antibodies against SARS-CoV-2 Are Independent of Symptoms of Non-Severe COVID-19 in Young Adults. Viruses 2021, 13, 284. https://0-doi-org.brum.beds.ac.uk/10.3390/v13020284
Jonsdottir HR, Bielecki M, Siegrist D, Buehrer TW, Züst R, Deuel JW. Titers of Neutralizing Antibodies against SARS-CoV-2 Are Independent of Symptoms of Non-Severe COVID-19 in Young Adults. Viruses. 2021; 13(2):284. https://0-doi-org.brum.beds.ac.uk/10.3390/v13020284
Chicago/Turabian StyleJonsdottir, Hulda R., Michel Bielecki, Denise Siegrist, Thomas W. Buehrer, Roland Züst, and Jeremy W. Deuel. 2021. "Titers of Neutralizing Antibodies against SARS-CoV-2 Are Independent of Symptoms of Non-Severe COVID-19 in Young Adults" Viruses 13, no. 2: 284. https://0-doi-org.brum.beds.ac.uk/10.3390/v13020284






