Smuggling Drugs into the Brain: An Overview of Ligands Targeting Transcytosis for Drug Delivery across the Blood–Brain Barrier
Abstract
:1. Introduction
2. Blood–Brain Barrier—Morphology and Function
2.1. The BBB as a Physical Barrier
2.2. The BBB as a Functional Barrier
3. Passive and Active Mechanisms of Transport across the BBB

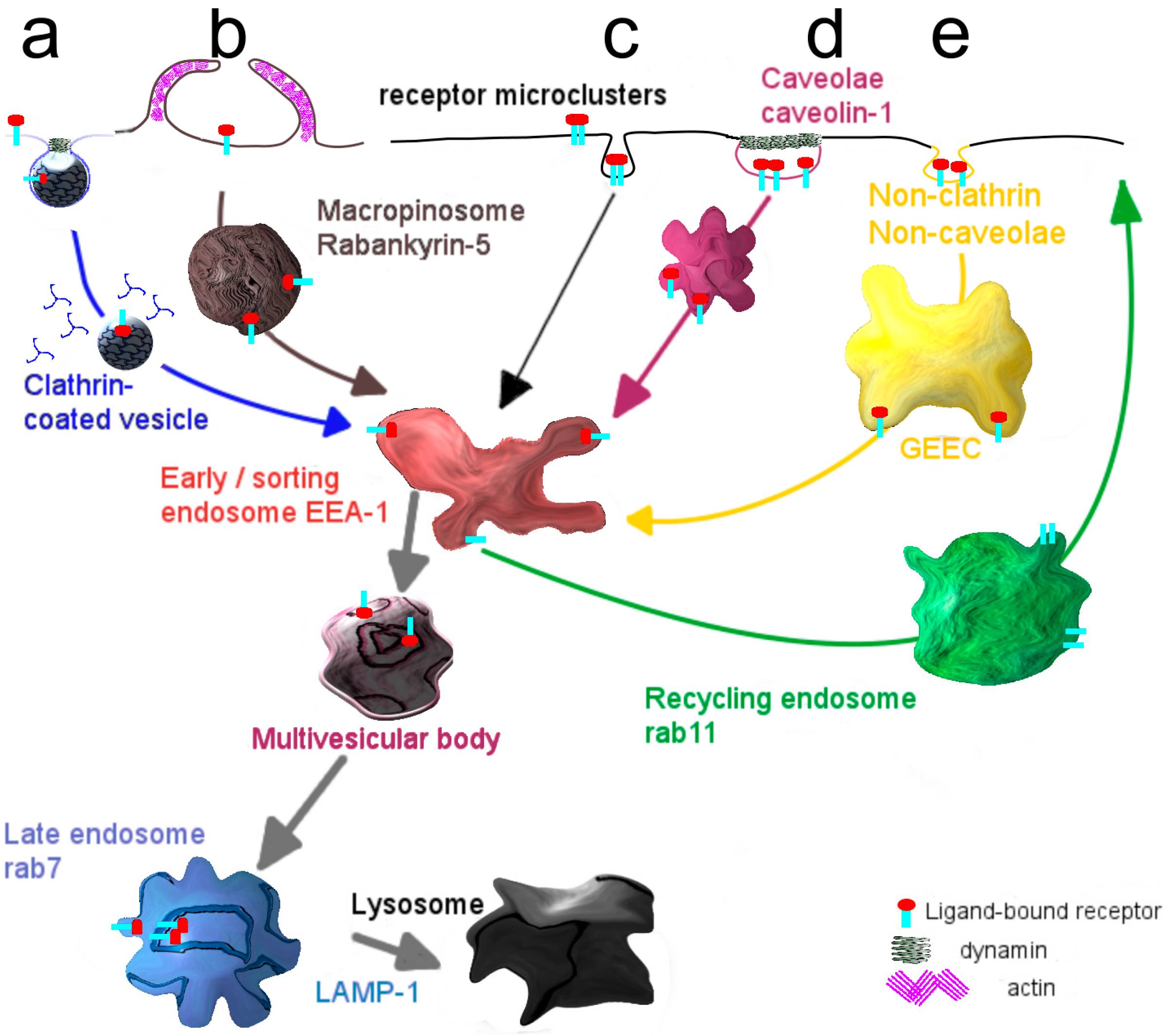
Box 1. Entry of molecules and particles into cells: Modes of internalization.
- Internalization via the endocytic mechanism is subdivided into clathrin-dependent, caveolin-dependent and clathrin- and caveolin-independent pathways (Figure 2). Clearly, the mode of cellular cargo entry is crucial for its eventual fate, as will be discussed below.
- Clathrin-mediated endocytosis involves the assembly of a clathrin coat at membrane regions enriched in receptor-ligand complexes, just underneath the plasma membrane, which subsequently triggers its inward budding. The small GTPase dynamin closes the neck of the invaginating membrane surface, leading to the formation of the clathrin-coated vesicle, which subsequently pinches off [19]. Shortly thereafter clathrin molecules dissociate from the vesicles, which then merge homotypically and/or with preexisting compartments that become enriched in EEA-1, a typical marker of early sorting endosomes [20]. From this compartment recycling may occur either directly to the plasma membrane, the so-called rapid recycling route [21,22] or via a recycling endosome, the slow recycling route [21]. Alternatively, early endosomes deliver their cargo to late endosomes, either via maturation [23] or by means of a vesicular transport mechanism [24] and eventually to lysosomes where its degradation may take place. Along the transition from early to late endosomes, the compartmental pH gradually drops [25].
- A better known but still poorly defined pathway capable of transferring cargo across the endothelial cell lining, constituting the blood-brain barrier, appears to rely on entry via caveolae [26,27]. As an advantage, lysosomal delivery as often occurs for entry along the clathrin-mediated pathway, may be avoided thus promoting secretory transport, rather than capture of cargo in a digestive compartment [28]. Accordingly, targeting specific receptors associated with caveolae may therefore well represent a far more appropriate strategy in facilitating transcellular transport, including that of targeted nanodevices. Caveolae are plasma membrane invaginations, commonly described as structures with a flask-like shape [29]. They are predominantly present at the surface of adipocytes [30], lung epithelium [31] and vascular endothelium [32,33]. A major constituent of caveolae is caveolin-1, which localizes at the inner leaflet of the plasma membrane [34]. Supported by the cavins-1, -2, -3 and -4, caveolae sustain their characteristic morphology, but gradually flatten [35]. Caveolae are considered platforms, where trafficking and signaling events take place [36,37,38].
- Stimulation of caveolar receptors triggers their internalization involving formation of a vesicular structure, named cavicle [39]. Cargo taken up by caveolae might be delivered to the Golgi apparatus or the endoplasmic reticulum, as reported for the cholera and shiga toxin B subunits in epithelium [40].
- A caveolar transcytotic route has been proposed to operate in endothelial cells of the BBB. Specifically, this pathway has been described for LDL particles which, prior to their release at the basolateral surface (brain side), localize at multivesicular bodies [41].
- The molecular organization of caveolae is strongly dependent on the presence of cholesterol, and agents that perturb the cholesterol content and organization, such as cyclodextrin and filipin, frustrate caveolae-mediated internalization [42,43]. The kinase inhibitor genistein is also known for its capacity to block caveolar uptake [44].
4. Nanocarriers and Active Targeting Systems (Ligands)
4.1. Transferrin
4.2. Melanotransferrin
4.3. Insulin
4.4. LDL
4.5. Angiopeps
4.6. Leptin
4.7. Thiamine
4.8. Glutathione
4.9. Synthetic Opioid Peptides
4.10. RVG Peptide
4.11. Tetanus Toxin, Tet1 and G23
4.12. Diphteria Toxin
4.13. TAT peptide
5. Concluding Remarks
Acknowledgments
Conflicts of Interest
References
- Abbott, N.J.; Rönnbäck, L.; Hansson, E. Astrocyte-endothelial interactions at the blood–brain barrier. Nat. Rev. Neurosci. 2006, 7, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Ballabh, P.; Braun, A.; Nedergaard, M. The blood–brain barrier: An overview: Structure, regulation, and clinical implications. Neurobiol. Dis. 2004, 16, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Bhaskar, S.; Tian, F.; Stoeger, T.; Kreyling, W.; de la Fuente, J.M.; Grazú, V.; Borm, P.; Estrada, G.; Ntziachristos, V.; Razansky, D. Multifunctional nanocarriers for diagnostics, drug delivery and targeted treatment across blood–brain barrier: Perspectives on tracking and neuroimaging. Part. Fibre Toxicol. 2010, 7, 3. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, C.; Soni, U.; Patravale, V. Nano-interventions for neurodegenerative disorders. Pharmacol. Res. 2010, 62, 166–178. [Google Scholar] [CrossRef] [PubMed]
- Yong, V.W. Metalloproteinases: Mediators of pathology and regeneration in the CNS. Nat. Rev. Neurosci. 2005, 6, 931–944. [Google Scholar] [CrossRef]
- Hori, S.; Ohtsuki, S.; Hosoya, K.; Nakashima, E.; Terasaki, T. A pericyte-derived angiopoietin-1 multimeric complex induces occludin gene expression in brain capillary endothelial cells through Tie-2 activation in vitro. J. Neurochem. 2004, 89, 503–513. [Google Scholar] [CrossRef]
- Wosik, K.; Cayrol, R.; Dodelet-Devillers, A.; Berthelet, F.; Bernard, M.; Moumdjian, R.; Bouthillier, A.; Reudelhuber, T.L.; Prat, A. Angiotensin II controls occludin function and is required for blood brain barrier maintenance: Relevance to multiple sclerosis. J. Neurosci. 2007, 27, 9032–9042. [Google Scholar] [CrossRef] [PubMed]
- Daneman, R.; Zhou, L.; Kebede, A.A.; Barres, B.A. Pericytes are required for blood-brain barrier integrity during embryogenesis. Nature 2010, 468, 562–566. [Google Scholar] [CrossRef] [PubMed]
- Armulik, A.; Genové, G.; Mäe, M.; Nisancioglu, M.H.; Wallgard, E.; Niaudet, C.; He, L.; Norlin, J.; Lindblom, P.; Strittmatter, K.; et al. Pericytes regulate the blood–brain barrier. Nature 2010, 468, 557–561. [Google Scholar] [CrossRef]
- Ben-Zvi, A.; Lacoste, B.; Kur, E.; Andreone, B.J.; Mayshar, Y.; Yan, H.; Gu, C. Mfsd2a is critical for the formation and function of the blood–brain barrier. Nature 2014, 509, 507–511. [Google Scholar] [CrossRef] [PubMed]
- Celia, C.; Cosco, D.; Paolino, D.; Fresta, M. Nanoparticulate devices for brain drug delivery. Med. Res. Rev. 2010, 31, 716–756. [Google Scholar] [PubMed]
- Candela, P.; Gosselet, F.; Saint-Pol, J.; Sevin, E.; Boucau, M.-C.; Boulanger, E.; Cecchelli, R.; Fenart, L. Apical-to-basolateral transport of amyloid-β peptides through blood-brain barrier cells is mediated by the receptor for advanced glycation end-products and is restricted by P-glycoprotein. J. Alzheimers Dis. 2010, 22, 849–859. [Google Scholar] [PubMed]
- Kageyama, T.; Nakamura, M.; Matsuo, A.; Yamasaki, Y.; Takakura, Y.; Hashida, M.; Kanai, Y.; Naito, M.; Tsuruo, T.; Minato, N.; et al. The 4F2hc/LAT1 complex transports l-DOPA across the blood–brain barrier. Brain Res. 2000, 879, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Ohtsuki, S.; Terasaki, T. Contribution of carrier-mediated transport systems to the blood-brain barrier as a supporting and protecting interface for the brain; importance for CNS drug discovery and development. Pharm. Res. 2007, 24, 1745–1758. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Fan, W.; Chen, H.; Yao, N.; Tang, W.; Tang, J.; Yuan, W.; Kuai, R.; Zhang, Z.; Wu, Y.; et al. In vitro and in vivo investigation of glucose-mediated brain-targeting liposomes. J. Drug Target. 2010, 18, 536–549. [Google Scholar] [CrossRef] [PubMed]
- Xie, F.; Yao, N.; Qin, Y.; Zhang, Q.; Chen, H.; Yuan, M.; Tang, J.; Li, X.; Fan, W.; Zhang, Q.; et al. Investigation of glucose-modified liposomes using polyethylene glycols with different chain lengths as the linkers for brain targeting. Int. J. Nanomed. 2012, 7, 163–175. [Google Scholar] [CrossRef]
- Gromnicova, R.; Davies, H.A.; Sreekanthreddy, P.; Romero, I.A.; Lund, T.; Roitt, I.M.; Phillips, J.B.; Male, D.K. Glucose-coated gold nanoparticles transfer across human brain endothelium and enter astrocytes in vitro. PLoS One 2013, 8, e81043. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Xin, H.; Ren, Q.; Gu, J.; Zhu, L.; Du, F.; Feng, C.; Xie, Y.; Sha, X.; Fang, X. Nanoparticles of 2-deoxy-d-glucose functionalized poly(ethylene glycol)-co-poly(trimethylene carbonate) for dual-targeted drug delivery in glioma treatment. Biomaterials 2014, 35, 518–529. [Google Scholar] [CrossRef] [PubMed]
- Schmid, S.L. Clathrin-coated vesicle formation and protein sorting: An integrated process. Annu. Rev. Biochem. 1997, 66, 511–548. [Google Scholar] [CrossRef] [PubMed]
- Mu, F.-T.; Callaghan, J.M.; Steele-Mortimer, O.; Stenmark, H.; Parton, R.G.; Campbell, P.L.; McCluskey, J.; Yeo, J.-P.; Tock, E.P.C.; Toh, B.-H. EEA1, an early endosome-associated protein. EEA1 is a conserved α-helical peripheral membrane protein flanked by cysteine “fingers” and contains a calmodulin-binding IQ motif. J. Biol. Chem. 1995, 270, 13503–13511. [Google Scholar] [CrossRef] [PubMed]
- Maxfield, F.R.; McGraw, T.E. Endocytic recycling. Nat. Rev. Mol. Cell Biol. 2004, 5, 121–132. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, A.; Sharma, D.K.; Marks, D.L.; Pagano, R.E. Elevated endosomal cholesterol levels in niemann-pick cells inhibit rab4 and perturb membrane recycling. Mol. Biol. Cell 2004, 15, 4500–4511. [Google Scholar] [CrossRef] [PubMed]
- Murphy, R.F. Maturation models for endosome and lysosome biogenesis. Trends Cell Biol. 1991, 1, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, G.; Gruenberg, J. The arguments for pre-existing early and late endosomes. Trends Cell Biol. 1991, 1, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Mellman, I. The importance of being acid: The role of acidification in intracellular membrane traffic. J. Exp. Biol. 1992, 172, 39–45. [Google Scholar] [PubMed]
- Schnitzer, J.E.; Oh, P.; Pinney, E.; Allard, J. Filipin-sensitive caveolae-mediated transport in endothelium: Reduced transcytosis, scavenger endocytosis, and capillary permeability of select macromolecules. J. Cell Biol. 1994, 127, 1217–1232. [Google Scholar] [CrossRef] [PubMed]
- Dehouck, B.; Fenart, L.; Dehouck, M.P.; Pierce, A.; Torpier, G.; Cecchelli, R. A new function for the LDL receptor: Transcytosis of LDL across the blood-brain barrier. J. Cell Biol. 1997, 138, 877–889. [Google Scholar] [CrossRef] [PubMed]
- McIntosh, D.P.; Tan, X.-Y.; Oh, P.; Schnitzer, J.E. Targeting endothelium and its dynamic caveolae for tissue-specific transcytosis in vivo: A pathway to overcome cell barriers to drug and gene delivery. Proc. Natl. Acad. Sci. USA 2002, 99, 1996–2001. [Google Scholar] [CrossRef]
- Anderson, R.G.W. The caveolae membrane system. Annu. Rev. Biochem. 1998, 67, 199–225. [Google Scholar] [CrossRef]
- Thorn, H.; Stenkula, K.G.; Karlsson, M.; Örtegren, U.; Nystrom, F.H.; Gustavsson, J. Strålfors, P. Cell surface orifices of caveolae and localization of caveolin to the necks of caveolae in adipocytes. Mol. Biol. Cell 2003, 14, 3967–3976. [Google Scholar] [CrossRef] [PubMed]
- Newman, G.R.; Campbell, L.; von Ruhland, C.; Jasani, B.; Gumbleton, M. Caveolin and its cellular and subcellular immunolocalisation in lung alveolar epithelium: Implications for alveolar epithelial type I cell function. Cell Tissue Res. 1999, 295, 111–120. [Google Scholar] [CrossRef]
- Simionescu, N.; Simionescu, M.; Palade, G.E. Permeability of intestinal capillaries pathway followed by dextrans and glycogens. J. Cell Biol. 1972, 53, 365–392. [Google Scholar] [CrossRef] [PubMed]
- Parton, R.G.; Joggerst, B.; Simons, K. Regulated internalization of caveolae. J. Cell Biol. 1994, 127, 1199–1215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rothberg, K.G.; Heuser, J.E.; Donzell, W.C.; Ying, Y.-S.; Glenney, J.R.; Anderson, R.G.W. Caveolin, a protein component of caveolae membrane coats. Cell 1992, 68, 673–682. [Google Scholar] [CrossRef] [PubMed]
- Hansen, C.G.; Nichols, B.J. Exploring the caves: Cavins, caveolins and caveolae. Trends Cell Biol. 2010, 20, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Shaul, P.W.; Anderson, R.G. Role of plasmalemmal caveolae in signal transduction. Am. J. Physiol. 1998, 275, L843–L851. [Google Scholar]
- Smart, E.J.; Graf, G.A.; McNiven, M.A.; Sessa, W.C.; Engelman, J.A.; Scherer, P.E.; Okamoto, T.; Lisanti, M.P. Caveolins, liquid-ordered domains, and signal transduction. Mol. Cell. Biol. 1999, 19, 7289–7304. [Google Scholar] [PubMed]
- Marx, J. Caveolae: A once-elusive structure gets some respect. Science 2001, 294, 1862–1865. [Google Scholar] [CrossRef] [PubMed]
- Mundy, D.I.; Machleidt, T.; Ying, Y.; Anderson, R.G.W.; Bloom, G.S. Dual control of caveolar membrane traffic by microtubules and the actin cytoskeleton. J. Cell Sci. 2002, 115, 4327–4339. [Google Scholar] [CrossRef] [PubMed]
- Nichols, B.J.; Kenworthy, A.K.; Polishchuk, R.S.; Lodge, R.; Roberts, T.H.; Hirschberg, K.; Phair, R.D.; Lippincott-Schwartz, J. Rapid cycling of lipid raft markers between the cell surface and Golgi complex. J. Cell Biol. 2001, 153, 529–542. [Google Scholar] [CrossRef] [PubMed]
- Candela, P.; Gosselet, F.; Miller, F.; Buee-Scherrer, V.; Torpier, G.; Cecchelli, R.; Fenart, L. Physiological pathway for low-density lipoproteins across the blood–brain barrier: Transcytosis through brain capillary endothelial cells in vitro. Endothelium 2008, 15, 254–264. [Google Scholar] [CrossRef] [PubMed]
- Neufeld, E.B.; Cooney, A.M.; Pitha, J.; Dawidowicz, E.A.; Dwyer, N.K.; Pentchev, P.G.; Blanchette-Mackie, E.J. Intracellular trafficking of cholesterol monitored with a cyclodextrin. J. Biol. Chem. 1996, 271, 21604–21613. [Google Scholar] [CrossRef] [PubMed]
- Orlandi, P.A.; Fishman, P.H. Filipin-dependent inhibition of cholera toxin: Evidence for toxin internalization and activation through caveolae-like domains. J. Cell Biol. 1998, 141, 905–915. [Google Scholar] [CrossRef] [PubMed]
- Aoki, T.; Nomura, R.; Fujimoto, T. Tyrosine phosphorylation of caveolin-1 in the endothelium. Exp. Cell Res. 1999, 253, 629–636. [Google Scholar] [CrossRef] [PubMed]
- Simionescu, M.; Popov, D.; Sima, A. Endothelial transcytosis in health and disease. Cell Tissue Res. 2008, 335, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Pardridge, W.M. Biopharmaceutical drug targeting to the brain. J. Drug Target. 2010, 18, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Gupta, Y.; Jain, A.; Jain, S.K. Transferrin-conjugated solid lipid nanoparticles for enhanced delivery of quinine dihydrochloride to the brain. J. Pharm. Pharmacol. 2007, 59, 935–940. [Google Scholar] [CrossRef] [PubMed]
- Pardridge, W.M.; Buciak, J.L.; Friden, P.M. Selective transport of an anti-transferrin receptor antibody through the blood–brain barrier in vivo. J. Pharmacol. Exp. Ther. 1991, 259, 66–70. [Google Scholar]
- Boado, R.J.; Tsukamoto, H.; Pardridge, W.M. Drug delivery of antisense molecules to the brain for treatment of Alzheimer’s disease and cerebral AIDS. J. Pharm. Sci. 1998, 87, 1308–1315. [Google Scholar] [CrossRef] [PubMed]
- Pang, Z.; Lu, W.; Gao, H.; Hu, K.; Chen, J.; Zhang, C.; Gao, X.; Jiang, X.; Zhu, C. Preparation and brain delivery property of biodegradable polymersomes conjugated with OX26. J. Control. Release 2008, 128, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Ulbrich, K.; Hekmatara, T.; Herbert, E.; Kreuter, J. Transferrin- and transferrin-receptor-antibody-modified nanoparticles enable drug delivery across the blood-brain barrier (BBB). Eur. J. Pharm. Biopharm. 2009, 71, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Paris-Robidas, S.; Emond, V.; Tremblay, C.; Soulet, D.; Calon, F. In vivo labeling of brain capillary endothelial cells after intravenous injection of monoclonal antibodies targeting the transferrin receptor. Mol. Pharmacol. 2011, 80, 32–39. [Google Scholar] [CrossRef]
- Moos, T.; Morgan, E.H. Restricted transport of anti-transferrin receptor antibody (OX26) through the blood–brain barrier in the rat. J. Neurochem. 2001, 79, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Gosk, S.; Vermehren, C.; Storm, G.; Moos, T. Targeting anti-transferrin receptor antibody (OX26) and OX26-conjugated liposomes to brain capillary endothelial cells using in situ perfusion. J. Cereb. Blood Flow Metab. 2004, 24, 1193–1204. [Google Scholar] [CrossRef]
- Yu, Y.J.; Zhang, Y.; Kenrick, M.; Hoyte, K.; Luk, W.; Lu, Y.; Atwal, J.; Elliott, J.M.; Prabhu, S.; Watts, R.J.; et al. Boosting brain uptake of a therapeutic antibody by reducing its affinity for a transcytosis target. Sci. Transl. Med. 2011, 3, 84ra44. [Google Scholar] [CrossRef] [PubMed]
- Niewoehner, J.; Bohrmann, B.; Collin, L.; Urich, E.; Sade, H.; Maier, P.; Rueger, P.; Stracke, J.O.; Lau, W.; Tissot, A.C.; et al. Increased brain penetration and potency of a therapeutic antibody using a monovalent molecular shuttle. Neuron 2014, 81, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Food, M.R.; Rothenberger, S.; Gabathuler, R.; Haidl, I.D.; Reid, G.; Jefferies, W.A. Transport and expression in human melanomas of a transferrin-like glycosylphosphatidylinositol-anchored protein. J. Biol. Chem. 1994, 269, 3034–3040. [Google Scholar] [PubMed]
- Demeule, M.; Poirier, J.; Jodoin, J.; Bertrand, Y.; Desrosiers, R.R.; Dagenais, C.; Nguyen, T.; Lanthier, J.; Gabathuler, R.; Kennard, M.; et al. High transcytosis of melanotransferrin (P97) across the blood–brain barrier. J. Neurochem. 2002, 83, 924–933. [Google Scholar] [CrossRef] [PubMed]
- Karkan, D.; Pfeifer, C.; Vitalis, T.Z.; Arthur, G.; Ujiie, M.; Chen, Q.; Tsai, S.; Koliatis, G.; Gabathuler, R.; Jefferies, W.A. A unique carrier for delivery of therapeutic compounds beyond the blood-brain barrier. PLoS One 2008, 3, e2469. [Google Scholar] [CrossRef] [PubMed]
- Pardridge, W.M.; Eisenberg, J.; Yang, J. Human blood–brain barrier insulin receptor. J. Neurochem. 1985, 44, 1771–1778. [Google Scholar] [CrossRef] [PubMed]
- Boado, R.J.; Hui, E.K.-W.; Lu, J.Z.; Zhou, Q.-H.; Pardridge, W.M. Selective targeting of a TNFR decoy receptor pharmaceutical to the primate brain as a receptor-specific IgG fusion protein. J. Biotechnol. 2010, 146, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Schlachetzki, F.; Zhu, C.; Pardridge, W.M. Expression of the neonatal Fc receptor (FcRn) at the blood-brain barrier. J. Neurochem. 2002, 81, 203–206. [Google Scholar] [CrossRef] [PubMed]
- Ohshima-Hosoyama, S.; Simmons, H.A.; Goecks, N.; Joers, V.; Swanson, C.R.; Bondarenko, V.; Velotta, R.; Brunner, K.; Wood, L.D.; Hruban, R.H.; et al. A monoclonal antibody–GDNF fusion protein is not neuroprotective and is associated with proliferative pancreatic lesions in Parkinsonian monkeys. PLoS One 2012, 7, e39036. [Google Scholar] [PubMed]
- Alyautdin, R.; Gothier, D.; Petrov, V.; Kharkevich, D.; Kreuter, J. Analgesic activity of the hexapeptide dalargin adsorbed on the surface of polysorbate 80-coated poly (butyl cyanoacrylate) nanoparticles. Eur. J. Pharm. Biopharm. 1995, 41, 44–48. [Google Scholar]
- Alyautdin, R.N.; Petrov, V.E.; Langer, K.; Berthold, A.; Kharkevich, D.A.; Kreuter, J. Delivery of loperamide across the blood-brain barrier with polysorbate 80-coated polybutylcyanoacrylate nanoparticles. Pharm. Res. 1997, 14, 325–328. [Google Scholar] [CrossRef] [PubMed]
- Alyautdin, R.N.; Tezikov, E.B.; Ramge, P.; Kharkevich, D.A.; Begley, D.J.; Kreuter, J. Significant entry of tubocurarine into the brain of rats by adsorption to polysorbate 80-coated polybutylcyanoacrylate nanoparticles: An in situ brain perfusion study. J. Microencapsul. 1998, 15, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Gulyaev, A.E.; Gelperina, S.E.; Skidan, I.N.; Antropov, A.S.; Kivman, G.Y.; Kreuter, J. Significant transport of doxorubicin into the brain with polysorbate 80-coated nanoparticles. Pharm. Res. 1999, 16, 1564–1569. [Google Scholar] [CrossRef] [PubMed]
- Olivier, J.C.; Fenart, L.; Chauvet, R.; Pariat, C.; Cecchelli, R.; Couet, W. Indirect evidence that drug brain targeting using polysorbate 80-coated polybutylcyanoacrylate nanoparticles is related to toxicity. Pharm. Res. 1999, 16, 1836–1842. [Google Scholar] [CrossRef] [PubMed]
- Kreuter, J.; Ramge, P.; Petrov, V.; Hamm, S.; Gelperina, S.E.; Engelhardt, B.; Alyautdin, R.; von Briesen, H.; Begley, D.J. Direct evidence that polysorbate-80-coated poly (butylcyanoacrylate) nanoparticles deliver drugs to the CNS via specific mechanisms requiring prior binding of drug to the nanoparticles. Pharm. Res. 2003, 20, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Alyaudtin, R.N.; Reichel, A.; Löbenberg, R.; Ramge, P.; Kreuter, J.; Begley, D.J. Interaction of poly (butylcyanoacrylate) nanoparticles with the blood-brain barrier in vivo and in vitro. J. Drug Target. 2001, 9, 209–221. [Google Scholar] [CrossRef] [PubMed]
- Lück, M.; Schröder, W.; Harnisch, S.; Thode, K.; Blunk, T.; Paulke, B.R.; Kresse, M.; Müller, R.H. Identification of plasma proteins facilitated by enrichment on particulate surfaces: Analysis by two-dimensional electrophoresis and N-terminal microsequencing. Electrophoresis 1997, 18, 2961–2967. [Google Scholar] [CrossRef] [PubMed]
- Lemke, A.; Kiderlen, A.F.; Petri, B.; Kayser, O. Delivery of amphotericin B nanosuspensions to the brain and determination of activity against Balamuthia mandrillaris amebas. Nanomedicine 2010, 6, 597–603. [Google Scholar] [CrossRef] [PubMed]
- Wilson, B.; Samanta, M.K.; Santhi, K.; Kumar, K.P.S.; Paramakrishnan, N.; Suresh, B. Targeted delivery of tacrine into the brain with polysorbate 80-coated poly (n-butylcyanoacrylate) nanoparticles. Eur. J. Pharm. Biopharm. 2008, 70, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Göppert, T.M.; Müller, R.H. Polysorbate-stabilized solid lipid nanoparticles as colloidal carriers for intravenous targeting of drugs to the brain: Comparison of plasma protein adsorption patterns. J. Drug Target. 2005, 13, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Zensi, A.; Begley, D.; Pontikis, C.; Legros, C.; Mihoreanu, L.; Wagner, S.; Büchel, C.; von Briesen, H.; Kreuter, J. Albumin nanoparticles targeted with Apo E enter the CNS by transcytosis and are delivered to neurones. J. Control. Release 2009, 137, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Hamidi, M.; Azadi, A.; Rafiei, P. Pharmacokinetic consequences of pegylation. Drug Deliv. 2006, 13, 399–409. [Google Scholar] [CrossRef] [PubMed]
- Kreuter, J.; Hekmatara, T.; Dreis, S.; Vogel, T.; Gelperina, S.; Langer, K. Covalent attachment of apolipoprotein A-I and apolipoprotein B-100 to albumin nanoparticles enables drug transport into the brain. J. Control. Release 2007, 118, 54–58. [Google Scholar] [PubMed]
- Zensi, A.; Begley, D.; Pontikis, C.; Legros, C.; Mihoreanu, L.; Büchel, C.; Kreuter, J. Human serum albumin nanoparticles modified with apolipoprotein A–I cross the blood–brain barrier and enter the rodent brain. J. Drug Target. 2010, 18, 842–848. [Google Scholar] [CrossRef] [PubMed]
- Dehouck, B.; Dehouck, M.P.; Fruchart, J.C.; Cecchelli, R. Upregulation of the low density lipoprotein receptor at the blood-brain barrier: Intercommunications between brain capillary endothelial cells and astrocytes. J. Cell Biol. 1994, 126, 465–473. [Google Scholar] [PubMed]
- Fillebeen, C.; Descamps, L.; Dehouck, M.P.; Fenart, L.; Benaïssa, M.; Spik, G.; Cecchelli, R.; Pierce, A. Receptor-mediated transcytosis of lactoferrin through the blood-brain barrier. J. Biol. Chem. 1999, 274, 7011–7017. [Google Scholar] [CrossRef] [PubMed]
- Pan, W.; Kastin, A.J.; Zankel, T.C.; van Kerkhof, P.; Terasaki, T.; Bu, G. Efficient transfer of receptor-associated protein (RAP) across the blood-brain barrier. J. Cell Sci. 2004, 117, 5071–5078. [Google Scholar] [CrossRef] [PubMed]
- Benchenane, K.; Berezowski, V.; Ali, C.; Fernández-Monreal, M.; López-Atalaya, J.P.; Brillault, J.; Chuquet, J.; Nouvelot, A.; MacKenzie, E.T.; Bu, G.; et al. Tissue-type plasminogen activator crosses the intact blood–brain barrier by low-density lipoprotein receptor-related protein-mediated transcytosis. Circulation 2005, 111, 2241–2249. [Google Scholar] [CrossRef] [PubMed]
- Bell, R.D.; Sagare, A.P.; Friedman, A.E.; Bedi, G.S.; Holtzman, D.M.; Deane, R.; Zlokovic, B.V. Transport pathways for clearance of human Alzheimer’s amyloid beta-peptide and apolipoproteins E and J in the mouse central nervous system. J. Cereb. Blood Flow Metab. 2007, 27, 909–918. [Google Scholar]
- Kounnas, M.Z.; Moir, R.D.; Rebeck, G.W.; Bush, A.I.; Argraves, W.S.; Tanzi, R.E.; Hyman, B.T.; Strickland, D.K. LDL receptor-related protein, a multifunctional ApoE receptor, binds secreted beta-amyloid precursor protein and mediates its degradation. Cell 1995, 82, 331–340. [Google Scholar] [PubMed]
- Demeule, M.; Régina, A.; Ché, C.; Poirier, J.; Nguyen, T.; Gabathuler, R.; Castaigne, J.-P.; Béliveau, R. Identification and design of peptides as a new drug delivery system for the brain. J. Pharmacol. Exp. Ther. 2008, 324, 1064–1072. [Google Scholar] [CrossRef] [PubMed]
- Demeule, M.; Currie, J.-C.; Bertrand, Y.; Ché, C.; Nguyen, T.; Régina, A.; Gabathuler, R.; Castaigne, J.-P.; Béliveau, R. Involvement of the low-density lipoprotein receptor-related protein in the transcytosis of the brain delivery vector angiopep-2. J. Neurochem. 2008, 106, 1534–1544. [Google Scholar] [CrossRef] [PubMed]
- Régina, A.; Demeule, M.; Ché, C.; Lavallée, I.; Poirier, J.; Gabathuler, R.; Béliveau, R.; Castaigne, J.-P. Antitumour activity of ANG1005, a conjugate between paclitaxel and the new brain delivery vector Angiopep-2. Br. J. Pharmacol. 2008, 155, 185–197. [Google Scholar] [CrossRef] [PubMed]
- Gabathuler, R. An engineered peptide compound platform technology incorporating angiopep for crossing the BBB. In Drug Delivery to the Central Nervous System; Jain, K.K., Ed.; Humana Press: Totowa, NJ, USA, 2010; Volume 45, pp. 249–260. [Google Scholar]
- Lin, N.U.; Schwartzberg, L.; Kesari, S.; Elias, A.; Anders, C.; Raizer, J.; Kozloff, M.; Amiri-Kordestani, L. Abstract B76: A phase II study of ANG1005, a novel, brain-penetrant taxane derivative, in breast cancer patients with brain metastases. Mol. Cancer Ther. 2013, 12, B76–B76. [Google Scholar] [CrossRef]
- Van Rooy, I.; Mastrobattista, E.; Storm, G.; Hennink, W.E.; Schiffelers, R.M. Comparison of five different targeting ligands to enhance accumulation of liposomes into the brain. J. Control. Release 2011, 150, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Neuwelt, E.A.; Bauer, B.; Fahlke, C.; Fricker, G.; Iadecola, C.; Janigro, D.; Leybaert, L.; Molnár, Z.; O’Donnell, M.E.; Povlishock, J.T.; et al. Engaging neuroscience to advance translational research in brain barrier biology. Nat. Rev. Neurosci. 2011, 12, 169–182. [Google Scholar] [CrossRef] [PubMed]
- Tamaru, M.; Akita, H.; Fujiwara, T.; Kajimoto, K.; Harashima, H. Leptin-derived peptide, a targeting ligand for mouse brain-derived endothelial cells via macropinocytosis. Biochem. Biophys. Res. Commun. 2010, 394, 587–592. [Google Scholar] [CrossRef] [PubMed]
- Malyukova, I.; Murray, K.F.; Zhu, C.; Boedeker, E.; Kane, A.; Patterson, K.; Peterson, J.R.; Donowitz, M.; Kovbasnjuk, O. Macropinocytosis in Shiga toxin 1 uptake by human intestinal epithelial cells and transcellular transcytosis. Am. J. Physiol. Gastrointest. Liver Physiol. 2009, 296, G78–G92. [Google Scholar] [CrossRef] [PubMed]
- Ragnarsson, E.G.E.; Schoultz, I.; Gullberg, E.; Carlsson, A.H.; Tafazoli, F.; Lerm, M.; Magnusson, K.-E.; Söderholm, J.D.; Artursson, P. Yersinia pseudotuberculosis induces transcytosis of nanoparticles across human intestinal villus epithelium via invasin-dependent macropinocytosis. Lab. Investig. 2008, 88, 1215–1226. [Google Scholar] [CrossRef] [PubMed]
- Bjørbaek, C.; Elmquist, J.K.; Michl, P.; Ahima, R.S.; van Bueren, A.; McCall, A.L.; Flier, J.S. Expression of leptin receptor isoforms in rat brain microvessels. Endocrinology 1998, 139, 3485–3491. [Google Scholar] [CrossRef] [PubMed]
- Van Heek, M.; Compton, D.S.; France, C.F.; Tedesco, R.P.; Fawzi, A.B.; Graziano, M.P.; Sybertz, E.J.; Strader, C.D.; Davis, H.R., Jr. Diet-induced obese mice develop peripheral, but not central, resistance to leptin. J. Clin. Investig. 1997, 99, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Lockman, P.R.; Oyewumi, M.O.; Koziara, J.M.; Roder, K.E.; Mumper, R.J.; Allen, D.D. Brain uptake of thiamine-coated nanoparticles. J. Control. Release 2003, 93, 271–282. [Google Scholar] [CrossRef] [PubMed]
- Gaillard, P.J.; de Boer, A.G. 2B-Trans technology: Targeted drug delivery across the blood–brain barrier. Methods Mol. Biol. 2008, 437, 161–175. [Google Scholar] [PubMed]
- Gaillard, P.J.; Appeldoorn, C.C.M.; Dorland, R.; van Kregten, J.; Manca, F.; Vugts, D.J.; Windhorst, B.; van Dongen, G.A.M.S.; de Vries, H.E.; Maussang, D.; et al. Pharmacokinetics, brain delivery, and efficacy in brain tumor-bearing mice of glutathione pegylated liposomal doxorubicin (2B3–101). PLoS One 2014, 9, e82331. [Google Scholar] [CrossRef] [PubMed]
- Rip, J.; Chen, L.; Hartman, R.; van den Heuvel, A.; Reijerkerk, A.; van Kregten, J.; van der Boom, B.; Appeldoorn, C.; de Boer, M.; Maussang, D.; et al. Glutathione PEGylated liposomes: Pharmacokinetics and delivery of cargo across the blood–brain barrier in rats. J. Drug Target. 2014, 22, 460–467. [Google Scholar] [PubMed]
- Banks, W.A.; Kastin, A.J. Peptide transport systems for opiates across the blood–brain barrier. Am. J. Physiol. 1990, 259, E1–E10. [Google Scholar] [PubMed]
- Zlokovic, B.V. The blood-brain barrier in health and chronic neurodegenerative disorders. Neuron 2008, 57, 178–201. [Google Scholar] [CrossRef] [PubMed]
- Zlokovic, B.V.; Mackic, J.B.; Djuricic, B.; Davson, H. Kinetic analysis of leucine-enkephalin cellular uptake at the luminal side of the blood–brain barrier of an in situ perfused guinea-pig brain. J. Neurochem. 1989, 53, 1333–1340. [Google Scholar] [CrossRef] [PubMed]
- Rousselle, C. Improved brain uptake and pharmacological activity of dalargin using a peptide-vector-mediated strategy. J. Pharmacol. Exp. Ther. 2003, 306, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Tsuzuki, N.; Hama, T.; Hibi, T.; Konishi, R.; Futaki, S.; Kitagawa, K. Adamantane as a brain-directed drug carrier for poorly absorbed drug: Antinociceptive effects of (D-Ala2) Leu-enkephalin derivatives conjugated with the 1-adamantane moiety. Biochem. Pharmacol. 1991, 41, R5–R8. [Google Scholar] [CrossRef] [PubMed]
- Polt, D.R.; Palian, M.M. Glycopeptide analgesics. Drugs Future 2001, 26, 561. [Google Scholar] [CrossRef]
- Egleton, R.D.; Bilsky, E.J.; Tollin, G.; Dhanasekaran, M.; Lowery, J.; Alves, I.; Davis, P.; Porreca, F.; Yamamura, H.I.; Yeomans, L.; et al. Biousian glycopeptides penetrate the blood–brain barrier. Tetrahedron Asymmetry 2005, 16, 65–75. [Google Scholar] [CrossRef]
- Costantino, L.; Gandolfi, F.; Tosi, G.; Rivasi, F.; Vandelli, M.A.; Forni, F. Peptide-derivatized biodegradable nanoparticles able to cross the blood–brain barrier. J. Control. Release 2005, 108, 84–96. [Google Scholar] [CrossRef] [PubMed]
- Tosi, G.; Costantino, L.; Rivasi, F.; Ruozi, B.; Leo, E.; Vergoni, A.V.; Tacchi, R.; Bertolini, A.; Vandelli, M.A.; Forni, F. Targeting the central nervous system: In vivo experiments with peptide-derivatized nanoparticles loaded with Loperamide and Rhodamine-123. J. Control. Release 2007, 122, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Vergoni, A.V.; Tosi, G.; Tacchi, R.; Vandelli, M.A.; Bertolini, A.; Costantino, L. Nanoparticles as drug delivery agents specific for CNS: In vivo biodistribution. Nanomed. Nanotechnol. Biol. Med. 2009, 5, 369–377. [Google Scholar] [CrossRef]
- Tosi, G.; Fano, R.A.; Bondioli, L.; Badiali, L.; Benassi, R.; Rivasi, F.; Ruozi, B.; Forni, F.; Vandelli, M.A. Investigation on mechanisms of glycopeptide nanoparticles for drug delivery across the blood–brain barrier. Nanomedicine 2011, 6, 423–436. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Wu, H.; McBride, J.L.; Jung, K.-E.; Kim, M.H.; Davidson, B.L.; Lee, S.K.; Shankar, P.; Manjunath, N. Transvascular delivery of small interfering RNA to the central nervous system. Nature 2007, 448, 39–43. [Google Scholar] [PubMed]
- Alvarez-Erviti, L.; Seow, Y.; Yin, H.; Betts, C.; Lakhal, S.; Wood, M.J.A. Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat. Biotechnol. 2011, 29, 341–345. [Google Scholar] [PubMed]
- Sinha, K.; Box, M.; Lalli, G.; Schiavo, G.; Schneider, H.; Groves, M.; Siligardi, G.; Fairweather, N. Analysis of mutants of tetanus toxin Hc fragment: Ganglioside binding, cell binding and retrograde axonal transport properties. Mol. Microbiol. 2000, 37, 1041–1051. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, D.M.; Hallewell, R.A.; Chen, L.L.; Fairweather, N.F.; Dougan, G.; Savitt, J.M.; Parks, D.A.; Fishman, P.S. Delivery of recombinant tetanus-superoxide dismutase proteins to central nervous system neurons by retrograde axonal transport. Exp. Neurol. 1997, 145, 546–554. [Google Scholar] [CrossRef] [PubMed]
- Coen, L.; Osta, R.; Maury, M.; Brûlet, P. Construction of hybrid proteins that migrate retrogradely and transynaptically into the central nervous system. Proc. Natl. Acad. Sci. USA 1997, 94, 9400–9405. [Google Scholar] [CrossRef]
- Fishman, P.S.; Savitt, J.M.; Farrand, D.A. Enhanced CNS uptake of systemically administered proteins through conjugation with tetanus C-fragment. J. Neurol. Sci. 1990, 98, 311–325. [Google Scholar] [CrossRef] [PubMed]
- Townsend, S.A.; Evrony, G.D.; Gu, F.X.; Schulz, M.P.; Brown, R.H., Jr.; Langer, R. Tetanus toxin C fragment-conjugated nanoparticles for targeted drug delivery to neurons. Biomaterials 2007, 28, 5176–5184. [Google Scholar] [CrossRef] [PubMed]
- Bearer, E.L.; Breakefield, X.O.; Schuback, D.; Reese, T.S.; LaVail, J.H. Retrograde axonal transport of herpes simplex virus: Evidence for a single mechanism and a role for tegument. Proc. Natl. Acad. Sci. USA 2000, 97, 8146–8150. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.K.; Teng, Q.; Garrity-Moses, M.; Federici, T.; Tanase, D.; Imperiale, M.J.; Boulis, N.M. A novel peptide defined through phage display for therapeutic protein and vector neuronal targeting. Neurobiol. Dis. 2005, 19, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Federici, T.; Liu, J.K.; Teng, Q.; Yang, J.; Boulis, N.M. A means for targeting therapeutics to peripheral nervous system neurons with axonal damage. Neurosurgery 2007, 60, 911–918. [Google Scholar]
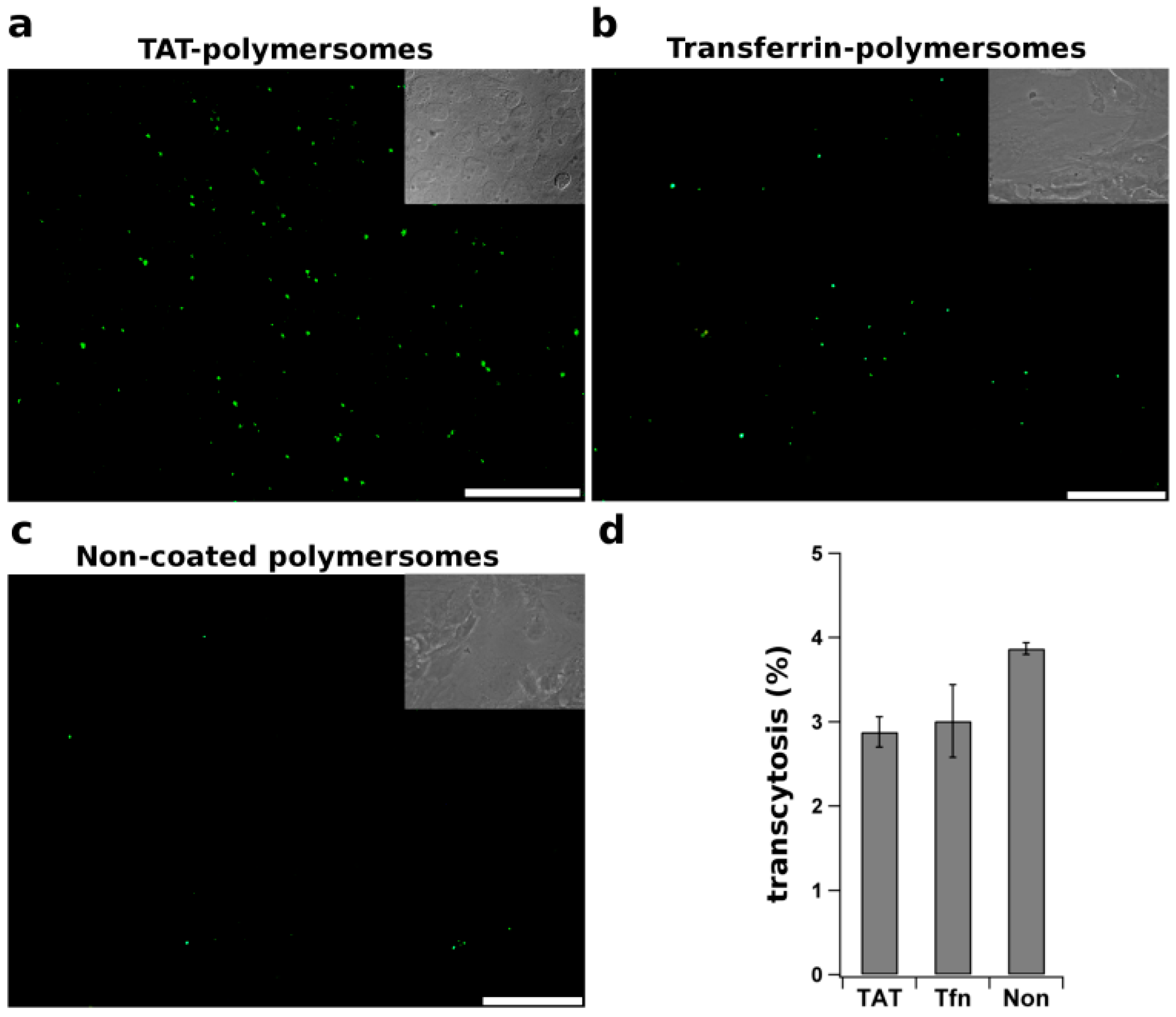
- Georgieva, J.V.; Brinkhuis, R.P.; Stojanov, K.; Weijers, C.A.G.M.; Zuilhof, H.; Rutjes, F.P.J.T.; Hoekstra, D.; van Hest, J.C.M.; Zuhorn, I.S. Peptide-mediated blood–brain barrier transport of polymersomes. Angew. Chem. Int. Ed. 2012, 51, 8339–8342. [Google Scholar] [CrossRef]
- Gaillard, P.; Brink, A.; Deboer, A. Diphtheria toxin receptor-targeted brain drug delivery. Int. Congr. Ser. 2005, 1277, 185–198. [Google Scholar] [CrossRef]
- Gaillard, P.J.; Visser, C.C.; de Boer, A.G. Targeted delivery across the blood-brain barrier. Expert Opin. Drug Deliv. 2005, 2, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Patel, L.N.; Zaro, J.L.; Shen, W.-C. Cell penetrating peptides: Intracellular pathways and pharmaceutical perspectives. Pharm. Res. 2007, 24, 1977–1992. [Google Scholar] [PubMed]
- Schwarze, S.R. In vivo protein transduction: Delivery of a biologically active protein into the mouse. Science 1999, 285, 1569–1572. [Google Scholar] [CrossRef] [PubMed]
- Lindgren, M.; Hällbrink, M.; Prochiantz, A.; Langel, U. Cell-penetrating peptides. Trends Pharmacol. Sci. 2000, 21, 99–103. [Google Scholar] [CrossRef] [PubMed]
- Santra, S.; Yang, H.; Stanley, J.T.; Holloway, P.H.; Moudgil, B.M.; Walter, G.; Mericle, R.A. Rapid and effective labeling of brain tissue using TAT-conjugated CdS: Mn/ZnS quantum dots. Chem. Commun. Camb. 2005, 3144–3146. [Google Scholar] [CrossRef]
- Simon, M.J.; Kang, W.H.; Gao, S.; Banta, S.; Morrison, B. Evaluation of the cell-penetrating peptide TAT as a trans-blood-brain barrier delivery vehicle. In Bioengineering Conference, Proceedings of the 2010 IEEE 36th Annual Northeast, New York, NY, USA, 26–28 March 2010; pp. 1–2.
- Rao, K.S.; Reddy, M.K.; Horning, J.L.; Labhasetwar, V. TAT-conjugated nanoparticles for the CNS delivery of anti-HIV drugs. Biomaterials 2008, 29, 4429–4438. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Guo, K.; Lu, J.; Venkatraman, S.S.; Luo, D.; Ng, K.C.; Ling, E.-A.; Moochhala, S.; Yang, Y.-Y. Biologically active core/shell nanoparticles self-assembled from cholesterol-terminated PEG-TAT for drug delivery across the blood-brain barrier. Biomaterials 2008, 29, 1509–1517. [Google Scholar] [CrossRef] [PubMed]
- Georgieva, J.V.; Kalicharan, D.; Couraud, P.O.; Romero, I.A.; Weksler, B.; Hoekstra, D.; Zuhorn, I.S. Surface characteristics of nanoparticles determine their intracellular fate in and processing by human blood-brain barrier endothelial cells in vitro. Mol. Ther. 2011, 19, 318–325. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Georgieva, J.V.; Hoekstra, D.; Zuhorn, I.S. Smuggling Drugs into the Brain: An Overview of Ligands Targeting Transcytosis for Drug Delivery across the Blood–Brain Barrier. Pharmaceutics 2014, 6, 557-583. https://0-doi-org.brum.beds.ac.uk/10.3390/pharmaceutics6040557
Georgieva JV, Hoekstra D, Zuhorn IS. Smuggling Drugs into the Brain: An Overview of Ligands Targeting Transcytosis for Drug Delivery across the Blood–Brain Barrier. Pharmaceutics. 2014; 6(4):557-583. https://0-doi-org.brum.beds.ac.uk/10.3390/pharmaceutics6040557
Chicago/Turabian StyleGeorgieva, Julia V., Dick Hoekstra, and Inge S. Zuhorn. 2014. "Smuggling Drugs into the Brain: An Overview of Ligands Targeting Transcytosis for Drug Delivery across the Blood–Brain Barrier" Pharmaceutics 6, no. 4: 557-583. https://0-doi-org.brum.beds.ac.uk/10.3390/pharmaceutics6040557



